Abstract
A 2-y-old Brahman bull was presented with progressive hindlimb ataxia and paraparesis that led to recumbency. Postmortem examination revealed scattered pinpoint, red-brown foci within the brainstem and gray matter of the spinal cord, and a larger lesion within the spinal cord at the level of T13. Histology of the section of T13 contained cross-sections of nematodes consistent with Parelaphostrongylus tenuis. Evidence of inflammation was present in other affected areas of the spinal cord and brain. DNA extraction and nested PCR were performed, which demonstrated 98% identity and 100% coverage to both P. tenuis and P. andersoni. Our case highlights the utility of DNA sequencing in parasite identification.
Parelaphostrongylus tenuis (i.e., meningeal worm) is a protostrongylid nematode parasite that rarely causes clinical disease in the definitive host, the white-tailed deer (Odocoileus virginianus). 12 However, when P. tenuis infects aberrant hosts, including domestic ungulates and other cervid species, it can migrate extensively through the spinal cord and brain, causing severe mechanical damage and inflammation. Cerebrospinal nematodosis is reported in cervids, camelids, small ruminants, horses, and calves.2,6,11,17 The classic signs are progressive asymmetrical ataxia of the hindlimbs, although a wide range of neurologic deficits are possible, depending on the area of the central nervous system affected.8,10 To date, the only antemortem test suggestive of P. tenuis in patients with compatible clinical signs is cerebrospinal fluid (CSF) analysis, and affected animals typically have eosinophilic pleocytosis.4,11,13 Definitive diagnosis has relied on autopsy, with identification of the nematode or characteristic tissue changes histologically, although PCR has become increasingly useful in reaching a diagnosis.
A 2-y-old Brahman bull was presented to the Texas A&M Large Animal Hospital (College Station, TX) with a 1-wk history of hindlimb ataxia and paraparesis and a 1-d history of recumbency. The bull lived on pasture with 7 other similarly aged bulls. Bulls had been dewormed with topical moxidectin 3 mo previously and were reportedly up-to-date on vaccinations (bovine viral diarrhea virus, bovine herpesvirus 1, bovine parainfluenza 3 virus, bovine respiratory syncytial virus, and a multivalent clostridial vaccine). Another bull in the pasture had developed similar signs 6 mo previously and was euthanized after prolonged recumbency, with no postmortem examination performed.
On presentation, the bull was in sternal recumbency with normal mentation and cranial nerve reflexes. Vital parameters (heart rate, respiratory rate, and temperature) were within normal limits. Auscultation of the thorax and abdomen was normal. Body condition score was 5 of 9. A patellar reflex could not be elicited in either limb, but appropriate withdrawal reflexes were present in the hindlimbs. Sensation and deep pain were present in all limbs. Anal tone, tail tone, and perineal reflex were normal. The forelimbs were apparently normal at admission but became weaker, as evidenced by the inability to move from lateral to sternal recumbency and support this position during hospitalization.
Radiographs of the thoracolumbar spine revealed no fractures or narrowing of the spinal canal. CSF was collected from the lumbosacral space, and analysis revealed an increased nucleated cell count of 0.056 × 109/L (56/µL; reference interval [RI]: < 0.003 × 109/L, < 3/µL), red blood cell count of 0.120 × 109/L (120/µL; RI: 0 × 109/L, 0/µL), total protein of 1.35 g/L (135 mg/dL; RI: 0.2–0.4 g/L, 20–40 mg/dL), and eosinophilia (75% eosinophils, 17% small lymphocytes, 8% large mononuclear cells). 15 Bovine leukemia virus serum ELISA was negative. The bull was treated with 0.2 mg/kg dexamethasone intramuscularly and 50 mg/kg fenbendazole orally once daily but was euthanized on the second day of hospitalization because of progression of clinical signs and poor prognosis.
At autopsy, the principal gross changes included right-sided subcutaneous edema and necrosis of the musculature of the right hindlimb, consistent with prolonged recumbency. The spinal cord, vertebral column, and brain were grossly normal on external examination. Numerous 5 mm diameter, white, firm masses were on the serosal surface of the jejunum, consistent with Oesophagostomum species infection. Collected tissues were fixed in 10% neutral-buffered formalin. After 48–72 h of fixation, the brain and spinal cord were sectioned every ~ 5 mm. Scattered pinpoint, red-brown foci were within the brainstem and gray matter of the spinal cord. A 2 × 2 mm, red-brown focus was within the left ventrolateral spinal cord at the level of T13 (Fig. 1).
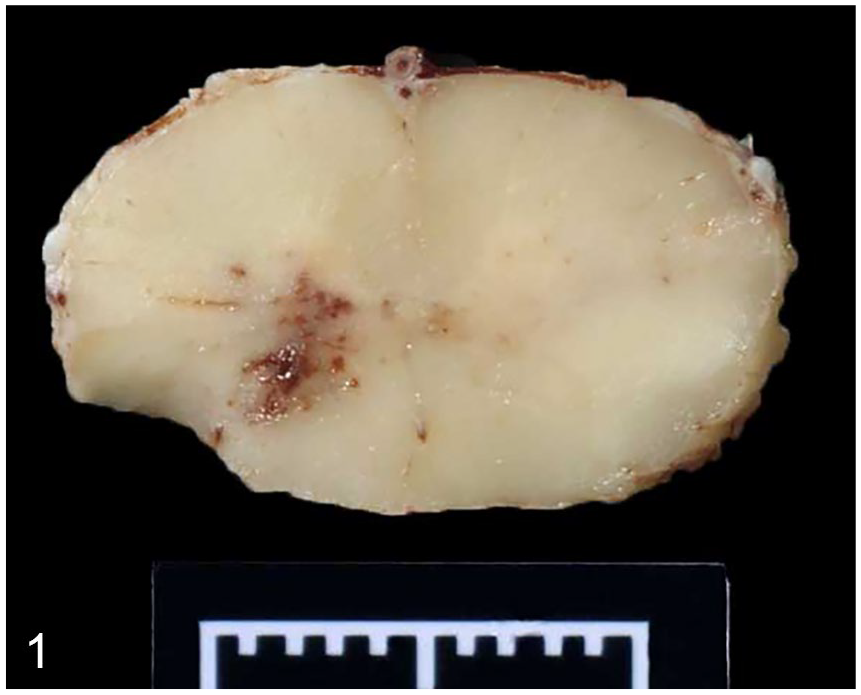
Transverse section of the spinal cord at the level of T13. An irregular area of red-brown discoloration is in the gray matter. Marker = 1 cm.
For histologic evaluation, formalin-fixed tissues were embedded in paraffin, sectioned at 5 μm, and stained with hematoxylin and eosin. On histologic examination of T13, there were several cross-sections of one or more degenerate, adult nematodes within the dilated central canal, surrounded by degenerate eosinophils and epithelioid macrophages (Figs. 2, 3). Nematodes had an eosinophilic cuticle, coelomyarian–polymyarian musculature, pseudocoelom, lateral cords, a gastrointestinal tract lined by multinucleate epithelial cells, and a reproductive tract. Cross-sections of the parasite averaged 162 μm diameter. Moderate numbers of lymphocytes, eosinophils, plasma cells, and macrophages surrounded adjacent blood vessels and extended into the meninges (Fig. 4). Multifocally within the peripheral white matter tracts were areas of moderately to markedly dilated myelin sheaths containing spheroids, macrophages, or cellular debris (Fig. 5). Multifocal areas of hemorrhage were within the gray matter. Other sections of the brainstem and spinal cord had areas of hemorrhage, mild eosinophilic inflammation, and degeneration of the white matter, but no evidence of parasites.
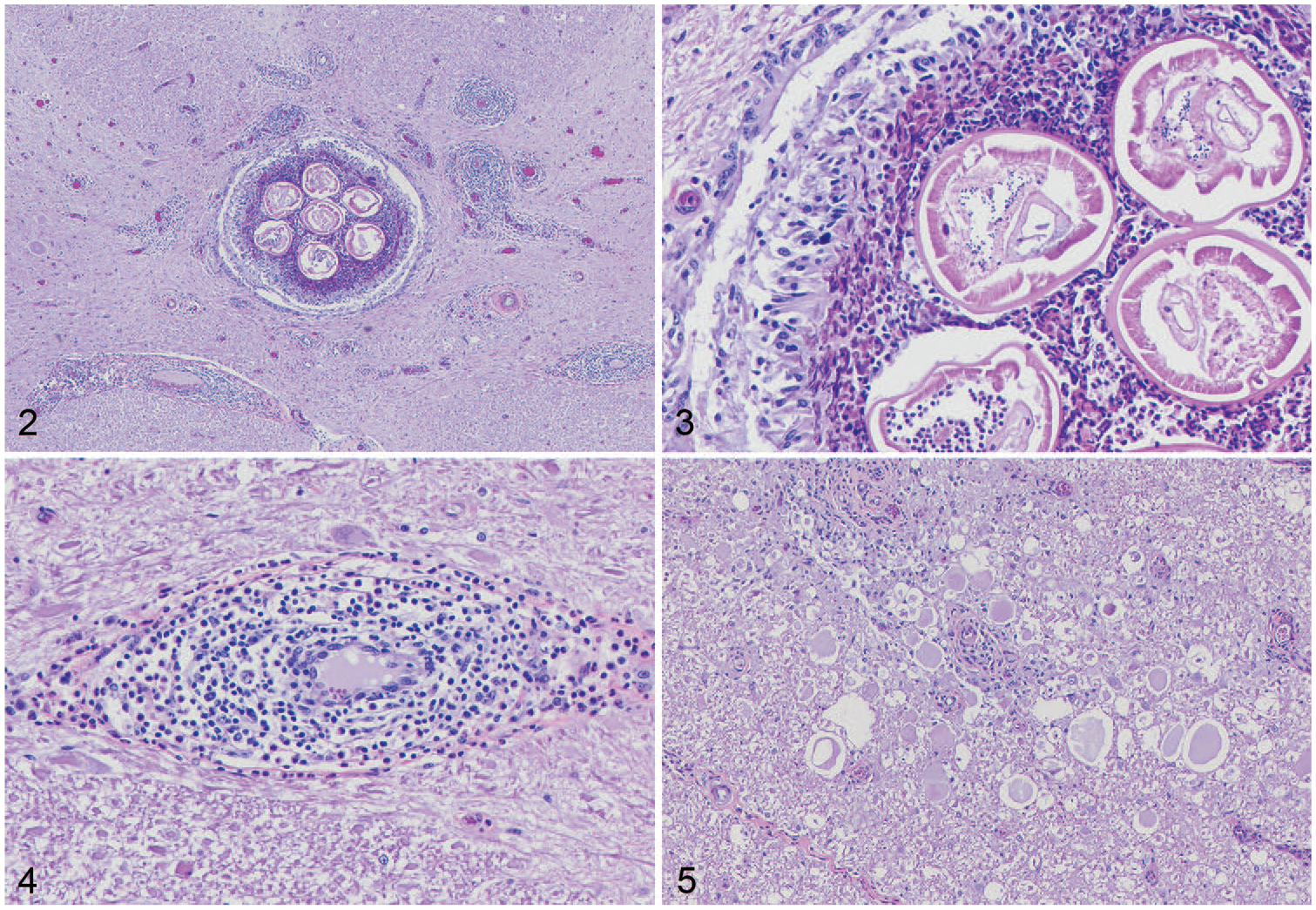
Photomicrographs of the spinal cord at the level of T13.
Ten 5-μm sections of paraffin-embedded tissue from T13 were submitted for PCR at the molecular parasitology laboratory at the University of Tennessee College of Veterinary Medicine (Knoxville, TN). DNA extraction and nested PCR were performed as described previously. 11 PCR products were sequenced using nested PCR primers, and resultant sequences had a 98% identity and 100% coverage to both P. tenuis and P. andersoni.
P. tenuis is endemic in central and eastern regions of North America where there are large white-tailed deer populations. The adult nematode lives within the subarachnoid space and typically causes no damage or clinical signs in deer (nematodiasis). Eggs are deposited into the venous sinuses and travel to the lungs, where they hatch into first-stage larvae that are coughed up and swallowed. The larvae pass through the gastrointestinal tract to eventually become third-stage larvae in the gastropod intermediate hosts, which are then ingested by grazing ungulates. Once ingested by an aberrant host, the larvae migrate from the abomasum to the central nervous system, causing trauma and inflammation as they pass through the spinal cord and brain. 12
Presumptive antemortem diagnosis of P. tenuis infection relies on clinical signs and CSF analysis. Eosinophilic pleocytosis of CSF in camelids is considered a sensitive laboratory test for P. tenuis and is reported in acute cases of infection in cattle.11,13 Postmortem diagnosis of nematodosis has previously relied on identification of larvae or compatible histopathologic changes. Nested PCR has been used to confirm the presence of Parelaphostrongylus DNA in fixed nervous tissue, and the presence of P. tenuis and related species have been confirmed with sequencing.4,11,16,17
Previous reports of Parelaphostrongylus spp. infection in cattle are rare, and only reported in calves < 1 y old.5,11,18 Adult cattle have been considered resistant to infection with P. tenuis; other species, such as camelids and wild hoofstock, are considered more susceptible.3,7 The clinical signs of ataxia, paraparesis, and recumbency coupled with eosinophilic pleocytosis of CSF were consistent with those reported in other acute cases, although some calves with chronic infection (> 1 mo duration of signs) were reported to have lymphocytic pleocytosis.11,18
Previous confirmation of nematodosis in cattle has been made based on identification of nematodes and eosinophilic and lymphoplasmacytic meningomyelitis on histologic examination of spinal cord sections,5,18 although PCR and DNA sequencing were utilized as confirmation in 2 calves with compatible clinical signs and histologic lesions. 11 P. tenuis larvae mature within the dorsal gray column of the definitive host before migrating to the subarachnoid space, and nematodes have been identified in the dorsal gray column and all areas of the spinal white matter of affected cattle.5,11,18 Our case notably contained nematodes within the central canal, which has not been previously reported in cattle, to our knowledge.
Treatment protocols utilizing an anti-inflammatory medication (typically flunixin meglumine or dexamethasone) paired with an anthelminthic (fenbendazole or ivermectin) have been reported with variable success.2,7,12 Although clinical signs reportedly improved after treatment in a group of affected calves after confirmation of P. tenuis infection, 11 no response was seen in our case.
Although the sequence results in our case had 98% identity to both P. tenuis and P. andersoni, the width of the nematode averaged 162 µm, which is larger than P. andersoni (males: 87–140 µm, females: 92–130 µm), but consistent with P. tenuis (males: 92–200 µm, females: 120–250 µm). 9 P. andersoni infects the musculature of multiple cervid species and has been documented in several areas of the United States and Canada, including the southeastern United States.1,14 It has not been documented as a cause of cerebrospinal nematodosis in animals. The morphology of the parasite was consistent with P. tenuis, and the clinical course of disease was similar to that described in other species.
To our knowledge, Parelaphostrongylus nematodosis has not been reported previously in an adult bovid. Lack of previous diagnosis in adult cattle may have less to do with low prevalence, and more to do with the challenges of transporting weak or recumbent cattle, performing an autopsy, and successfully sectioning the affected area of spinal cord to reach a diagnosis. Although rare, P. tenuis infection should be included as a differential diagnosis of neurologic disease in adult cattle with compatible signs in endemic regions of North America.
Footnotes
Acknowledgements
We thank the University of Tennessee sequencing facility and Megan Miller for assistance with PCR.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
