Abstract
The new Analyst™ GT is a multi-mode plate reader supporting all five leading technologies: fluorescence polarization (FP), fluorescence intensity, time-resolved fluorescence (TRF), absorbance and luminescence. It can be easily integrated into both workstation and robotic environments for HTS for all major non-radiometric applications. Analyst™ GT is based on second-generation SmartOptics™, a patented system that combines light sources, optical components and detectors together for fast high-precision measurements in 96-, 384-, and 1536-well microplates. Applications include kinase and photophase assays, receptor ligand binding assays, protease assays, cAMP, and cGMP, signal transduction, reporter gene assays and many others.
In this study, we not only optimized measurement parameters on Analyst™ GT for all five modalities but also compared instrument settings and performance with the first generation Analyst HT and Acquest.
Introduction
Analyst GT is the second generation of Analyst Systems from Molecular Devices Corp. Its performance, in terms of its speed, sensitivity and precision has been greatly improved. Analyst GT delivers the highest precision available for FP assays in 96-through 1536-well formats. The result is greater Z (or Z′)-factor values even for challenging assays with low fluorophore concentrations. Analyst GT supports multiple-wavelength assays, enabling users to measure up to eight different wavelengths per well. New to the Analyst GT are time-course measurements that enable entire sets of plates to be read and data to be stored between reads.

SoftMax® Pro, Molecular Devices' industry-standard data analysis software, offers comprehensive data reduction and graphing capabilities for Analyst GT, including predefined protocols for determining calibration curves, percent activity and total intensity vs.mP for FP assays.
Materials and Methods
For absorbance, a serial dilution of tartrazine was prepared from 442 mg/L stock solution with water. It had 200mL/well for a 96-, 80 mL/well for a 384-, and 8 mL/well for a 1536-well plate (96- and 384-well plate from Corning Costar, 1536-well plate from Greiner). They were all clear bottom, black. Eight replicates in 96-well, 16 replicates in 384-well, and 8 replicates in 1536-well plates were made for each tartrazine concentration and water only for background subtraction. The plates were read on Analyst GT using an excitation filter of 405, and a 50% transmission mirror.
For fluorescence intensity. a 1 to 3 log serial dilution of fluorescein was prepared with PBS buffer (pH 7.4). It had 200mL/well for a 96-, 80 mL/well for a 384- and 8 mL/well for a 1536-well plate (plates were from Corning Costar and Greiner). Numbers of replicates were the same as in absorbance measurement. For TRF, a 1 to 3 log serial dilution of europium solution was prepared with DELFIA, Enhancement solution (PerkinElmer, Cat.#1244–105). Corning Costar's 96- and Greiner's 384- and 1536-well plates were used. Numbers of replicates were the same as in absorbance measurements.
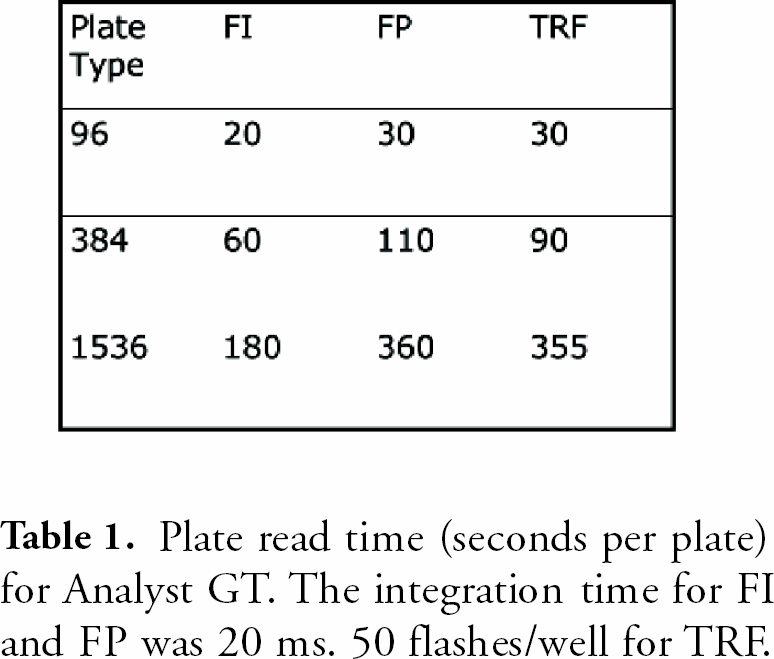
For FP, a dilution serial of fluorescein in a 384-well plate and 1nM, 10 nM fluorescein solutions in 384-well plate were prepared and plates were read on Analyst GT.
For luminescence, the substrate and buffer for luciferase titration were from MDC. The luciferase and alkline phosphatase were from Sigma. White Packard (96-well) or Greiner (384- and 1536- well) plates were used.
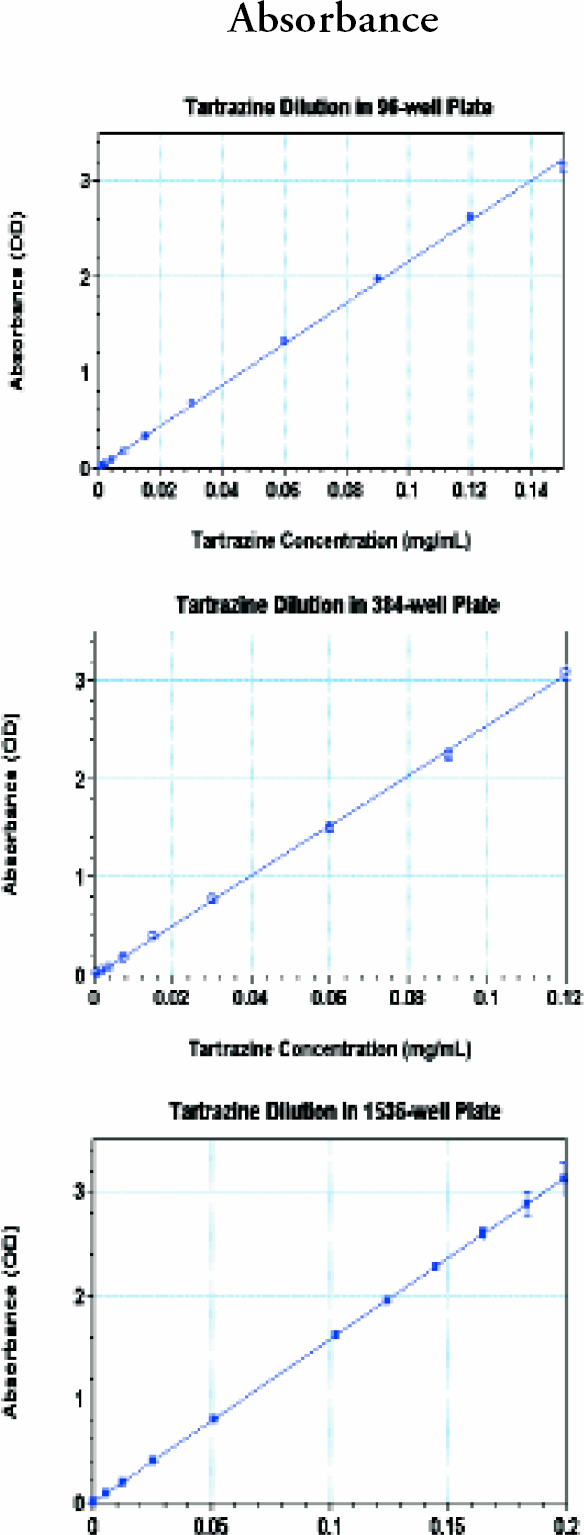
(upper), 2 (middle) and 3 (bottom): Analyst GT absorbance performance.
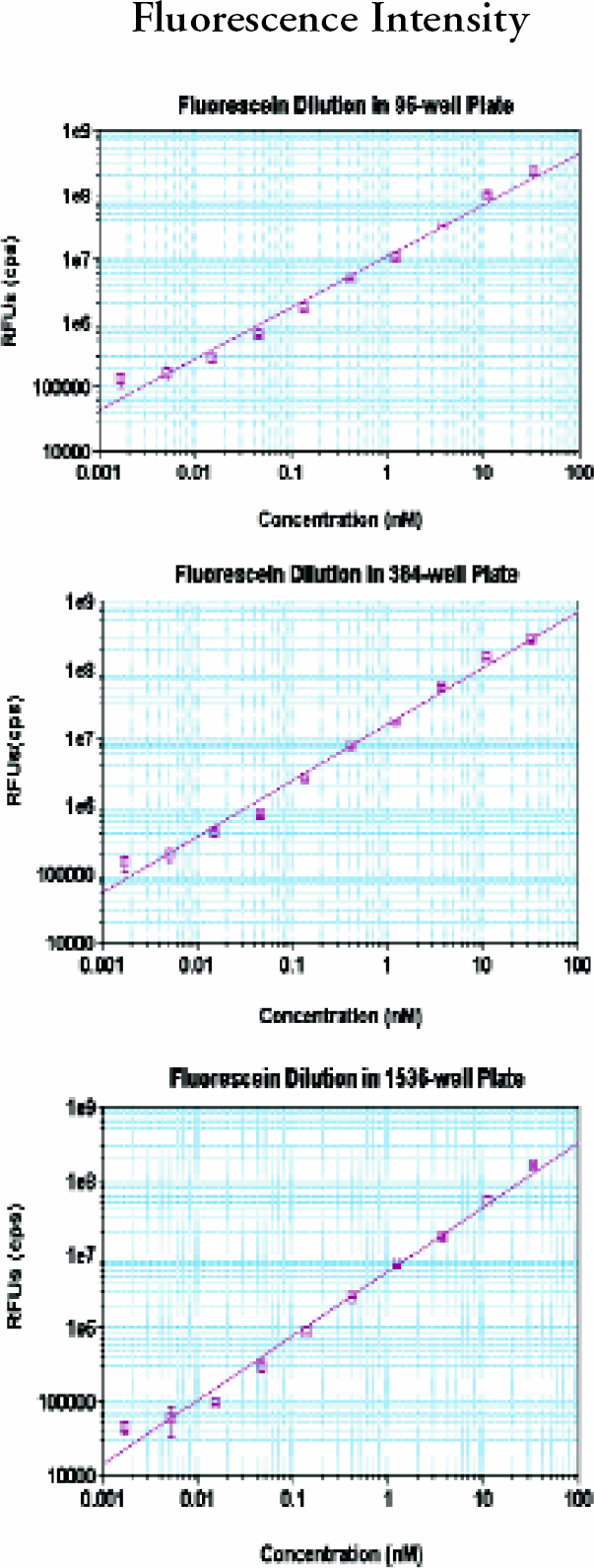
(upper), 5 (middle) and 6 (bottom): Analyst GT fluorescence intensity performance.
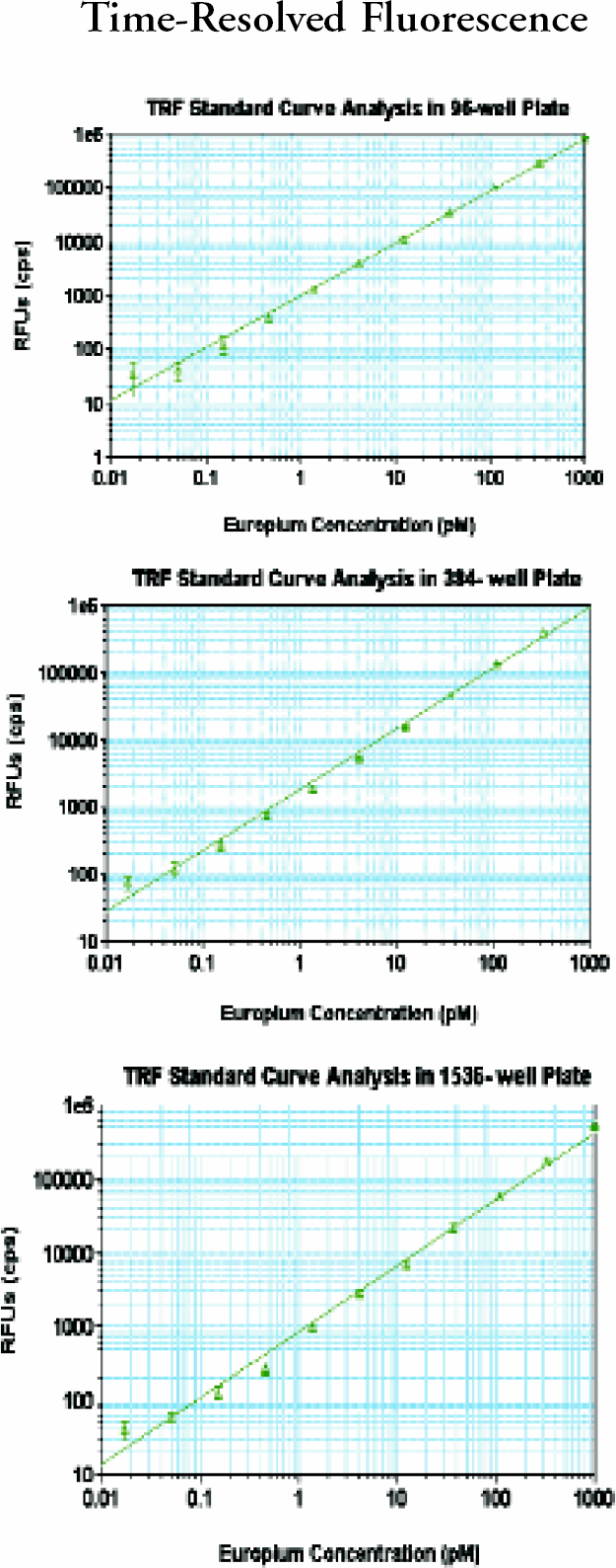
(upper), 9 (middle) and 10 (bottom): Analyst GT TRF performance.
Plate read time (seconds per plate) for Analyst GT. The integration time for FI and FP was 20 ms. 50 flashes/well for TRF.
Results
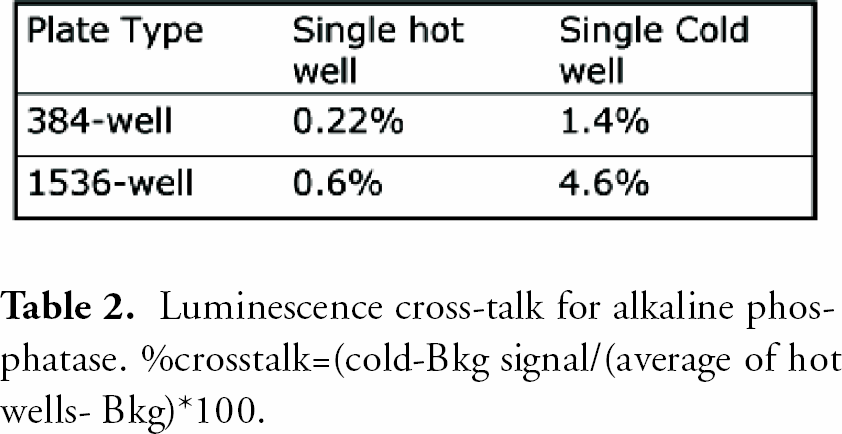
In all three plate formats, absorbance was linear up to 3 OD with very small standard deviations (SDs). Fluorescence intensity dynamic ranges were greater than 3 magnitudes. The lower limits of detection were 2–5 pM. The precision for FP at 10 pM was less than 1 mP and less than 2 mP at 1 nM fluorescein for 384- well plate. The dynamic ranges for TRF were greater than 4 magnitudes with very small SDs. The lower limits of detection in TRF were less than 100 fM europium. Cross-talk for luminescence was 1.4% in 384-well and 4.6% in 1536-well plate in the worse case scenario; a cold(negative) well surrounded by hot (positive) wells.
(upper), 12 (middle) and 13 (bottom): Analyst GT FP performance. Fig. 13 is showing a FP kinase (IMAP%) assay read on Analyst HT (blue) and GT (green). Analyst GT was twice as fast as HT for this 384-well plate assay, but with similar assay results.
Conclusion
After this thorough study, we conclude that Analyst GT brings sensitivity, precision, robustness, and speed for a multimode plate reader to new high standards. It is sufficient to use only 20 ms integration time for FI and F P, and 50 flashes for TRF, without sacrificing sensitivity. The speed of TRF has increased more than fourfold compared to Analyst first generation instruments. The performance range for absorbance is greater than 3 ODs for all 96-, 384-, and 1536-well plates.
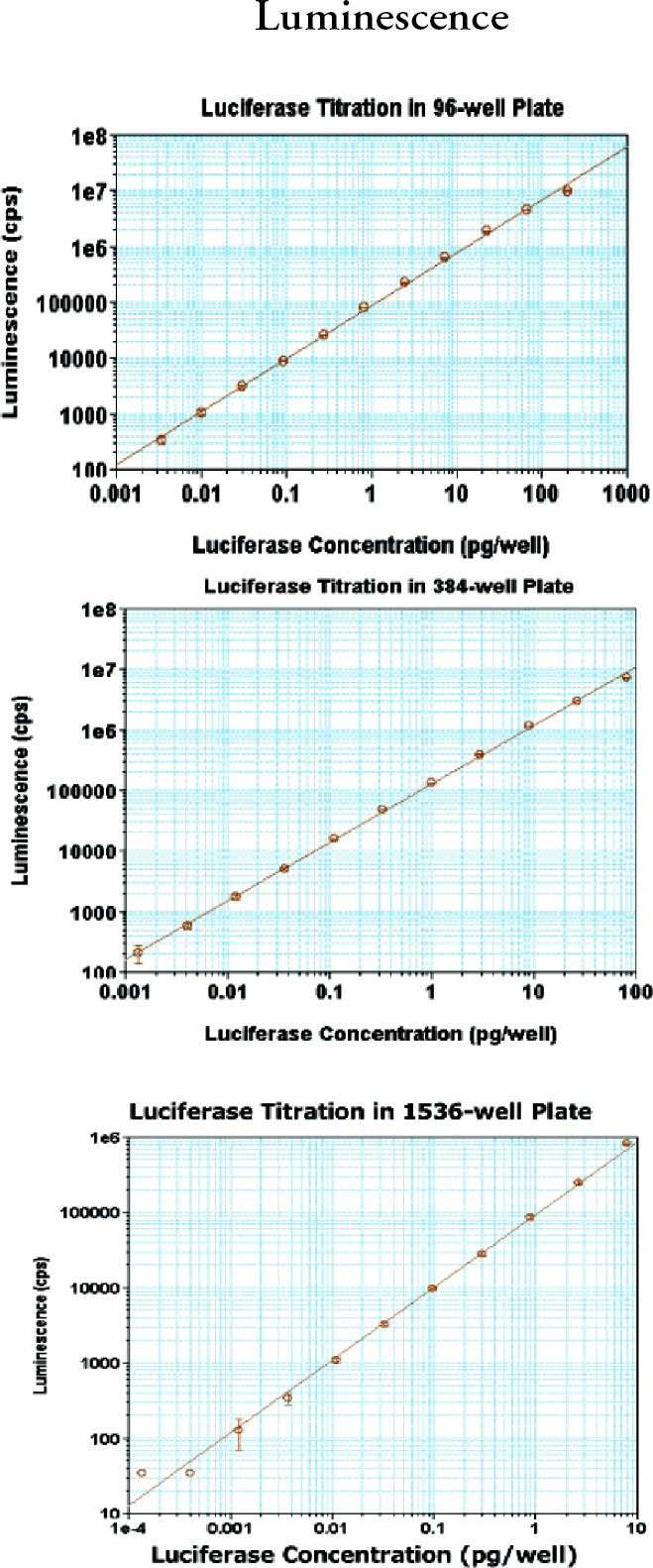
(upper), 15 (middle) and 16 (bottom): Analyst GT luminescence performance.
Luminescence cross-talk for alkaline phosphatase. %crosstalk=(cold-Bkg signal/(average of hot wells-Bkg)*100.