Abstract
Traditional organic synthesis is often cumbersome and time consuming. Significant effort by manufacturers of automated systems has been directed at increasing the speed, efficiency, and consistency of performing chemical reactions. To date, the numbers of integrated systems that combine all the relevant steps of compound preparation are few.
Using the five automated systems in tandem can successfully streamline research and development of potential drug candidates. The features of these workstations offer a unique approach for supporting the convenient synthesis and workup of diverse compounds without compromising reagent types or conditions used for synthesis.
This poster details an example of the automated drug discovery approach to high throughput organic synthesis using the solution phase synthesis of a series of 1-indanones as building blocks and subsequent reductive amination reactions to generate a diverse group of amines. The purpose of each automated system is outlined along with its role in generating the small library as an illustration of the importance of time saving devices in laboratories.
Workflow
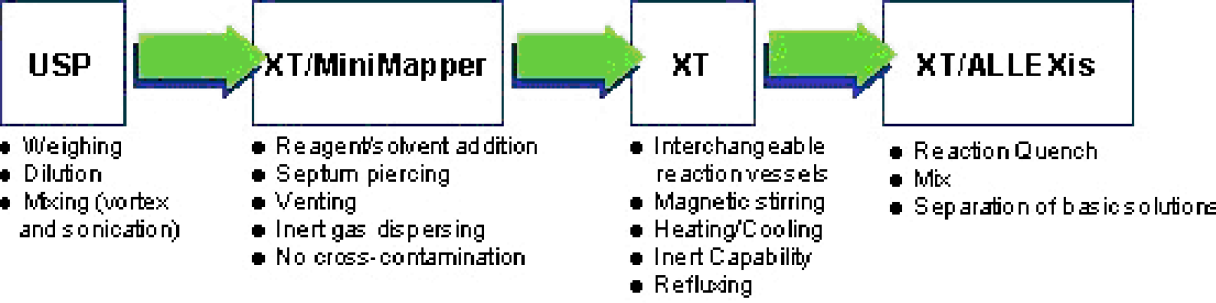
AutoChem Automated Systems
The automated workstations utilized in this project are commercially available and meet the requirements to support most of the varied chemical processes performed day to day in laboratories.
REAGENT PREPARATION
The USP workstation, shown in Figure 1, enables cost effective automation that is reliable in addition to offering increased throughput for several laboratory processes. Its features include:
Tare and gross weighing
Tracking laboratory vessels using barcodes
Preparing solutions by converting measured weights of reactants to exact concentrations by calculating solvent addition
Dissolution by mixing via vortexing and/or sonicating

USP Workstation.
MAPPING OF REAGENTS
The MiniMapper, shown in Figure 2, automates addition of all reagents and solvents into the MiniBlock XT, previously setup for parallel synthesis. Its features include:
Accurate liquid handling
Variable solvent availability
Multiple reagent vials
Handles typical organic building blocks and reagents
Compact workstation
3-channel cannula that facilitates septum piercing, venting, and inert gas dispersing and prevents cross-contamination

MiniMapper.
REACTION SYNTHESIS
The MiniBlock XT, shown in Figure 3, is a parallel solution phase reaction block that is fully integrated with the AutoChem automated workstations. The MiniBlock XT features:
Readily interchangeable reaction vessels
Magnetic stirring for agitation
Heat provided through a heat dispersion adapter and regulated by feedback control electronics
Cooling
Inert capability
Refluxing

XT with Temperature Control.
REACTION WORKUP
The ALLEXis workstation, shown in Figure 4, automates liquid-liquid extractions and facilitates reaction quenching. Its features include:
Compact workstation that integrates with MiniBlock XT and other reaction vessels
Addition of solvents and quench reagents
Sample mixing
Phase separation by automated phase boundary detection technology
Phase distribution

ALLEXis.
Synthesis
This synthesis of diverse amines represents typical organic derivatives with potential biological activities and demonstrates AutoChem's automated workstations usage in generating varied libraries under difficult reaction conditions.
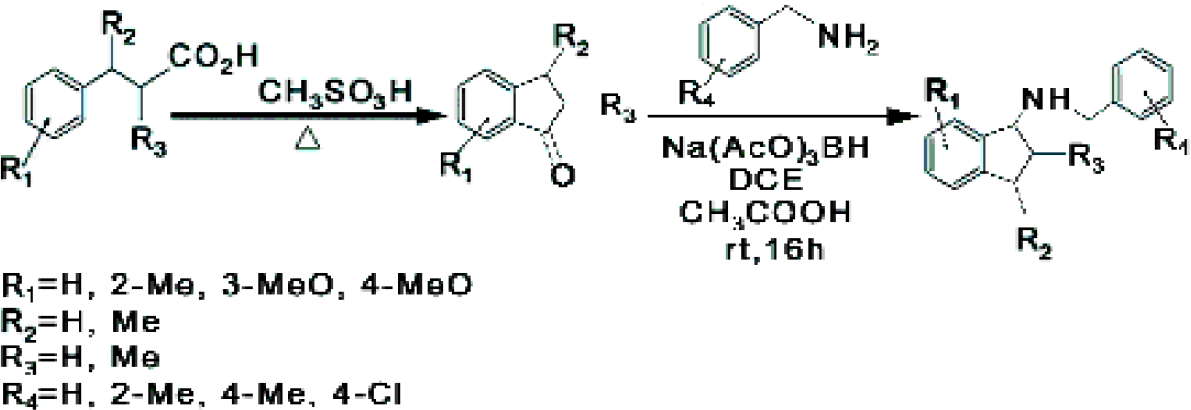
Experiment
INTRAMOLECULAR CYCLIZATION
MiniBlock XT
This experiment illustrates the advantages of the XT reaction block, shown in Figure 3, to handle corrosive agents. The 6-position MiniBlock XT solution phase vessels were utilized, enabling gram scale quantities of 1-indanone building blocks to be synthesized.
Phenylpropionic acids (6g) and Methanesulfonic acid (60g) were mixed and stirred at 40°C under nitrogen for 16 hours.
Reactions were monitored by HPLC until complete.
The contents of the reaction vessels were slowly poured into 200mL water and extracted with methylene chloride (3×50mLs).
The organic layers were washed with 10% sodium bicarbonate, separated, dried over anhydrous magnesium sulfate, and filtered to provide clear solutions.
The samples were evaporated to dryness in vacuo on an evaporator to get HPLC purities and crude yields. (The 5-Methoxy-1-Indanones were obtained from Sigma-Aldrich.)
REDUCTIVE AMINATION INTEGRATED WITH AUTOMATED SYSTEMS
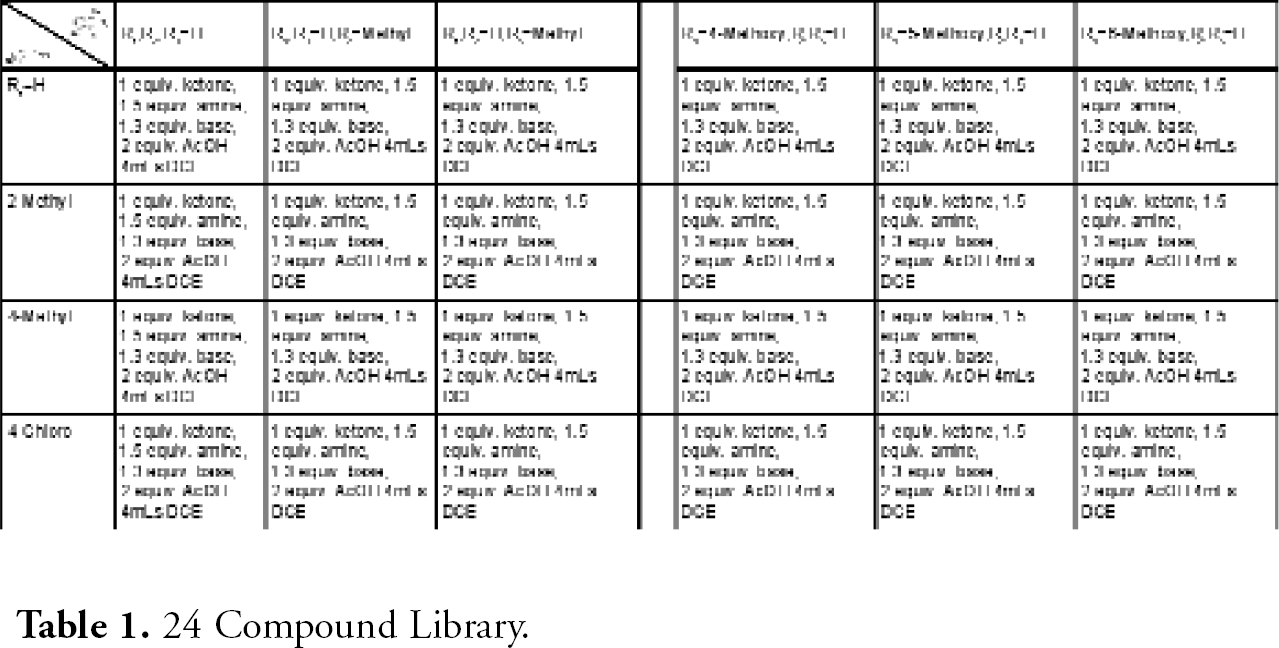
This reaction illustrates the advantages of using the automated systems in tandem. The systems were used to prepare a range of amines from 1-Indanones, shown in Table 1.
24 Compound Library.
USP - Universal Sample Preparation and Label Automator
An input file was generated containing bar-codes and weights of vessels for synthons, reagents, and final extracts. Solutions of 1-indanones and benzylamines in 1,2-dichloroethane (DCE) were generated using the input file.
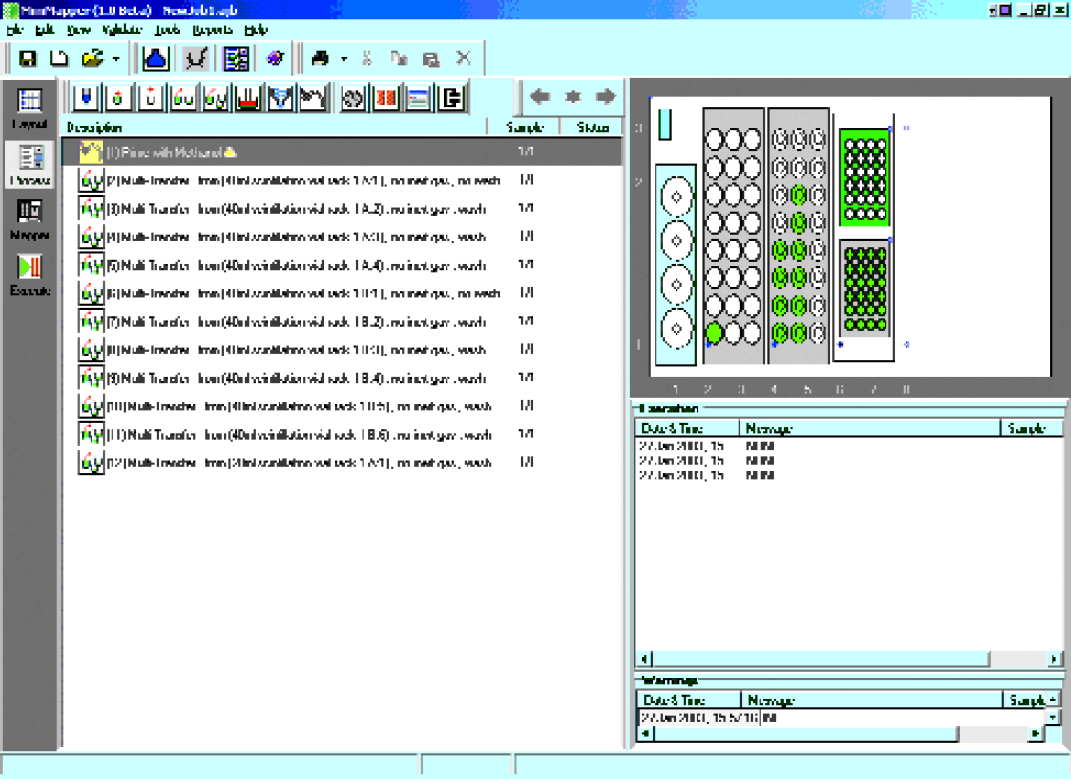
MiniMapper and MiniBlock XT
MiniMapper transferred Na(OCH3)3BH, 1-indanones, amines, and acetic acid to MiniBlock XT, generating the 24 compound array, as illustrated in Figure 5. See Table 1 for the details of the array. Then, the reactions were magnetically stirred at room temperature off-line.
ALLEXis
The XT reaction block was transferred to ALLEXis for reaction workup by quenching, extracting, and distributing extracts to bar-coded scintillation vials, shown in Figure 6 ALLEXis screenshot.
USP - Universal Sample Preparation
Vials were returned to USP, shown in Figure 1, after dry down for gross weights.
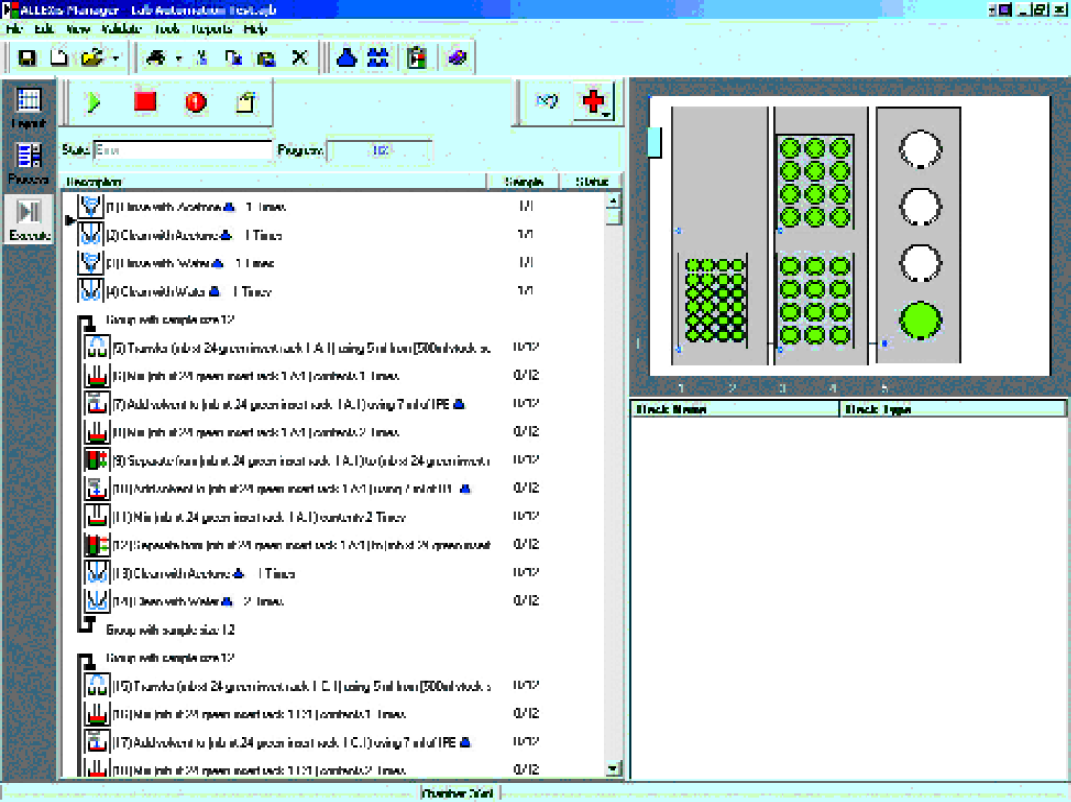
ALLEXis Screenshot.
Results
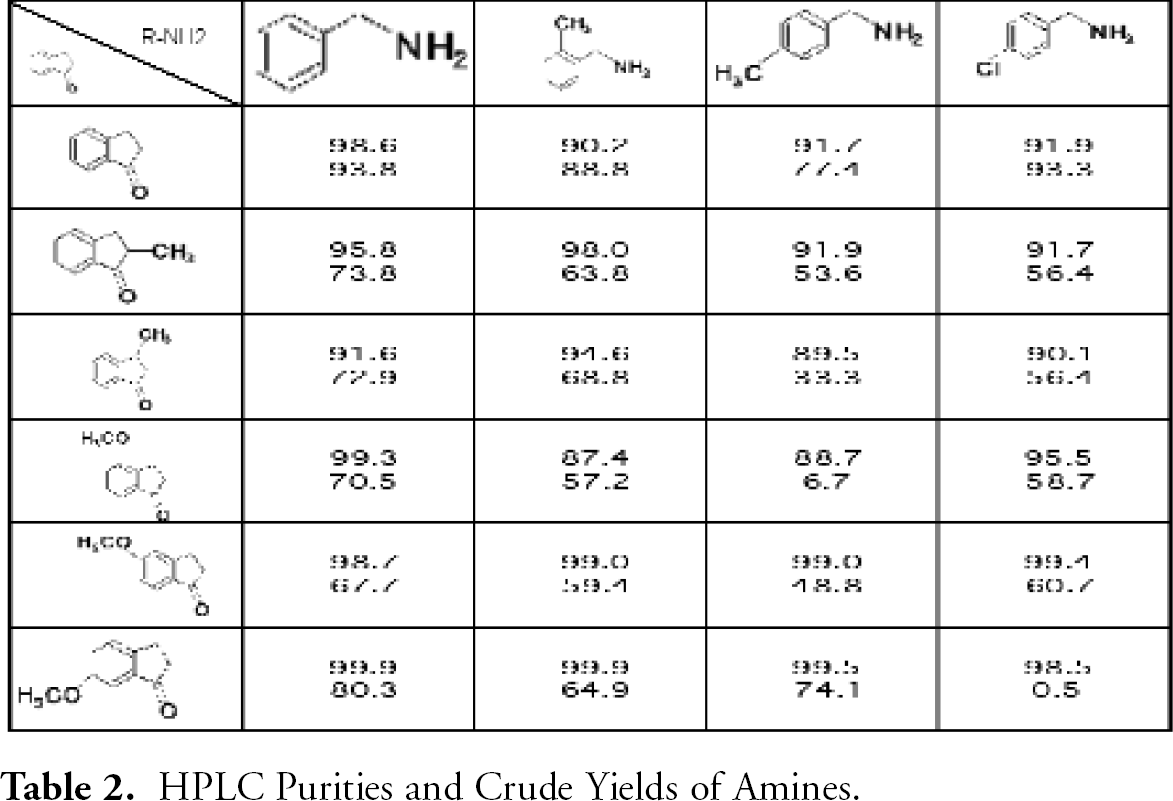
The reductive amination of various substituted 1-Indanones with primary amines was very successful, generating the desired products in very good yields and exhibiting excellent purities by HPLC. See Table 2. HPLC Purities and Crude Yields of Amines.
HPLC Purities and Crude Yields of Amines.
Conclusion
The synthesis of a library of potentially biologically active amines was successfully carried out by solution phase chemistry and expedited by the use of five automated workstations to increase the speed, efficiency, and consistency of the process. The AutoChem workstations utilized in tandem were able to alleviate bottlenecks, errors, and manipulations usually experienced in semi-automated or manual processes.
Footnotes
Acknowledgments
Shannon Lewis-Hillstrom, Tom Osborne, and Thierry Boxus.


