Abstract
The RoboMicroScope II is an image capture and analysis system, which enables the automated analysis of protein crystallization trials. The RoboMicroscope II couples robust hardware with intuitive software that tracks and assigns an objective score to each instance of a crystallization experiment. This machine can also be used in a manual mode, to increase the reliability, objectivity and efficiency of human scoring of crystallization trials.
Background
Structural biology has been widely embraced by the pharmaceutical and biotech industries as it provides an atomic description of macromolecules, and interactions between macromolecules and small (drug-like) molecules. X-ray crystallography is the most widely used technique for macromolecular structure elucidation, but does require the production of diffraction quality crystals. We present here a system that speeds and simplifies the observation and analysis of the myriad of crystallization trials that are set up in order to produce crystals of data collection quality.
Protein (or any other macromolecule) crystal growth involves screening crystallization space for conditions under which the protein forms large (100μm) single crystals. Protein crystal space is the collection of possible combinations of chemicals that may induce protein crystal growth, and is generally sampled by setting up 50–1000 trial experiments simultaneously. This space is huge, and spans hundreds of different chemicals, at varying concentrations and pH. The results from this battery of experiments are used to refine the crystallization conditions in second and subsequent rounds of crystallization. Crystallization trials are traditionally set up as vapor diffusion experiments, where a drop (0.5–5μl) of a concentrated protein solution is mixed with a similar quantity of crystallization cocktail, then equilibrated against a reservoir of the crystallization cocktail. Batch methods, where the protein and the cocktail are mixed, but not equilibrated against a reservoir, are also used.
The key to this iterative process of crystallization is to observe the results of the crystallization trials, and to correlate the results back to the cocktail of chemicals that was used in that experiment. Furthermore, we want to predict from these observations where a crystallization optimum would be. Crystallization is a time dependent event, so each trial must be observed many times, over a long time span (days to months). Temperature can be an important factor in protein crystallization, so often the trials are set up and stored in a 4°C cold room, which adds a measure of difficulty to the process of viewing the trials.
SYSTEM LEVEL ARCHITECTURE
RoboDesign has brought a complete systems (Figure 1) approach to automation of protein crystal analysis. This ensures the seamless integration of the many facets of the process: plate handling, image collection, image storage and analysis, crystallization data collection and analysis. Most importantly, it allows for the objective analysis of all crystallization trials

The key components of the system include experiment plate storage in the RoboIncubator, imaging of the crystallization trials on the RoboMicroScope II, image and data archiving on the Image dB, automatic image analysis (CPXO), and crystallization data management (CrystalTrak). Each step in the process is linked through barcode tracking. An intuitive user interface completes the package. Offline software allows for the remote viewing of all acquired images and scoring results. It also provides an interface for entering plate setup information, printing barcodes, and manually scoring images.
ROBOINCUBATOR
The RoboIncubator (Figure 2) is a fully automated, random access, temperature and humidity controlled storage device, which can hold up to 602 SBS footprint plates, 396 Linbro plates, or a combination of the two in a footprint of 1m. 2 The plates are stored in easily removable magazines and permit the reconfiguration of the device to handle many different types of plates. The interior temperature can be maintained at any specific setting from 4° C to ambient temperature and the relative humidity is controllable within a range of 45% to 75%. There is a small, automatic door that allows the RoboIncubator to load plates into the RoboMicroScope II, or other instruments, whilst maintaining the controlled environmental within the RoboIncubator. Crystallization plates can be loaded or removed via Input/Output (I/O) magazines that are placed or removed through the I/O doors at the front of the device. Upon request, the RoboIncubator will fill an I/O magazine in the output area with plates to be removed from storage, or will automatically place plates into storage from an I/O magazine placed in the input area. The RoboIncubator reads the barcodes of plates when they are put into storage as well as any time they are accessed for inspection or removal.

The plate storage area is isolated from vibrations of the environmental conditioning equipment and the speed, acceleration, and deceleration values of the plate handling robotics are carefully controlled. In addition, when a plate is requested, only the plate being accessed is moved. This is unlike the majority of incubators that must rotate all stored plates whenever any one plate is being accessed. These features provide the perfect environment and handling characteristics for protein crystal growth.
ROBOMICROSCOPE II
The RoboMicroscope II (Figure 3) is a robust crystal imaging system whth many features, including automatic drop location, automatic zoom level selection, automatic focusing, polarization analysis, barcode tracking, and image storage. The RoboMicroScope II images a 96 well hanging or sitting drop plate in less than five minutes. It collects an image with the full well in the field of view, and uses this image to automatically locate the drop. It then collects a number of “slice” images at various focal planes to image the entire depth of the drop. It has the option to combine in-focus information from all slices into a single, composite image that is particularly useful for scoring. All images - including the intermediate slices - are stored in the Image Database Server and may used for later viewing or scoring.

The RoboMicroScope II also comes equipped with a powerful suite of tools useful for manual exploration of a particular well or drop. This allows a crystallographer who is interested in a particular drop to zoom in on particular crystals, take higher magnification images, take pseudo-color polarized images, increase lighting, and make annotations associated with the image. Although the user has complete control over the system, the RoboMicroScope still keeps track of the images, and the custom images are stored along with the automatic images in the Image Database Server.
IMAGE QUALITY AND ROBOMICROSCOPE II
Image quality is of paramount importance for an automatic imaging system, particularly to ensure the consistency and reliability of an associated automatic scoring algorithm. Factors which contribute to image quality include:
Repeatability of measurements over long time frames
Uniformity of image contrast and gray level
Minimal shading across the image
Minimal dust and imaging artifacts
High ratio of signal contrast to shading contrast
Sharpness of image
Tuned depth of focus
Controlled extinction imaging for polarized imaging
Adequate resolution and pixel depth
Many combinations of lighting and lenses were tested to produce a system that performs well according to the criteria above. The figures below present a comparison of images from the RoboDesign imager and images that were provided from a facility that has designed its own image inspection system prototype (IISP).
Figure 4 shows four individual images of wells in a Greiner CrystalQuick flatbottom plate taken by the IISP. Each of the wells has a clear protein solution that has not yet formed precipitate or crystals. The four images exhibit a high degree of shading across each well (gray scale variation). The shading is probably a result of the imaging lens and the lighting. These four images present a real challenge to automated analysis.

Figure 5 shows a clear protein solution in the same Greiner plate as Figure 4. However, these images were taken with the RoboMicroScope II system. The images exhibit a high degree of uniformity in the lighting and in the gray scale across each well. These images are much easier to analyze automatically than the ones shown in Figure 4. Figures 6 and 7 provide further comparison between the IISP and RoboMicroscope II systems.

Figure 8 shows sitting drops of approximately 1ul total volume in the CrystalQuick plate and imaged with the IISP system. These images look much better than those in Figures 4 and 6 from an automatic analysis point of view. However, the background around the drops is saturated, this complicates automatic analysis, as it prevents accurate determination of grey scale value within the drop.
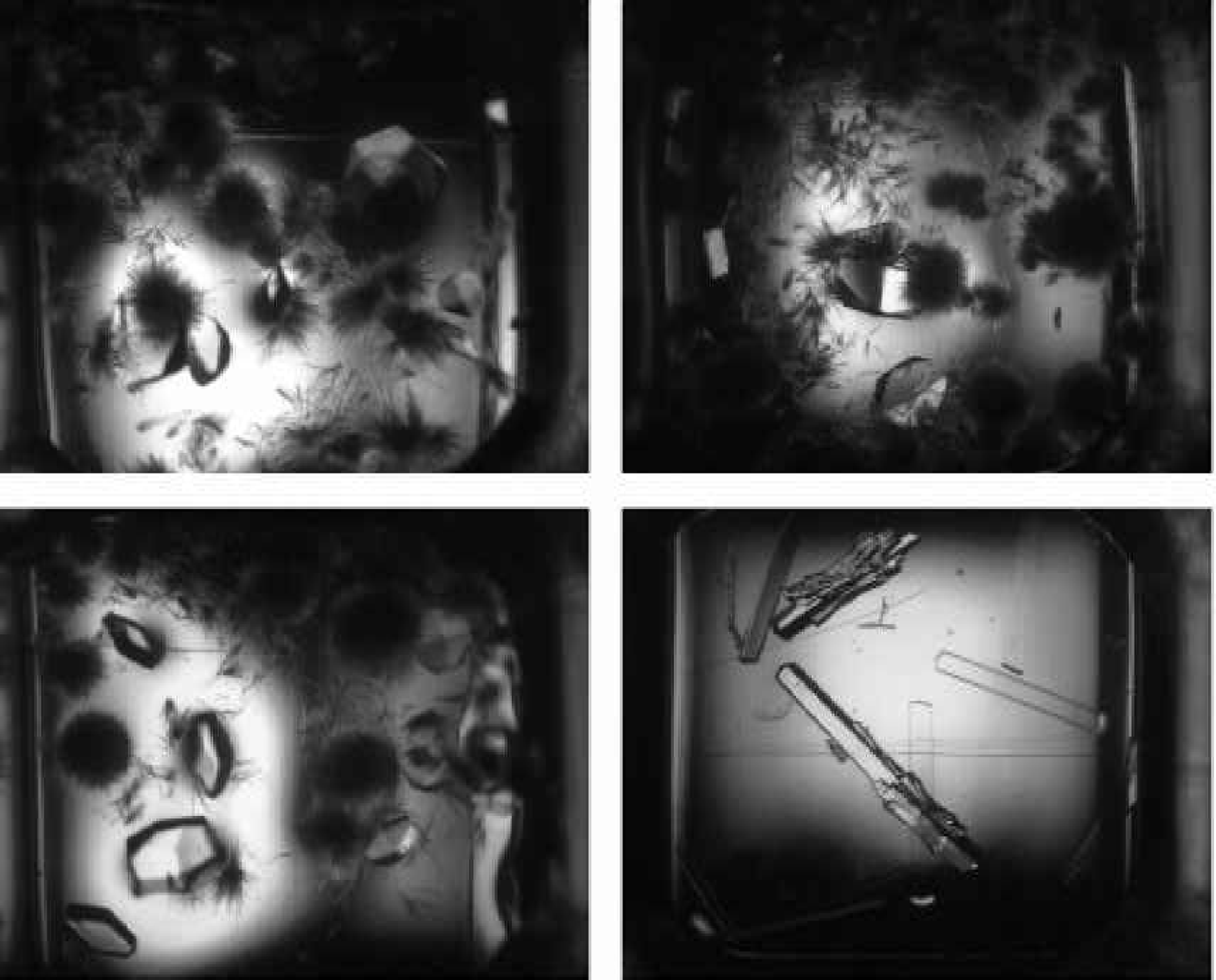
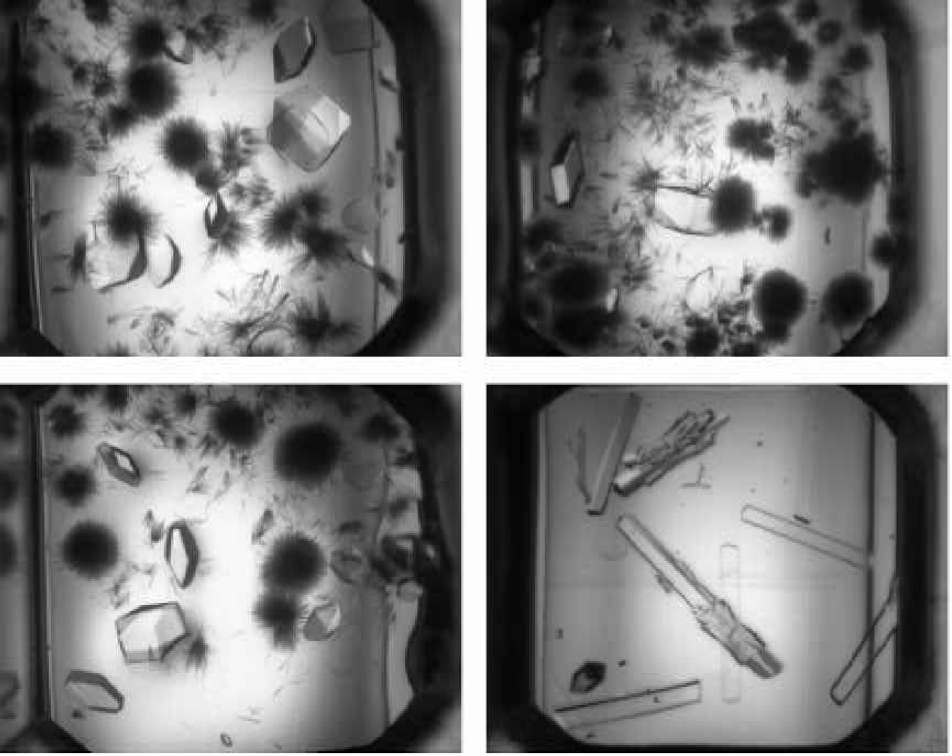
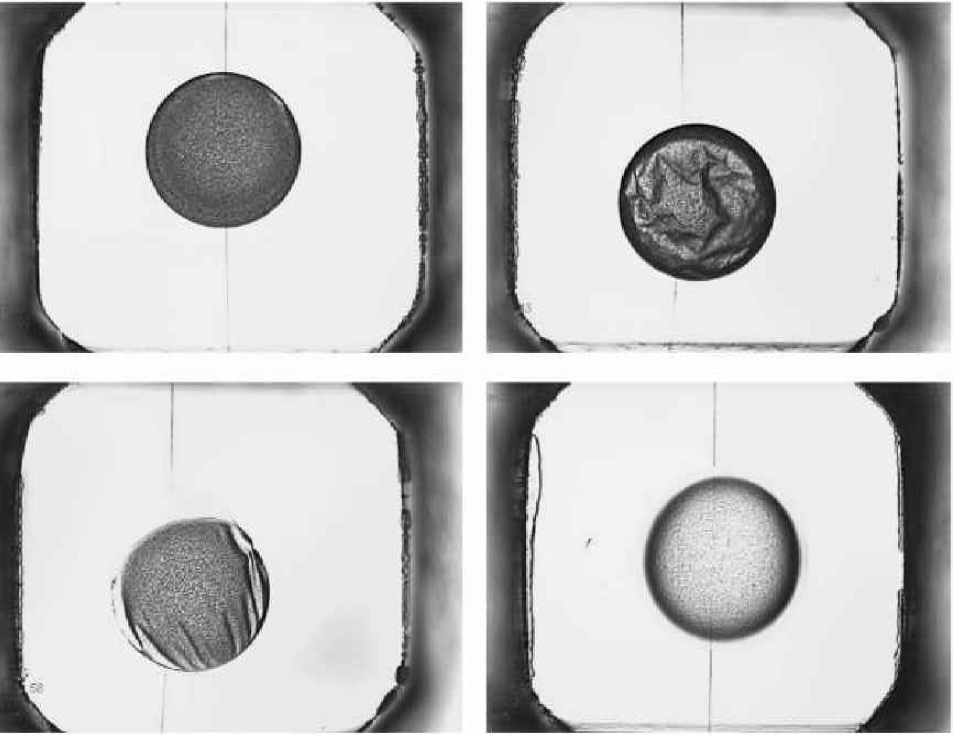
Figure 9 shows similar sitting drops in the CrystalQuick plate and imaged with the RoboMicroScope™ II system. These images look much better from an automatic analysis point of view with non-saturated gray scale and uniform lighting.

Figure 10 compares a hanging drop image from the IISP (left) with one from the RoboMicroScope II. In the IISP image, over half of the area of the drop is so heavily shaded that it is not useful for automatic analysis, whereas the RoboMicroScope™ II image is usable almost to the very edge of the drop.
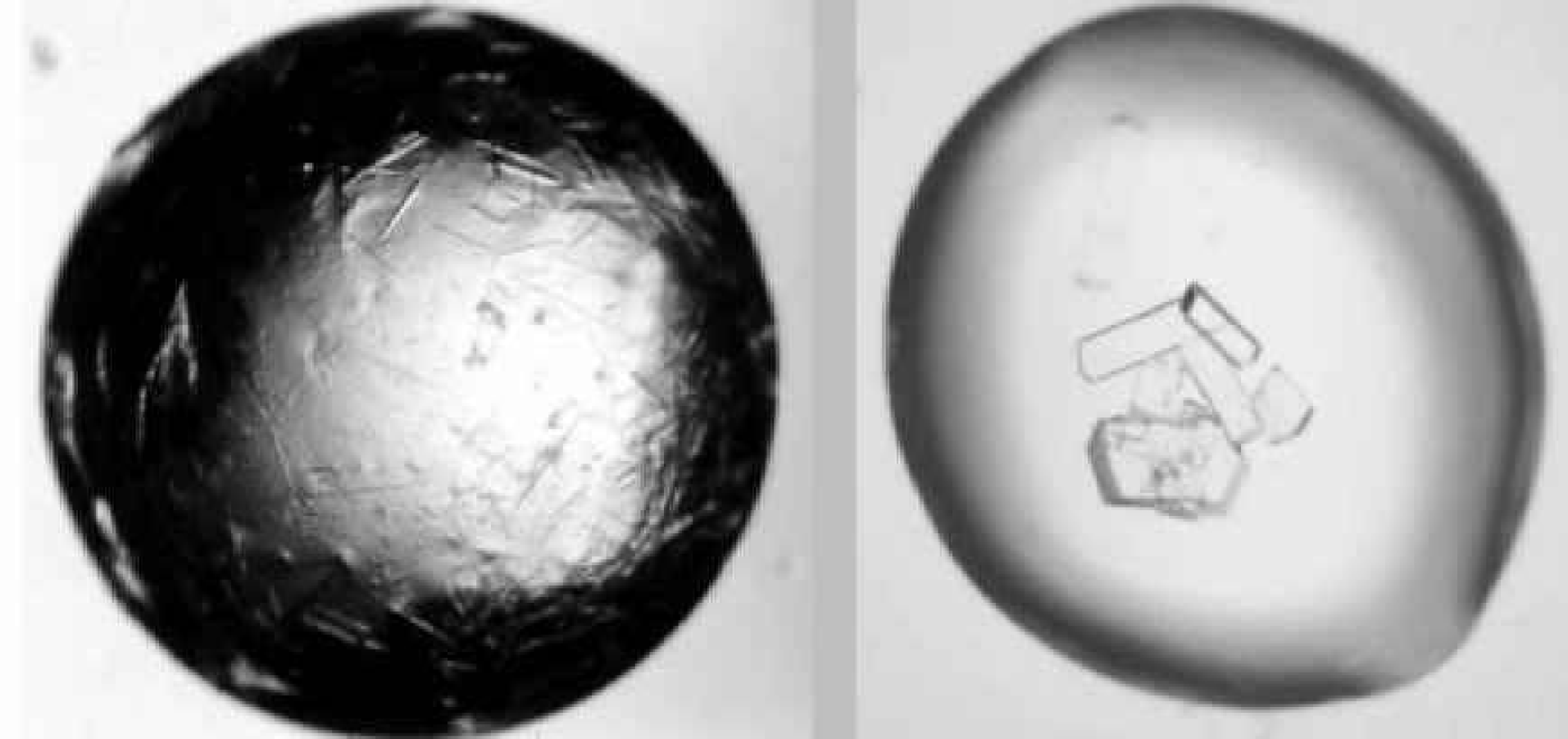
Figure 11 shows the same sitting drop experiments as Figure 9, but imaged with polarizing technology to give pseudo-color imaging. Under the standard lighting conditions shown in Figure 9, it is difficult to determine if any crystals are present in the drops, whereas the pseudo-color images clearly distinguish between drops containing only precipitate, and those with crystals and precipitate.
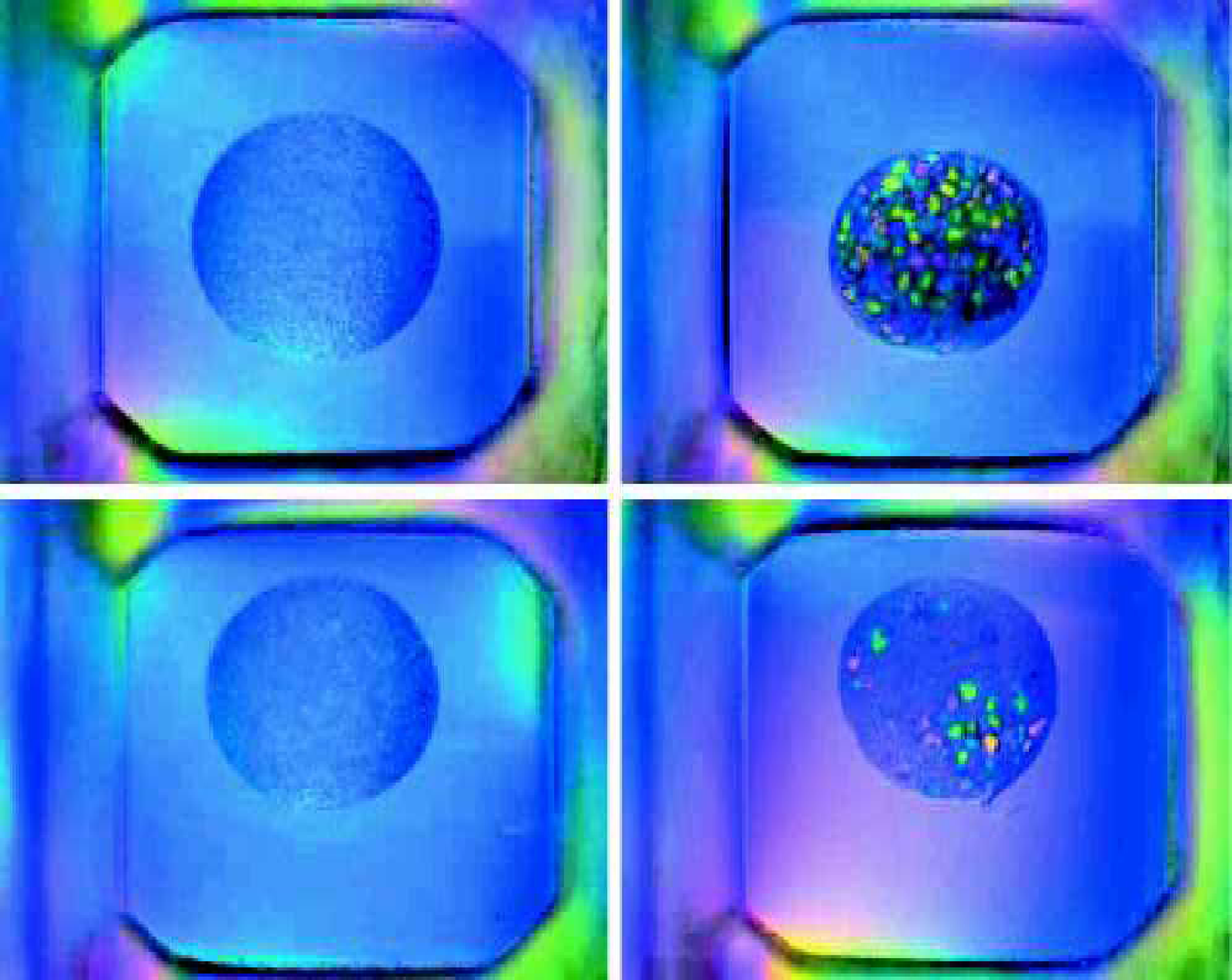
IMAGE DATABASE SERVER
The Image Database server is software designed to accommodate customers' need to merge data produced by the RoboMicroScope II into their Enterprise Databases or Laboratory Information Systems (LIMS). The RoboMicroScope II stores data to XML files. The Image Database Server reads these files and scripts are generated to insert the data into an Access, Oracle, or SQL Server table in the format of the customers' choosing.
Additionally, the Image Database Server distributes work orders to computers running the CPXO software described below. This distributed architecture allows for scalability to process the very large volume of images.
CPXO SCORING SOFTWARE
RoboDesign's CPXO™ software automatically measures and classifies protein crystallization trials. The algorithm classifies drops into four classes: clear, precipitate, crystals and other (C, P, X, O). Both the hardware and control software design of the fully automated RoboMicroScope II have been optimized to ensure reliable performance of the scoring software. A benefit of this is that images generated by the RoboMicroscope are easier to analyze than images captured from a conventional stereo microscope.
The computer analysis of protein crystallization trials has been an intensive and challenging task both at RoboDesign International and at several other companies over the past few years. Our aim from the outset was to ensure that the system does the following:
Normalizes image parameters for measurement stability
Extracts meaningful physical measurements from the image
Reduces the feature vector space to a manageable number
Runs a rule-based classifier to score the images.
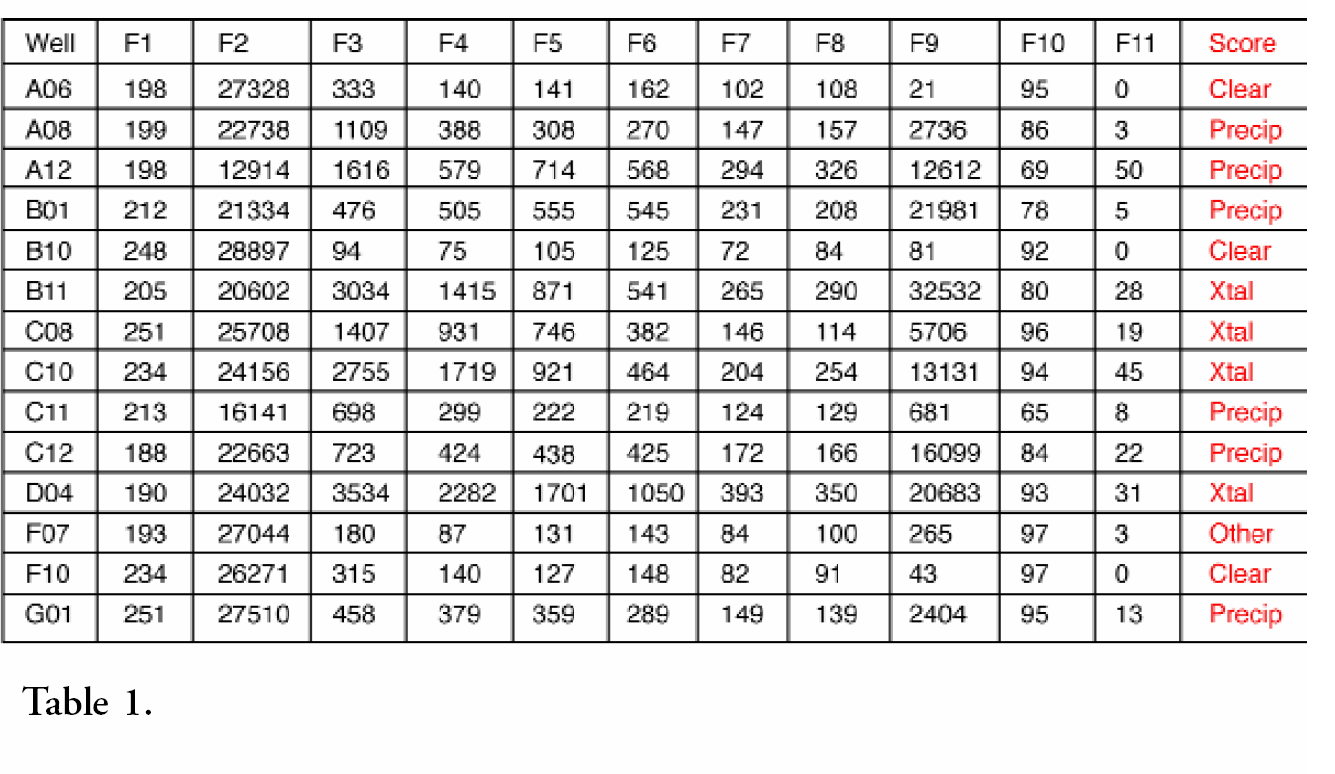
As a demonstration of the automatic analysis feature extraction program, a set of 14 hanging drop images were presented to the CPXO™ program. The images were presented in gray-scale only. Eleven features were extracted from each drop and are labeled F1 through F11 in Table 1 below. These measurements were then presented to the rule-based classification program and resulted in classification of all of the 14 drops.
CM VIEW OFFLINE IMAGE VIEWER
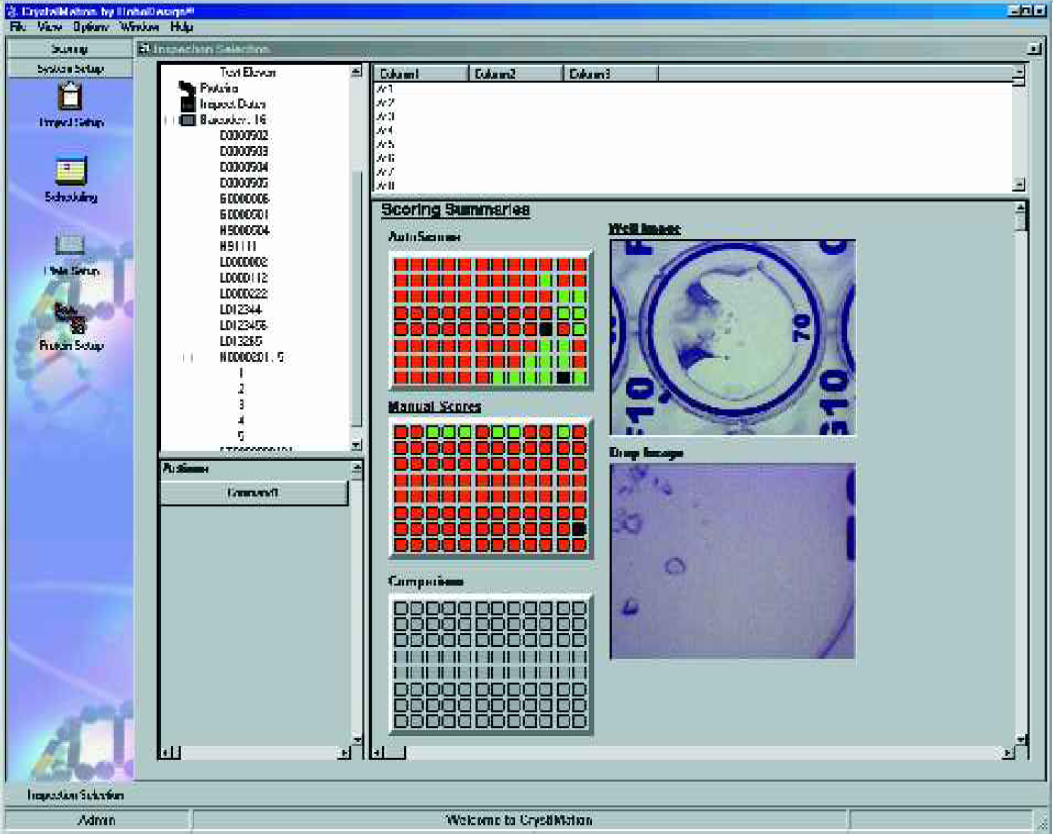
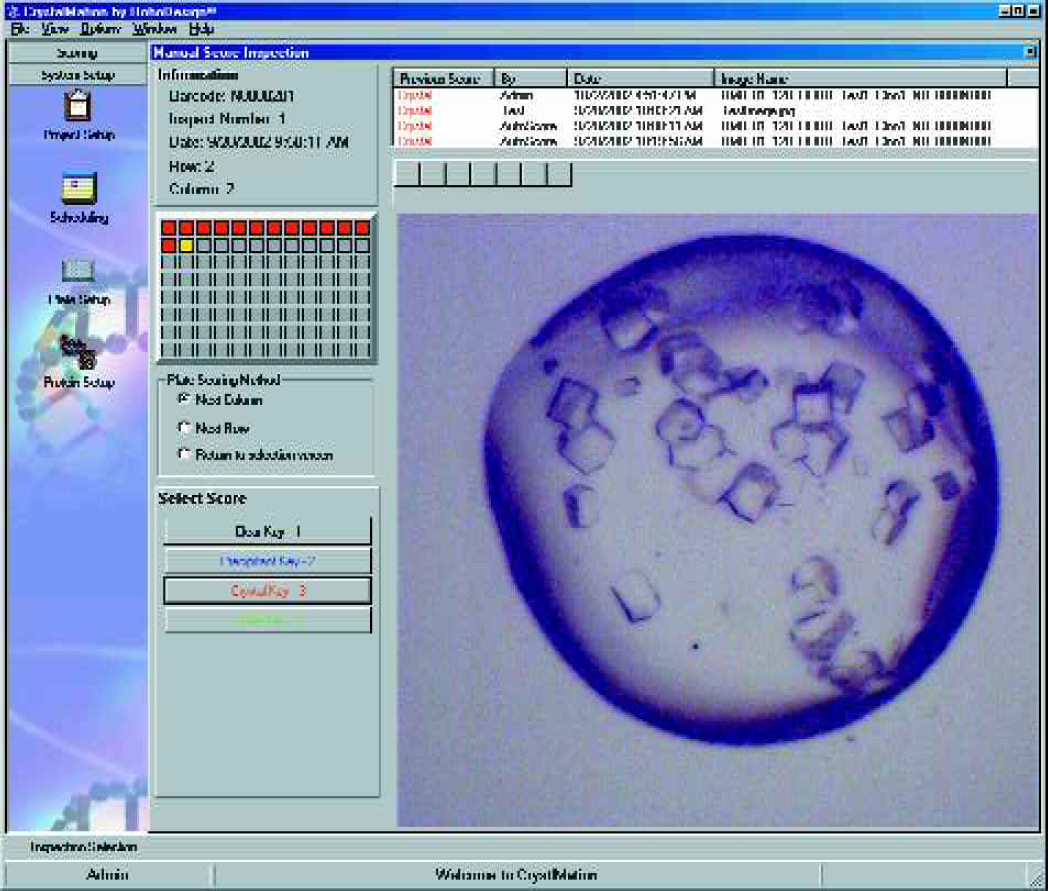
The RoboMicroscope II system also provides software for the viewing and manual scoring of crystal inspections, and this software can be run on any computer that has network access to the Image Database Server. This allows tracking of projects, proteins and other pertinent information related to the design of the experiment. Scheduling features of the software enables users to create time-based list of plates to be inspected with email alerts or it can be interfaced to the incubator for automatic inspection. Furthermore, RoboDesign can optionally provide a barcode printer which is controlled from the same software, enabling the user to generate the unique barcode that tracks a plate through the RoboMicroscope II suite. Figures 12–14 shows various screenshots of the image viewer, manual scoring, and scheduling modules.
CRYSTALTRAK
This is an Oracle® based database which can capture the information about any crystallization experiment, and is flexible enough to allow the efficient description of post-setup modifications, such as seeding, or reservoir manipulation. Relevant information is included with the database, for example, all the commercially available screens, and their constituent chemicals, are preloaded into the database. This database links into the imaging system, so that the results of the experiments are coupled and can be sorted or filtered on the crystallization conditions, as well as allowing the scoring of the images to drive the development of the next round of crystallization experimentation.
Conclusions
Crystallization of macromolecules is a complicated and poorly understood process, and progress in the field has been slow. Until now, there has been no effective way to collect information about all the crystallization trials set up for any given project, nor any general way to collate and mine the patchy information that was collected 1 . The vast majority of crystallization experiments do not result in crystals, and even if a crystal is found in initial screening, it is very unlikely to be suitable for X-ray analysis without further refinement of the growth conditions. Carter and Carter 2 have shown that the application of standard statistical analyses to the results of a set of crystallization trials can lead to well diffracting crystals quite efficiently, but this approach has been stymied by the requirement that the result of each experiment needs to be recorded and included in the analysis. The assignment of crystallization trials into four categories by the CPXO software allows for the implementation of such statistical analyses, and may well lead to a large increase in the efficiency of the crystallization process, independent of the improvement obtained simply by decoupling crystallization trial inspection from its analysis. RoboMicroscopeII provides a system which can produce timely and robust descriptions of crystallization trials, and for the first time allows the easy implementation of statistical methods to this arcane yet fascinating science.
