Abstract
The BIOPHILE Individual Vial Retriever (IVR) system has been developed to provide automated access to vials stored at ultra-low temperatures. The IVR performs storage, retrieval, sorting, cataloging, volume estimation (weighing), barcode reading, and re-racking. All racking operations are performed in an environment designed to keep samples frozen at their optimal temperature. Operating temperatures are −80°C, −40°C, −20°C, and room temperature. Laboratory Information Management System (LIMS) integration, automation integration, chain of custody tracking, and FDA 21CFR Part 11 compliance are supported. This article introduces the IVR and provides information on its characteristics.
Introduction
Biological materials and other materials used in the life sciences are commonly stored in vials of various types and then placed in standard laboratory freezers or in tanks of liquid nitrogen. Screw-cap ‘cryovials’, 2D barcoded matrix tubes, and one dram ‘amber’ glass vials are all used for storage of compounds for high throughput screening (HTS). Researchers storing samples collected during toxicology studies, ADME studies, or clinical trials also use these types of vials. Depending on the type of material under storage, the stability of the material may be affected by temperature, humidity, and exposure to oxygen. The laboratory process determines which vials are retrieved from cold storage at any given time. Typical manual processes require laboratory personnel to locate the sample vials in storage, verify their identification codes, retrieve them, and return them to storage once they have been used. Ultimately, vials may be discarded due to exhaustion of the sample or completion of a study. Because the vial ‘pick list’ may be known well in advance, ad-hoc optimization schemes are often used to group samples together that may be later retrieved en masse. For example, some racks, containing vials, may be only partially filled so that they may be filled with vials received later. While this approach allows for some optimization, it can waste large amounts of storage space. Other problems that manual storage and retrieval of temperature-sensitive samples can cause are temperature variance, loss of sample integrity due to excessive handling, loss of samples due to accidental mis-handling, and large variances in the time required to manually fill an order for vials from storage.
The temperature at which life science materials are stored is based on three factors: the type of material in question, the profile of usage, and institutional norms. Often, dry compounds and certain genomic material may be stored at room temperature. Enzymes and organic compounds in DMSO are often stored at −20°C. Proteins (in particular RNA), genomic material in solution, and certain tissue samples are often stored at −80°C. Finally, living cells and reproductive materials are often stored at −140°C. 1 For every situation that fits these general guidelines, one may find examples where an institution has chosen to use a different storage temperature.
As HTS becomes more focused, higher standards for stability of compound libraries will be required. Of particular interest are recent studies indicating that compounds stored in DMSO may actually be more stable at −80°C. 2 Those entities wishing to transfer particularly important compounds or libraries to automated storage and retrieval systems operating at these colder temperatures will find that their choices for equipment will be severely limited. The types of automation that will operate reliably at −80°C differ drastically from those designed for −20°C.
THE BIOPHILE IVR
The BIOPHILE IVR is an automated storage, retrieval, and processing system that operates at temperatures as low as −80°C. Samples in the BIOPHILE system are kept within secure freezer chambers called BIOPHILE Storage Units (BSUs). The BSUs are cooled using dual-cascade compressor technology. Liquid nitrogen backup systems ensure that long-term loss of compressor power does not compromise temperature stability.
Coupled to that proven technology is a high-speed conveyor system that shuttles samples to one or more Vial Handlers (VHs). The Vial Handlers re-rack, scan, weigh, and sort vials, as well as provide a stacking capability to allow up to 500 vials to be loaded or unloaded at one time. See Figure 1.

An Individual Vial Retriever with two BIOPHILE Storage Units (left and center) being served by one Vial Handler (right).
The BIOPHILE IVR is a scalable system (Figure 2). A system may consist of one or multiple BSUs and one or multiple Vial Handlers. The system allows BSUs to be freely added to or removed from the system without any software reconfiguration as the user's needs evolve. Similarly, if extra vial processing capability is needed, additional Vial Handlers can be daisy-chained onto the system. The added Vial Handlers will automatically assume a subordinate role to the original (master) Vial Handler. If the master Vial Handler is brought off-line, one of the subordinate Vial Handlers will automatically assume master status.

The IVR system is scalable, allowing for expansion as needed. This system, consisting of 6 BSUs and 2 Vial Handlers are capable of storing over 240,000 2D-barcoded minitubes.
Vials move between the BSUs and the Vial Handlers in racks of 54 cryovials or 96 minitubes. Within the Vial Handler, the racks are placed in a small freezer chamber for re-racking. After use, racks of vials are then returned to storage; racks that have been filled for output but are not yet needed are also returned to a BSU until they are needed.
Within the VH are barcode scanners for both linear and 2D data matrix codes. The VH is capable of scanning and identifying an entire rack of minitubes in one operation.
Also inside the VH is an electronic balance that automatically weighs each vial as it enters (or returns to) the system. The system is capable of taring and converting the measured weight to calculate an estimated sample volume in each vial.
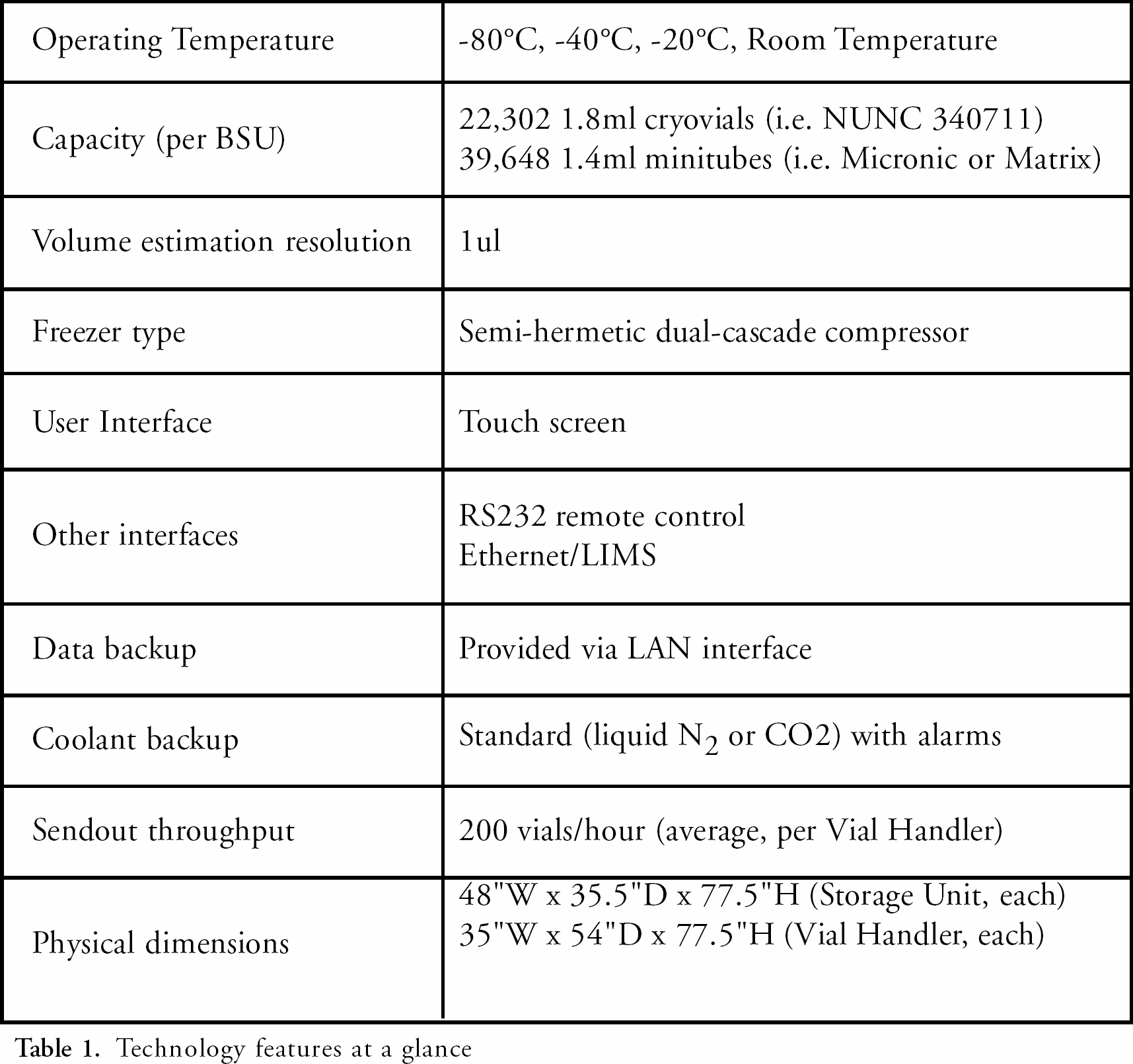
The IVR is built upon the BIOPHILE microplate storage and retrieval system. 3 The BIOPHILE microplate system proved that the technology of automatically handling labware at −80°C was feasible and cost effective. The BIOPHILE IVR uses the original BIOPHILE system as the core storage and retrieval mechanism (Figure 3). Other system specifications are presented in Table 1.

The IVR is based on the proven BIOPHILE Microplate Storage and Retrieval System, shown here retrieving a microplate for a lab technician.
Technology features at a glance
BASIC SYSTEM MODES
Some common operation modes of the IVR include:
Storing containers - There are two alternatives when storing vials: storing them as racks (whole racks being stored as a group), or storing them pre-sorted according to criteria chosen to improve the efficiency of future retrieval. When storing, the IVR scans the barcode of each vial in the rack. This information is transmitted to the central database and/or LIMS. In an IVR system with more than one VH, multiple racks may be processed simultaneously, even though all the racks were loaded into one VH. This is accomplished by the VHs passing the racks between themselves on the conveyor. If the volume estimation component is available in the VH, each vial will also be weighed, and the estimated volume will be made available to the LIMS, if available.
Re-racking of stored vials - either for fulfilling a sendout worklist, or for re-organizing stored samples to improve future retrieval efficiency.
Unloading Containers — Whole racks may be requested from the master VH touchscreen. Larger groups of containers may be requested via the LIMS interface.
Unloading a work list — After a work list has been fulfilled, the user may use the master VH touchscreen to request the vials. This may happen immediately after the fulfillment, a day later, or perhaps at a much later date. In any case, the user logs in and specifies which work list is needed. The racks comprising that work list are then placed into the stacker.
SECURITY AND VALIDATION
The BIOPHILE IVR is intended for storage of highly critical samples in validated environments. It was designed according to the GaMP 3.0 (Good Automated Manufacturing Practices) specification. The software system conforms to the FDA Guidelines for Electronic Signatures and Electronic Records (21CFR Part 11). All records are password protected and encrypted, and chain of custody information is tracked indefinitely. The system will accommodate biometric identification devices for user login.
ENTERPRISE INTEGRATION
Sample inventory databasing is performed either locally to the system, or the IVR may use an external database server. Data base communication is performed using an XML interface, allowing communication with Oracle and a wide variety of other commercial and custom databases. If multiple IVR systems are present, they share a common inventory database. In this way, BSUs containing samples may be moved between IVR systems without the need to perform any database updates. The IVR also integrates with external automation through the same XML interface.
The XML interface is used to generate worklists that result in sample pre-sorting, worklist fulfillment, re-racking, and worklist retrieval.
SUMMARY
The IVR provides rapid access to individual cryovials or minitubes in ultra-low temperature storage while:
Maintaining sample integrity - Samples are at reduced risk of accidental damage or unsealing
Maintaining sample quality - Samples are not subjected to unnecessary handling or temperture variation
Improving container security - Samples can only be accessed by authorized persons
Improving container accessibility - Samples qare available quickly and easily
Improving sample/data visibility - Information on sample location, chain of custody, temperature, and ownership is easily obtained.
The IVR is a reliable, accurate, rapid, and easy to use system. Very little training is needed to ensure that the operator will have trouble-free operation. It integrates fully with external robotic automated laboratory systems. Integration options allow the IVR to be controlled and accessed (loaded or unloaded) by other automation systems. Also, IVR allows the exchange of data with external information systems. The IVR reduces the rate of frost build up within a storage unit to the point where no defrosting is required for normal operation. Finally, the BIOPHILE IVR provides an affordable, scalable solution that can add vial storage capacity and processing capacity, as the customer's needs increase.
