Abstract
We describe a reagent system and robotic methods for the purification of BigDye™ Terminator sequencing reactions prior to automated fluorescent sequence analysis. The methods use MagneSil™ paramagnetic particles to isolate sequencing extension products from unincorporated dye-labeled terminators and exchanges sequencer loading solution for reaction buffer. Processed samples give usable data that is greater than 98% accurate from primer plus 5–15 bases to over 700 bases. Typical Phred greater than 20 quality scores range from 600 to over 700 bases. This process has been adapted to a number of liquid handling robotic platforms in both 96- and 384-well formats. One method using a single POD Beckman Biomek® FX can process up to four plates in approximately 40 minutes.
Introduction
Automated fluorescent sequencing using the ABI PRISM® BigDye™ Terminator chemistry is the method of choice for high-throughput DNA sequence determination. If not removed prior to analysis, certain reaction components can interfere with data collection. Salts in the reaction mix can cause injection failure (capillary systems) and /or aberrant electrophoretic migration of ladder fragments. Unincorporated dye-labeled terminators and related species overlap and obliterate the legitimate signal from sequence extension products (Figure 1). Common purification methods based on gel filtration and /or precipitation by ethanol are problematic for high throughput applications, in that they require (multiple) centrifugation and /or vacuum steps.

A Comparison of Clean-Up Methods. ABI Prism® BigDye™ Terminator sequencing reactions, version 1, were prepared as described in Methods (Columns 1–4) and with 1/4 the recommended volume of the Terminator Ready Reaction Mix (Columns 5–8). Un-purified sequencing reaction (O), Purification by gel filtration (G), Purification by ethanol precipitation (E), Purification by Wizard® MagneSil™ Sequencing Reaction Clean-Up System (W).
Promega has developed a number of ‘silica-paramagnetic’ particles (MagneSil™) for the purification of nucleic acids 1 under defined conditions. Particle structure and solution composition can be altered to selectively adsorb nucleic acids based on size and type. These parameters have been optimized to develop reagent systems for the purification of plasmid 2 and genomic 3 4 DNAs, as well as PCR* fragments 5 based on a three step–bind, wash, elute process. This technology can also be used for the purification of BigDye™ Terminator DNA Sequencing reactions 6 prior to automated fluorescent sequence analysis. Methods have been developed for a variety of robotic workstations including the Biomek® FX and 2000 Laboratory Automation Workstations (Beckman Coulter Inc. Fullerton, CA) and the Genesis RSP Series (Tecan AG, Männedorf, CH).
Methods and Materials
Sequencing reactions were prepared using the ABI PRISM® BigDye™ Terminator Cycle Sequencing Ready Reaction Kit (Applied Biosystems, Foster City, CA) according to the manufacturer's instructions and with the following modifications. Standard reactions for 96-well format contained 8μl Terminator Ready Reaction Mix, 350ng pGEM®-3zf(+) (Promega, Madison, WI), and 3.2pmole pUC/M13 forward primer (Promega, Madison, WI) in a final volume of 20μl. When the amount of the Terminator Ready Reaction Mix used was less than recommended, the buffering capacity and magnesium concentration were maintained at 50mM (pH 8.9) and 2mM, respectively. One quarter reactions (1/4 dilution) were prepared as above, but with 2μl Terminator Ready Reaction Mix and 3μl of 5x Sequencing Buffer (250mM Tris. Cl [pH8.9], 10mM MgCl2) (Promega, Madison, WI). BigDye™ Terminator version 3 chemistry was used unless indicated otherwise. Reactions for 384-well format were scaled proportionately for 5 or 10μl final volume. MagneSil™ samples purified in 384-well format were eluted in a final volume of 20μl.
Purified sequencing reactions were stored at −20°C prior to shipment to a custom sequencing facility for analysis on an ABI Prism® 3700 DNA Sequencer (SeqWright, Houston, TX). Run parameters were as follows: Injection Voltage: 1000V, Injection Time: 50 seconds, Post Injection Voltage: 5250V, Step Interval: 50V, Interval Time: 40 seconds. POP-6 polymer was used.
THE SEQUENCING REACTION CLEAN-UP PROCESS
The Wizard® MagneSil™ Sequencing Reaction Clean-Up System uses a particle type designed for high recovery of small fragments 7 to purify BigDye™ Terminator sequencing reactions prior to automated fluorescent sequence analysis. This formulation has been optimized to selectively adsorb ssDNA extension products from the sequencing reaction mix (Figure 2). After capture, an external magnetic field is applied to localize the particle and adherent sequencing extension products at the side of the well, and nonbound material is removed to waste. Residual contaminants are eliminated with a particle wash and purified extension products are eluted in sequencer loading solution (water or formamide (+/-) Blue Dextran / EDTA), ready for analysis.
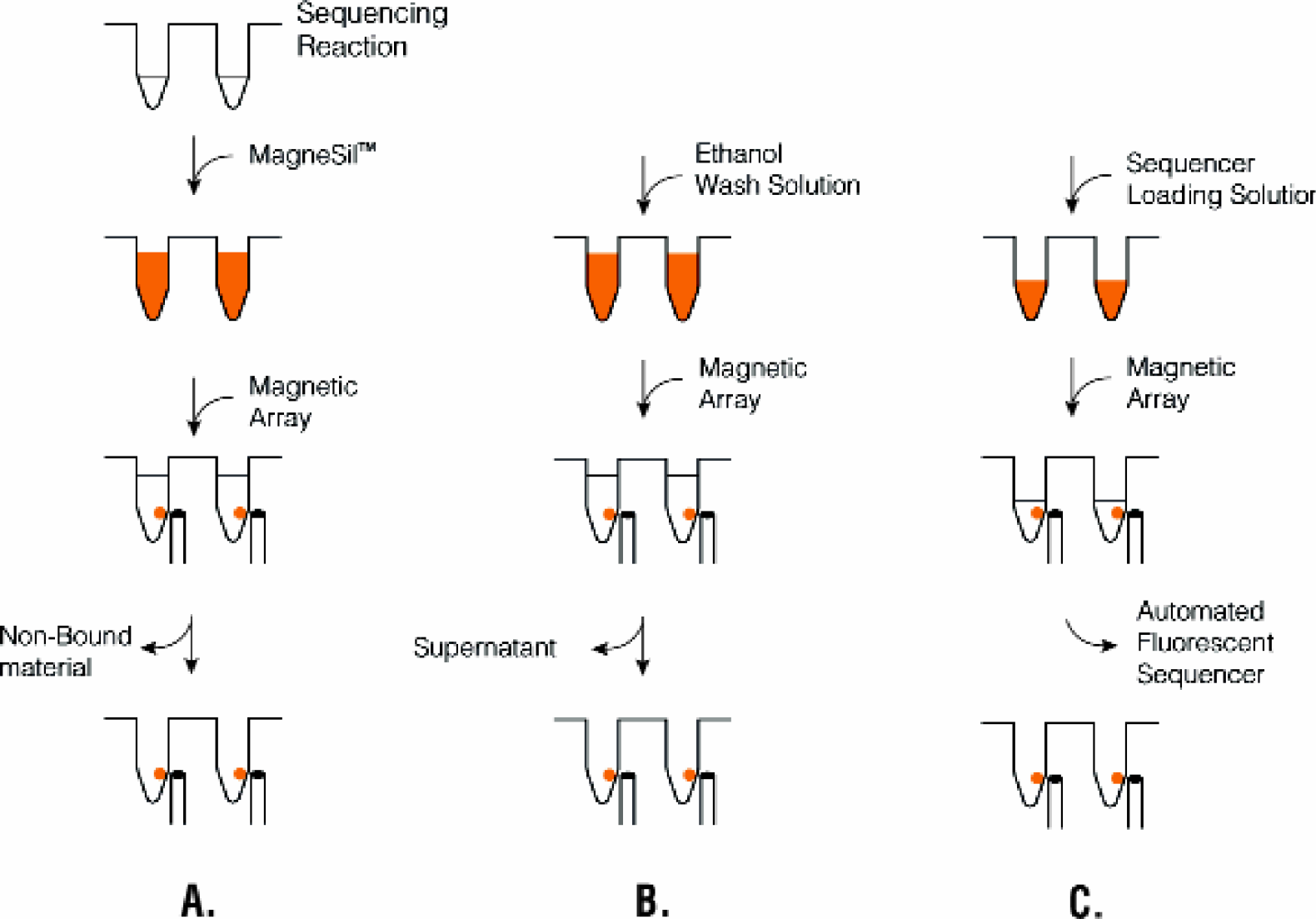
The Purification Process. Panel A: Sequencing reactions are incubated with MagneSil™ binding resin. The sequence extension product/particle complex is captured by application of an external magnetic field. Non-bound material is removed to waste. Panel B: Residual contaminants are removed by washing with ethanol. Panel C: Sequencing extension products are eluted in sequencer loading solution, ready for downstream processing.
GENERIC METHOD
The following sequence of steps form the basis for both 96- and 384-well robotic methods:
Resuspend and add nine parts MagneSil™ particle slurry to one part sequencing reaction sample.
Incubate two minutes at room temperature. Mix at zero, one and two minutes.
Place onto MagnaBot® magnetic separation device to capture the particles.
Remove and discard the liquid.
Remove the plate from the MagnaBot® device.
Add five volumes ethanol wash solution.
Incubate two minutes at room temperature. Mix at zero, one and two minutes.
Place onto MagnaBot® magnetic separation device to capture the particles.
Repeat steps 4–8 for a total of three washes.
Remove and discard the liquid.
Incubate five minutes to dry particles.
Remove the plate from the MagnaBot® device.
Add 20ul appropriate elution/loading solution.
ABI 377: formamide / blue dextran / EDTA solution.
ABI 310: Template Supressor Reagent.
ABI 3100: Hi-Di™ formamide (Perkin Elmer, Foster City, CA).
ABI 3700: Hi-Di™ formamide or water.
Incubate two minutes at room temperature.
Place onto MagnaBot® magnetic separation device to capture the particles.
Transfer purified sequencing reactions to destination plate.
MAGNETIC ARRAY HARDWARE
Custom magnetic arrays have been developed for high-throughput processing in 96- and 384-well plate formats (Figure 3). These devices have an SBS standard footprint to allow for maximum usefulness among liquid handling workstations. The magnets are orientated such that the MagneSil™ particles are captured to the side of each well. This allows quantitative removal of liquids with minimal particle loss.

Magnetic Arrays. Panel A: MagnaBot® - magnetic array for pro cessing 96-well plates. Panel B: MagnaBot® 384 - High-density magnetic array for processing in 384-well plates.
AUTOMATED METHODS
The MagneSil™ purification process has been adapted to a number of robotic platforms including the Beckman Biomek® FX (Figure 4). Maximizing usage of available deck positions with a single POD (processing arm) instrument configuration allows for parallel processing of four plates (Figure 5). Each four-plate run cycle takes approximately 40 minutes before re-populating the deck is necessary.

Dual POD Biomek® FX Laboratory Automation Workstation.

“Loaded Deck” Configuration for a Single POD Biomek® FX - This arrangement allows for simultaneous processing of four 96-well plates in approximately 40 minutes. Labware elements are 20μl and 200μl tip boxes, reagent reservoirs for MagneSil™ particles, wash reagent, and elution solution, four MagnaBot® magnetic array units, four sample (processing) plates and four destination plates. The deck layout for 384-well method is the same with the following exceptions: substitute 384-well microtiter plates and MagnaBot® 384, processing is with a single box of tips. Cycle time is unchanged.
SEQUENCE QUALITY
The MagneSil™ Clean-Up System is QC qualified to provide usable data from less than or equal to primer plus 25 bases to over 600 bases with at least 98% accuracy. A reference template of a known sequence was purified by the MagneSil™ as described in methods in order to assess sequence quality using these parameters. When the start position is defined as the first base in the first run of four correct base calls, the average for the four-plate method is primer plus 16 (+/-) 2 bases (Figure 6, Panel A). Average accuracy for the four plates at 600 bases is 99.4% (+/-) 0.8 (Figure 6, Panel B). The accuracy specification is achieved at 700 bases as well. For experimental samples the most common method for assessing sequence quality is by Phred score. 8 The average Phred > 20 quality score for 96 samples purified using the MagneSil™ 1-Plate FX method is 619 (Figure 7).
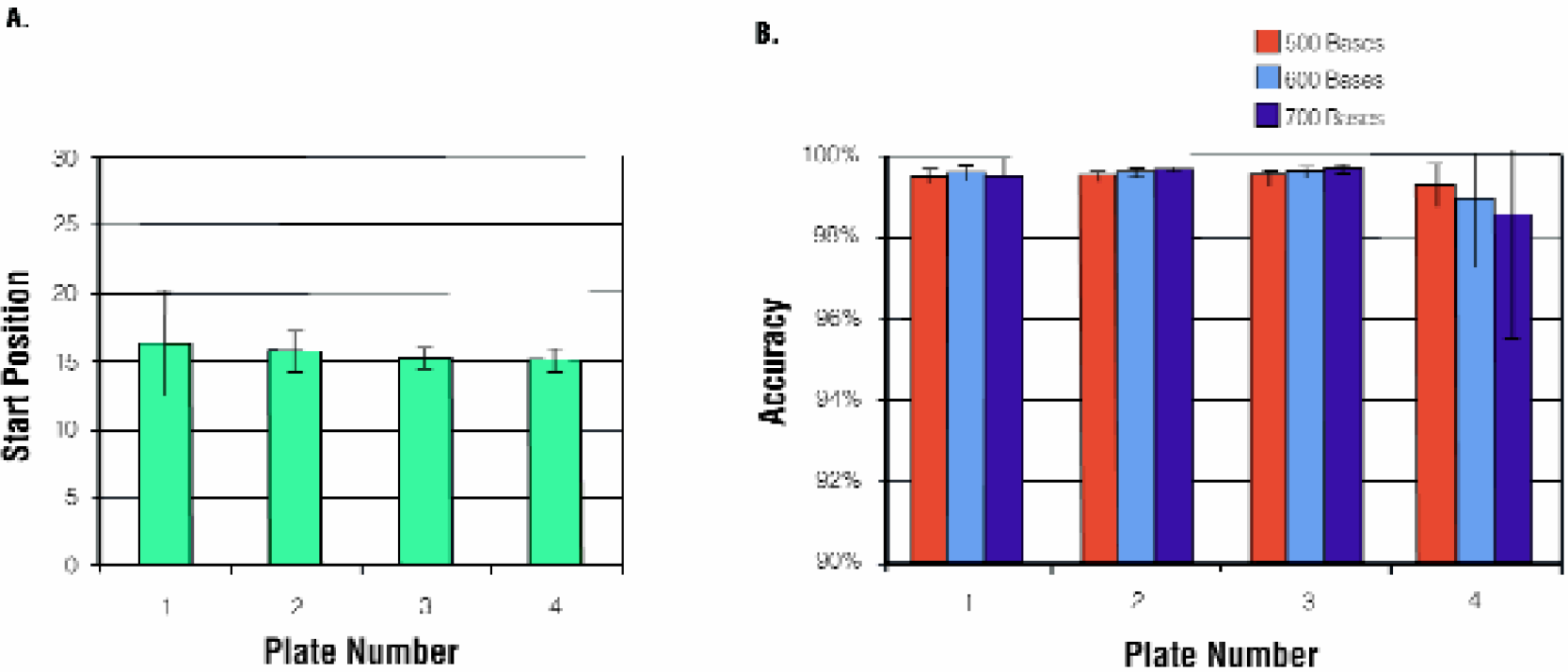
Start Position and Accuracy of MagneSil™ System Purified Samples. BigDye™ Terminator chemistry version 3 sequencing reactions (1/4 dilution) were prepared as described in Methods, 96-well plate format. Samples were purified using the MagneSil™ 4-Plate Biomek® FX method and analyzed with an ABI PRISM® 3700 DNA Sequencer. (Panel A): Average start position. (Panel B): Average accuracy at 500, 600 and 700 base read lengths.
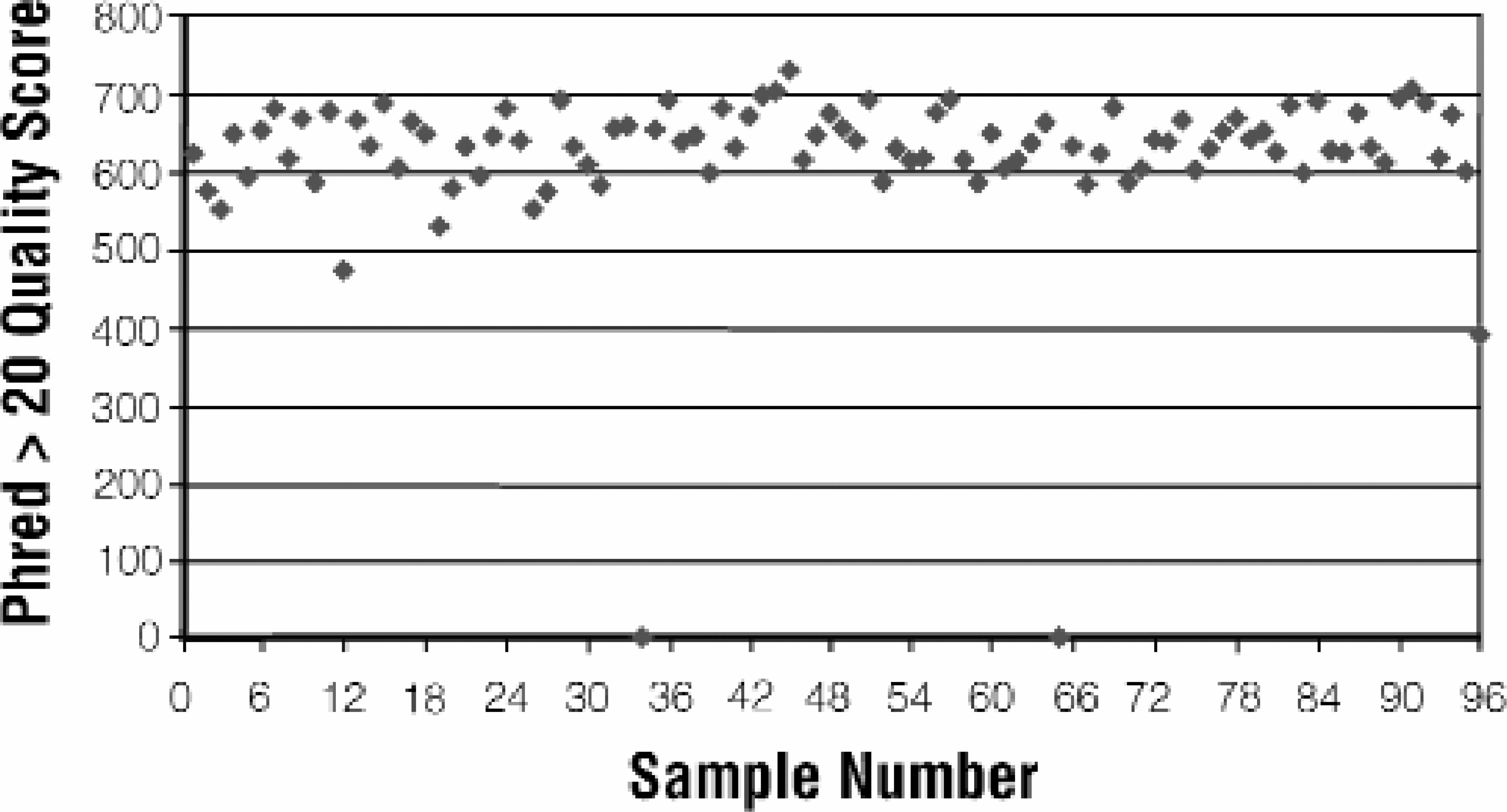
Phred > 20 Quality Scores for Full Plate of MagneSil™ System Purified Samples. Version 3 BigDye™ sequencing reactions (1/4 dilution) were prepared from plasmid templates according to the manufacturer's instructions, purified with MagneSil™ method and analyzed on an ABI Prism® 3700 DNA Sequencer. Data is a generous gift of Dr. Ulrich Broeckel, Human and Molecular Genetics Center, Medical College of Wisconsin, Milwaukee, WI.
COMPATIBILITY WITH BIGDYE™ VERSION 1, 2 AND 3 DYE SETS
Applied Biosystems has developed multiple versions of the BigDye™ Terminator Chemistry. The version 1 dye set is based on a 5-carboxy-d-rhodamine acceptor with isomers of 4'-aminomethyl fluorescine donors. 9 Version II uses this same dye set, but has reduced ddNTP/dNTP ratios to give longer read lengths. Recently, ABI developed the version 3 chemistry with a different dye set. The MagneSil™ Clean-Up method was tested for compatibility with ABI Prism® BigDye™ Terminator chemistry versions 1, 2 and 3 (Figure 8). The results indicate that the MagneSil™ particles are transparent to BigDye™ chemistry version.
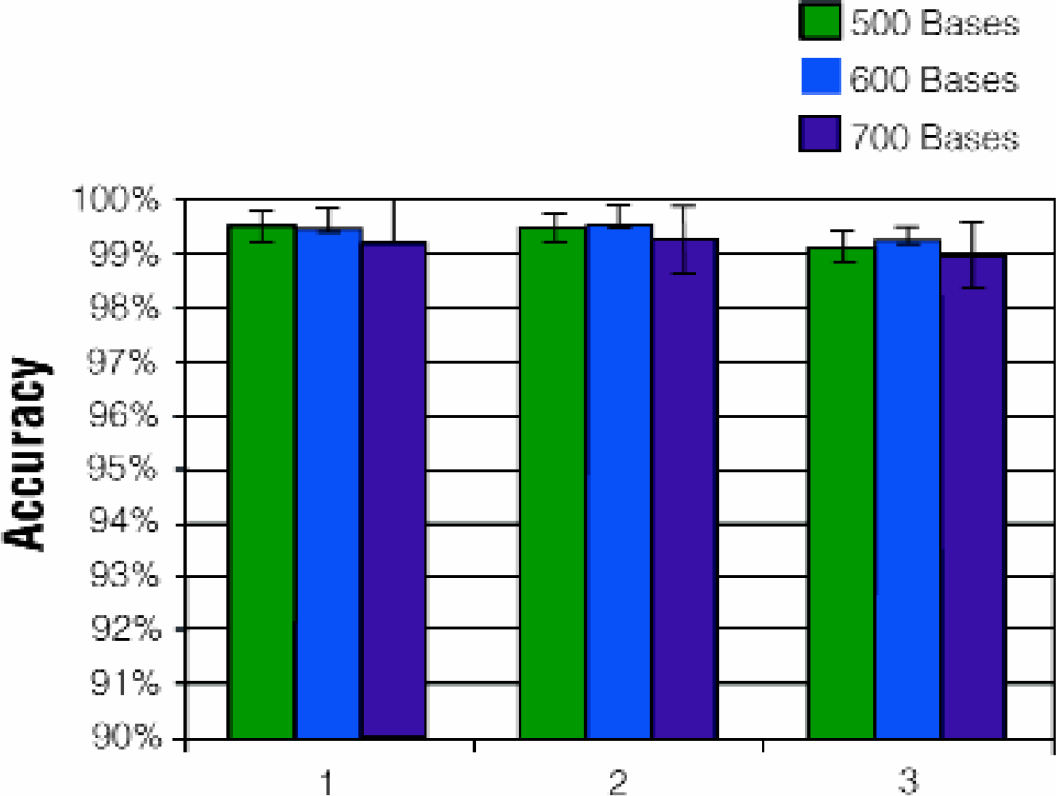
MagneSil™ Compatibility with BigDye™ Terminator Version I, II and 3 Dyes. BigDye™ sequencing reactions (1/4 dilution) were prepared and purified as described in Methods. Analysis was performed using an ABI Prism® 3700 Automated DNA Sequencer. Accuracy was assessed as described for Fig. 6.
384-WELL PLATE FORMAT
The MagneSil™ purification method has been adapted to a 384-well format (Figure 9). Quality scores were unchanged when reagent volumes were scaled to process the reduced sample volumes. The higher-density format significantly reduced reagent cost while maintaining the quality and quantity of data.
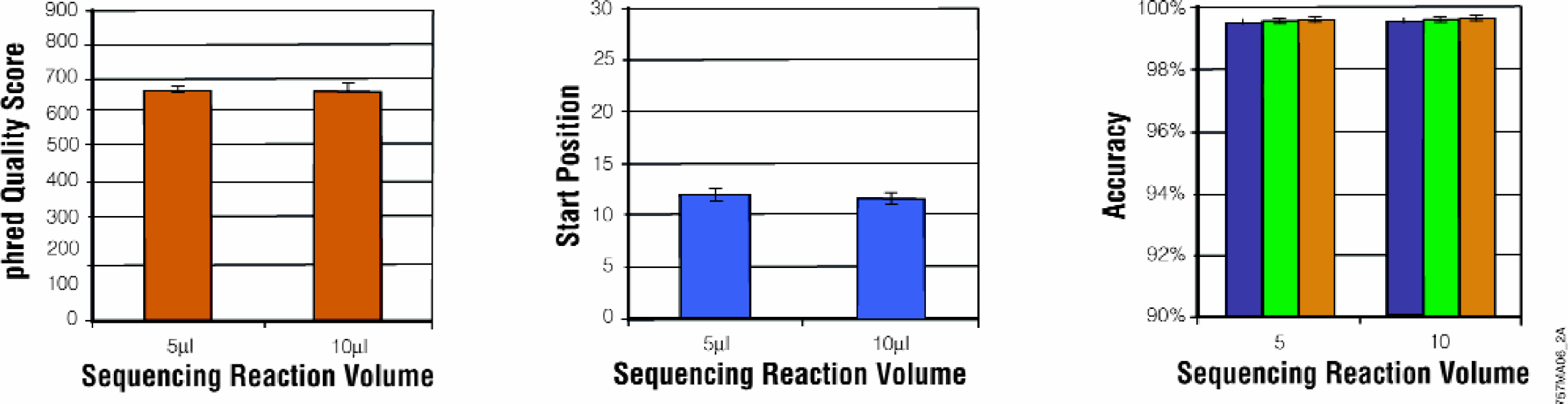
Quality Scores for the MagneSil™ Method in 384-Well Plate Format. Version 3 BigDye™ sequencing reactions (1/4 dilution) were prepared as described in Methods. Samples were purified by the MagneSil™ 384-well Biomek® FX method. Five or 10μl reaction volumes were processed. Purified samples were eluted in 20μl water and analyzed with an ABI PRISM® 3700 DNA Sequencer. Accuracy was assessed as described for Fig. 6.
Conclusion
The MagneSil™ Sequencing Reaction Clean-Up System provides an automated, hands-free solution for high-throughput purification of BigDye™ Terminator sequencing reactions. Robotic methods have been developed that can process four plates in less than 40 minutes. Sequence quality scores such as start position, accuracy and Phred > 20 are similar to or better than other manual, semi-automated or automated methods. This matrix is scalable to sample size and offers a distinct price advantage over other methods.
Footnotes
*The PCR process is covered by patents issued and applicable in certain countries. Promega does not encourage or support the unauthorized or unlicensed use of the PCR process. All trademarks are property of their respective owners
