Abstract

INTRODUCTION
The task of automating organic chemical reactions is a multi-step one including synthesis, work-up and purification of desired compounds. The required work-up procedure depends on the type of chemistry performed and is crucial in terms of the quality of the compounds isolated. For example, in the case of solid phase chemistry, multi-step filtration and washing can be employed to remove impurities and excess reagents. Automating such tasks is perfectly feasible and has been documented extensively.
In the case of solution phase chemistry, where all of the components in the reaction container are present in the same phase, simple filtration and isolation of components is not an option. This presents challenges for automation in terms of isolation of the reaction components in high yields and purities.
USE OF SOLID PHASE RESINS
One approach to the problem is the use of alternative reagents, such as scavenger resins or solid supported reagents. This approach was used as early as 1957 when the first review on catalysis using ion exchange resins was published. 1 There have since been numerous publications on this subject. 2,3 Such reagents are highly suitable for automated procedures, providing a simplified work-up by filtration, combined with the easy monitoring of solution phase chemistry. However, there are some disadvantages in using solid phase reagents:
Reaction times can be significantly longer than for the corresponding solution phase reaction.
Resins will not swell in all solvents and the reaction solvent may have to be changed to accommodate the resin, although resins are now available with low swelling properties.
Physical properties of the resin may limit use, as resins can be thermally and mechanically fragile.
Loading level of the resins may be low and limit the scale at which the reaction can be carried out.
Resin costs can be prohibitive.
Of all of the solution phase work-up techniques, traditional liquid-liquid extraction is probably the most frequently used. In addition to isolating the crude reaction product, it can often aid purification, particularly if the desired compound is acidic or basic. Considering the prevalence of this technique in the chemical literature, the development of a technology capable of performing automated parallel extractions would greatly enhance the breadth of solution phase chemistry.
AUTOMATED LIQUID-LIQUID EXTRACTION TECHNIQUES
The separatory funnel is one of the most common pieces of glassware in the chemical laboratory. Extractions are an intensive manual procedure, requiring the user to perform several actions to ensure a successful separation. Any successful automation of this procedure will have to address each action and find a procedure that mimics it. Such actions could include:
Repeated extractions, since the extraction process is an equilibrium controlled by the partition coefficient of the desired compound.
Agitating the separating vessel contents by vigorous shaking and swirling to ensure that the equilibrium is established.
Releasing of any vapor pressure built up when using volatile solvents.
Draining the lower layer and visually determining the boundary between the two layers.
Scientists can often use a combination of their experience, an expert eye and knowledge of the mixture components to determine whether a clear phase separation exists. On the other hand, automation requires a much more strict criteria to achieve reproducible separations. One such method is the use of hydrophobic frits, which allow the organic layer to pass through but retain the aqueous layer. However, this immediately restricts the organic solvents that can be used for such a procedure to those heavier than water, e.g., dichloromethane and chloroform. It is also limited to a single extraction per sample.
The manual Lollipop system, developed at GlaxoSmithKline, is able to separate organic liquids both heavier and lighter than water from the aqueous layer by freezing the water layer. It is specifically designed for use with small volumes, i.e., multi-well plates and tubes. Small plastic pegs are withdrawn, leaving only the organic, non-aqueous phase. 4 This method can be used in conjunction with automation.
A relatively new technique, Solid Supported Liquid-liquid Extraction (SLE), utilizes a solid phase medium loaded with an appropriate liquid reagent/solvent to achieve separations. 5 –7 Hence, the liquid phase is kept stationary on the surface of the solid media and as such allows the introduction of liquid reagents, through a solid medium in a controlled manner, without risk of contamination of the sample. For example in acylation reactions, excess imidazole reagents can be removed by 1.0N H2SO4, supported on diatomaceous earth. 8,9
Another solution for parallel work-up on larger volumes uses ‘dead-reckoning’, which involves withdrawing a pre-programmed amount of liquid from the two-phase reaction mixture. This is often used on synthesis platforms employing a cannula-based liquid handling system. 10,11 Such systems are not limited to one extraction but are often inaccurate if the boundary between the layers is poorly defined; evaporation has occurred during the reaction; or solvents are partially soluble in each other so that the actual volume of each layer is not known.
Techniques involving automated phase boundary detection have recently been developed. 12 In these systems, two immiscible liquids are separated by using sensor technology to determine the boundary layer. Unlike systems using dead-reckoning, this eliminates the need to pre-program the volumes. This technique most closely resembles the manual method. A variety of sensor technologies have been investigated based on different physical properties of the phases including electrical properties (e.g., conductivity), refractive index, UV absorbance and light reflection from the phase boundary.
The principle challenge is to develop a technology that works robustly and reliably on the widest possible range of two phase mixtures and is also tolerant to “messy” solutions that may have poorly defined boundaries or contain suspended precipitates. This paper discusses results obtained by using a new liquid-liquid extraction workstation developed by Mettler-Toledo, which employs a proprietary boundary sensor.
EXPERIMENTAL
All results were obtained using the ALLEX liquid-liquid extraction system (Figure 1). The unit employs a sensor to reliably separate and fully automate all liquid-liquid extraction steps, including:
Solvent addition
Vigorous agitation of mixtures
Repeated extractions
Vapor pressure release
Phase boundary detection
Selective drainage to waste
Figure 1 shows the separation chamber and sensor with a two-phase mixture being separated. The sample is passed through the sensor, lower phase first, until the solvent boundary is detected. At this point, the workstation stops dispensing and moves to another vial where the top phase is dispensed.

Mettler-Toledo Automated Liquid-Liquid Extraction (ALLEX) Workstation, showing the phase boundary sensor and settling chamber.
METHODOLOGY FOR NEUTRAL EXTRACTIONS
The partition coefficient of a compound between two immiscible solvents is fixed by the chemical properties of the compound. Therefore, recovery of the desired product can be improved by performing multiple extractions or by addition of dissolved salts such as sodium chloride. The use of brine increases the partition coefficient as the solubility of organic compounds in the aqueous layer is considerably reduced. Brine is often avoided in automated syringe based systems due to its corrosive properties, but is easily handled on ALLEX as all of the wetted surfaces on the sensor are made from PTFE. The automation of multiple extractions to optimize recovery is demonstrated in Figure 2.
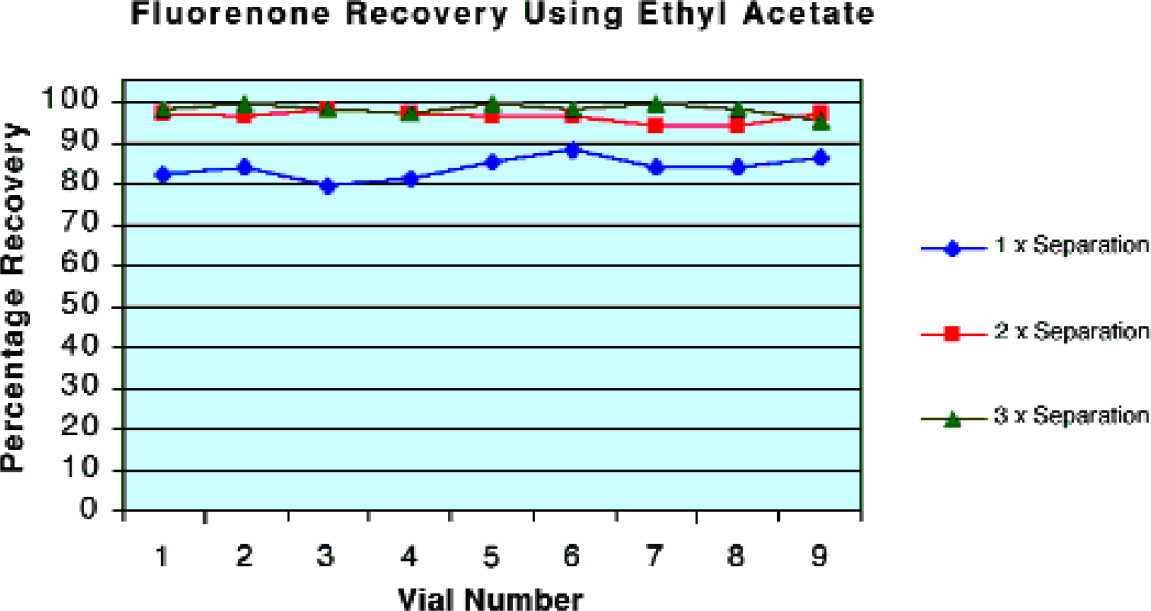
Recovery of fluorenone using ethyl acetate.
The recovery of a known quantity of 9-fluorenone from an ethyl acetate / water mixture was determined gravimetrically. Three extractions were found to be optimal. Increasing the number of extractions did not further improve the yield.
The ideal organic solvents for liquid-liquid extraction are of course immiscible with water and also have good solubility for a wide range of organic compounds. They should also have a different density from water and high volatility, so that they can be easily removed from the product by evaporation. Both dichloromethane and diethyl ether fulfill these criteria and an ideal automation system should accommodate their use. The graph in Figure 3 illustrates the handling of ether on ALLEX. The two lines on the graph show how priming the system with solvent vapor before dispensing improves liquid handling. Priming saturates the system with vapor, reducing evaporation of the solvent during dispensing and preventing dripping during transfer.
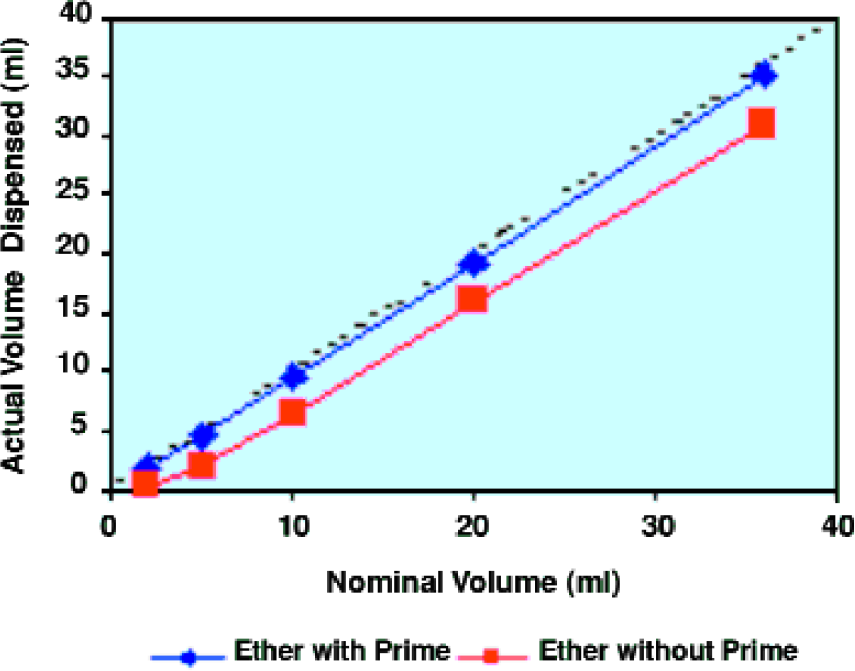
Effect of solvent priming on dispensing of volatile solvents.
A major difference between manually processing one reaction and automating multiple parallel reactions is the need to prevent cross-contamination between samples. This is not an issue with any system using individual components for each separation, e.g., hydrophobic frits. Since robotic systems process many samples, efficient washing steps must be included to prevent carryover between samples. This usually requires water to remove traces of aqueous solutions (e.g., brine, acid and base solutions) and a wash using an organic solvent to remove organic impurities.
The washing efficiency of ALLEX was tested by spiking samples with known amounts of 9-fluorenone (500mg) and performing a separation, then performing a second separation and determining the amount of 9-fluorenone carried into the second sample, using HPLC. Washing steps were then included between the two separations and their effect was carefully monitored. Where no washing was used, approximately 5% carry-over of 9-fluorenone into the second sample was observed, but the addition of a single ethyl acetate wash reduced the level to only 0.02%. This was further improved by a second wash, resulting in no measurable carryover.
METHODOLOGY FOR CHEMICALLY ACTIVE EXTRACTIONS
A major advantage of the liquid-liquid extraction procedure is the ability to separate acidic and basic compounds from neutral compounds. In such a separation, the intimate mixing of the two layers is of vital importance. Manually, this is achieved by a vigorous shaking of the separatory funnel. Other automated workstations such as the Mettler-Toledo Universal Sample Prep (USP), use vortex mixing and have grippers to carry individual vials to this part of the unit. This type of mixing is highly efficient. Other automated methods include orbital shaking, inversion of sealed reaction tubes and magnetic stirring. ALLEX uses the “aspire and dispense” approach to retract the entire sample and then expel it rapidly so that the two layers are intimately combined.
The results in Figure 4 show the efficiency of the mixing process in the extraction of benzoic acid from dichloromethane, using 2M sodium hydroxide. The basic solution was then neutralized and the acid re-extracted into clean organic solvent. With only one mixing cycle performed on the sample, an extraction efficiency of 81% was obtained. Mixing multiple times only improved the extraction efficiency to 88%, showing that the technique is very effective even with just a single mixing cycle.
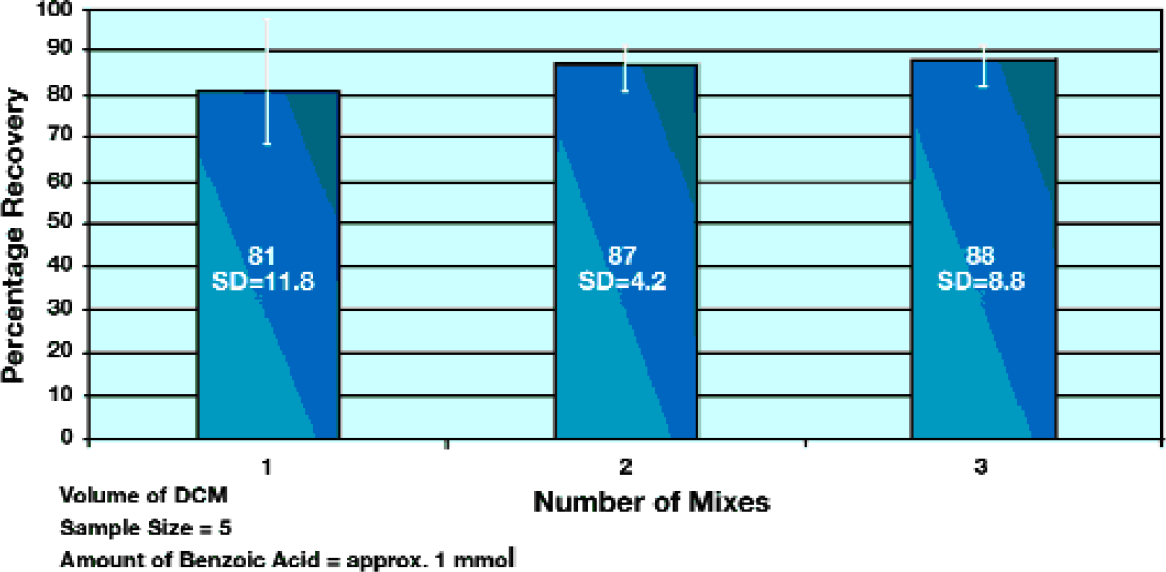
Extraction efficiency using the aspirate and dispense method of mixing.
RESULTS AND DISCUSSION
The principles mentioned above were applied in the automated work-up of a lithium aluminum hydride reduction of acetophenone and 4-methyl acetophenone, as depicted in Figure 5.
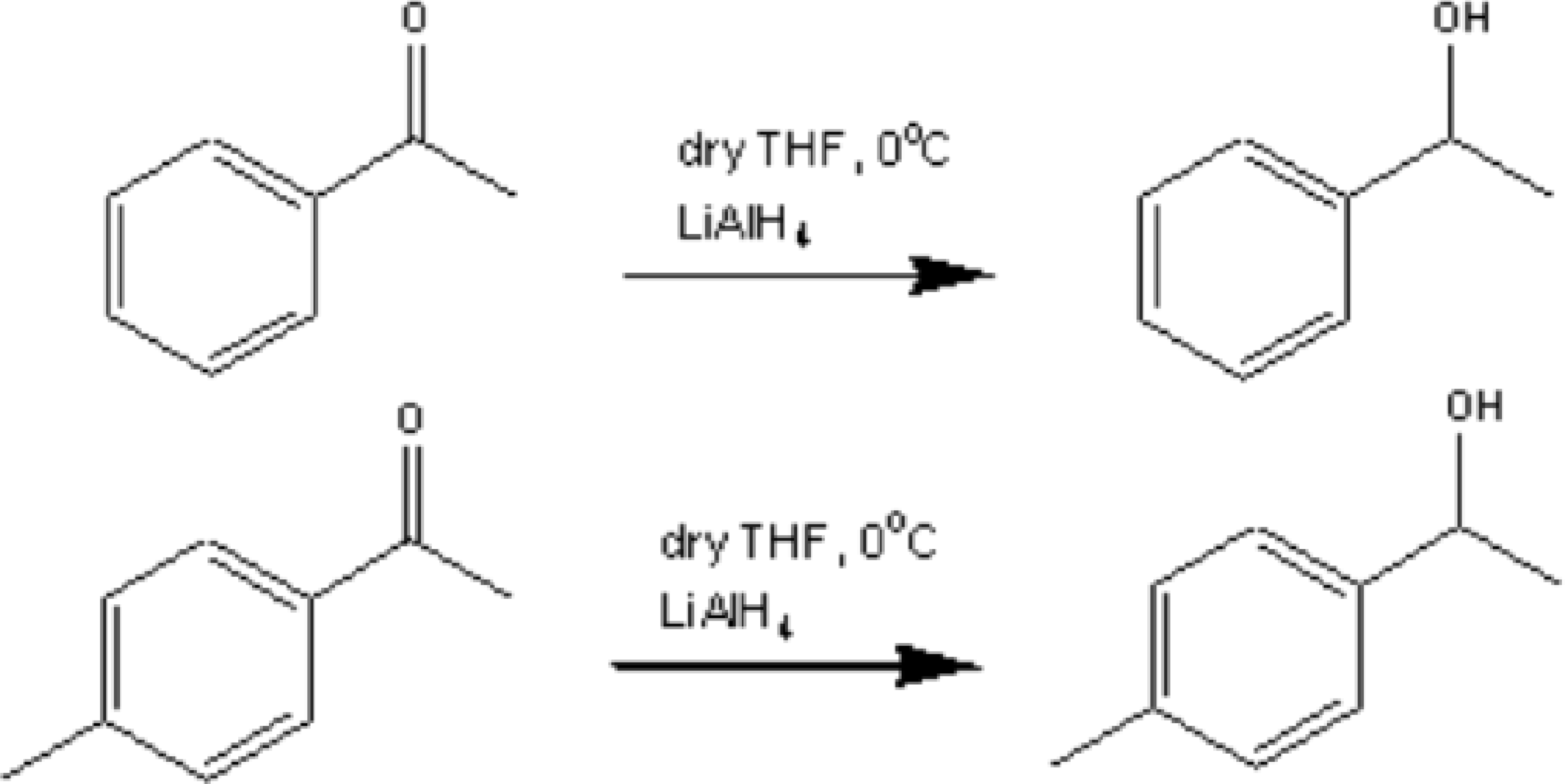
Acetophenone and 4-methyl acetophenone reduction using LAH.
The two reactions were set up for alternate work-up so that any cross-contamination could be monitored. The reaction was performed in dry THF in triplicate and then quenched by the careful addition of 2M hydrochloric acid, to destroy the excess reducing reagent. Ether was added and the mixture mixed twice rapidly. The two layers were separated and the aqueous layer extracted twice more in this fashion with ether. The combined ether layers were then washed with saturated brine to help remove any water dissolved in the organic layer. The system was programmed to wash with ethyl acetate and water, before commencing the next sample, in order to remove any carry-over between samples. All HPLC purities were determined by comparison with calibration curves prepared using authentic samples. Purities were found to be greater than 98% in all cases, demonstrating that cross-contamination can be effectively eliminated using the system.

In a final application, DIBAL reduction of γ-butyrolactone was carried out and the reaction mixture was then quenched by the careful addition of methanol, which caused the formation of a thick gel-like precipitate (Figure 6).
Figure 6. DIBAL Reduction of γ-butyrolactone
The work-up was then carried out on ALLEX by adding water and rapidly mixing three times to break up the solid. The reactions were left for five minutes to allow the precipitate to settle. Ethyl acetate was added and the mixture was rapidly mixed twice and the two layers were separated. The aqueous layer was extracted twice more in this fashion with ethyl acetate. The combined ethyl acetate layers were then washed with water and evaporated to dryness. The overall yield from the synthesis, including the work-up, is shown in the table below.
The results show that even in the case of difficult matrices, such as those containing solids or gel-like precipitates, separation steps can be automated by using instrumentation specifically designed for liquid-liquid extraction.

CONCLUSION
A perceived drawback of solution phase chemistry for the parallel synthesis of chemical libraries is that it creates a bottleneck at the work-up stage. The numbers of compounds generated using parallel synthesis means that without appropriate automated work-up techniques, the prospect of using solution phase chemistry is not always attractive.
As the work-up step plays a crucial role in the synthesis process, it can easily impact yields and purities of final products. For this reason, work-up needs to be addressed carefully and methodically. As shown in the applications above, the ability to automate the work-up step significantly extends the utilization of solution phase chemistry in combinatorial and medicinal chemistry laboratories. The Mettler-Toledo ALLEX system can be successfully applied to methodologies which would benefit from unattended liquid-liquid extraction.
