Abstract

Currently, within the Pharmaceutical drug pipeline, a “bottleneck” has resulted from an unprecedented number of new compounds generated by combinatorial chemistry coming together with an equally unprecedented number of new receptor (drug) targets from the Human Genome Project. As a class, G-protein coupled receptors (GPCRs) are historically the single richest receptor targets for drug discovery, accounting for nearly 60% of all prescription drugs on the market today including 12 of the top 20 drugs. These drugs cover most therapeutic areas and represent many billions in annual sales worldwide. There is a need for direct and accurate means for screening potential drug compounds against GPCR targets. The potential for GPCR-based drugs is largely untapped because only a fraction of these important drug targets are functionally known. It is estimated that about 1,000 GPCRs are “orphans” (functionally unknown). Because of the historical productivity of GPCR targets and the huge market value of related drugs, most pharmaceutical and biotechnology companies have active GPCR drug discovery programs that are in need of enabling technologies.
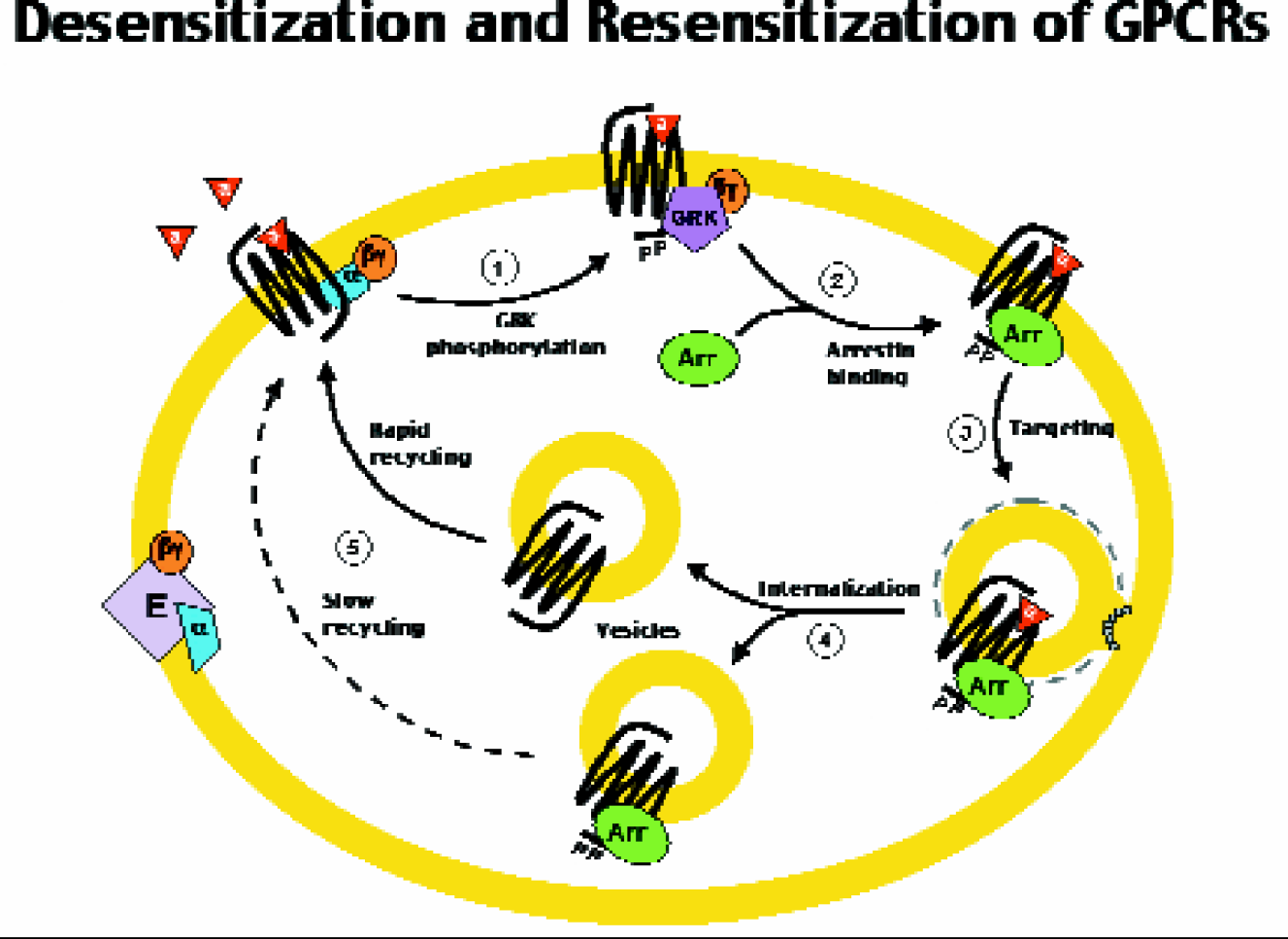
Upon activation by ligand binding, virtually all GPCRs rapidly undergo deactivation or “desensitization” by a common pathway (figure 1). An early step in this pathway is the binding of the cytoplasmic protein arrestin to the activated receptor at the plasma membrane. Arrestin binding turns off the GPCR, which results in the initiation of a process of internalization that brings the receptor into the cell where the ligand is removed before the receptor is recycled back to the membrane. By attaching a fluorescent label to arrestin, the recycling of the receptor-arrestin complex may be monitored. Since desensitization only occurs with an activated receptor, monitoring the redistribution of arrestin within the cell provides a method to detect the activation of any GPCR. Using human cell lines genetically engineered to express both the labeled arrestin and the GPCR of interest, one can screen for natural or synthetic ligands, whether agonists (activators) or antagonists (inhibitors).
Marc Caron, Ph.D., Robert Lefkowitz, M.D., and Larry Barak, M.D., Ph.D. of Duke University, leading authorities in the field and the scientific founders of Norak Biosciences developed the Transfluor™ technology, which consists of the fluorescently labeled arrestin. Norak Biosciences, Inc., a private biotechnology company based in Research Triangle Park, NC, is utilizing its proprietary Transfluor™ technology to become a world leader in the discovery and development of drugs that regulate G protein-coupled receptors (GPCRs). Transfluor™ is a cell-based fluorescence bioassay used to screen for GPCR ligands.
Norak's Transfluor™ technology monitors receptor activity by detecting arrestin movement in a cell. Arrestin is fused to green fluorescent protein (GFP) and is stably expressed in various cell lines. These cells can be transfected with a GPCR of interest either transiently or stably. Resting cells (figure 2) display fluorescence throughout the cytoplasm with no observable fluorescence in the nucleus. Activation of the transfected receptor induces, within seconds, a mass movement of the fluorescence to the membrane (figure 3). With a longer 15-minute stimulation, the GFP-labeled arrestin is found to redistribute with many GPCRs into intracellular vesicles (figure 4). Depending upon the binding affinity of the ligand and receptor, the complex can dissociate quickly or form internalized vesicles. The fluorescent vesicle state is stable for hours before the live cells will revert back to the resting state. The desensitization mechanism is fully reversible allowing screening for both agonists and antagonists of GPCRs. Cells can be permanently fixed and viewed up to one month later.
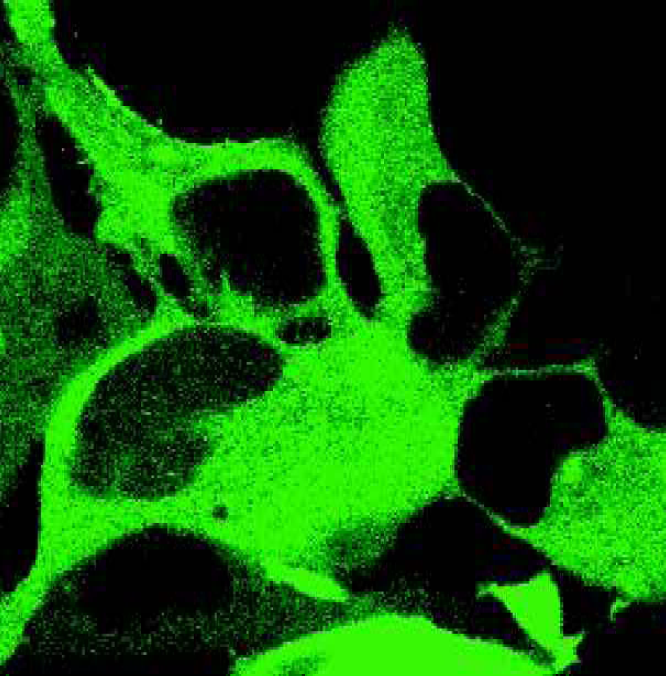
shows cells expressing GFP-labeled arrestin and a GPCR of interest under resting conditions.
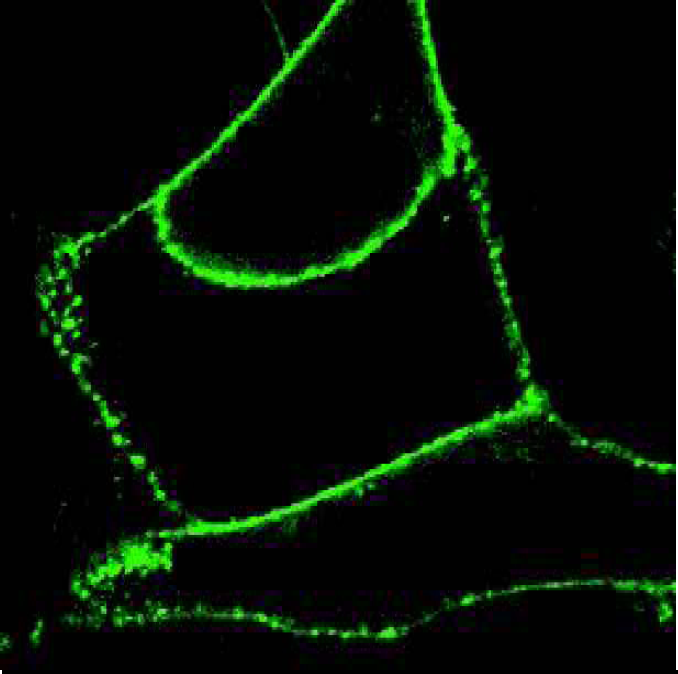
shows the movement of GFP-labeled arrestin to the activated GPCR at the plasma membrane 1 minute after stimulation.
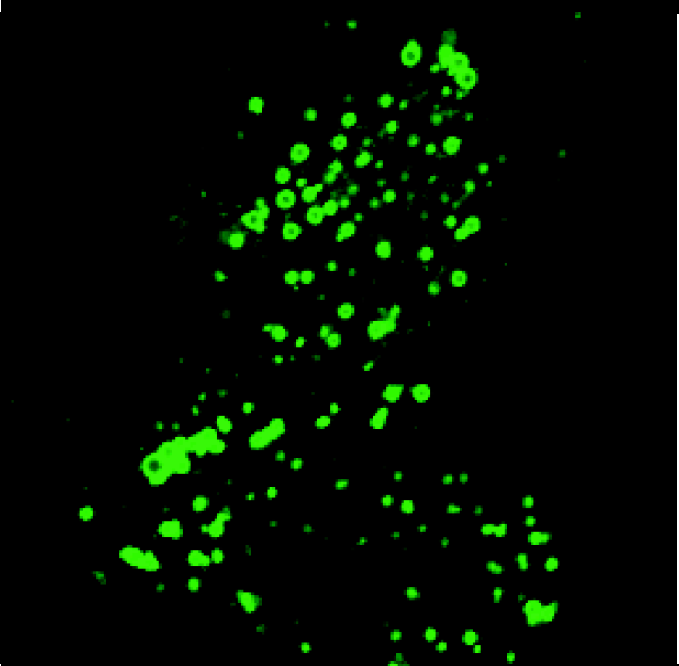
shows the movement of GFP-labeled arrestin with the activated GPCR into intracellular vesicles 15 minutes after stimulation (100:1 S/N)
“The key differentiating feature of Transfluor from other such technologies is that it is universally applicable to all GPCRs, is highly quantitative, and has an extremely high signal to noise.” commented Dr. Roger Blevins, Norak's CEO and President. “In addition, it is the most direct means of detecting receptor activation, and subsequent desensitization, so there are few, if any, false positives or false negatives generated during compound screening.”
Now commercially available, Transfluor™ appears to offer significant advantages over other GPCR-based assays, including, compatibility with modern high-throughput screening (HTS) technology, high sensitivity for GPCR ligands (few if any false negatives), and high specificity for GPCR ligands (few if any false positives). It is apparent that this technology is universally applicable to GPCRs whether functionally known or orphan. Norak is working to identify new drugs by high throughput screening of large compound libraries using an automated Transfluor™ assay. The assay will screen both known and orphan GPCRs as well as the proteins active in the cell mechanism of receptor desensitization. Since GPCRs are well conserved throughout the plant and animal kingdom, Transfluor™ provides numerous other potential applications and related business opportunities, including veterinary medicines (i.e., animal GPCRs), agrichemical products (i.e., plant and insect GPCRs), foods, beverages and cosmetics (i.e., human sensory GPCRs), diagnostic applications (i.e., detection of GPCR-based ligands). This technology may even find a useful niche as a basis for environmental biosensors (i.e., detection of GPCR-based toxins).
