Abstract
While high density MicroWell plates, including 384 well and 1536 well plates, offer advantages of low volume reaction or growth vessels in a standardized footprint allowing robotic manipulation, the greatest benefits lie in the increased well surface area to reaction volume ratios. This surface area to volume benefit can be utilized to optimize assay development by utilizing the molecular surface as a reactant. The well geometry and optical properties are also enhanced in high-density plates, allowing microscopic imaging and increased signal detection for fluorescence and luminescence.
The molecular characteristics of high density MicroWell plates were examined in regard to base materials and induced surface modification. Various modification methods including injection-molding parameters, exposure to high and low energy sources and exposure to specific physical conditions were studied. The surfaces were molecularly characterized and then utilized as reactants in high throughput and low volume in vitro assays. Polystyrene plates exhibited a natural relatively hydrophobic surface due to the hydrocarbon backbone of the polystyrene along with the repeating benzene ring. Exposure to direct electrical discharge, and several forms of irradiation in the presence of air and oxygenated atmosphere resulted in surfaces exhibiting various levels of oxidation. These levels were measured and the molecular species were calculated.
These specific surfaces were utilized as reactants in solid phase, cell based and cell culture assays. Specific surfaces demonstrated specific efficacy in regard to particular applications. For example, a highly charged surface exhibiting hydroxyl, carbonyl, and some carboxyl moieties was best for adherent cell culture. Utilizing passive adsorption procedures, a variant surface exhibiting more hydroxyl and carbonyl groups did not bind IgG well but did bind phospholipids, providing a surface “high-binding” for lipids while maintaining low background due to non-specific adsorption. A surface, commonly referred to as “high binding,” having slightly lower surface energy than the other two studied and characterized by aromatic hydrocarbons, hydroxyl and some few carbonyl groups did not bind hydrophobic antigens, but did bind glycoproteins such as IgG very well under slightly basic, passive conditions.
The geometries of wells were also examined. Well geometries contributed significantly by eliminating reagent wicking or creeping, enhancing detection of signals, and increasing the active surface area to reagent volume, allowing the utilization of decreased substrate concentrations along with low reagent volumes without loss of sensitivity.
This presentation was given at the 2000 International Symposium for Laboratory Automation and Robotics (ISLAR) held in Boston, MA, October 15-18, 2000. The full manuscript is available on CD-Rom and can be acquired by contacting Christine O'Neil, 508-497-2224; email
INTRODUCTION
When developing assays in high density, low volume MicroWell® plates, it is important to understand how the plate, characterized in part by the molecular surface properties, interacts as an assay reactant rather than a passive container. In addition to the analyte molecules or organisms, reagents and detection system, the reaction vessel itself is a critical component of an in vitro assay. MicroWell plate material, surface properties, format, especially as encountered as well surface area to volume ratio, and optical properties are critical parameters in assay development.
The most commonly utilized plastic for assay plates or culture vessels is polystyrene because of its optical clarity and range of surface properties. As a long chain hydrocarbon, non-modified polystyrene tends to repel water and hydrophilic molecules and attract hydrophobic molecules (Figure 1).
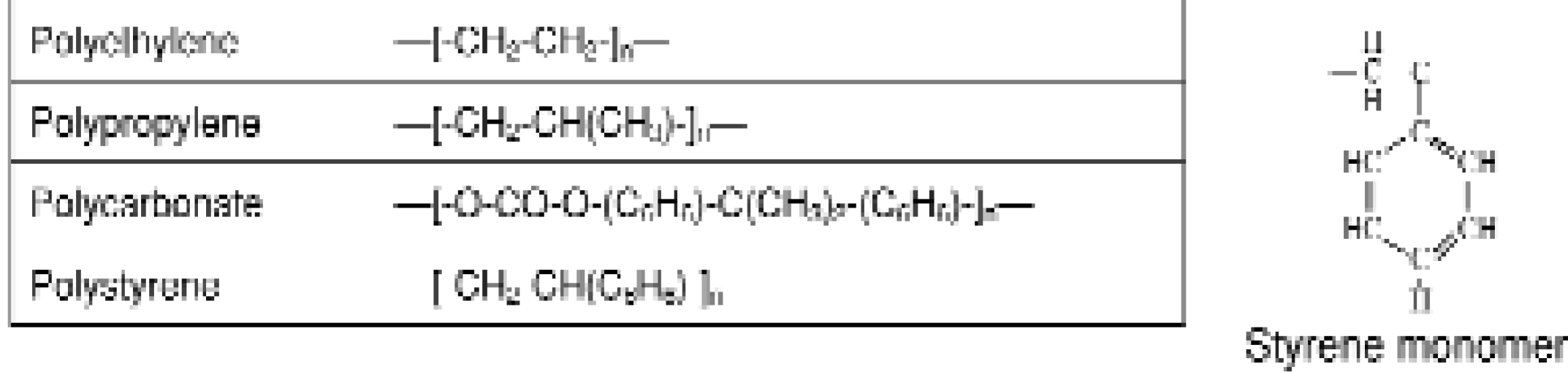
Polymers commonly utilized in medical devices and reaction vessels.
Hydrophobic molecules adsorb to the polystyrene surface as a result of intermolecular interaction, mainly van der Waals forces. 1 In general, van der Waals mediated bonds are about 100 times weaker than ionic and covalent bonds. Large biomolecules considered hydrophilic will inevitably have stretches of hydrophobic regions that allow the molecule to adsorb to the polystyrene surface. But to enable stable binding of hydrophilic molecules, assay conditions such as high molecule concentration, long incubation time and critical temperature conditions might be required to prevent the molecule from washing off the polystyrene surface. 2
The molecular nature of plastic medical devices and reaction vessels such as injection molded or MicroWell plates are produced through the manipulation of key parameters during each stage of formation (Fig. 2). These parameters include: the material itself (polystyrene, polypropylene, polycarbonate, along with additives, for example), the design of the mold or device used to shape the reaction vessel format and geometry, and manufacturing processes utilized (temperature, pressure, time). Changes in these parameters can cause changes in the polymer such as cleaving double bonds. In addition, these processes influence gross structural characteristics such as crystal structure, lamellar structure and deformation. Thus, a 384 well plate of a singular design made from the same base polymer can have different molecular characteristics.
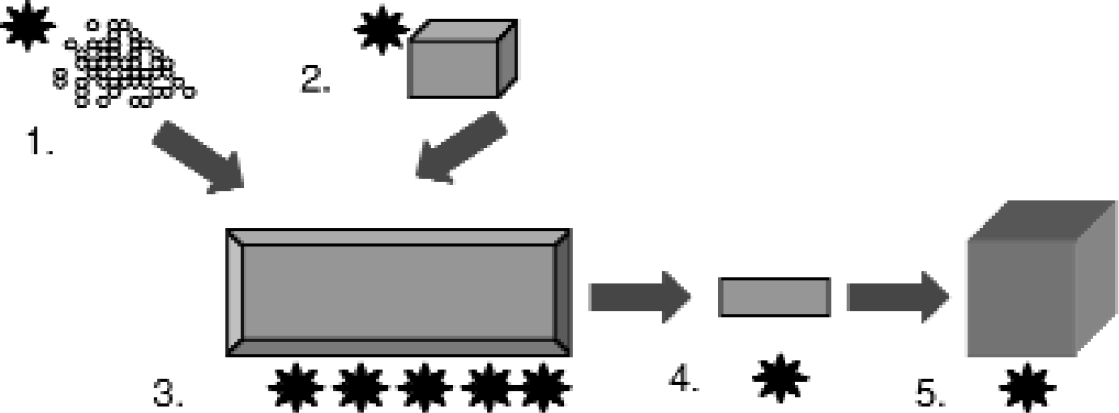
A reaction vessel's molecular characteristics can be manipulated through (1.) choice of the polymer material, (2.) the design of the forming device (mold) (3.) manufacturing processes such as injection molding, temperatures, pressures and timing, as well as (4.) and (5.) physical, chemical, and biogical secondary and tertiary modification processes.
Processes secondary to the manufacturing methods such as injection molding can further modify the molecular nature of a plastic device or vessel. The surface of polystyrene devices can be physically modified through a number of methods. 1,3,4 By applying energy (electrical discharge, UV irradiation, e-beam, and gamma irradiation), free radicals are formed in the polystyrene molecule, breaking bonds in the benzene ring and eventually the carbon backbone. When air or oxygen is added during the process, the polystyrene is oxidized, making it more hydrophilic. Polystyrene modification procedures involve addition of energy from a controlled source under strict environmental conditions resulting in a well-characterized, consistent surface.
High binding capacity surfaces, such as the MaxiSorp™ surface (Nalge Nunc International, Naperville, IL), are created by such a method. By introducing oxygen molecules to the molded polystyrene, a fine mosaic of hydrophilic and hydrophobic binding groups is generated which attract hydrophilic as well as hydrophobic regions on molecules. Both types of regions are common in antibody molecules. This mixture of binding capabilities makes the MaxiSorp surface ideal for applications such as ELISA in which antibodies are absorbed to the surface and function as capture agents in the assay.
METHODS AND RESULTS
By utilizing different energy sources at different intensities, under different environmental conditions or in a different atmosphere, the same MicroWell format formed from the same polystyrene, utilizing the same mold and injection molding parameters will exhibit distinct molecular surfaces. These molecular surfaces interact with various molecules in an in vitro assay in distinct manners. The MaxiSorp™ and MultiSorp™ surfaces demonstrate specific molecular surfaces induced by controlled and distinct, permanent, physical secondary modification processes.
ESCA ANALYSIS
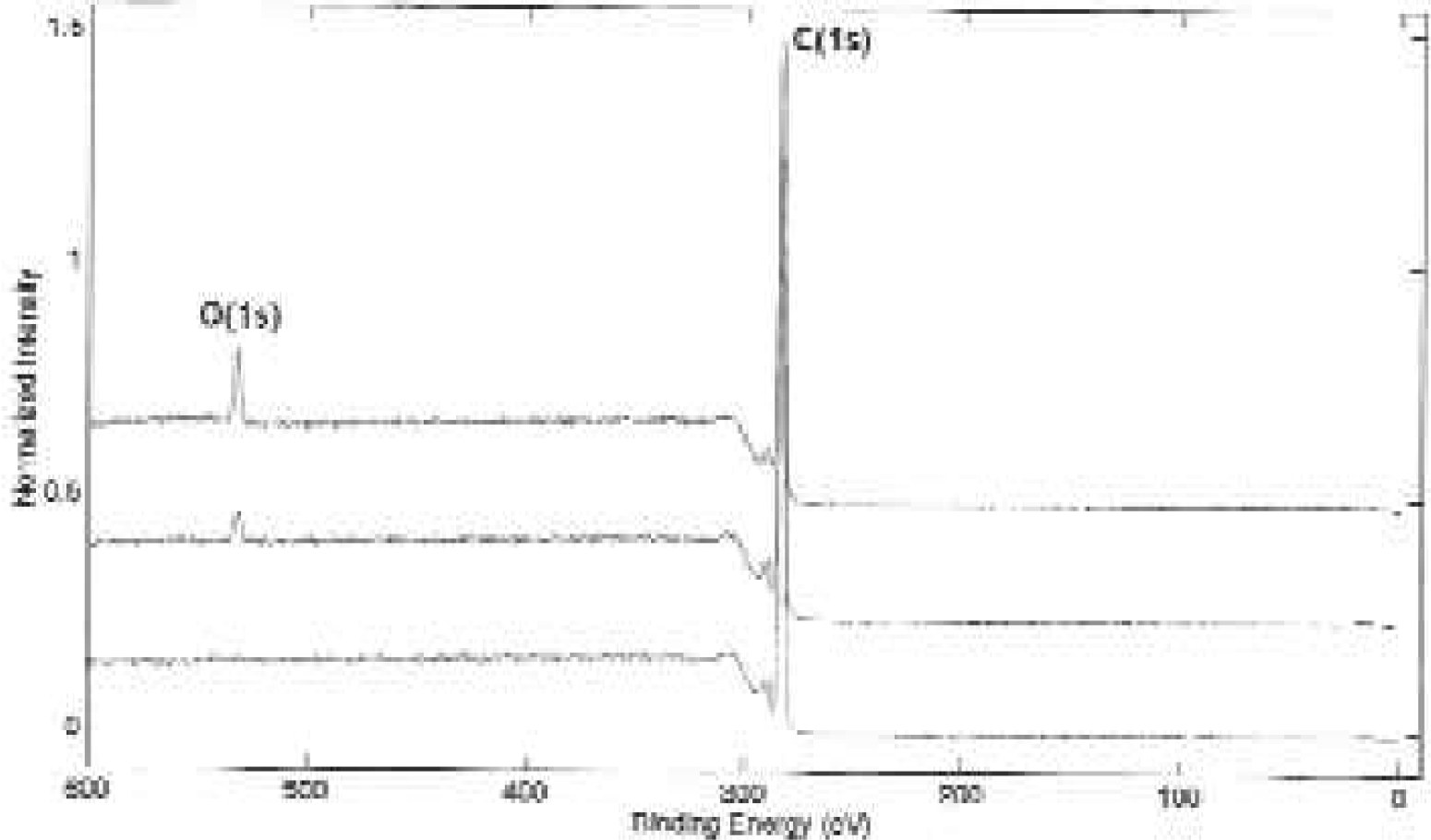
Through electron spectroscopy for chemical analysis (ESCA, also known as XPS), the composition and atomic concentration of elements of the MaxiSorp and MultiSorp surfaces were determined. Results indicated that only carbon and oxygen were detected on the MicroWell surfaces. Through measurement of the carbon 1s and oxygen 1s spectra on three different polystyrene surfaces (non-modified, MaxiSorp and MultiSorp), the atomic concentration of oxygen was calculated. Surface oxygen concentration varies among the different surfaces.
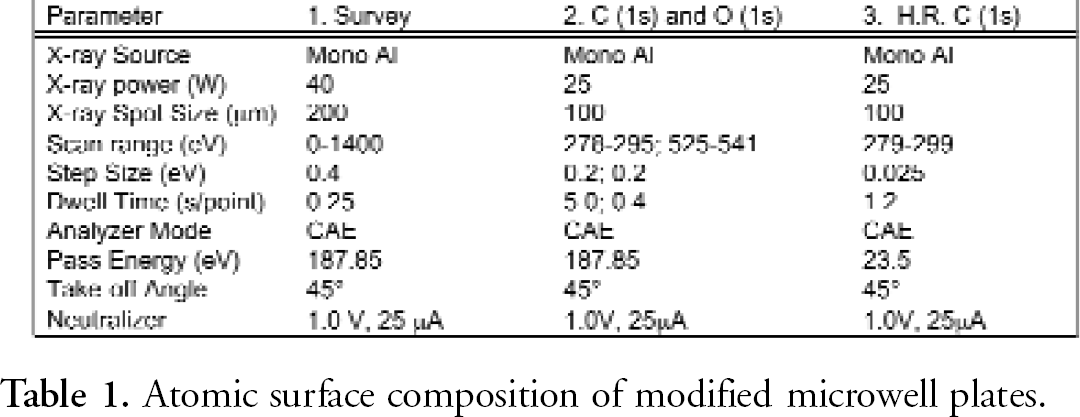
ESCA scans were obtained utilizing a Physical Electronics Quantum 2000 XPS instrument equipped with a monochromatic Al X-ray source. A survey spectrum was conducted to detect the elemental composition of each sample. The atomic concentration of oxygen was determined through the measurement of the carbon 1s and oxygen 1s spectra of triplicate samples. The functional group determination consisted of collecting high-resolution carbon 1s spectrum from triplicate samples. The analysis conditions for each type of analysis are presented in Table 1. The results indicated that only carbon and oxygen were detected on the samples (Figure 3).
Atomic surface composition of modified microwell plates.
XPS spectra of surfaces.
Measurements of the carbon 1s and oxygen 1s spectra in triplicate samples were used to determine the atomic concentration of oxygen of each surface. The results demonstrate an increased oxidation of the MaxiSorp and the MultiSorp surface.
These spectra were peak fit to determine the carbon-oxygen functional groups concentrations on the surfaces of the samples (Table 2).
Carbon Peak Fitting.

Further analysis of carbon-oxygen functional groups was performed using high-resolution carbon 1s spectrum from three samples. A representative spectrum for each surface type is depicted in Figure 4. Carbon oxygen functional groups appear as shoulders at higher binding energy than the main, aromatic/aliphatic carbon peak. The feature in the spectrum at ∼291.5 eV is due to a π → π* transition characteristic of aromatic rings in samples.
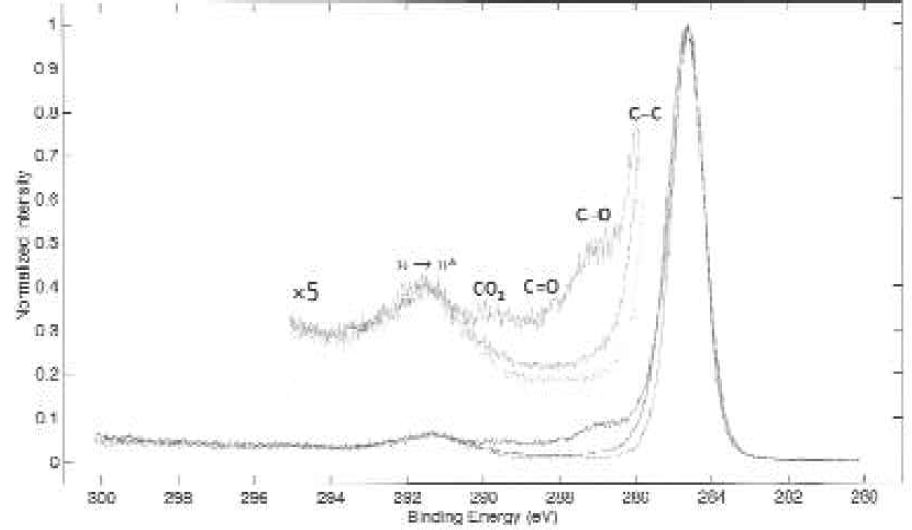
Carbon 1s spectra of surfaces.
These spectra were analyzed to determine the carbon-oxygen functional groups on each surface (Table 4). These percentages were determined through carbon peak performed on each spectrum. It is apparent from the data that the MultiSorp surface modification results in a greater degree of oxidation when compared with the control and MaxiSorp samples.
Atomic surface composition of modified MicroWells plates.
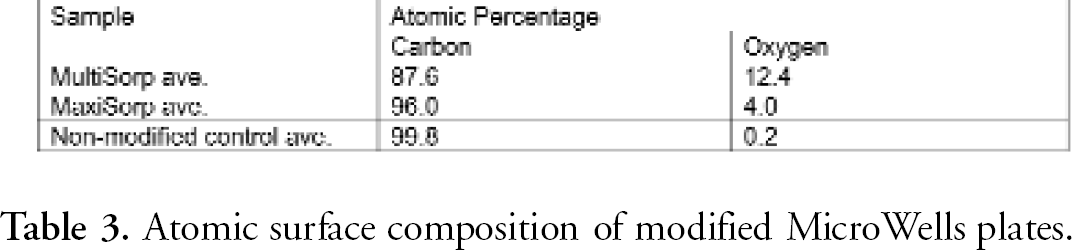
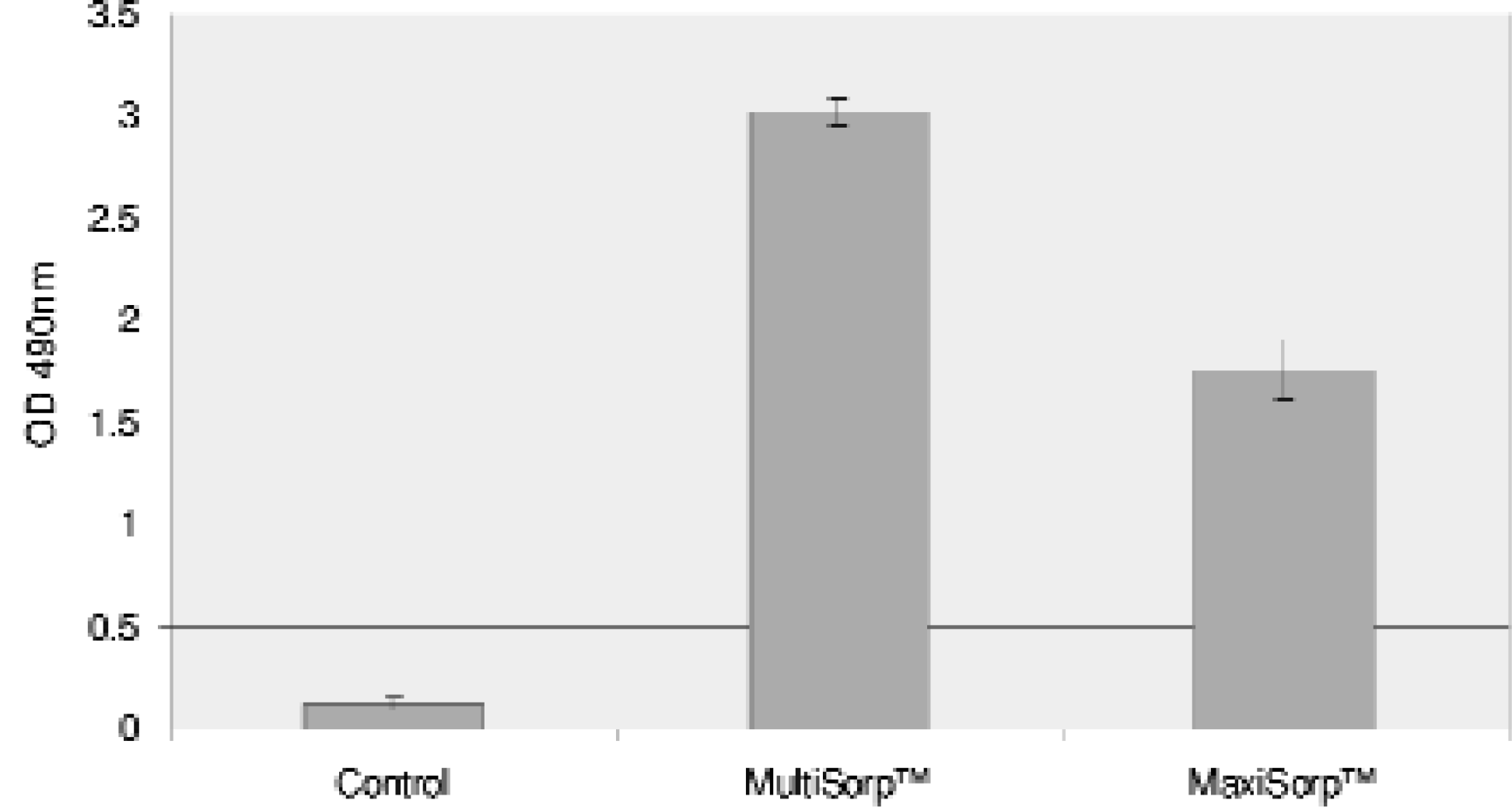
PASSIVE BINDING UTILIZING MODIFIED SURFACES IN A 96 WELL PLATE
BASIC PROTEIN BINDING ASSAY
The 96 well plates were coated with 200 μL per well of an antibody mixture containing 3600 ng/mL rabbit anti-sheep IgG and 33 ng/mL anti-alpha 1-feto-protein horseradish peroxidase conjugate in 0.05M carbonate buffer, pH 9.6. The plates were sealed with Nunc sealing tape to prevent evaporation and incubated overnight, protected from light, at room temperature. The plates were washed three times with 0.15M PBS, pH 7.2, containing 0.2M NaCl and 0.05% Triton X-100. The washed plates were then incubated with 200 μL per well of a substrate solution containing 0.6 mg OPD and 0.5μL 30% H2O2 per mL in 0.1M citrate phosphate buffer, pH 5.0. The colorimetric reaction was allowed to continue for five minutes before the addition of 150 μL of 2N H2SO4. The optical density at 490nm was determined using an Elx 800 microplate reader.
ACIDIC PROTEIN BINDING ASSAY
The 96 well plates were coated with 150 μL of a solution containing 2.5 μg/mL BSA, and 2.5 μg/μL human alpha 1-feto-protein in an acetate buffer (pH 4). The plates were sealed with Nunc sealing tape to prevent evaporation and incubated overnight, protected from light, at room temperature. The plates were washed three times with a washing solution (PBS containing 0.05% Triton X-100). To decrease non-specific binding, the plates were blocked with a 1 hour, room temperature incubation with 1% (w/v) of gelatin in PBS solution containing 0.05% Tween-20. The blocking solution was decanted and 150 μL of an antibody solution containing 1.4 μg/mL Rabbit anti-human alpha 1-feto-protein horseradish peroxidase conjugate and 9 μg/mL Rabbit anti-human alpha 1-feto-protein in PBS containing 0.05% Tween-20 was added. The plates were again washed three times with the washing solution. Finally, 150 μL of the substrate solution containing 0.6 mg OPD and 0.5μL 30% H2O2 per mL in 0.1M citrate phosphate buffer, pH 5.0 was added to each well. The colorimetric reaction was allowed to continue for four minutes before the addition of 100 μL of 2N H2SO4. The optical density at 490 nm was determined using an Elx 800 microplate reader.
PHOSPHOLIPID BINDING ASSAY
NBD-labeled phophatidylserine and non-labeled phosphatidylserine, at a ratio of 1:20 in chloroform:methanol (v/v 1:4), were mixed in a glass tube. Different amounts of phoshatidylserine were dispensed into a 96 well plate. The solutions were dried under nitrogen to minimize oxidation. The plates were washed three times with 100 μL of PBS. Fluorescence was detected using excitation and emission wavelengths of 485 and 530 nm, respectively.
CELL CULTURE
L929 cells were plated at a density of 2.5 × 105 cells/mL into 96 well plates with various surface modifications. The cells were allowed to proliferate for 24 hours in minimal essential media supplemented with 10% bovine calf serum. The morphology of the cells was examined microscopically utilizing a Zeiss inverted microscope equipped with a 10X objective and 10 X oculars. Images were captured digitally for analysis.
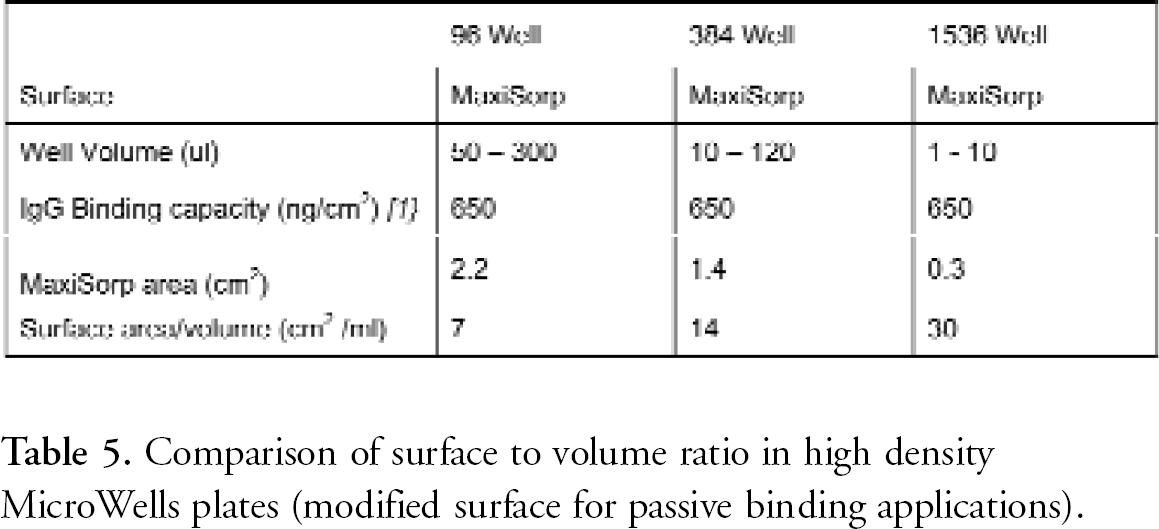
SURFACE AREA TO VOLUME RATIO
The 384 Well Plate surface to volume ratio is nearly double that of the 96 well plate. Therefore, the 384 Well Plate gives enhanced signal in solid phase reactions in which surface plays an important role in the assay.
CELL CULTURE IN HIGH DENSITY MICROWELL PLATES
As important as plastics have been to solid phase assays, plastic ware has been more essential to the area of cell culture. Often misunderstood, the cell-binding surface is far from just a sterile environment. Another misconception is that a cell culture surface is equivalent to a highly charged “high binding” plate. Unlike molecules binding a plastic surface as a result of hydrophobic or hydrophilic interaction, some types of cells require adherence to a surface or support in order to grow and function in vitro. Cell culture plates feature a charged, hydrophilic surface of higher energy than a hydrophilic, high binding MaxiSorp surface. The cell culture surface provides cells with a patchwork of oxidized molecules creating an environment that promotes stable attachment and growth. Since cells adhere better to this specialized surface, cells are not lost during washing or buffer changes. The effectiveness of cell culture surface compared to non-treated surface is demonstrated in Figure 9. 7
Passive Adsorption to Modified Polystyrene Surfaces under Basic pH Conditions.
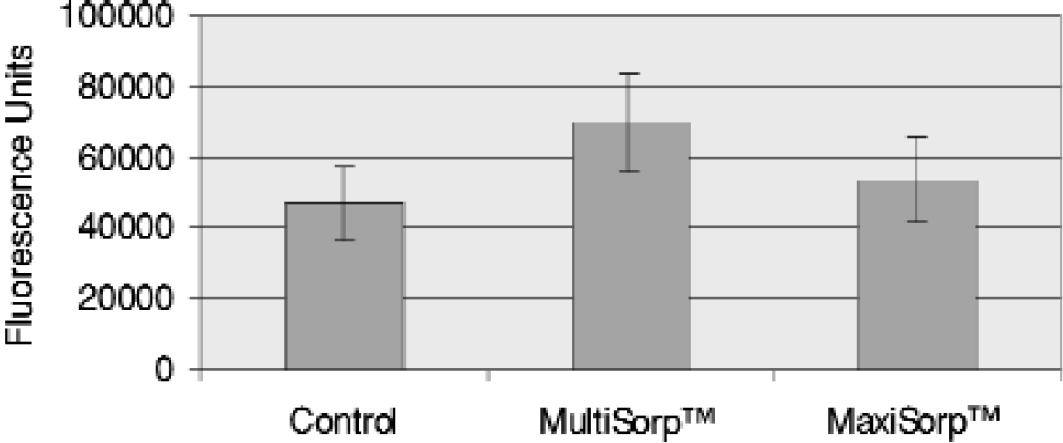
Passive Adsorption of Human alpha-Fetoprotein to Modified Polystyrene Surfaces under acidic pH Conditions.
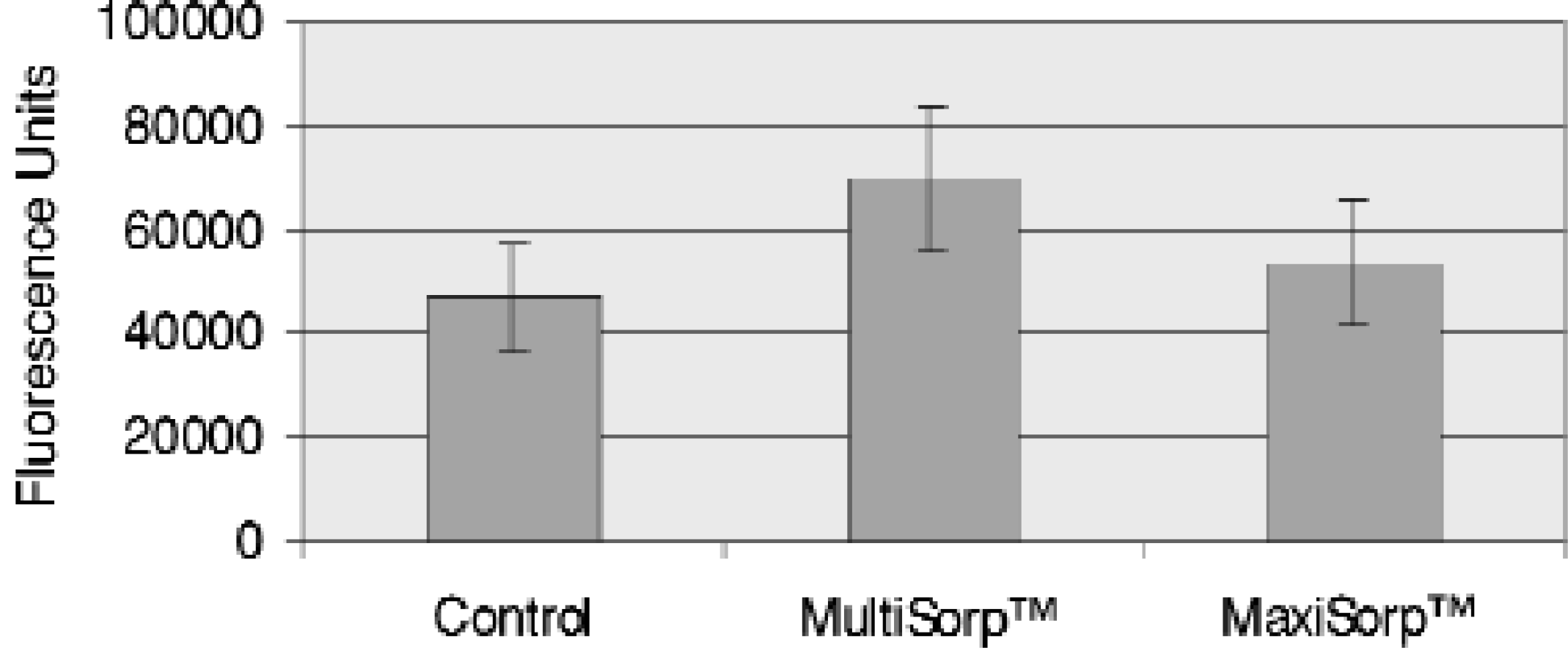
Passive Adsorption of Phosphatidylserine to Modified Polystyrene Surfaces
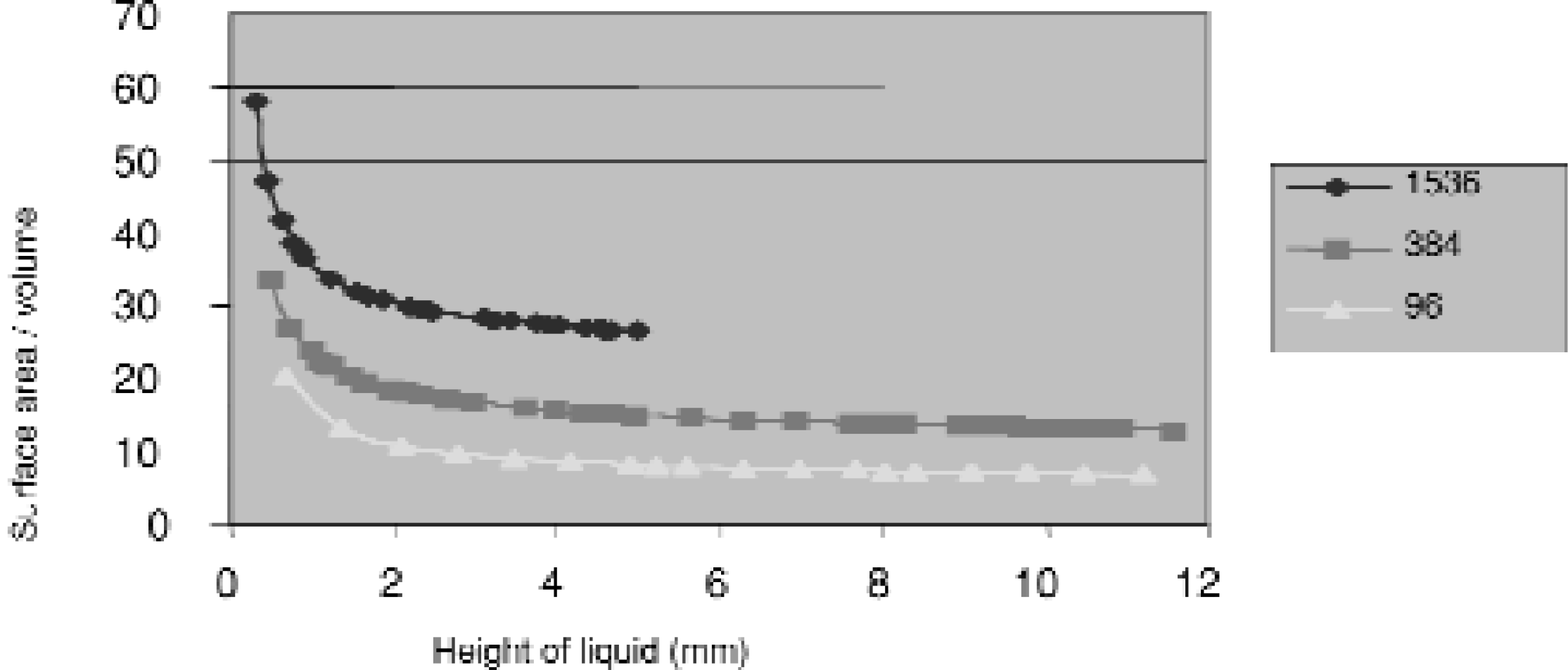
Well design and Surface: Surface Area to Volume Ratio of High Density MicroWell Plates. In many MicroWell Plates, the entire interior well surface is modified. This aspect of active surface area is extremely beneficial for solid phase assays. At equal liquid heights, the active surface to reagent volume of a 384 well plate is nearly twice that of a 96 well plate, and that of a shallow 1536 well plate is double again. This allows the use of very low reagent volumes without loss of sensitivity.
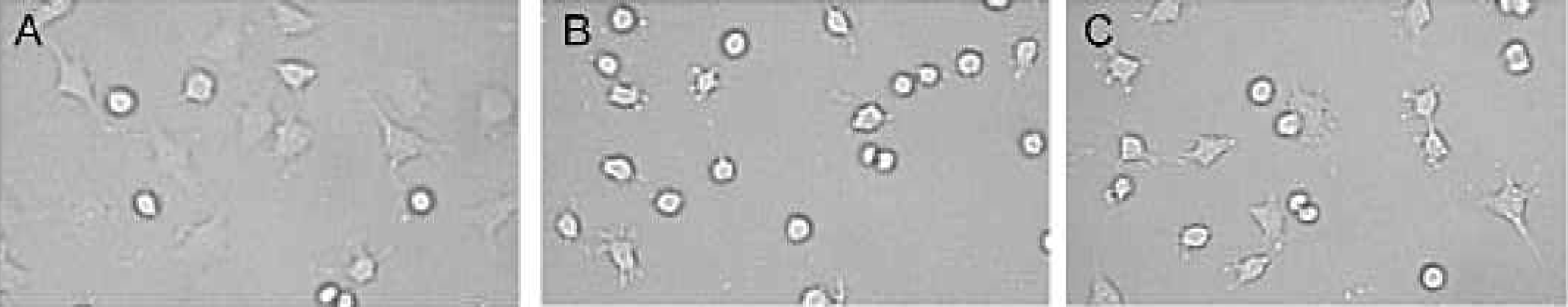
L929 Cell Culture on Polystyrene Surfaces. Cell culture treated plates allow for better cell adhesion than the non-treated plates. A. Cell culture modified surface; B. Unmodified, non-sterile surface; C: Unmodified, sterile surface. The physically modified cell culture surface supports cell adhesion and spreading typical of proliferating cells.
Comparison of surface to volume ratio in high density MicroWells plates (modified surface for passive binding applications).
WELL GEOMETRY & SURFACE MODIFICATION: PREVENTION OF WICKING AND INCREASED CHEMICAL RESISTANCE OF POLYSTYRENE
“Wicking” is the creeping of liquid up the walls of the wells. This results in the accumulation of liquid at the top of the wells and causes well-to-well contamination, crosstalk and inaccurate readings. Wicking is dependent on the surface tension and viscosity of the liquid. The presence of detergents or proteins decreases the surface tension of aqueous solutions and often contributes to wicking.
Wicking is influenced by the plate well geometry and surface energy. The rounded square well geometry of the Nunc brand 384 well plates prevents wicking. A consistently low surface energy over the plate is also critical in the prevention of wicking. In order to maintain the reflective properties of white crystal polystyrene, a secondary modification process is necessary to prevent wicking and cracking when incubating or storing plates containing harsh solvents such as those contained in most scintillation cocktails.
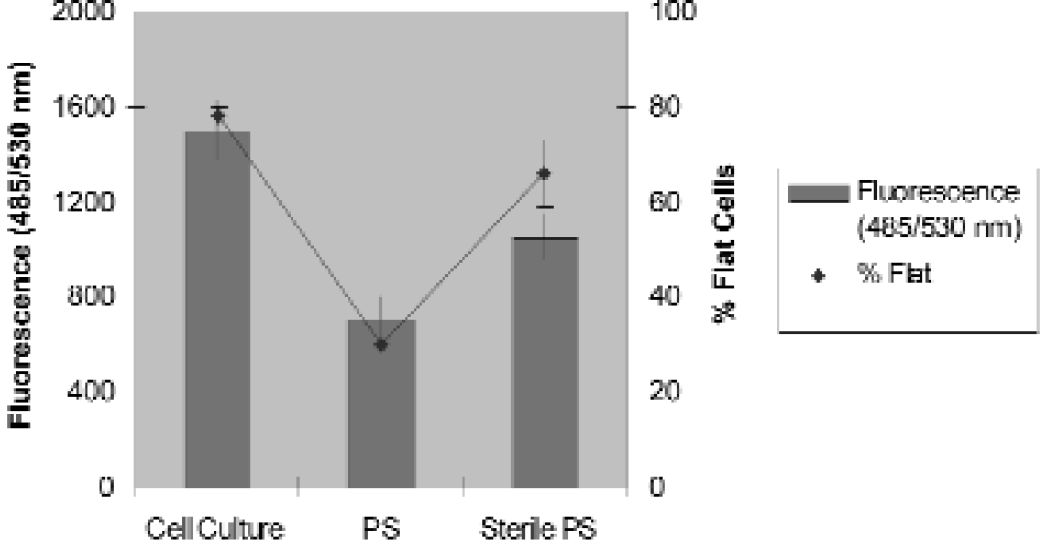
Cell culture treated and non-treated plates (PS) and non-treated, sterile plates (Sterile PS) were plated with L929 cells. Cell suspension (40 μl) was added to each well. Cells were seeded at a density of 2 × 10 cells/well and incubated at 37°C in a CO2 incubator for 24 hours. The plates were then washed and filled with fluorescein diacetate (20 μg/ml). After incubation for the indicated times at room temperature, the plates were read at 485 nm excitation and 535 nm emission in the Wallac Victor™ 1420. Fluorescein diacetate is a fluoregenic ester that is non-fluorescent in solution and freely diffuses into live cells. Within cells, esterase activity cleaves the ester from fluorescein diacetate causing it to become non-membrane permeable and fluorescent. Therefore, live cells will accumulate the fluorescent molecule over time. Esterase activity in serum containing media has a similar effect and, therefore, media should be removed before addition of label to the cells. Physically modified cell culture plates show significant increase in live cells over unmodified, sterile plates (typically utilized for bacteriology) and unmodified, non-sterile plates. The fluorescein results were compared to the percent of flat cells observed in the well. Flat cells indicate adherent, proliferating cells as opposed to round cells that are not adhering or proliferating.
In Figure 12, the top two plates demonstrate wicking or creeping of the scintillant to the top of the wells. Scintillation cocktail was pipetted into the wells of a 384 well plate. The plate was sealed with a solid, optically clear adhesive tape. After incubating at room temperature overnight (12–16 hours), the plates were illuminated with a UV lamp (366 or 254 nm). The top two plate images show the top of the plates. The Nunc brand plate, a white, relatively hydrophobic, highly reflective polystyrene material has low surface energy. Combined with the rounded square well geometry and columnar shape, the Nunc brand plate prevents wicking. The lighter, irregular outlines of some wells in Plate B is wicking or accumulation of the scintillant at the top of the wells.
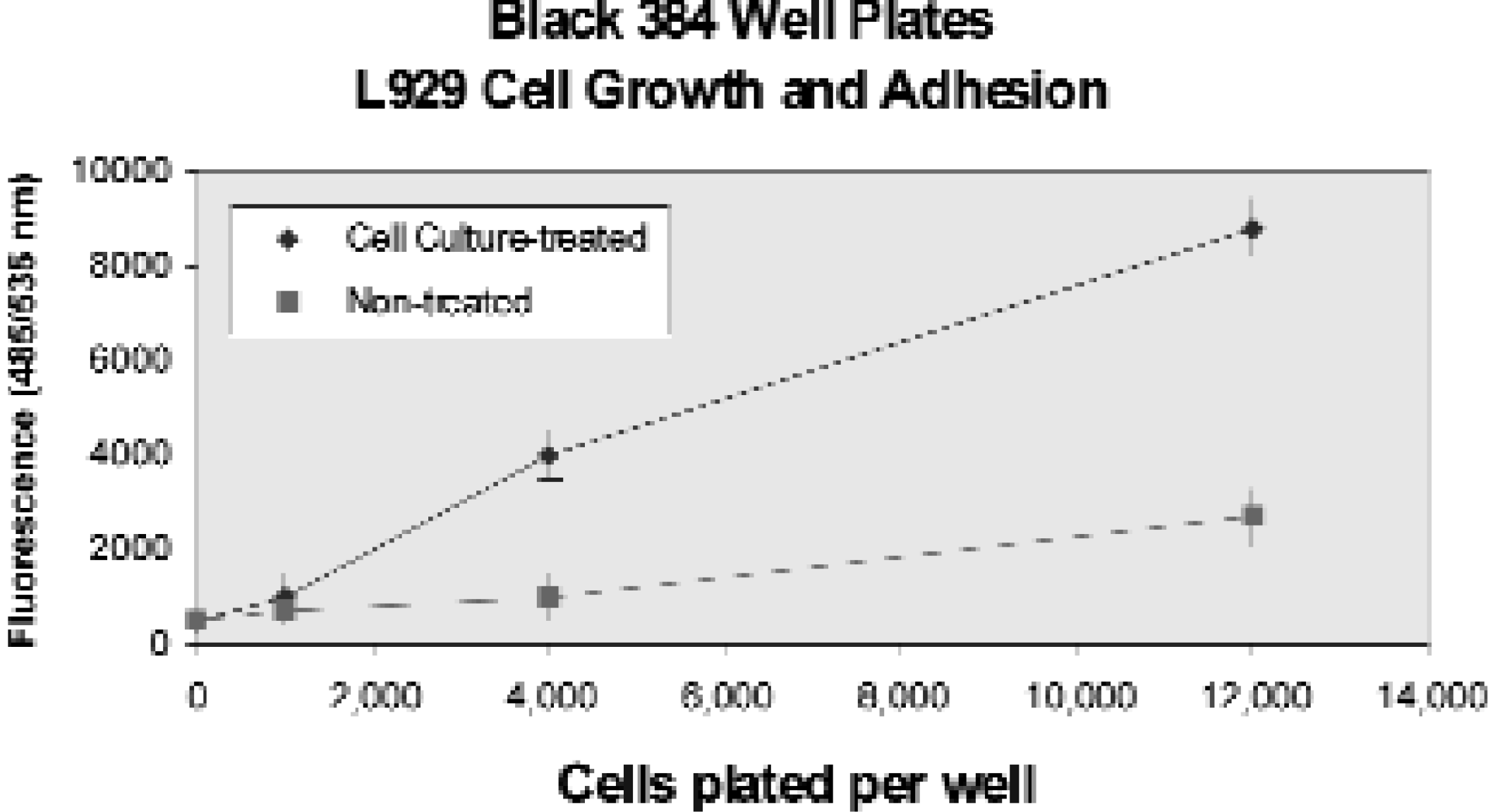
L929 Cell Culture on Polystyrene Surfaces. Cell culture treated plates allow for better cell adhesion than the non-treated plates. Five concentrations of L929 cells were seeded and incubated for 24 hours. Plates were washed with Hank's Balanced Salt Solution (HBSS) and filled with 40 μl/well fluorescein diacetate (50 μm) in HBSS. The plates were read at 485 nm excitation and 535 nm emission.

Example of wicking in 384 well plates with rounded square well geometry (Nunc Brand plate) and square well geometry (Plate B).
The bottom view shows the chemical resistance afforded by the modification process. The wells of the Optical Bottom Nunc brand plate are separate and distinct. The material of Plate B was not chemically resistant and deteriorated after 6 hours, slowing cross contamination between wells.
THERMAL STABILITY
By and large, polystyrene is an ideal material for most applications because of the clarity of the material and the ability to modify the polystyrene surface to optimize it for use. However, one property that polystyrene lacks is thermostability. Maximum use temperature for polystyrene is 90°C, which makes it unsuitable for PCR* applications. Polypropylene and polycarbonate are thermostable resins that have been widely used in PCR, but they lack the optical clarity of polystyrene. Typically, after PCR, amplicons are transferred to another reaction vessel for detection. Increased handling often results in lower recovery or carryover contamination. Another surface is available that possesses the attributes of an optically clear well bottom, thin wall construction for thermal conductance, covalent binding capability and thermostable resin. Under the product name NucleoLink™ (Nalge Nunc International, Naperville, IL), this surface covalently binds PCR primers oriented in a 5′-3′ direction. Solid phase asymmetric amplification is performed in the NucleoLink well; after amplification, the PCR reactants are removed from the well, and the immobilized amplicons are hybridized to a labeled probe and detected in the same well 12 . Advantages of a single vessel for reaction and detection include improved specificity and sensitivity, reduced contamination, and higher throughput of sample processing
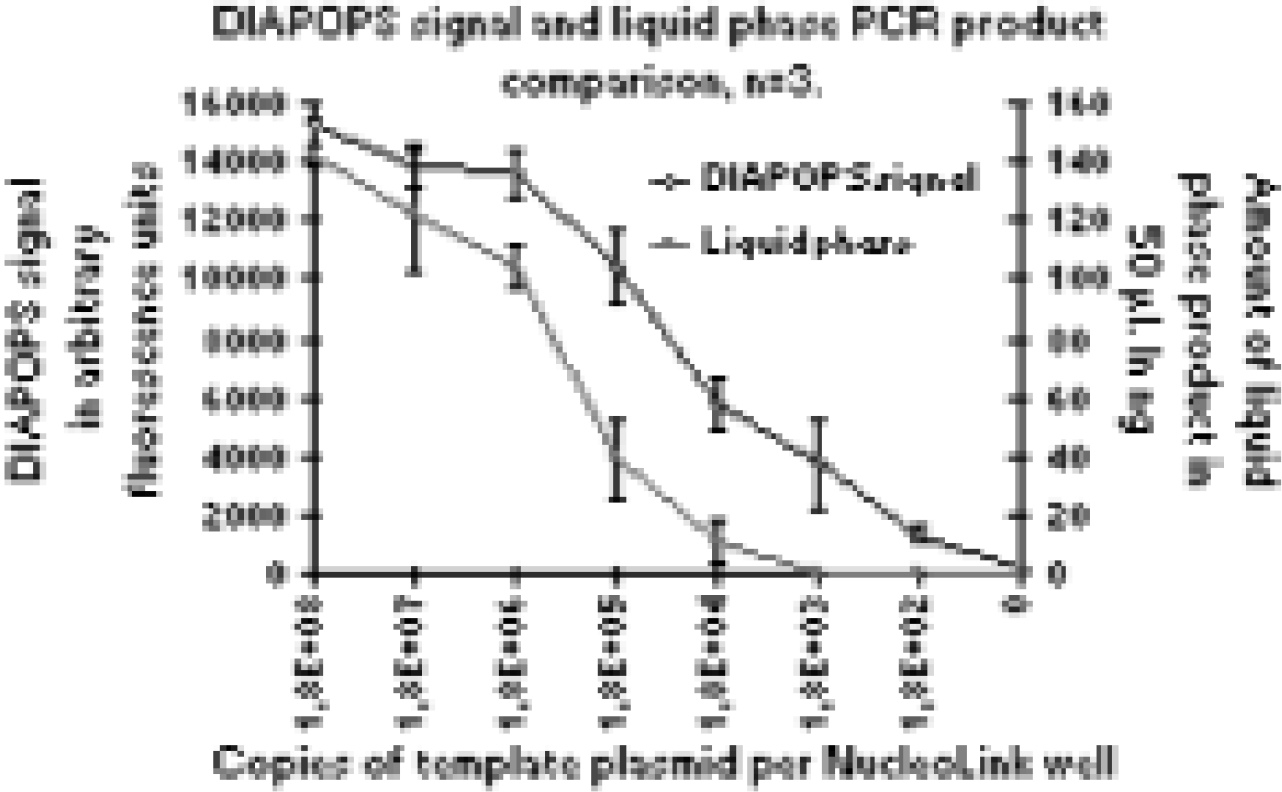
Comparison of amplification and detection of amplicons from a Liquid Phase PCR Reaction to a Solid Phase, DIAPOPS (
SUMMARY
It is essential to understand that while standardizing reaction vessel formats into a “96 well footprint” is important for the development of consistent, high throughput assays, there are also other benefits to a high density MicroWell plate. The molecular nature of the plate material, and exquisite modification through injection molding parameters, the design of the mold and tooling and secondary processes affect assay performance extensively. The molecular nature of a MicroWell plate can be utilized to benefit assay development and consistency.
The preferential passive binding of various macromolecules to the non-modified and various types of modified surfaces can be summarized as follows:
The optimum surface for any application depends on (1) whether the application calls for a passively or covalently immobilized component, (2) the chemical nature of component being immobilized and (3) the nature of the other reagents in an assay. Chemical resistance, surface energy, well geometry and optical properties must also be considered and optimized depending upon the application. One plate cannot fit all. The good news is that consistent, modified surfaces can be produced regardless of resin color; therefore black, white and clear plates are available with molecularly specific surfaces in order to benefit the enormous variety of indispensable assays.

