Abstract
Two non-competitive, antibody-capture enzyme-linked immunosorbent assays, a commercial Kit VZIG ELISA and an in-house developed, amplified VZIG ELISA were automated by using the liquid handling and plate processing abilities of the Microlab AT plus 2 and Microlab F.A.M.E. instruments, respectively. The evaluation of the experimental data has also been automated by using the F.A.M.E.-specific data import converter and PLA software program for data capture / uploading and analysis. The automation increased the economy of analytical operations and traceability of the experimental data thus better supporting the production and clinical studies on a therapeutic Varicella Zoster Immune Globulin manufactured at Cangene.
Keywords
INTRODUCTION
Varicella Zoster Virus (VZV) causes chickenpox (primary varicella) usually a benign childhood disease and the recurrent infections, shingles (herpes zoster) that may be associated with painful acute lesions, postherpetic neuralgia or congenital malformations 1 . Recently it has, however, been shown that VZV infections in children may result in serious life-threatening brain damage. 2,3,4 Varicella Zoster Immune Globulins (VZIGs) have an important role in the management of susceptible high-risk patients. 5 To support the manufacture of a novel VZIG therapeutic at Cangene Corporation (VariZIG™), a commercial Kit VZIG ELISA, and an in-house developed, amplified ELISA, were validated for the quantitation of the VZV-specific antibodies in the finished product 6,7 and patient sera, 8 respectively.
Both these ELISA methods involve assaying the test samples over a range of serial dilutions and the liquid handling steps are therefore, repetitive, lengthy and mundane. It has recently been shown for the amplified VZIG ELISA that the use of the automated Microlab AT plus 2 liquid handler streamlined all liquid handling steps and increased their accuracy and precision. It has also been shown that the use of the BioSwift data capture and evaluation software program decreased the time required for data documentation and analysis and increased the reliability and security of the ELISA data. 9
In the current study, the manual commercial Kit VZIG ELISA was automated for liquid handling steps by using the Microlab ATplus 2 liquid handler. Moreover, the Microlab F.A.M.E. instrument and PLA software program were used to automate microtitre plate processing and data analysis of both VZIG ELISA methods.
It has been shown that the implemented automation elements improved the performance of the assays and traceability of the experimental data while, at the same time, increasing the economy of analytical operations.
EXPERIMENTAL
MATERIALS AND EQUIPMENT
Analytical labware including analytical balance, barcodes, beakers, floppy diskettes, magnetic stirrers, magnetic stirring bars and pH meter.
ELISA Amplification System (Life Technologies™, Gaithersburg, MD, U.S.A.).
Immulon 2 and Immulon 4 Microtiter® 96 Wells Plates (Dynex Technologies Inc., Chantilly, VA, U.S.A.).
Microlab ATplus 2® Liquid Handler with SUNRISE plus® (V 3.3.1) Software Program (Hamilton Company, Reno, NV, U.S.A).
Microlab F.A.M.E.® Fully Automated Microplate ELISA Analyser with F.A.M.E.® (V 1.3) Software Program (Hamilton Company, Reno, NV, U.S.A).
Microsoft Excel® Spreadsheet Program (Microsoft Corporation, Redmond, WA, U.S.A.)
PLA© (V 1.2.03) Software Program and F.A.M.E.-specific data import converter© (Stegmann Systemberatung, Rodgau, GE).
Statistica® High Performance Statistical Software (Stat Soft Inc., Tulsa, OK, U.S.A.).
Varicella Zoster Virus IgG ELISA Kit (Wampole Laboratories™, Dist., Division of Carter Wallace, Inc., Cranbury, NJ, U.S.A).
Other materials, reagents and buffers required for the commercial Kit and Amplified VZIG ELISAs as described in the respective STM documents. 10,11
TEST SAMPLES
WHO International Reference Preparation of anti-varicella Zoster immunoglobulin, WHO Standard, stock solution at 50 IU/mL, (Central Laboratory of the Netherlands Red Cross Blood Transfusion Service, Amsterdam, ND).
Human VZIG positive plasma, NIBS ICS, stock solution at 2 IU/mL, (National Institute for Biological Standards and Control, Hertfordshire, U.K).
Human VZIG negative serum (A component of the VZV IgG ELISA kit, Wampole Laboratories™, Dist., Division of Carter Wallace, Inc., Cranbury, NJ, U.S.A).
METHODS
Automated preparation of two-fold dilutions performed with the help of the Microlab ATplus 2 liquid handler according to the designed, assay-specific plate-pipetting protocols.
Automated processing of the ELISA plates performed with the help of the Microlab F.A.M.E. instrument according to the designed, assay-specific plate-processing methods.
Manual data analysis performed with the help of the Excel and Statistica software programs as detailed in the STM documents for Kit 10 and Amplified 11 VZIG ELISA methods.
Automated data capture and analysis performed with the help of the F.A.M.E.- specific data import converter and PLA soft ware program according to the designed, assay-specific data-evaluation templates.
TESTING
The Kit VZIG ELISA was preformed either manually by following the described procedure 6,7,10 or automatically, by modifying it for use with the Microlab ATplus 2 and F.A.M.E. instruments as detailed in the Experimental Protocol 1:
Processing steps available for selection in the FAME method editor.
The Amplified VZIG ELISA was performed either in a semi-automated manner by following the described procedure 11 or by modifying it for use with the Microlab F.A.M.E. instrument as detailed in the Experimental Protocol 2:
Data analysis steps available for selection in the PLA data evaluation template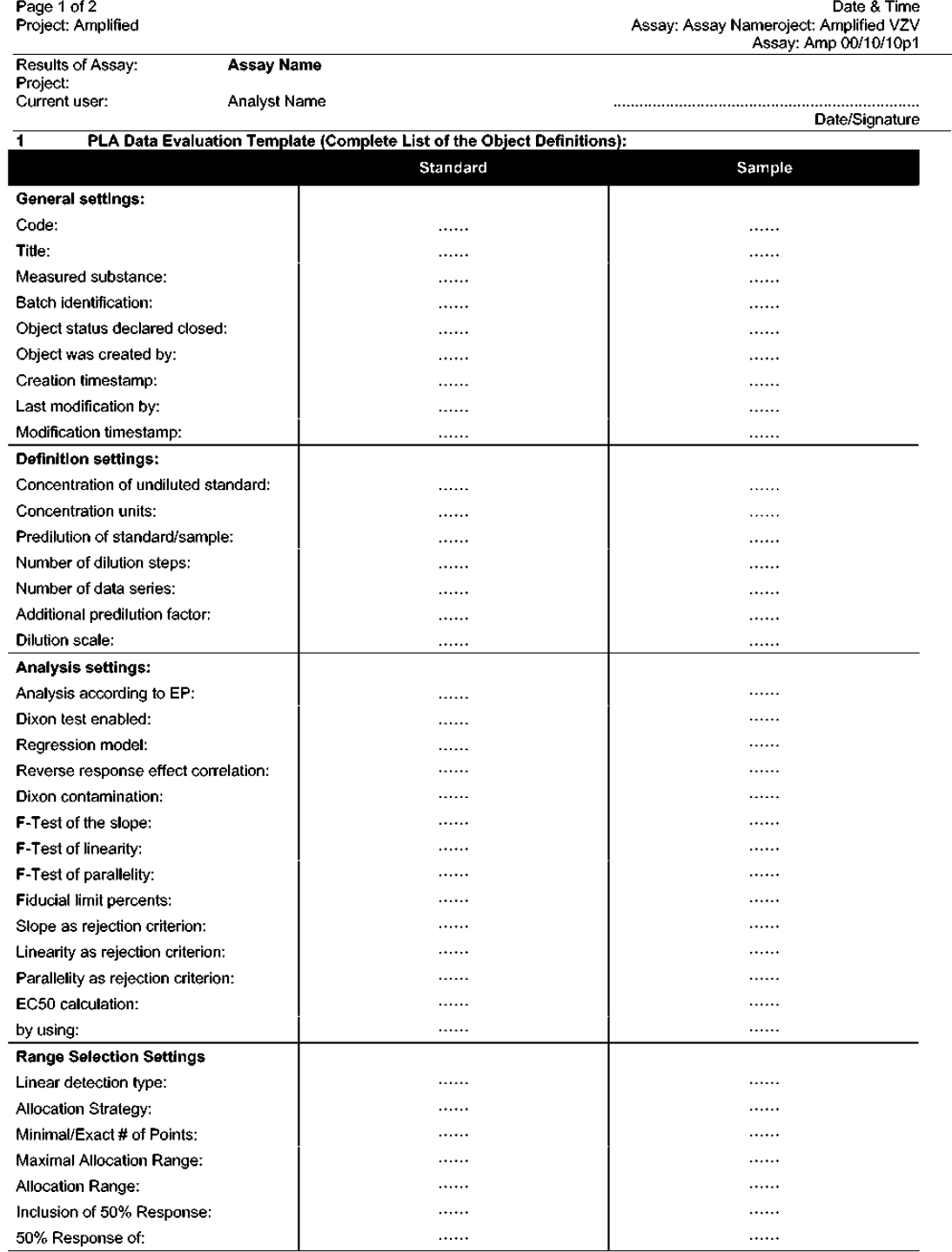
RESULTS
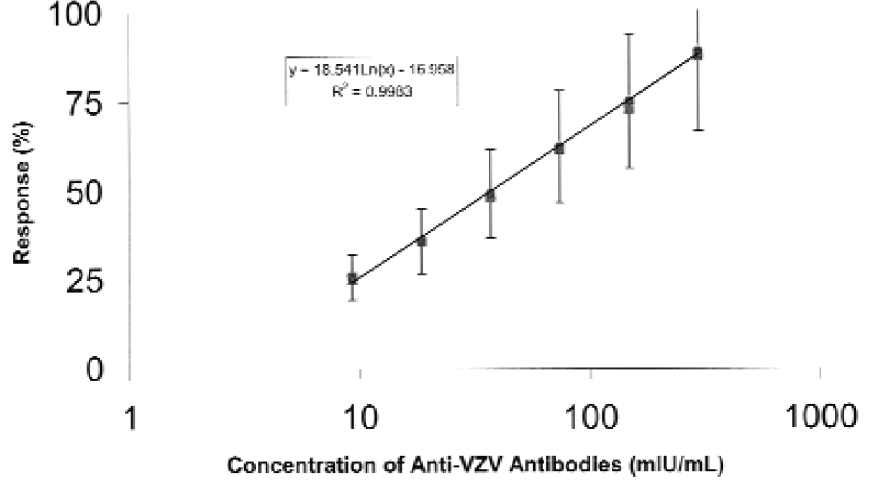
The automation of the Kit VZIG ELISA has been achieved by modifying the sample preparation, plate processing and data analysis steps of the manual assay as described in Experimental Protocol 1. Four automated ELISA experiments were performed, each assaying the reference and internal control standards as well as four quality control samples. The performance of the automated Kit VZIG ELISA has comparatively been evaluated on the basis of the cumulative standard curves generated in four currently performed ELISA experiments or in five ELISA experiments performed during validation studies. 6,7 The results obtained show that the analytical range of the automated ELISA (Figure 3) is similar to that of the manual assay (Figure 4) extending from ca. 9 to 595 mIU/mL. The results also show that the slopes and R2 values of the cumulative standard curves generated in the automated ELISA are higher than those of the curves generated in the manual assays and that there is substantially less variability in the concentration of the VZIG standard determined at each of the tested dilutions by the automated than by the manual ELISA (Figures 3 and 4). These results show that the automated Kit VZIG ELISA has the same analytical range as the manual assay and that its sensitivity, linearity and variability improved with automation.
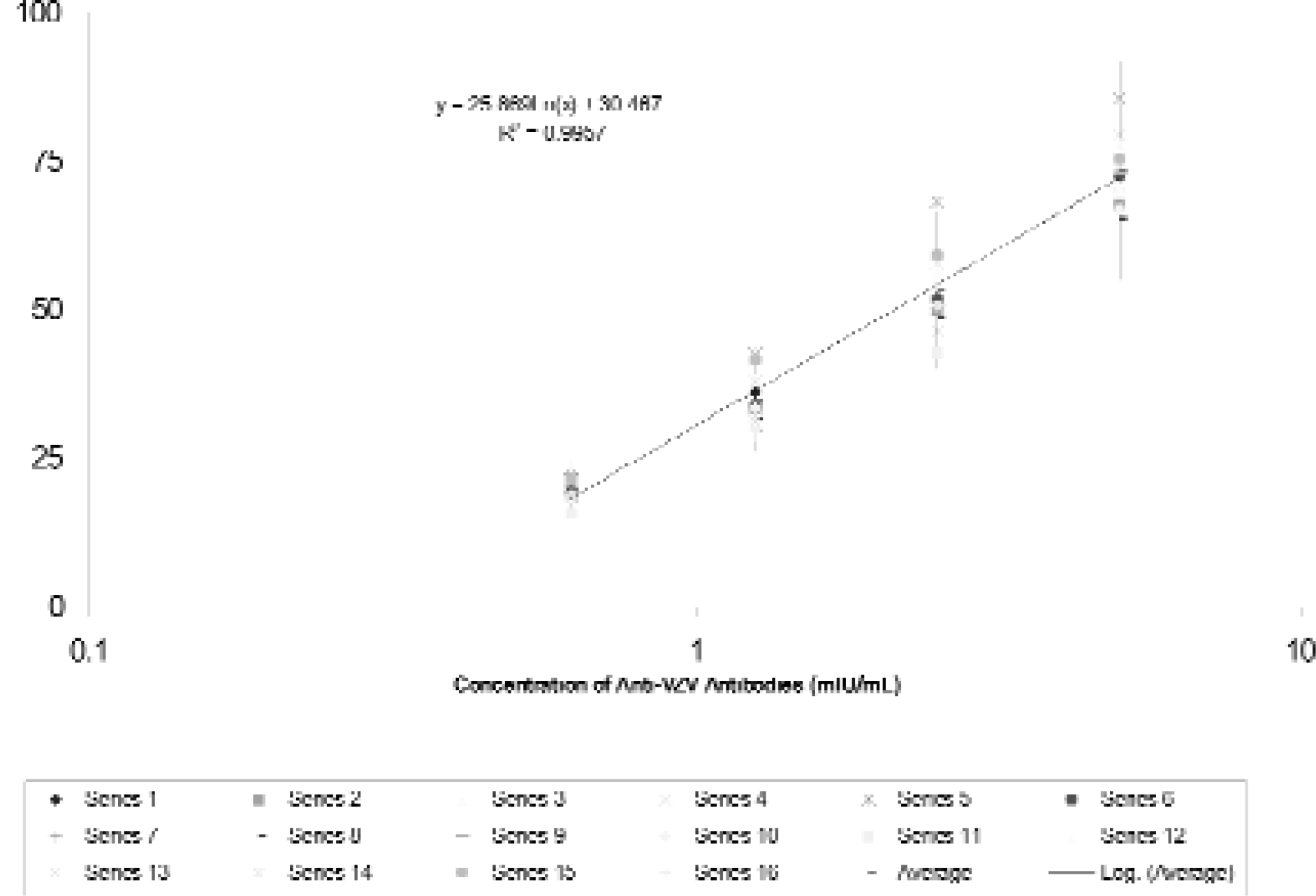
Further automation has been achieved for the partially automated Amplified VZIG ELISA 9 by modifying, as described in Experimental Protocol 2, some of the plate processing and data analysis steps. Sixteen ELISA experiments modified in this manner were performed, each assaying the reference and internal control standards as well as three test samples representing independently prepared aliquots of two patient sera. Similarly, as in case of the Kit VZIG ELISA, the performance of the automated Amplified VZIG ELISA has comparatively been evaluated on the basis of the cumulative standard curves generated in 16 currently performed ELISA experiments or in 27 ELISA experiments performed during validation studies. 8 The results obtained show that the analytical range of the current ELISA (Figure 5) is similar to that of the previous assay (Figure 6) extending from ca. 0.6 to 5 mIU/mL. The results also show that the slopes and R2 values of the cumulative standard curves of both assay types are high and similar. There is somewhat less variability in the concentration of the VZIG standard determined at each of the tested dilutions by the current further automated ELISA then by the previous, less modified assay (Figures 5 and 6). These results show that both the previous partially automated and the current further automated Amplified VZIG ELISA have very similar performance characteristics with the further automated ELISA being less variable.
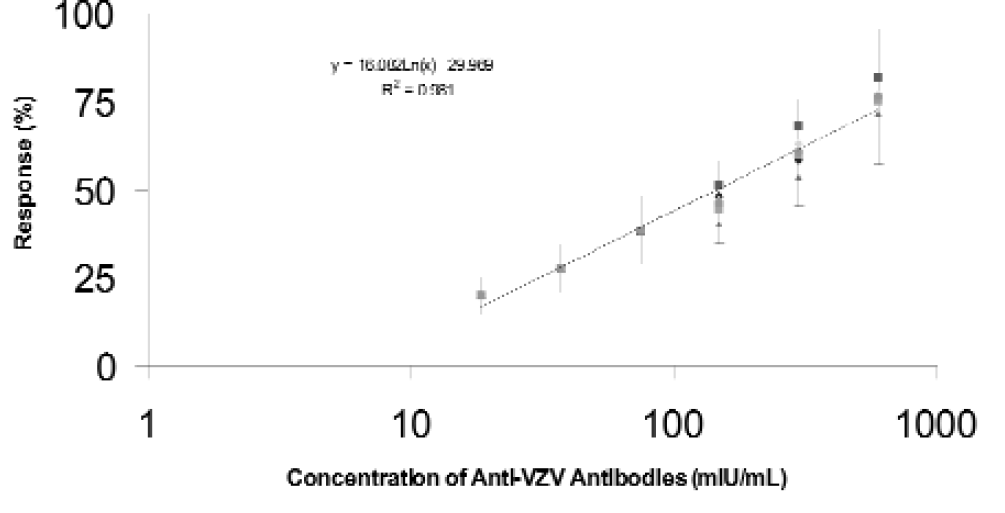
Cumulative standard curve obtained for WHO VZIG assayed by the automated amplified VZIG ELISA.
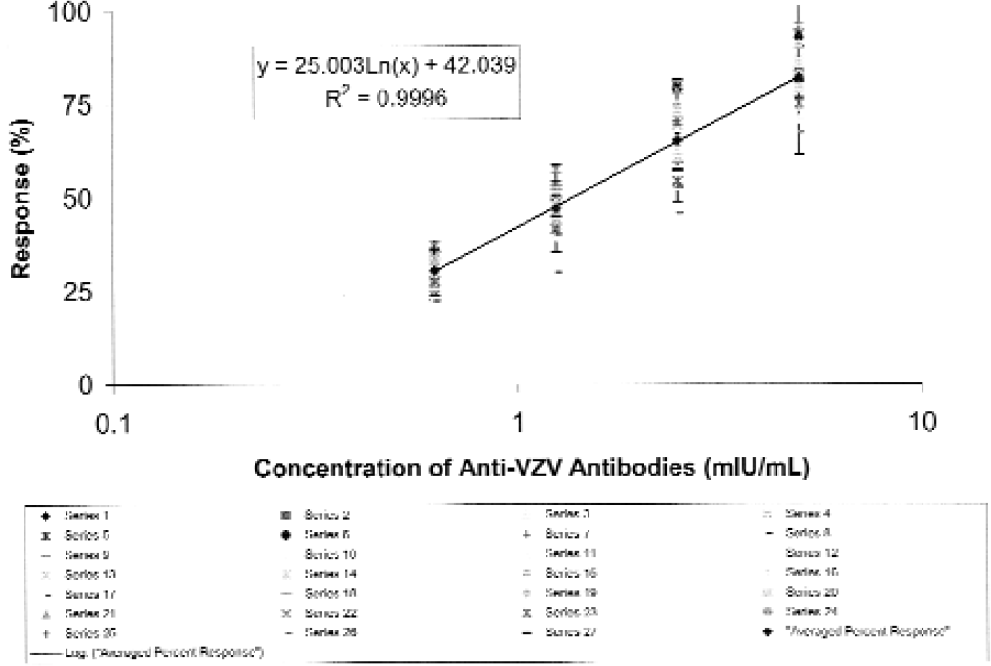
Cumulative standard curve obtained for WHO VZIG assayed by the manual amplified VZIG ELISA.
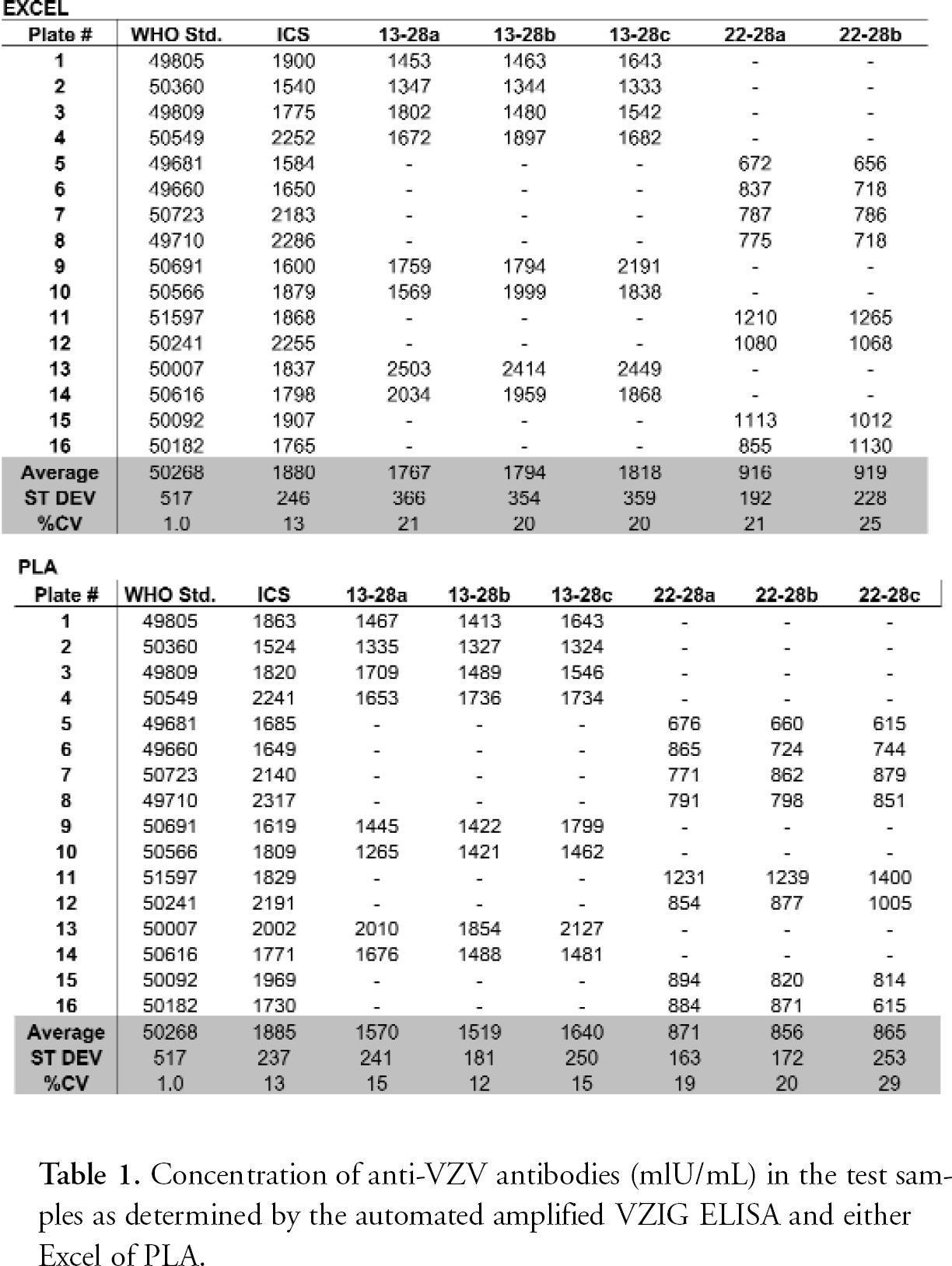
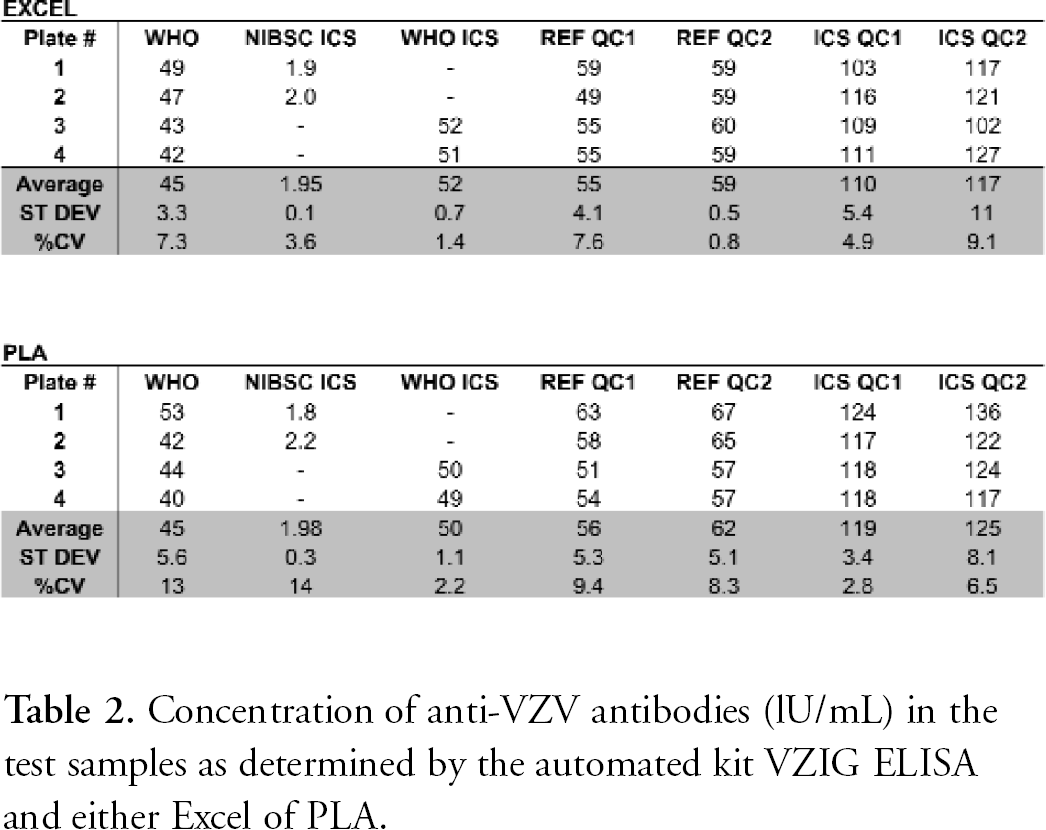
The experimental data obtained in the four Kit VZIG and 16 Amplified VZIG ELISA experiments performed during the current studies were analysed with the help of Excel and Statistica by following the standard validated procedures of data plotting and analysis. 10,11 A comparative data analysis was also done with the help of the PLA software program. As shown in Tables 1 and 2, both methods of analysis lead to similar results. This shows that the PLA program may be useful to perform, in a fully automated manner, the evaluation of the experimental data obtained by both VZIG ELISAs.
Overall the results obtained in the current studies show that the automation facilitated by a tandem of the Microlab ATplus 2 and F.A.M.E. instruments streamlined the sample preparation and plate processing of both studied ELISAs whereas the PLA program rapidly analysed and evaluated the experimental data obtained by these assays.
DISCUSSION
The ELISA methods are multi-step procedures that involve sample preparation, plate processing and data analysis. The sample preparation for the ELISA, and in particular for multiple-plate assays, may become very laborious as these methods commonly involve the testing of the analytes over a range of serial dilutions. The processing of the ELISA plates is also very laborious as it encompasses sequential application of different reagents into the plate and ultimately their thorough removal by repetitive washing. Much like other biological / immunological binding assays, the analysis of the data generated from the ELISA adds to the complexity of the method as the experimental data is often abundant and several statistical tests may be required. The automation of the ELISA may, therefore, be expected to increase its throughput and efficiency. We recently validated the performance of the Microlab ATplus 2 automated liquid handler and showed that the accuracy and precision of the automated pipetting were higher than those of the manual pipetting 9 . We also validated the BioSwift software program for an automated data capture and analysis. We showed that when both of these automation elements were implemented with a newly developed amplified VZIG ELISA 12 , they increased the analytical throughput of the assay and traceability of the experimental data as well as reduced human errors. 9
In the current studies, we advanced in our automation efforts and used the Microlab ATplus 2 automated liquid handler to similarly automate the sample preparation for the manually performed commercial Kit VZIG ELISA 6,7 , Furthermore, we automated the plate processing steps of both VZIG ELISA methods by using the Microlab F.A.M.E microtitre plate analyser. This instrument offers a rare possibility of processing the pipetted ELISA plates, according to the designed processing steps in a fully automated way, by transferring them inside the instrument into its different compartments (modules). The individual modules are dedicated for either plate incubation, reagent application / removal, or plate mixing and reading. The Kit VZIG ELISA plates were entirely processed within the F.A.M.E. whereas the unstable character of the amplified substrate and the economics of its use* called for the manual addition of the substrate, amplifier and stopping solutions. The results shown in Figures 3 to 6 document the impact of automation on the assay performance as the variability of both automated methods is smaller than that of their manual counterparts. In comparison to the automated Amplified VZIG ELISA the percent response variability of the automated Kit VZIG ELISA decreased to a much greater extent. These results are consistent with the observation that the precision of dispensing of the reagents most recently added to the plate, impacts the overall assay precision to the greatest extent. The smaller variability of the Kit VZIG ELISA may also be due to the fact that the quality of commercial Kit-based ELISAs is better controlled and assured. The amplified VZIG ELISA has been developed during Cangene's R&D studies and its performance entirely depends on the quality of the many materials and biologics. Some of them, like the Substrate Amplification System, have a long expiration period (six to 12 months) and its quality is not apparent as neither the manufacture nor the expiration date is indicated on the package of the purchased chemical. However, when comparing slopes between automated and manual assays, the amplified ELISA did not exhibit as great a variability as the kit VZIG ELISA therefore demonstrating that the performance of the amplified assay is more consistent.
In the current studies we also used the PLA software program to further streamline the data analysis of both VZIG ELISAs. The recently validated BioSwift program
9
prompted us to quest for further automation in the area of data analysis as this program was designed to automatically capture the data from the plate reader and not the F.A.M.E. instrument. Moreover, its current version allows merely for data calculation
9
but not further evaluation. The United States Pharmacopoeia
13
and European Pharmacopoeia
14
both emphasize, however, the importance of the evaluation of the parallelism between the dose response curves obtained for the reference standard and test samples when binding assays are used for measuring the biological potency of the tested substances. It is our standard approach, therefore, to use Statistica and Excel when testing the VZIG ELISA data for normality, homogeneity and parallelism
10,11
. The PLA program newly introduced at Cangene uses data automatically imported via a F.A.M.E.-specific import converter, calculates and graphs the data by using the parallel line model and statistically evaluates the linearity, slope and parallelism. In the current studies the experimental data were calculated with the help of PLA or Excel / Statistica and the results obtained were compared. We showed that the PLA program provides a powerful method to rapidly evaluate ELISA data and that the calculations obtained with the help of this program were by and large similar to the calculations obtained with the help of Excel and Statistica (
Concentration of anti-VZV antibodies (mlU/mL) in the test samples as determined by the automated amplified VZIG ELISA and either Excel of PLA.
Concentration of anti-VZV antibodies (lU/mL) in the test samples as determined by the automated kit VZIG ELISA and either Excel of PLA.
CONCLUSIONS
A tandem of the Microlab instruments, by offering a fully flexible and computerized platform, allowed a customized automation of both VZIG ELISA methods while facilitating their fully cGMP-compliant performance and documentation.
The use of the Microlab F.A.M.E. instrument allowed for processing of eight plates / day / analyst thus facilitating a throughput necessary for the clinical studies.
Once validated, the PLA program shall provide a powerful means to rapidly analyse, in a cGMP-compliant manner, the data obtained by both VZIG ELISA methods.
The automation elements implemented in the current studies for use with the Kit and Amplified VZIG ELISA improved assay and data management, reduced human errors and increased data traceability thus improving the performance and efficiency of both these methods.
ACKNOWLEDGMENTS
We would like to express our appreciation to Ms. Susan Dawson and Mr. Kirk Andree of Hamilton Company in Reno, NV, US.A. for assisting us in the implementation of the Microlab ATplus 2 and Microlab F.A.M.E. instruments for the VZIG ELISA methods. We would also like to thank Drs. Ralf Stegmann and Matthias Schmitt of Stegmann Systemberatung in Rodgau, Germany and Mr. Lutz Ankes of Hamilton Company in Bonaduz, Switzerland for their efforts on timely development of the F.A.M.E. -specific, PLA-compatible data import converter.
Footnotes
*
Two microtitre plates can be developed with single vials of a substrate and amplifier solutions whereas a small dead volume required for reagents dispensed on the F.A.M.E. calls for the use of two vials of each reagent.
