Abstract

INTRODUCTION
The rapid development in the field of microtechnology has opened up new possibilities of rapid screening protocols in pharmaceutical research and drug development. Today there is a strong need for high-throughput techniques, both for bio-interaction studies by immunoassays and for mass identification of biomolecules by the use of mass spectrometry (MS). Considering that it only takes microseconds for MS spectra generation, sample preparation from biological materials is currently the rate limiting step when running large sample numbers. The use of micromachined tools for sample handling has a number of advantages such as low manufacturing cost in batch processing, high mechanical strength and reproducible structures when fabricating well-defined and small flow channels, minute orifices, and thin membranes. In this respect monocrystalline silicon is an excellent material with mechanical properties in many aspects comparable to stainless steel. This combined with its chemical robustness and the amount of known surface modifications available, makes silicon a useful construction material for chemical microsystems. Miniaturization provides increased reaction kinetics in low volumes and the possibility to rapidly perform automated high precision sample-handling procedures at a high speed in micro/nanoliter systems. Since sample amounts in life science research fields are often very small and precious, it is desirable that every analysis consumes as little sample as possible while providing the desired information. This makes the combination of silicon micromachined analytical tools and matrix-assisted laser desorption/ionization time of flight mass spectrometry (MALDI-TOF MS) 1 ideal for rapid automated analysis of proteins and peptides.
EXPERIMENTAL
In Figure 1 the instrumental set-up of the automated on-line configured sample deposition system is depicted. The sample handling robot transfers the samples from 96 or 384-well plates by the injection needle via an injection port into the continuous carrier flow. The sample plug (typically 1 μl) will then reach the piezo-actuated flow-through microdispenser, where it is dispensed perpendicularly to the high-density nanovial arrays on the MALDI plate. The piezo-actuated flow-through microdispenser has been developed within our group and is capable of depositing single droplets with a volume of only 65 picoliter. The microdispenser was fabricated by anisotropic wet etching of monocrystalline silicon, using pn-etch stop technique as described earlier by Laurell et al 2,3 . The dispensing principle is based on a piezo-ceramic element, which elongates when a voltage pulse is applied, creating a pressure pulse that results in droplet ejection at the orifice. The unique microdispenser design features flow-through capability and a pyramid-shaped nozzle that ensures stable droplet directivity. The internal volume, from inlet to orifice of the microdispenser, was 600 nL. During operation the droplet frequency could be varied between 1–1000 Hz. Sample deposition on high density nanovial MALDI-target plates was accomplished by step-wise movement of the target plate using an in-house built computer controlled x-y stage, with 5 μm spatial resolution. Deposition of the sample was observed via a microscope (Leitz, Wetzlar, Germany) equipped with a video camera (CCD-72EX, DAGE-MTI Inc., Michigan City, IN, USA).
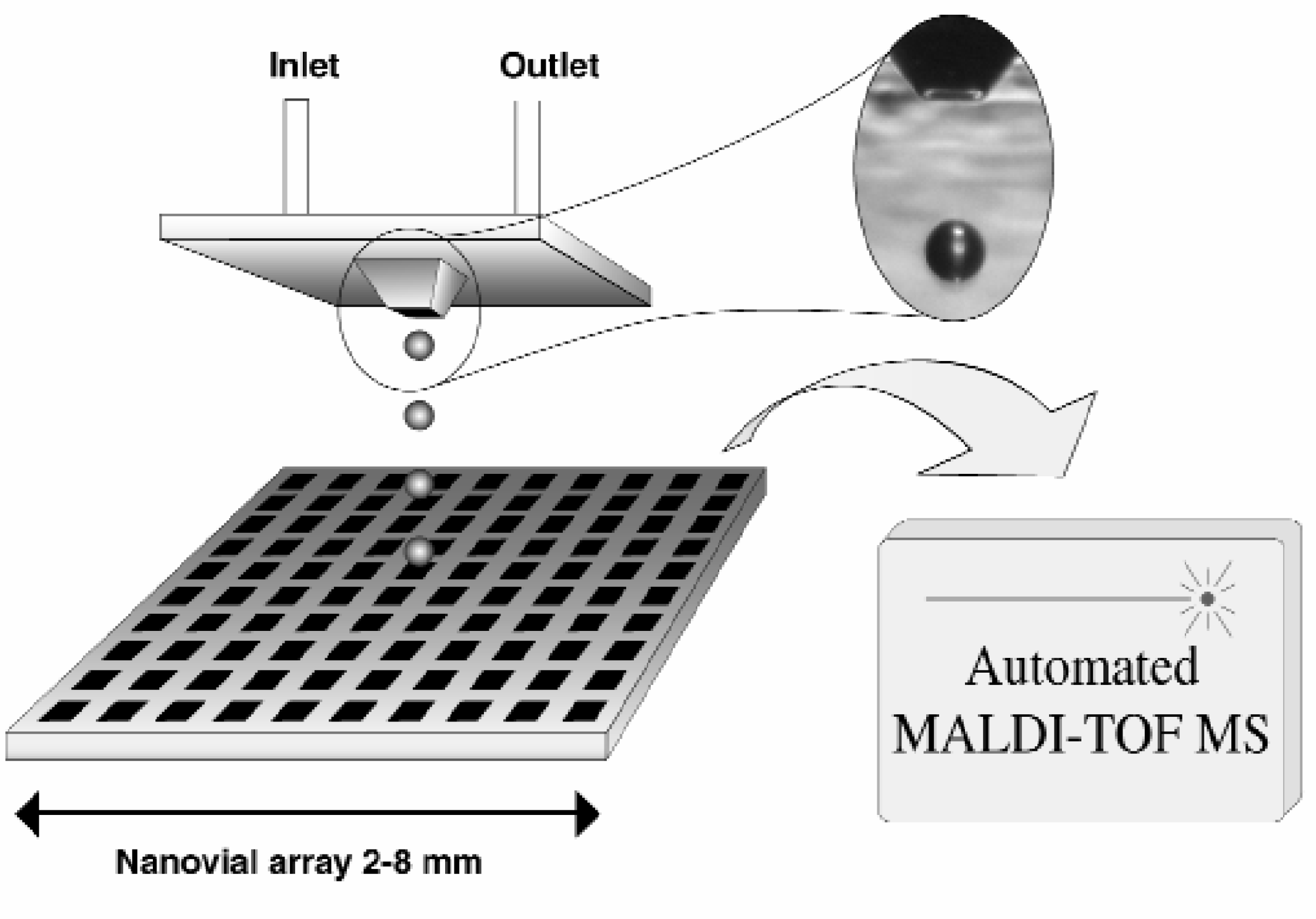
The instrumental setup used for sample deposition into the high-density nanovial arrays. The photo insert shows a 65 pL droplet in flight.
Vials ranging in size from 100×100 to 400×400 μm (inter-vial spacing of 100–400 μm respectively) were used for collection of deposited samples. Both pyramidal and shallow (20 μm deep) vials were manufactured in <100>-oriented p-type silicon. Figure 2 depicts a silicon wafer with etched nanovial arrays. Each nanovial chip had 100 sample positions in a 10×10 format and were mounted onto the MALDI-target plate by conductive epoxy or adhesive tape.

Photo of an etched silicon wafer with nanovial arrays (300×300 and 400×400 μm) arranged in 10 by 10 matrises.
The sample was either deposited directly onto a pre-deposited thin-layer matrix surface or matrix was dispensed after the sample. The pre-deposited matrix was made from CHCA 20 mg/ml in 4:1, acetone/2-propanol mixed 1:1 with nitrocellulose 10 mg/ml in 4:1, acetone/2-propanol to a final concentration CHCA/NC of 10/5 mg/ml. Alternatively the matrix, CHCA 8 mg/ml in 1:1, acetonitrile/0.5 % TFA (aq) was deposited after the sample. By heating the MALDI-target plate to a temperature of 50°C the evaporation time was shortened, thus increasing the amount of samples that could be deposited per time unit.
The MALDI-TOF instrument was a Voyager DE-RP (Perseptive Biosystems Inc., Framingham, MA, USA) mass spectrometer. The instrument, equipped with a delayed extraction ion source, uses a nitrogen laser at 337 nm and can be operated in both linear (L) and reflector (R) mode at accelerating voltages of 20–25 kV.
RESULTS AND DISCUSSION
The use of nanovials as MALDI-target plates has earlier been reported by Jespersen et al., 4 who presented picoliter volume vials etched in silicon wafers. The vials were inverse pyramids of 100×100 μm with a volume of 250 pL and low attomole levels of peptides and proteins were detected with MALDI-TOF MS. Nanovials have also been used for DNA analysis. 5 During previous work we have demonstrated that the combination of microdispensing and high-density nanovial arrays has many novel applications and advantages. 3,6 –8 The micro-vial size and geometry was found to influence the MALDI-TOF MS resolution. Experiments have revealed that the isotope resolution, when analyzing peptides, was much lower (up to a factor 10) for samples inside the deep pyramidal nanovials (>100 μm) than for samples that were deposited in shallow nanovials or on top of the silicon surface between the nanovials (Figure 3). This phenomenon originated from the spatial distribution of matrix-sample crystals along the vertical axis within the deep vials. A low mass resolution leads to a decreased mass accuracy. To avoid this, shallow planar nanovials with a vial depth of 20–50 μm should be used. The use of nanovials for sample collection ensures exact positioning of the sample spots and eliminates the need for “hot spot” searching. The MALDI laser spot diameter matches that of the nanovials, providing appropriate conditions for rapid automated MALDI-TOF MS analysis.
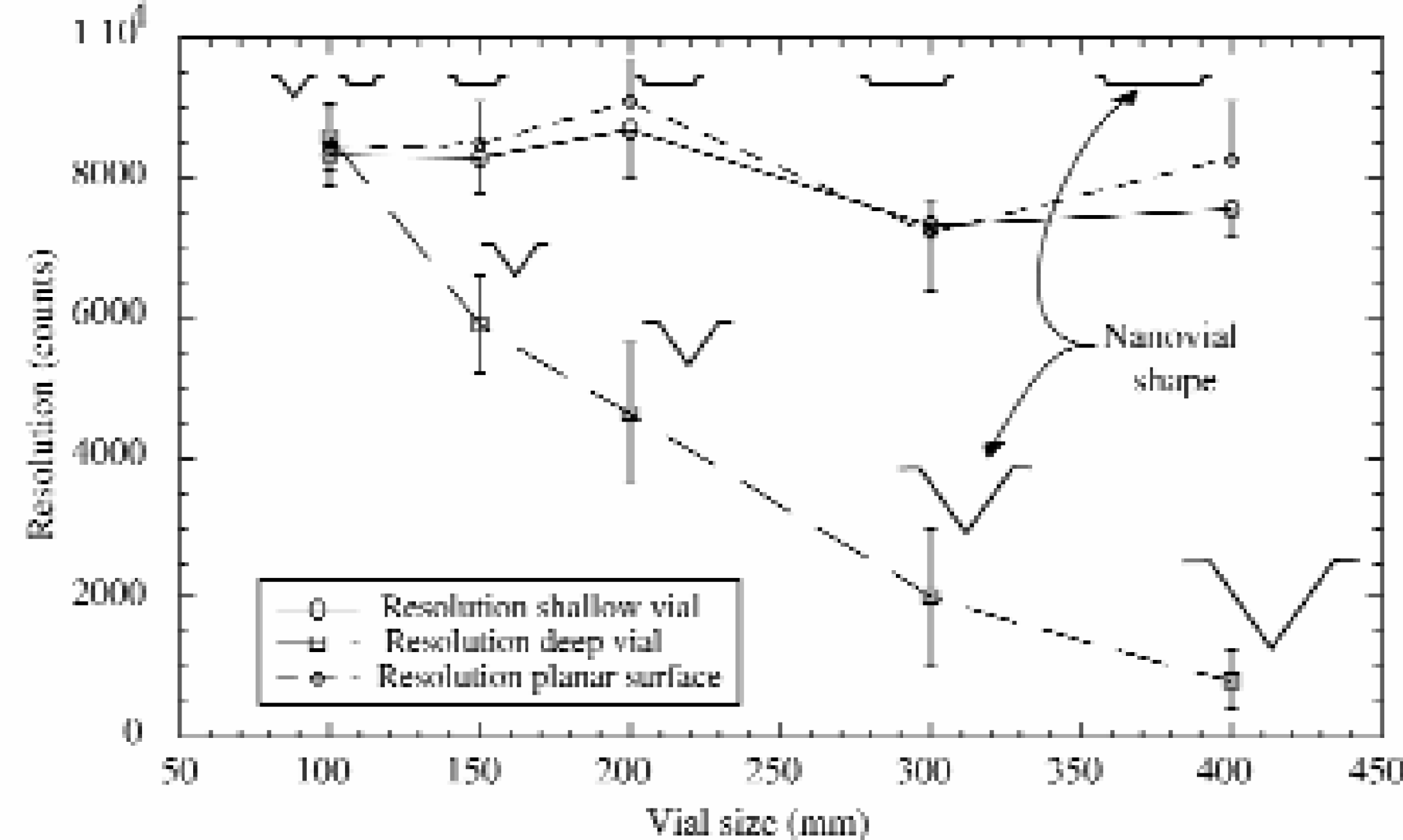
The graph shows how nanovial size and geometry affect the MALDI-TOF MS resolution. A sample solution of angiotensin I (100 fmol/μl) was used. Error bars show standard deviation, n=10. Matrix; 2,5-dihydroxybenzoic acid.
A big advantage of the microdispensing is that it can be used for “on-spot” enrichment of samples with low concentrations by making multiple sample depositions of the sample on the same position before the MALDI-TOF MS analysis (Figure 4). The enrichment allows the collection of a high analyte density in each sample position, which provides an increase in the observed MS signal intensity. This makes it possible to obtain mass spectra from sample solutions of such low concentration that ordinary dried droplet sample preparations 9 would not give usable mass spectra. The degree of this “signal amplification” is simply determined by the amount of deposited sample on the individual position.
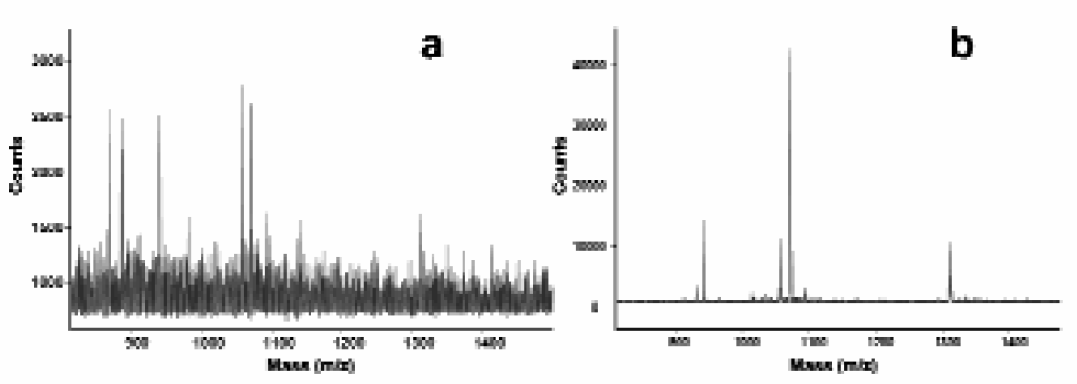
Effect of enrichment when analyzing a calibration mixture consisting of four peptides, at a concentration of 25 nM. The mass spectra a) shows an ordinary dried droplet sample preparation and mass spectra b) shows the signal amplification provided by dispensing 5000 droplets into one nanovial position.
The flow-through capability of the microdispenser also allows downstream fractionation at the flow outlet. Unidentified samples after MALDI-TOF MS or proteins of special interest can then be further analyzed by other methods, such as immunoassays or MS/MS sequencing using nanospray technology.
Deposition of replicate sample spots in 100 positions on a nanovial target can be achieved in less than 1 minute. The microdispenser can easily be used to deposit several thousand samples on each MALDI-plate and by making micromachined more “dense” nanovial MALDI-target plates it is possible to achieve several tens of thousands of sample positions on a standard MALDI target plate.
By using this automated sample preparation step lower standard deviations are obtained in comparison to manually prepared samples. In experiments we have been able to generate acceptable MS-spectra from over 90% of the sample spots by only using 8 laser pulses per spot. 7 This was more easily achieved by utilizing the micro-dispensing technology as compared to manual sample preparation. The small internal volume of the microdispenser enables efficient handling of small sample volume and this sample handling system can easily be coupled to upstream sample manipulations, such as separation techniques to provide the user with a microsystem capable of a multitude of different analytical possibilities. It can thus be concluded that the combination of microdispensing and nanovial arrays opens up the possibility to perform high throughput analysis of peptides/proteins by MALDI-TOF MS.
