Abstract
Sol-gel chemistry provides a novel production route for ceramics and composites that have a variety of applications in medicine, biology, and biochemistry. Advantages of sol-gel-derived materials in these applications include simple, low temperature production routes that are capable of achieving temperature, chemical, and radiation-inert porous materials with a wide range of structural and microstructural properties. Furthermore, sol-gel-derived materials display remarkable compatibility with biomacromolecules and are conveniently functionalized with a variety of coupling agents. The properties of these materials and their present and potential applications in medicine and biology are reviewed.
BACKGROUND
The term sol-gel indicates a low temperature production route whereby a ceramic or ceramic composite material is formed. Originally developed for the nuclear industry to aid in fuel processing, sol-gel-derived materials have found a wide range of applications including microelectronics, solar cells, intelligent coatings, 1 batteries, 2 and catalysis, as well as in clinical and analytical chemistry. The term sol refers to a colloidal suspension of solid particles that, under the proper conditions, condense with one another or with unreacted precursor to form an irregular, non-crystalline, three-dimensional network that spans the entire volume of a vessel. The resulting network can be described as a gel when a path can be traced from one boundary point of the vessel to another without leaving the solid phase, although further condensation may still be occurring.

The gel network is most commonly very porous (sometimes in excess of 99%), and is often denoted by the interstitial fluid. The fluid phase in hydrogels is thus water or aqueous solution, in alcogels it is alcoholic solutions, and in aerogels it is air or other gases. The gelation reaction can occur in any number of solvents and is commonly catalyzed by changes in pH or stoichiometry of the reactants. Unlike many organic polymer hydrogels, solvent exchange in inorganic gels produces minimal changes in the dimension of the gels. Swelling is typically only on the order of a few percent, and if it does occur, is likely to lead to breakage of the gel.
The solid backbone of the network can be made from any of a number of materials, but sol-gel processing is most common with metal oxides, and most specifically with silica, alumina, titania, and zirconia. The solid component is commonly derived from the hydrolysis/condensation of the respective alkoxy metallate (
An illustration describing a standard sol-gel process.
The physical microstructure of sol-gel-derived metal oxides is highly dependent upon the production conditions.
3
Reactant stoichiometry, temperature, catalyst, polarity of the reaction solution, and/or surfactant concentration all significantly influence the physical properties of the final gel. Although these effects are often specific to the particular reaction conditions, some general conclusions can be drawn. In general, gels formed under acidic conditions tend to be “polymeric” in nature, where condensation of unreacted alkoxy silicate occurs preferentially at the end of long chains that grow until they entangle and condense with one another. This type of gel tends to display smaller pore sizes and higher elastic moduli for a given density. On the other hand, sol-gels formed under basic conditions tend to form larger clusters that condense with one another as particles. Sol-gels formed under basic conditions tend to display larger pore sizes and lower elastic moduli. Surfactant concentration and solution polarizability can also have a significant influence upon gel microstructure, and phase separation may occur during the gelation reaction and this leads to unique microstructural features. SEM micrographs of three different large pore, phase-separated sample gels from our lab are shown in

Sample gel macrostructures obtained using the biocompatible production route described in the text. Although the densities of these samples are identical, the variable macrostructure will provide different physical and transport properties. The images show a clear transition between a polymeric (left) and a particulate (right) gel induced solely by surfactant (polyethylene glycol) concentration during condensation.
Functionalization of sol-gel-derived metal oxides is quite easy due to both the reactive surface and liquid precursor of the solid. The increased reactivity of sol-gel-derived materials removes the need for traditional activation steps, 7,8 and the liquid precursor guarantees even perfusion of a wide variety of silane coupling agents that can be introduced at various times during the sol-gel production process. Furthermore, by alternating condensation and functionalization steps, it is possible to pattern three-dimensional structures with functionalization domains. 9 The most common approach to surface functionalization involves silane coupling agents that, despite difficulty with reproducibility, are widely available and the subject of much research due to their commercial importance. 10
Liquid-filled gels have traditionally been dried either by simple evaporation or by supercritical drying of the liquid component. Evaporation is the most convenient and inexpensive drying route, but it is difficult to perform with anything other than thin films, microparticles, and small monoliths. The dry gel is called a xerogel to denote a change in gel structure. During evaporation, a capillary pressure develops within the pores of the gel and draws the pore walls together. On the microscale, this collapses the pores and reduces the mean pore diameter and bulk porosity. On the macroscale, gradients in capillary pressure are often strong enough to fracture the gel and large macroscopic monolithic pieces are difficult to obtain. Even though the dry volume of xerogels is commonly between 1/2 to 1/30th the original volume of the gel, xerogels often retain porosities in excess of 30% and they retain much of the surface reactivity of the undried gels.
Supercritical drying, on the other hand, essentially preserves the structure of the initial liquid-filled gel and is capable of producing large monolithic pieces. By taking the interstitial liquid supercritical, the capillary pressure is brought to zero and the gel micro- and macro-structure are preserved during removal of the supercritical fluid. The resulting material is called an aerogel and is one of the most extraordinary room temperature solids known to humanity. Aerogels have the lowest density, lowest thermal conductivity, lowest dielectric constant, lowest acoustic velocity, and lowest refractive index of any known solid. Although the primary commercial application of aerogels is thermal insulation in double pane windows, they have also been proposed in various microelectronic, acoustic, and personal care product 11 applications.
CURRENT APPLICATIONS
COLUMN CHROMATOGRAPHY STATIONARY PHASES:
In the past, the most widespread application of silicon chemistry to medicine and biology was in chromatography column stationary phases for clinical chemistry and bioanalytical chemistry. Despite stability over a limited pH range, modified silica particles and gels have been used as stationary phases in a wide range of chromatographic applications. 12 Recently, the sol-gel production route has begun to find novel applications in analytical chromatography. 13 - 15 Furthermore, the importance of the sol-gel production route may dramatically increase with the further development of microanalytical systems.
One of the earlier and more successful examples of a sol-gel-derived stationary phase is the Zorbax line originally developed at DuPont for size exclusion chromatography. 16 Zorbax is the trade name for a series of sol-gel-derived microspheres that are formed by causing the gelation reaction to occur in a “mold” consisting of reverse phase (water in oil) micelles. The geometry and size of the gel particles are determined by the microemulsion, and gel microstructure and surface functionality by the stoichiometry of the reactants. It should be noted that the commercially available Zorbax particles are commonly sintered prior to sale and thus do not display the high reactivity of the native gel without reactivation. 8
A more recent example of a sol-gel derived column stationary phase has been described by Guo and Colon for open tubular liquid chromatography. 13,14 Many capillary columns are functionalized (fixed) when they are solids, often with discouraging results. These chemically bonded stationary phases provide low phase ratios, poor pH stability, and the results are often unreliable. However, functionalization directly in the sol during gelation yields stationary phases with higher capacities and good pH stability. It has been postulated that the direct incorporation of the modification into the gel-derived silica is responsible for the increased performance and control of the reaction conditions.
In addition to such particulate chromatography stationary phases, a number of researchers have examined monolithic stationary phases for various HPLC and gas chromatography applications. Current research includes examining the effects of various surface modifications, developing larger pore diameter materials, 17 and developing hydrolysis-resistant materials other than silica for chromatographic applications. 18,19
CHEMICAL SENSORS:
Sol-gel-derived materials have recently spurred much interest in both medical and environmental chemical sensing. Sol-gel-derived silica is commonly used to entrap and/or covalently bind macromolecules and small molecule dyes within a highly porous solid network. An extensive body of literature has recently been reviewed. 20,21 The sol-gel matrix serves the dual purpose of confining large quantities of these molecules within a three-dimensional matrix yet allowing easy communication with the external environment by diffusion through the pores. Furthermore, the reactivity of biological macromolecules such as enzymes within sol-gel matrices has been shown to be largely preserved, and a wide range of such molecules have been incorporated into sol-gel-derived glasses.
One example is the entrapment of the proteins cytochrome c, copper-zinc superoxide dismutase, and myoglobin by Ellerby et al. and Yamanaka et al. 22,23 Simple admixing of the proteins in a prehydrolyzed sol followed by gelation resulted in a solid, three-dimensional, protein-containing matrix in which the reactivity and spectral characteristics of the proteins are only slightly changed. Similar approaches have been used for a variety of biopolymers, including redox proteins 24,25 and immunoglobulins. 26 Both monolithic and optical waveguide 27 devices have been produced using these approaches.
PERMSELECTIVE AND ION-EXCHANGE MEMBRANES:
Both permselective and ion-exchange membranes have been formed using sol-gel derived materials. The applications of these materials in medicine and biology include ion exchange during pharmaceutical production, ion selective electrodes for clinical diagnostics, and water treatment and desalinization. Functionalization of the metal oxide backbone with charged or reactive organic modifiers is required to achieve the requisite properties. 28,29
FUTURE APPLICATIONS
MICROANALYTICAL SYSTEMS:
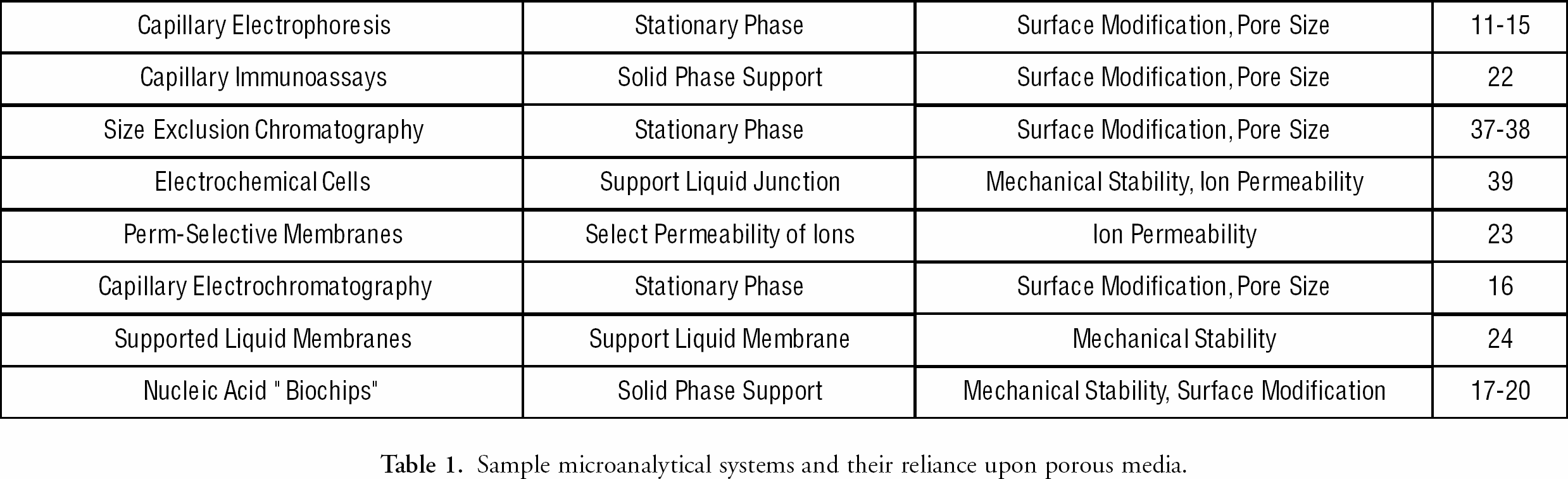
The relative importance of sol-gel-derived materials in analytical systems appears to be increasing with the development of micro-analysis, as sol-gel processing provides a convenient route for the production of porous media in a variety of applications. Microanalytical devices exploit microfabrication techniques from the semiconductor industry in order to create miniature analytical and bioanalytical devices for point-of-care medical diagnosis and environmental chemical analysis. The small sizes of these devices yield equipment that is inexpensive, fast, disposable, portable, and massively parallel, but at the same time raise novel production and handling issues for their components. For example, microscale analytical systems exhibit the same reliance upon porous media as their macroscale counterparts, and yet traditional, well-characterized porous materials and methods are incompatible with many microfabricated devices. This is due to handling difficulties, pH or temperature instability, the need for successive loading and/or washing steps, contamination by further microfabrication processing, or the need to access these materials with UV light. Current microscale analytical systems where porous media are of critical importance are outlined in
Sample microanalytical systems and their reliance upon porous media.
Sol-gel-derived silica possesses several material properties that make it a very attractive candidate for microanalytical systems, including covalent bonding with common microelectronic materials such as silica, temperature stability up to ∼700°C, an easy to handle liquid precursor state, a well-developed surface modification chemistry, and a microstructure that is largely tunable with production conditions. The gelation reaction is also particularly well-suited for incorporation into microanalytical devices. Gelation occurs at room temperature, which prevents damage to other thermally sensitive structures. Furthermore, gelation can be catalyzed by relatively mild changes in pH or water content and, under selected conditions, the catalysts need not be added in situ. The reaction conditions can be chosen such that gelation occurs in times ranging from seconds to weeks. Thus, a single liquid phase could be loaded into the device and used following a spontaneous gelation, without further rinsing, washing, addition of catalyst, or steps to promote adhesion to the microanalytical substrate.
A direct comparison of an organic polymer such as polyacrylamide and silica compatibility with microanalytical systems can be made based upon micro-gel electrophoresis systems. 30,31 In order to obtain sufficient adhesion of polyacrylamide to the capillary walls, a primer step was required. A low molecular weight polyacrylamide was bound to an alkoxy silicate and injected into the device for covalent immobilization. Only thereafter could the polyacrylamide precursor solution be injected. Furthermore, the catalyst for the polymerization (gelation), reaction had to be drawn into the device and diffusion mixed into the volume of interest. Diffusion is a relatively slow process, and can often result in large heterogeneity in the final gel. These multiple reactions and handling issues can be avoided by working with sol-gel-derived silica materials that provide covalent bonding of a ready-to-use porous medium in a single step.
CELL IMMOBILIZATION MATRICES:
The immobilization of cells within solid, three-dimensional matrices has been proposed for a number of cell-based applications including sensor systems, bioreactor supports, and tissue engineering. Sol-gel derived materials are likely candidates for these systems due to their high biocompatibility, large surface area, and open pore structure. Cells most likely interact with the matrix surface through hydrogen bonding and charge-charge interactions with ionized surface silanol groups.
In the implant community, the systemic and cyto-compatibility of melt alumina and silica are renown. 32,33 Indeed, the bio-compatibility of many silica-based glasses is so high that they are described as “bioactive.” 34,35 The host microenvironment inside bioactive glasses is so favorable that cell and fiber penetration occurs spontaneously after implantation, from both soft and hard tissue. 36 As mentioned earlier, sol-gel-derived silica and alumina are not identical to their melt cousins. However, melt silica and alumina are often “activated” in vivo through exposure to high pH conditions and yet retain their biocompatibility. It is thus strongly suspected that sol-gel-derived silica and alumina will provide similar cytocompatibility.
Traditional sol-gel methods involve extremes of pH and high concentrations of alcohol, both of which can destroy biological molecules. However, preparation of silica sols can be accomplished without the addition of alcohol at the start of the reaction. In addition, reaction conditions can be adjusted with a buffer to a biologically compatible pH, after the acid catalyzed hydrolysis of alkoxy metallate precursor and before the biological molecule is added. Although this work is still in its infancy, several groups have investigated cell and cell extract immobilization in sol-gel matrices.
Extracts of Thiobacillus thioxidans were doped into a sol-gel and the reduction of H2S was tested for and observed, indicating that the biological activity of the enzymes remained intact. 37 This group also trapped extracts of Pseudomonas stutzeri and Acinetobacter johnsonii, important organisms in anaerobic denitrification, in a sol-gel-derived monolith. 21 Enzymatic activity was monitored by following the conversion of nitrate to nitrite, albeit at somewhat lower levels than controls. In another study, Inama et al., 38 investigated the trapping of Saccharomyces cerevisiae in thin layers of silica sol-gel on glass surfaces. The authors showed that the trapped yeast cell survived and remained enzymatically active for at least 15 months. Braun et al., 39 entrapped whole E. coli cells in a sol-gel and observed that the cells are trapped as aggregates, the size of which increased with cell concentration. In this study however, cell viability was not reported.
In our laboratory, we added genetically engineered E. coli to a macroporous sol-gel-derived matrix
40
and subjected them to aerosols of bacteriophage virus containing the gene required to activate Green Fluorescent Protein in the entrapped bacteria (
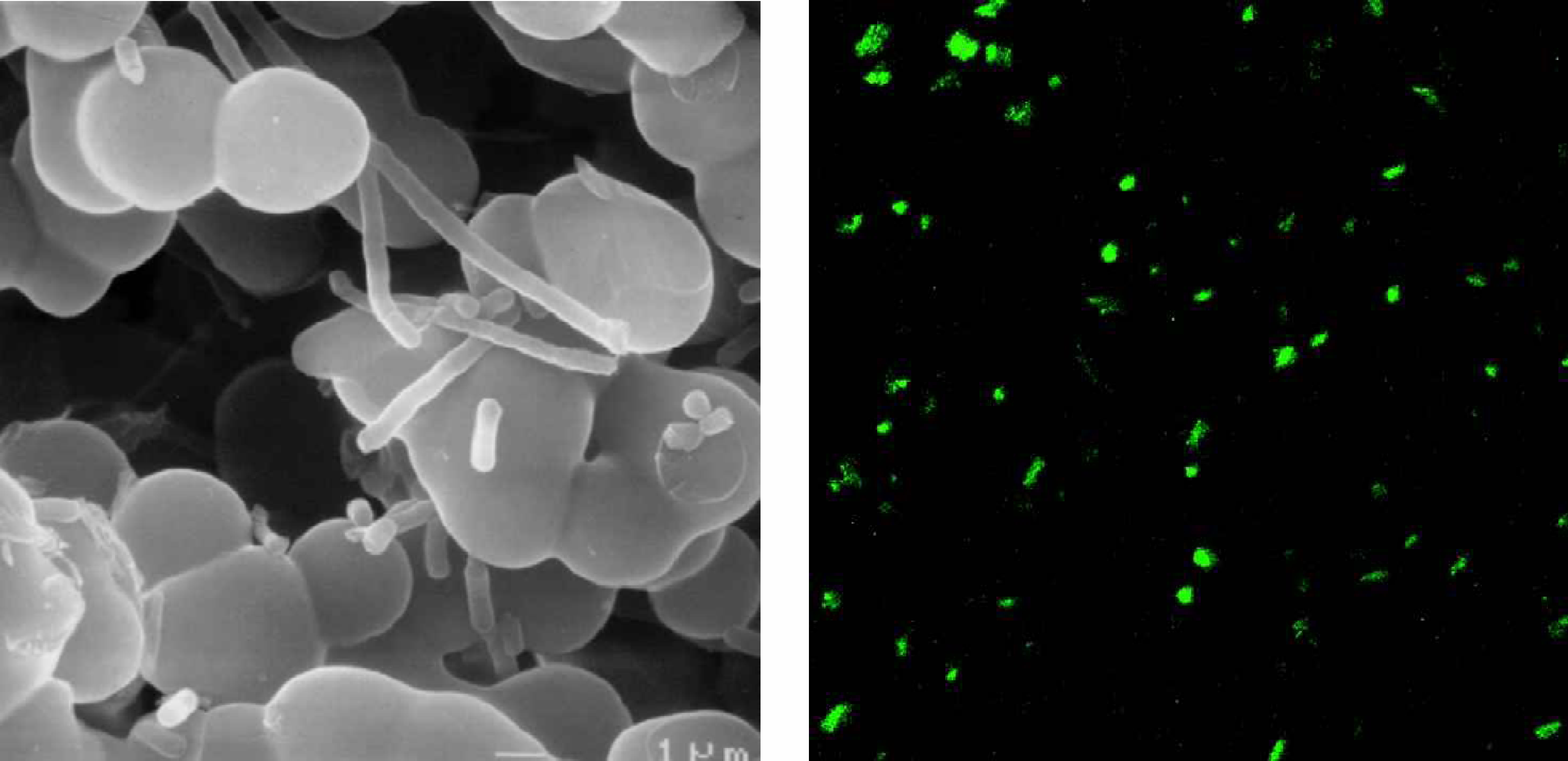
(A) SEM micrograph of E. coli (pET-gfp) adherent to and growing within the matrix of EBO8 aerogel. (B) SCLM micrograph of E. coli expressing GFP after contact with aerosolized bacteriophage λ.
ACOUSTIC APPLICATIONS
Supercritically-dried aerogels have been proposed for several applications such as impedance matching layers for air-coupled medical ultrasound imaging. 42 - 48 Since the acoustic impedance of aerogel is essentially tunable over the range between the acoustic impedance of air and many solid materials, it may provide a tailor-made material for maximizing acoustic transmission between different media. Such tuning has been done primarily through manipulation of the production reaction conditions, and a wide range of sol-gel densities and acoustic velocities have been achieved.
In our lab, we have conducted fundamental studies of acoustic propagation in the lightest of sol-gel-derived materials, namely aerogel.
49,50
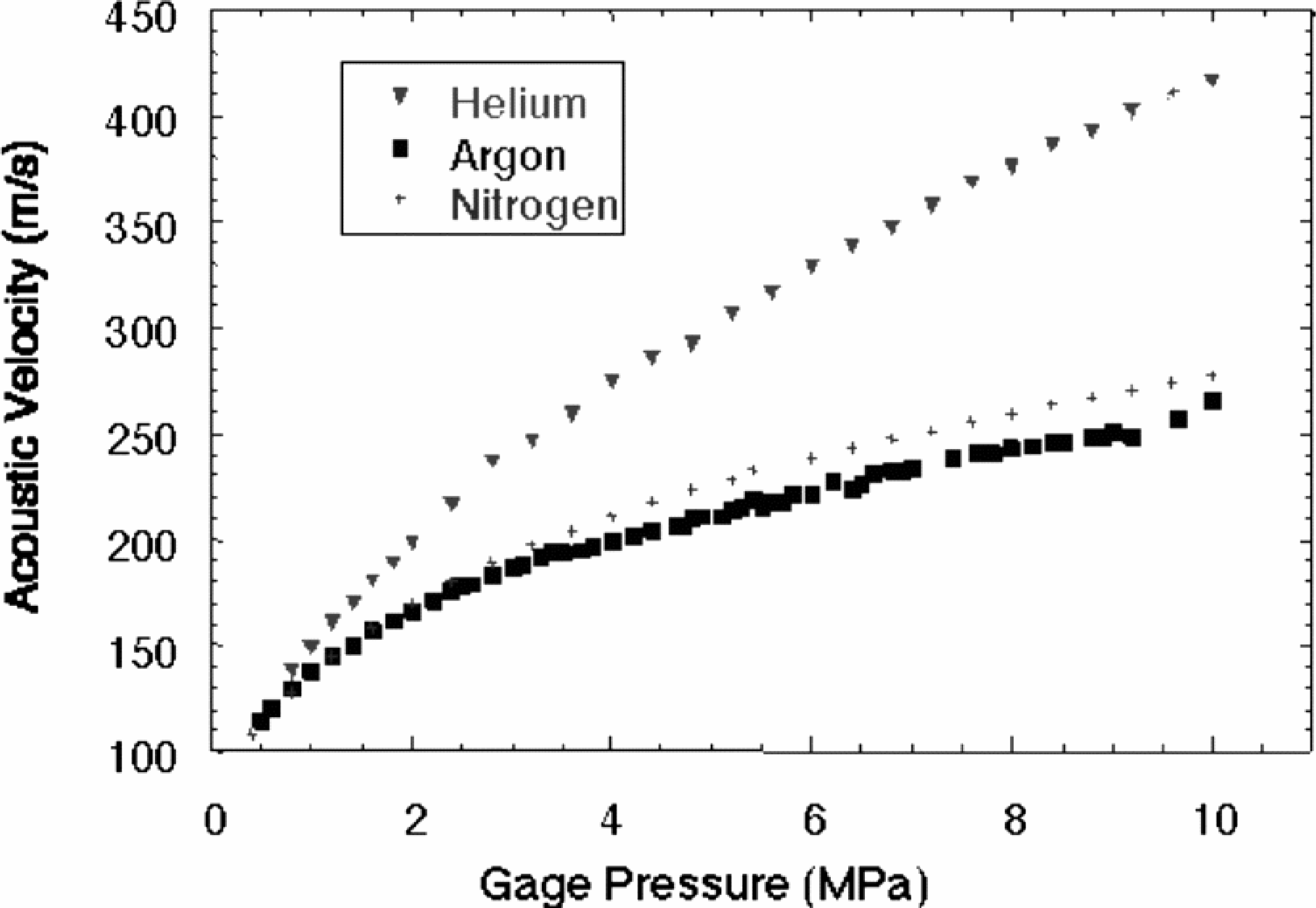
With a very low acoustic impedance, aerogel is easier to probe using air-coupled (as opposed to contact mode) transducers. Furthermore, given the extremely low density of common aerogels, the interstitial gas exerts a significant influence upon the bulk acoustic properties of the medium (
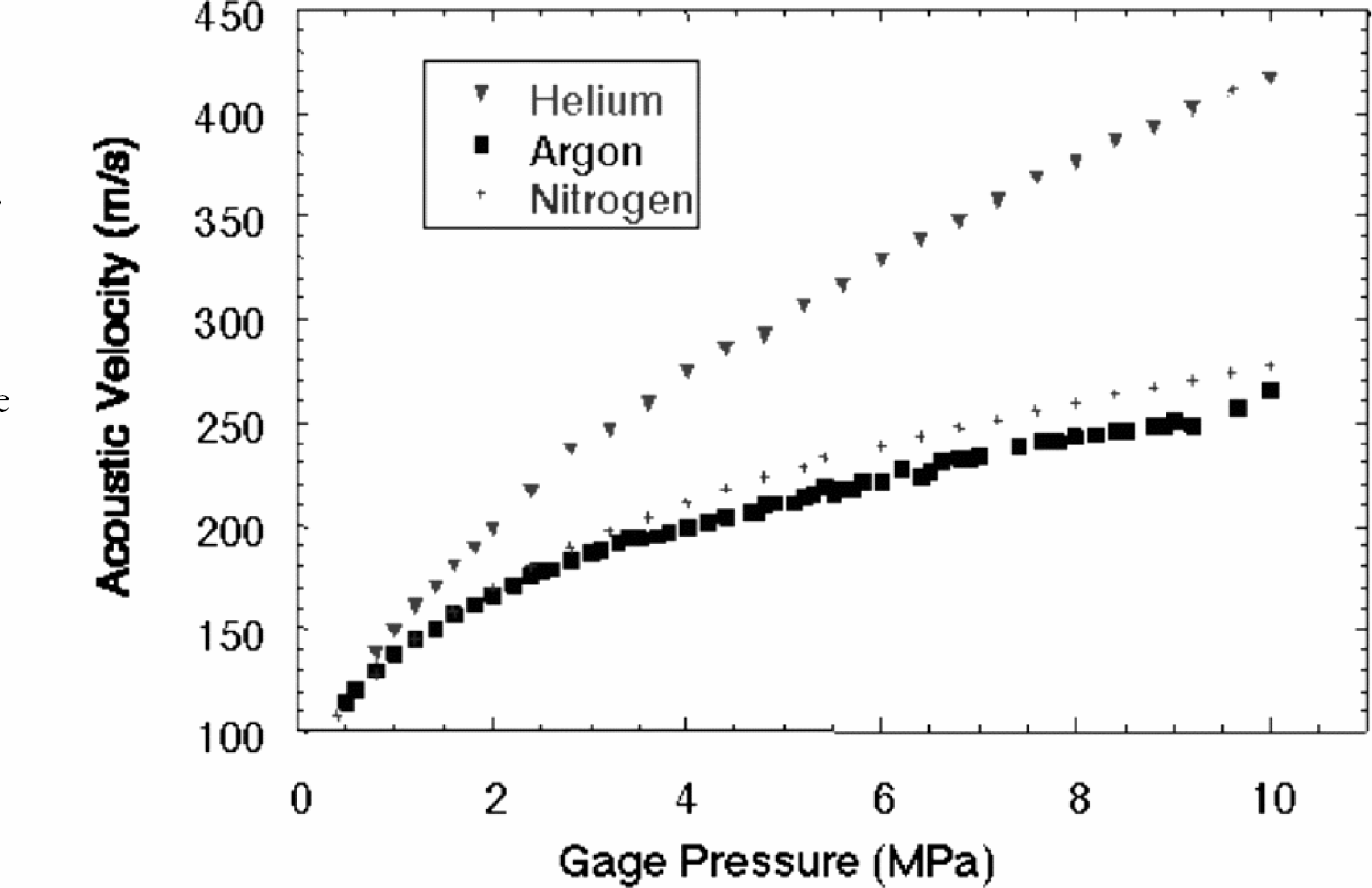
(Below) Acoustic velocity through an aerogel as a function of interstitial gas pressure. The acoustic velocity increases several fold with increased pressure, in contrast to ideal gasses where acoustic velocity is independent of pressure. The aerogel, in effect, has a heightened sensitivity to pressure changes in the molecular gas.
CONCLUSION
The sol-gel production route is a convenient method for the production of ceramics and ceramic composites for medicine and biology. The utility of sol-gel-derived materials is based upon their low temperature production route, tunable porosity and microstructure, ease of surface functionalization, and liquid precursor state that allows even perfusion of dopant molecules and the formation of complex shapes in situ by gelation molding. The high porosity of hydrogels is critical to wet applications like biopolymer immobilization and column chromatography, where a liquid-like environment is desirable. Likewise, in dry applications like acoustics, the high porosity of aerogels leads to an unexpectedly low acoustic velocity and several unique acoustic properties. Through a better understanding of the fundamental physical properties of sol-gel-derived materials, the engineering applications of these materials will increase and improved instrumentation and analytical techniques can be developed.
