Abstract


A review of any period in the history of medicine will present a picture of considerable achievement and an immense wealth of knowledge in relation to the pathology of disease, its diagnosis and its treatment. However, whilst in many ways there has been immense scientific achievement, the linkage between science and health is weak(1). The linkage becomes critically important in the face of an ever increasing pace of change both in terms of new challenges in disease as well as successes in diagnosis and treatment; thus the innovation must be harnessed and the change managed effectively. This need to develop the linkage and harness the innovation is essential as the financial pressures on healthcare delivery become greater mean, that an economic culture must also be integrated into our thinking and planning.
DEVELOPMENT OF LABORATORY MEDICINE
The earliest diagnostic tests were based on the use of the primary senses — namely sight, learning, smell and taste — including the analysis of body fluids. There then followed the use of simple chemical reactions as in the case of early methods for the measurement of reducing substances, in parallel with morphometric techniques. The early tests were undertaken at the bedside and then moved to a ward side room as chemicals, bunsen burners and microscopes began to be used.
Many of the early tests were developed in the academic or teaching hospitals and the reagents were produced locally (in house). There then followed the creation of the pathologist as a specialist medical professional, together with the inception of the multidisciplinary laboratory. As the workload grew greater specialisation ensued and automation was introduced into those areas where there was the greatest increase in work and where the methodology was amenable to automation. Out of this the specialist laboratory was born.
Whilst the diagnostics industry was born out of the need for specific instrumentation the development of the commercial reagent sector was inevitable with the advent of automated instrumentation. At the outset this development might have been regarded as a necessary piece of outsourcing combined with perceived economies of scale. However, in more recent times the innovation embodied in many of the modern diagnostic reagents places them outside of the realm of the routine laboratory both in terms of development, manufacture and quality assurance. Examples of this level of innovation include the selection of novel enzymes as reagents(2), the engineering of antibodies(3) and the creation of antibody mimics(4).
The transition of intellectual contribution to the analytical process is now complete with the advent of instrumentation that is designed for use with customised reagents, with no opportunity to “modify the chemistry” or employ a “homebrew” reagent. The integration of reagent and instrument are one of the ways that the manufacturers can control the performance of the system. This is exemplified in the development of point of care testing devices; early examples were prone to operator errors, in part because the operators did not always have the technical skills(5). The point of care testing device is designed as an integrated unit that can be operated by a technically lay individual(6).
It should be recognised that the backcloth to this pattern of development in laboratory practice is a continuous evolutionary cycle that includes the recognition of clinical need, research and development, the creation of new knowledge (eg what test is needed and what the results mean) and new technology (eg how to deliver the test) and the application of that knowledge (Figure 1).
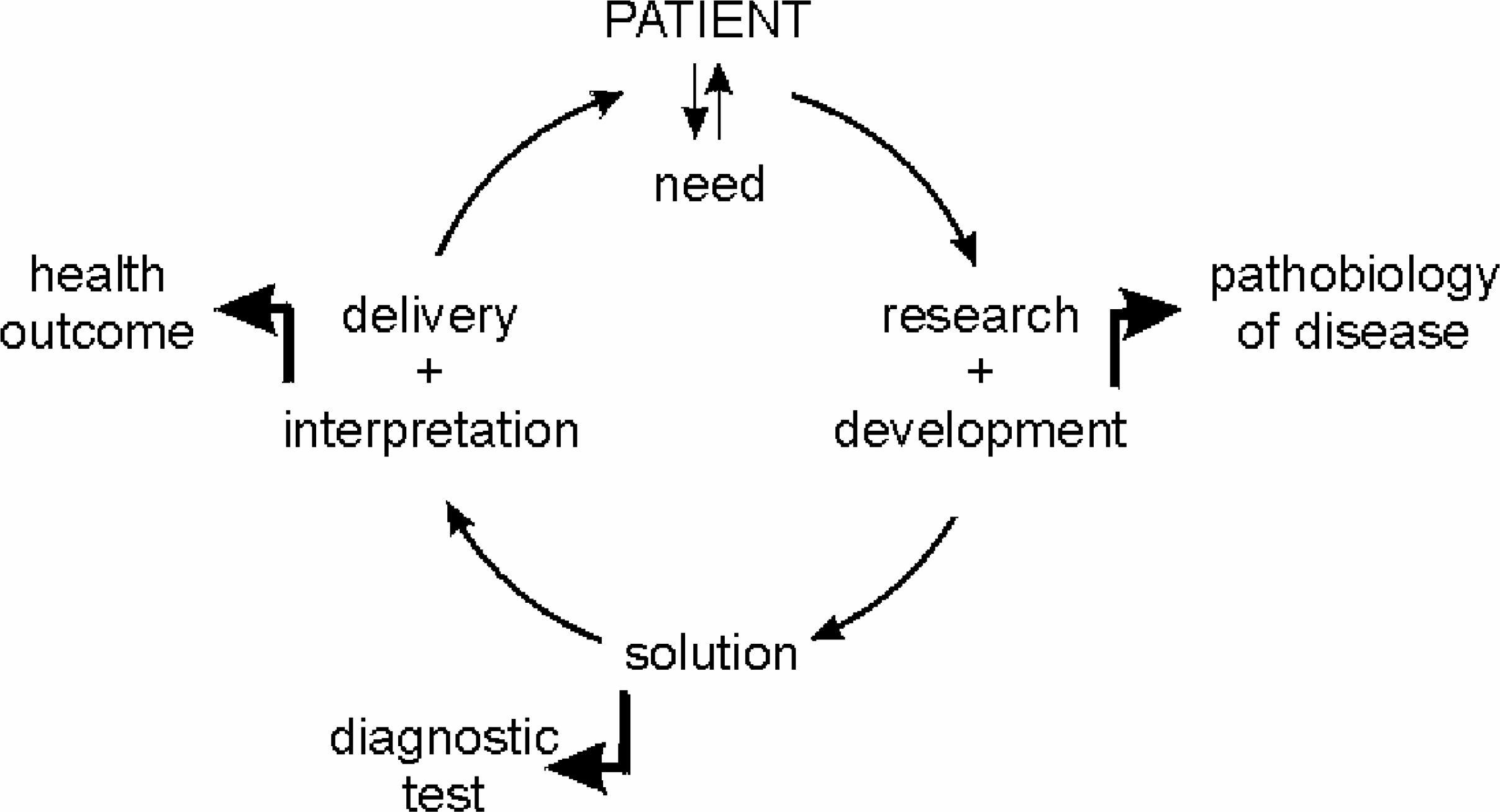
The evolutionary cycle in development of a new diagnostic test
CHANGES IN HEALTHCARE
There continue to be pressures on healthcare systems throughout; some of the major pressures are identified in table 1. One of the major changes is the increasing age of the population — perversely in the main as a consequence of the success of healthcare systems. This shift in age profile is bringing with it a change in the pattern of disease, with an increase in diseases associated with advancing years, eg osteoporosis, dementia. Life style changes have resulted in the increasing incidence of obesity, in a greater incidence of sexual transmitted diseases as well as the consequences of drug abuse.
Major Pressures on Modern Healthcare Systems

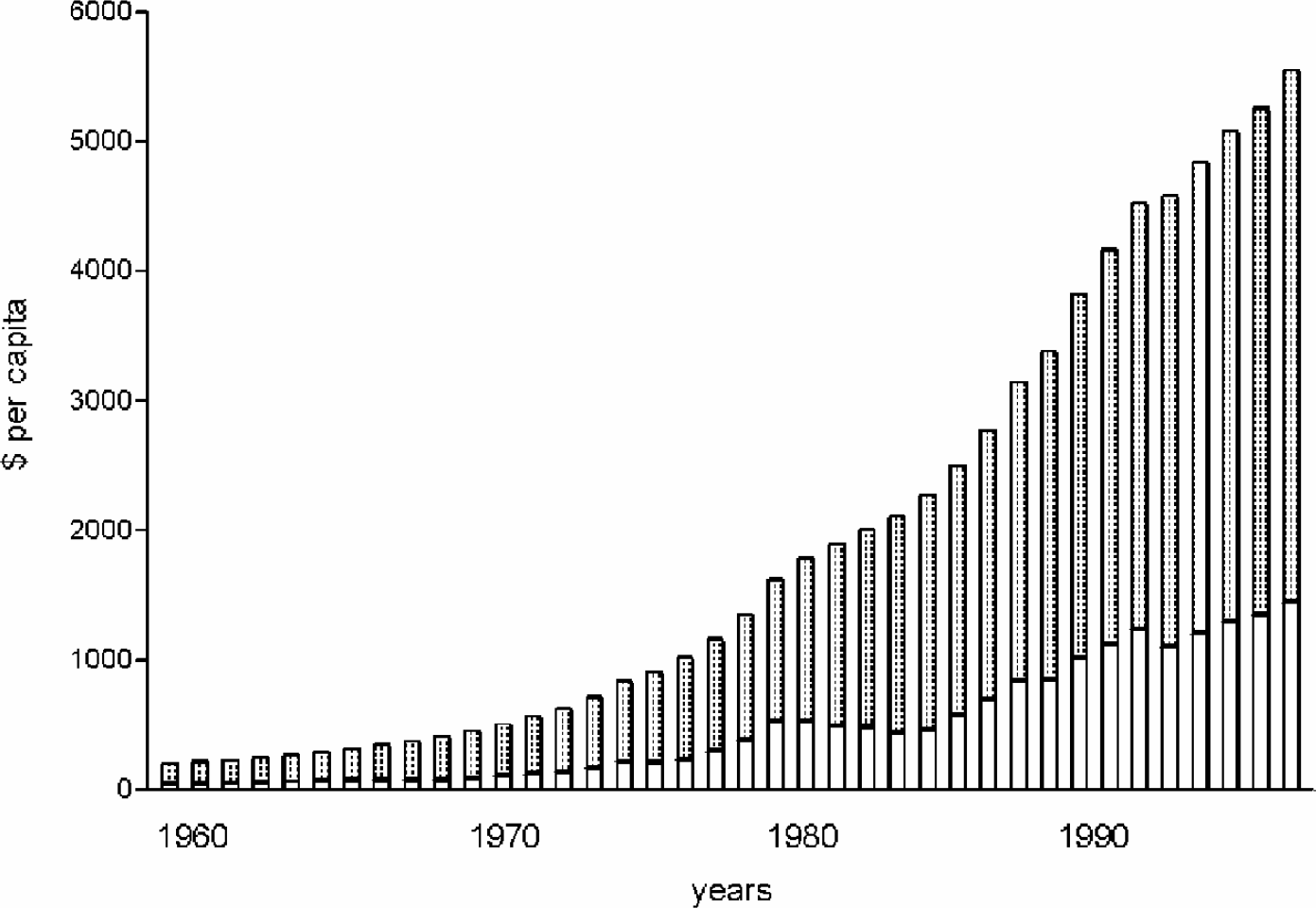
In many countries the highest profile is given to the economic pressures which result from the increasing burden of disease, the increasing cost of delivery and the enhanced expectation of the population. The per capita spend in the United Kingdom and the United States is illustrative of the trend in many developed countries of the world (Figure 2)(7).
The response to the financial pressures are very varied in their application and apparent success, but focus primarily on reducing the cost of hospital stay which is seen as the most expensive element. Thus there are initiatives to reduce length of stays(8), to devolve care back to the primary care physician(9) as well as to encourage the patient to use helplines rather than visit the primary care physician(10).
In the background to the current pressures and changes in healthcare provision is the huge amount of work being undertaken under the broad umbrella of the human genome project. It is sometimes difficult to comprehend the full impact of the project but trends are already being identified with opportunities to identify individuals at risk of developing a disease and the amenability of individuals to particular drug therapies offer major possibilities.
The genome project however illustrates one of the key issues for health services today and in the future — integration of information. It is said that the challenges for healthcare today and in the immediate future are a combination of a biological revolution as we learn more about the working of the human body and of disease, a technological revolution and solutions to problems are found and an information revolution — assimilating and utilising the knowledge that we gain(11).
RESEARCH AND DEVELOPMENT IN THE HEALTH SERVICE
In the light of this evolution and the transition of the intellectual base in analytical technology to the diagnostics industry, it is therefore important to determine what role there is for research and development in the health service provider sector.
Despite the many scientific achievements in medicine there remain many unmet needs both in terms of our understanding of the pathophysiology of disease, its recognition, diagnosis and treatment. As basic research increases our understanding of health and disease, so new diagnostic and treatment modalities are discovered. In addition, the normal process of maintaining clinical services, today strengthened by the commitment to clinical excellence and the practice of clinical audit(12), increases the focus on unmet needs as well as promoting the use of best practice guidelines(13).
Thus, whilst much of the development of new diagnostic tests may be undertaken in the diagnostics industry sector, the initial definition of clinical need and the basic research is undertaken in the hospital and at the bedside.
Whilst much research effort is committed to understanding the basis of disease and thus the search for diagnostic and therapeutic interventions, there is also an increasing effort directed toward the delivery of services with the development of new diagnostic techniques, new interventions and more recently in information technology to provide valuable infrastructure and manage data.
One of the important initiatives developed in response to the increasing pressures is the promotion of a more evidence based approach to practice. The link between the use of best evidence and the commitment to the highest quality of service through the use of clinical guidelines and stronger economic justification for the introduction of new technologies, will place a great demand to provide better evidence of effectiveness. This will be achieved through the greater emphasis on health technology assessment(14).
The starting point for health technology assessment is the identification of the clinical need — the purpose for which the technology is to be used. This is not the first stage in the evaluation of new technology, that being the assessment of technical and diagnostic performance. These characteristics must be established for each assay and piece of equipment; typically such performance is assessed by a manufacturer as part of a submission to a regulatory agency such as the Food and Drug Administration(15) or is established as part of an evaluation process such as by the Medical Devices Agency in the United Kingdom(16). Many of these evaluations are published in the scientific literature — often on more than one occasion. It is also the case that many laboratories responsible for test or equipment procurement wish to undertake their own evaluation rather than depending on the results of others; it could be argued that this is a waste of resources both on the part of the supplier and the evaluator.
There are, however, aspects of technical and diagnostic performance that will have a major impact on the clinical effectiveness of a test; a good example is the influence of the biological variation on diagnostic performance. Thus it is necessary to take more than one sample in several situations to gain reliable information, eg urinary measurements of albumin, catecholamines and collagen breakdown products.
However, the real value of the test lies in its clinical impact, be it in improved diagnosis, in improved patient management or in better health outcomes. Whilst the technical and diagnostic performance can be performed by a reagent manufacturer using banks of samples purchased from a clinical laboratory and are relatively straightforward studies, the examination of clinical benefit requires careful planning and can sometimes result in extensive (and expensive) studies. One of the recent studies that exemplifies this point is the Diabetes Control and Complications Trial; this represents a study that does not need to be repeated in many different geographical or cultural settings and in which the results can inform practice worldwide. On the other hand, where there are geographical and cultural factors that influence the presentation and prevalence of a disease, it may be necessary to undertake more studies. However, there is clear evidence, more from pharmaceutical studies, that: i) an unnecessarily large number of studies are sometimes undertaken; and ii) without a cumulative record kept the outcome does not become truly evident and implemented until long after the answer was known from the data available; this point was made in a metanalysis of thrombolytic therapy reported by Lau et al(17).
QUALITY OF EVIDENCE
In the sphere of diagnostic tests the production of good quality evidence is challenging and there are data to suggest that experimental design and reporting are not robust. Thus Reid et al(18) evaluated the papers concerned with diagnostic tests in a range of peer reviewed journals over a period of fifteen years to determine the adherence to guidelines drawn up to identify good quality evidence. In most cases less than fifty per cent of the papers met the criteria.
There are also only a limited number of systematic reviews of the use of diagnostic tests. Thus Olatidoye et al(19) undertook a metanalysis of the prognostic value of the troponins I and T as prognostic markers in patients with angina. The data presented in a summary ROC curve displayed wide variation between studies for which there could have been several reasons. The study however concluded that both tests were of similar prognostic value. The study demonstrated the value of using several well performed studies and drew important conclusions that would not necessarily have been arrived at from reading any of the individual papers.
A systematic review of the potential value of point of care testing in the primary care setting by Hobbs et al(20) illustrated what may be a common finding when more such studies are undertaken. Out of a total of 1062 papers that were considered to be relevant following electronic and hand searching only 92 were considered to contain extractable data. A large number of papers were concerned with technical evaluations – which may be more important in the case of point of care devices. The overall conclusion was that there was little evidence to demonstrate the benefits of point of care testing in the primary care setting.
In a systematic review of the use of urine dipsticks for the detection of urinary tract infection in children (conducted by the author) a total of 40 papers were found following an electronic search. After excluding papers that dealt with special patient groups or did not include a reference method, only 7 remained from which data could be extracted.
In a systematic review of the use of biochemical markers of myocardial damage in patients with acute chest pain, over 6500 papers were retrieved by an electronic search of English language papers since 1970. The review is covering several markers and focuses on diagnosis and prognosis in patients with acute chest pain. After review against strict inclusion criteria, primarily concerned with ensuring that there was a valid reference procedure used or outcome assessed with the evaluator blinded to the results, there remained a total of 382 papers from which data could be extracted. The bulk of the papers rejected were either because: a) they reported on technical evaluations; b) there was no reference procedure or appropriate control group; or c) the evaluator was not blinded with respect to test and reference results or outcomes. This observation on the small proportion of cited papers that appear to contain extractable data pose the question as to the level of wasted effort in the experimental work reported.
DISSEMINATION OF RESULTS
The metanalysis on thrombolytic therapy undertaken by Lau et al(17) clearly showed the benefit of regular systematic review of data to achieve an integrated picture. Absence of such a strategy leads to significant delay in implementation. There are many other examples where there have been delays in implementing findings; in practical terms this becomes more evident when additional investment is required, although the benefits to the patient or the organisation (operational and/or economic benefit) are evident.
It is also quite clear that there is significant variation in testing strategies between different laboratories and between different countries. Indeed that there is a dramatic variation between countries in the annual spend on diagnostics per head of population(21). A simple audit of the use of liver function tests, cardiac marker or thyroid function testing strategies will unveil considerable variation in test utilisation — both in range and number. Thus, at a time of increasing workload, an increasing repertoire of investigations available, together with an increasing burden of financial pressure, it is critically important to ensure that there is a regular dialogue between the provider and the user of the diagnostic services. In health technology assessment programmes dissemination of findings is one of the most challenging features(22).
PARTNERSHIP
The observations made in the preceding paragraphs provide a powerful argument for change and a radical review of the relationship that exists between the user of diagnostic tests and the provider of that technology. Indeed it is worth identifying all of the stakeholders in the diagnostic process and the role that they play (Figure 3). The most important stakeholder is the patient and he/she needs to be informed of the efficacy of diagnostic tests, based on good quality evidence and presented in an intelligible way for a lay person. This will be of increasing importance as point of care testing devices become available over the counter and as the focus on wellness screening increases(23).
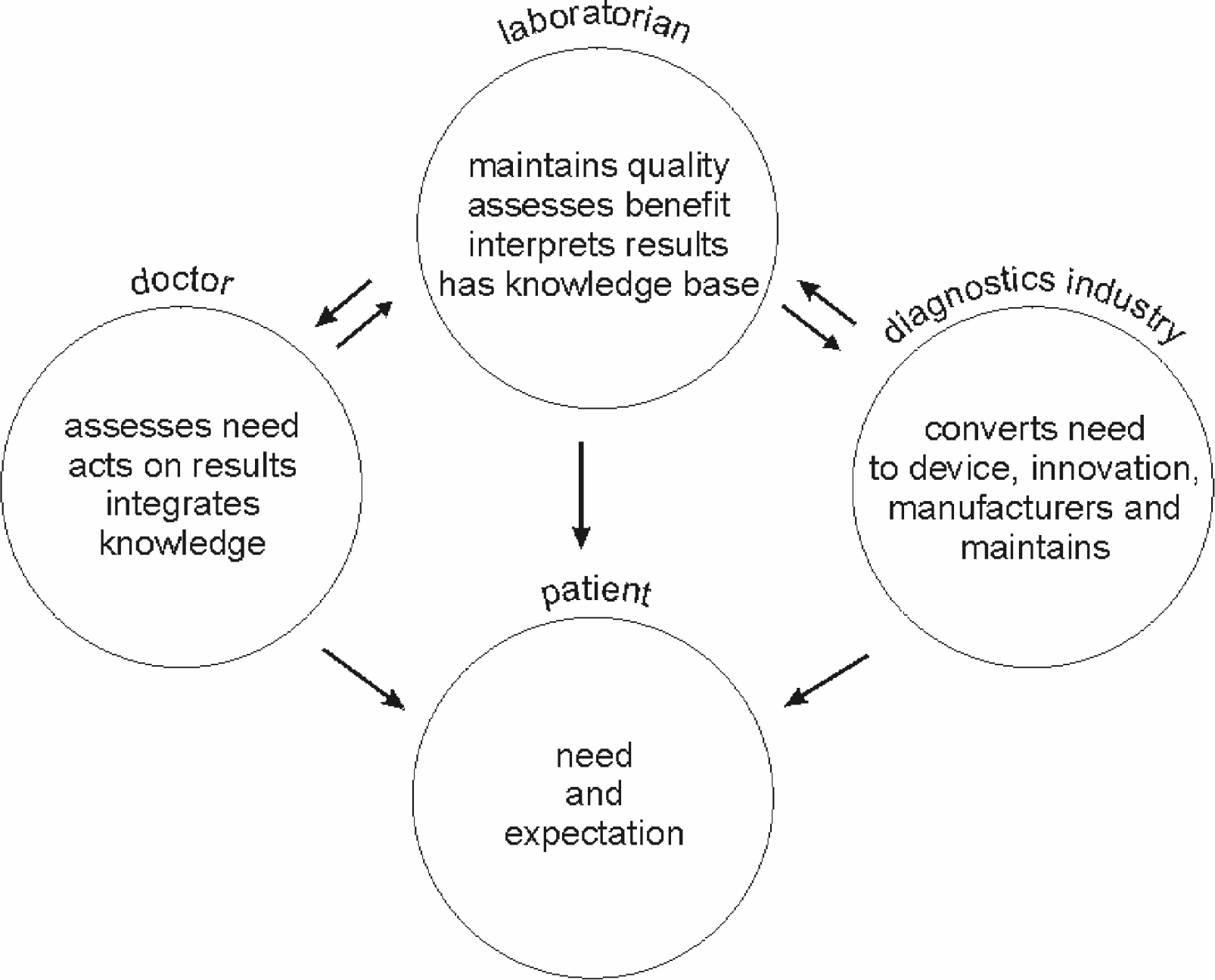
Stakeholders in diagnostic testing: needs, roles and responsibilities
The doctor faced with an increasing burden of information and with responsibility for the patient must be given information that meets his/her needs and distils out the best evidence — answering the questions that are being posed in his/her clinical practice. It must also be recognised that the patient:doctor interface is where the clinical need is recognised.
The laboratorian plays a pivotal role in the provision of diagnostic services either as informer, evaluator, deliverer, interpreter or auditor. Today the laboratorian invariably provides the technologically informed interface between the doctor/patient and the manufacturer. As the provider of service, whether directly or as agent for the point of care testing organisation, the laboratorian therefore is in a unique position to interpret the needs of the doctor and design the experimental protocols that will determine the efficacy of a test. However, the need may not be primarily clinical but more operational, eg rapid turnaround of tests to reduce length of hospital stay; again the laboratorian will be ideally placed to undertake these studies in order that operational decisions can be made.
The diagnostic industry now has the intellectual and manufacturing foundation to translate new analytical reagents or techniques into useable products which can be manufactured in a reproducible and reliable way. The importance of partnership at the interface between each of the stakeholders lies in understanding of the partners need, and delivering the product to meet that need, together with the provision of good evidence on the effectiveness of that product.
Experience of systematic reviews of the literature on diagnostic test would suggest that there needs to be greater clarity in defining need and then in the production of good evidence to support clinical, operational and economic effectiveness (or otherwise). The pressure on resources committed to the delivery of healthcare, and the requirement for the highest quality of service, demand close partnership between the manufacturer and the user of diagnostic tests and instrumentation.
CONCLUSIONS
In the face of an increasing pressure on healthcare providers it is critical to establish a closer partnership between those stakeholders who contribute to the recognition, diagnosis and management of disease namely clinical, laboratory and industrial professionals.
