Abstract
Cell-mediated immune responses have been found to play a crucial role in the protection from, or amelioration of, many human diseases. There are many vaccine trials being conducted where the most important immune assessment consists of measuring T cell responses. The most readily accessible source of T cells is the peripheral blood. Thus, collection, processing, cryopreservation, storage, and manipulation of human peripheral blood mononuclear cells (PBMCs) are all key steps for assessment of vaccine and disease-induced immune responses. Automation of these procedures is not currently practiced widely. This article reviews current practices and explores the need for automation.
Introduction: why t cells are so important and why we need functional viable t cells for immunogenicity assessments
Because of the human immunodeficiency virus-1 (HIV-1) epidemic, a new era in technology development has been ushered in. New discoveries not only for molecular biology but also for immunology and, in particular, for cellular immunology have occurred since HIV-1 was discovered 20 years ago. From studies in HIV-1 infected individuals, as well as individuals that were exposed to but not infected with HIV-1, it soon became apparent that CD8+ T cells were critical for protection. Thus, designing HIV vaccines and assessing HIV disease required a shift from not just examining antibody responses but, in addition, examining CD8+ T cell responses. 1,2 For other infectious diseases, such as malaria and tuberculosis, and cancers, disease is at least partially controlled by T cell immune responses. 3 –5 Thus, an important objective of human clinical trials of HIV, tuberculosis, malaria, and cancer vaccines has become the measurement of vaccine and disease-induced T cell responses. For measuring T cell responses, assays such as the traditional semiquantitative cytotoxic T cell (CTL) and lymphoproliferative assays (LPA) have historically been used. More recently, the tetramer, enzyme-linked immunospot assay (ELISPOT) and intracellular cytokine staining (ICS) assays have been employed. 6 –9 These assays, along with their potential for automation, are discussed elsewhere in this journal. Regardless of the assay used, in order to perform assays on T cells, we need to be able to isolate the appropriate functional T cells from pertinent tissues and process and store them in a manner that preserves their functional integrity. If performed correctly, the separation process from blood yields a pure population of mononuclear cells consisting of lymphocytes and monocytes, with high viability, minimal red blood cell and platelet contamination and optimum functional capacity. If cryopreserved and stored carefully, these PBMCs should maintain functional integrity for many years in storage prior to thawing.
Although PBMC processing may affect functional activity, we believe that cryopreservation is the most critical part of the procedure and the most likely to affect subsequent T cell function if performed suboptimally. This has led some to argue for real-time testing of T cell responses on freshly isolated PBMC without cryopreservation and storage. However, there are clear advantages to being able to batch assays from multiple time points from a clinical trial, and this requires the cryopreservation of PBMCs in a manner that maintains their functional capabilities. Under ideal conditions, fresh and cryopreserved PBMCs have been shown to have similar functional activity and preserve functional activity. Careful procedures used in PBMC cryopreservation and thawing result in minor losses in immune response. 10 –13 As such, most HIV vaccine trials now rely on cryopreserved PBMC for subsequent assessment of vaccine-elicited T cell immune responses. Inherent in the decision to go with cryopreserved cells is the knowledge that unless a mechanism exists to assure high viability and recovery of cryopreserved PBMC, there is no mechanism to evaluate immunogenicity in these clinical trials. Additionally, aspects of the cryopreservation process that do not affect viability can still adversely affect T cell function. Specifically, there is occasionally a population of cells with low forward scatter characteristics when PBMCs are thawed. While the presence of this cell population does not correlate with viability or recovery of cells, its presence does correlate with high background production of cytokines from CD4+ (and to a lesser degree CD8+) T lymphocytes, which is sufficient to obscure the detection of vaccine-induced T cell responses (Cox, Bailer, and Koup, unpublished). Considering the crucial aspect of blood processing and cryopreservation to the ultimate outcome of a vaccine trial, automation of these steps becomes an important goal. Currently there is very little in the way of automation in cellular immunology or even in most diagnostic immunology laboratories. 14,15
The absence of defined correlates of immune protection in HIV infection makes it difficult to assess the immunogenicity of vaccine candidates. Both CD4 and CD8 T lymphocyte-mediated immune responses might be important in the prevention of overt disease. Therefore, both CD4 and CD8 responses need to be examined during the early phases of assessment of a vaccine's immunogenicity (phase I and II clinical trials). Examples of assays that have been used to test vaccine immunogenicity along with the amount of blood required to perform them are reported in Table 1. All the trials were for assessing the immunogenicity of HIV recombinant canarypox vectors by the HIV Vaccine Trials Network (HVTN) and the AIDS Vaccine Evaluation Group (AVEG). 16 –18 The HVTN and AVEG 022 and 202 trials were performed approximately 5 years apart from trials 039 and 203. As shown, it is clear that the number of assays has increased progressively throughout the years. Initially, CD8 T cell responses were assayed by virtue of their cytotoxic activity using 51 Cr-release CTL assays, and CD4 responses were assayed in an LPA following antigen-specific stimulation. Both methods use radioactive reagents, making the assays more difficult to transfer to developing countries. These assays are very labor intensive, require fresh samples, and are difficult to perform on a large scale or in an automated fashion. Over the years, there have been many non-radioactive methods developed for detection of cytotoxic activity and for measurement of cell proliferation or growth. The pharmaceutical industry has developed rapid-through-put microtitration assays to screen new cytotoxic agents. 19,20 Other assays measure the effect of a drug on the growth of a population of cells, and the endpoint is an estimate of cell number. Use of a tetrazolium dye (MTT) as an indirect measure of cell number was first reported in the early 1980s. 21 However, for the immune assessment of vaccines, the gold standard radioactive CTL and LPA assays have until recently continued to be used. 18,22,23
Frequency of procedures used in phase I and II HIV vaccine trials
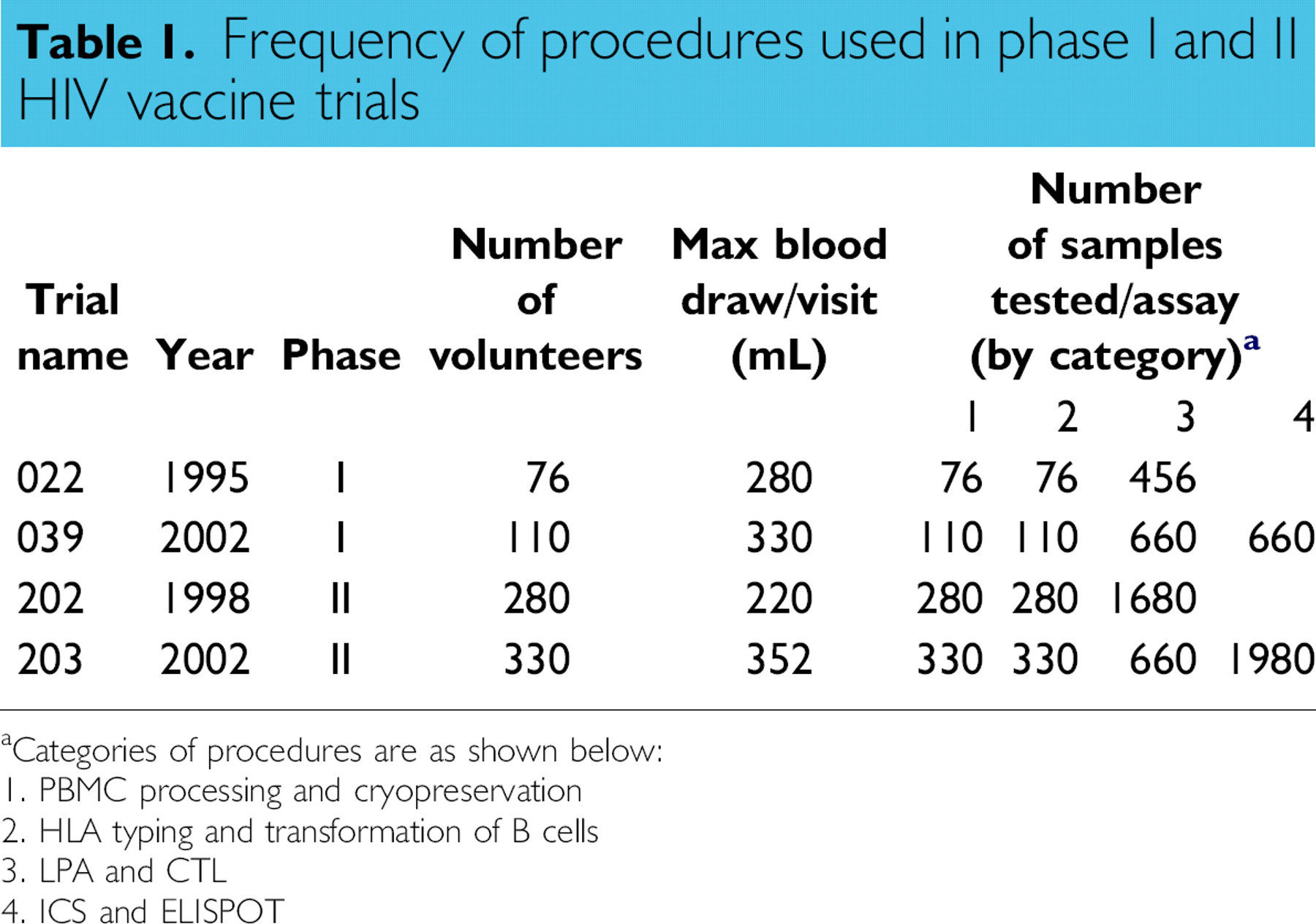
Categories of procedures are as shown below:
1. PBMC processing and cryopreservation
2. HLA typing and transformation of B cells
3. LPA and CTL
4. ICS and ELISPOT
The CTL and LPA assays typically require in vitro stimulation for 3 to 14 days with the antigen of interest and therefore do not measure the frequency of precursor cells in the peripheral circulation. During this in vitro stimulation, cells may expand at different rates, some may undergo antigen-induced apoptosis, and others may not expand under these conditions. The advantages of the ELISPOT, ICS, and tetramer assays are that they can be used to directly measure the frequency of T cells circulating in the blood. The ICS and tetramer assays offer the advantage of being able to directly phenotype cells as well as measure their ability to secrete cytokines. It should be pointed out that none of these assays are able in their present forms to measure all possible effector mechanisms of CD8+ or CD4+ T cell responses. Each assay has its limitations, since it is not really known in any given disease what is likely to be the main effector mechanism that correlates with protection from disease or with vaccine efficacy. Both the traditional CTL assay and non-radioactive cytotoxicity assays examine the death of target cells, not the CD8+ T cells that mediate killing. A novel, recently described assay measures the cytolytic potential of CD8+ T cells. Antigen-specific CD8+ T cells are enumerated using a marker expressed on the cell surface following activation-induced degranulation, a necessary precursor of cytolysis. 24,25 The assay measures the exposure of CD107a and b, present in the membrane of cytotoxic granules, onto the cell surface as a result of degranulation. This flow-cytometry-based assay may in the future be amenable to automation.
There have been some attempts to correlate the various types of assays used for immune assessment. Some assays correlate with each other; others do not. 26 –29 Furthermore, correlations between assays are likely to be dependent on the antigen, magnitude, and type of response as well as the disease or vaccine response being examined. An additional consideration for all the assays (CTL, LPA, ICS, ELISPOT, and tetramer) is the already sophisticated machinery that is required in order to conduct the assays. Another level of sophisticated machinery will be needed to implement automation. During the last 5 years, the ELISPOT and ICS assays have started to be implemented for assessing vaccine-induced immunogenicity. 30 Both assays can be performed on cryopreserved cell samples, do not use radioactive reagents, and may be more sensitive and quantitative than the previous assays. While the ELISPOT, ICS, and tetramer assays have some definite advantages, these assays remain to be standardized appropriately for use in clinical trials, and automation of these assays lies in the future. Some attempts at standardizing these assays are already being made. 10,31 –33 Some redundant testing is being performed during the time that we are switching from old assay formats (i.e., CTL and LPA) to the others (ELISPOT and ICS). However, the new assays have clear advantages in terms of data management and the possibility of automation. The use of cryopreserved samples with these assays has allowed batch testing of the samples and decreases the possibility of interassay variation of the results. As discussed earlier, the steady increase in sample processing and data management necessitates a switch to automated sample handling.
The potential for how many cellular immunological assays might be required in the near future can be illustrated by the requirements for projected phase I, II, and III HIV trials to be conducted by the Partnership for AIDS Vaccine Evaluation (PAVE) vaccine trials (Fig. 1). PAVE is a proposed international network for sharing information about AIDS vaccine development. The network will include government-funded organizations, pharmaceutical companies, and not-for-profit groups. A recent policy forum in Science magazine 34 has outlined this partnership and has highlighted the need for establishing central immune response laboratories: “Standardized protocols and immunogenicity measurements need to be broadly implemented at the preclinical and clinical stages of vaccine development to measure humoral and cell-mediated immunity and to provide a test bed for reproducibly assessing the immune response to HIV antigens and adjuvants. Laboratories should be established to develop and deploy robust, reproducible, and interpretable assays of immune response; to standardize reagents for such assays; and to incorporate quality-control measures for consistency.” Importantly, it is recognized that establishing central immune response laboratories might prove challenging to academic- based laboratories. Implicit in the development of one or more central immune response laboratories for testing all HIV vaccine candidates would be the requirement for a huge throughput of samples. 34 Most future PAVE trials will be conducted in Africa where there is an urgent need for an HIV vaccine. 35,36 There are many other HIV vaccine trials projected and ongoing. 37 The need for automation becomes evident by projections for personnel (Fig. 2) and equipment (Fig. 3) needs based on current throughput estimates without automation. Projections are based on a model that all trials have eight time points with eight test antigens. At each time point, there is blood processing, ICS, and ELISPOT performed on every sample. All trials are assumed to enroll at a constant rate over 6 months with follow-up for 1 year. The fraction of the trial enrolled on March 31 of a given fiscal year will have all testing assigned to that fiscal year (since the majority occurs in the first 6 months of the trial), with the remainder assigned to the next fiscal year. Work capacity for one FTE for blood processing, ICS, and ELISPOT are 1392, 2112, and 1800 samples, respectively. Equipment requirements for each blood processing FTE are 1 hood and a 0.33 centrifuge; for ICS, a 0.33 hood, centrifuge, and FACS; and for ELISPOT, a 0.33 hood, centrifuge, and 0.1 ELISPOT reader.
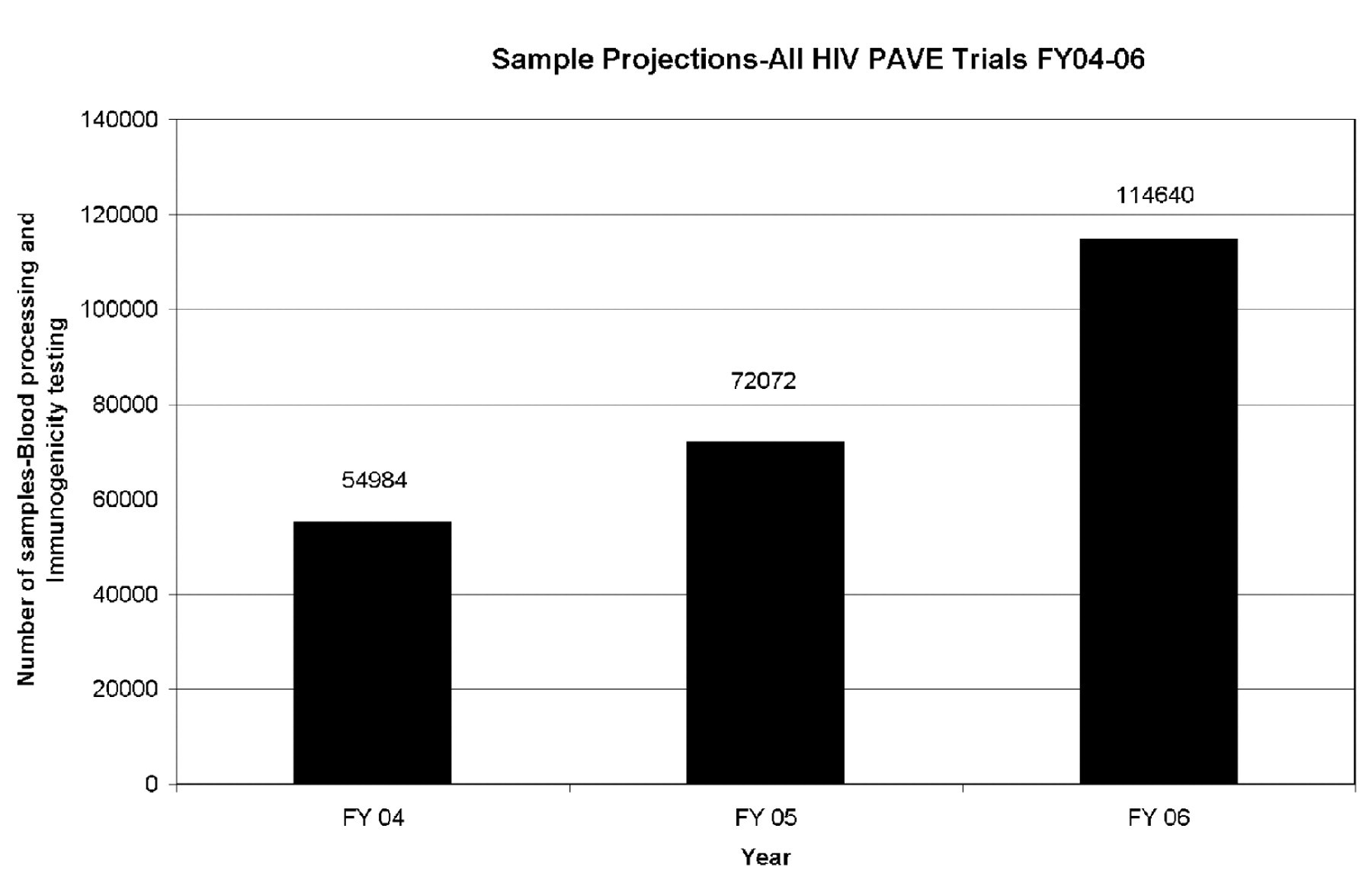
Sample projections PAVE Trials FY 2004–2006. Projections of the number of individual samples from all PAVE trials slated for Fiscal (FY) 2004–2006. Each time point for an individual within a trial is counted as an independent sample, with the assumptions that all samples will have immunogenicity testing. All projections are for reference purposes and are based on a model applied uniformly to all trials.
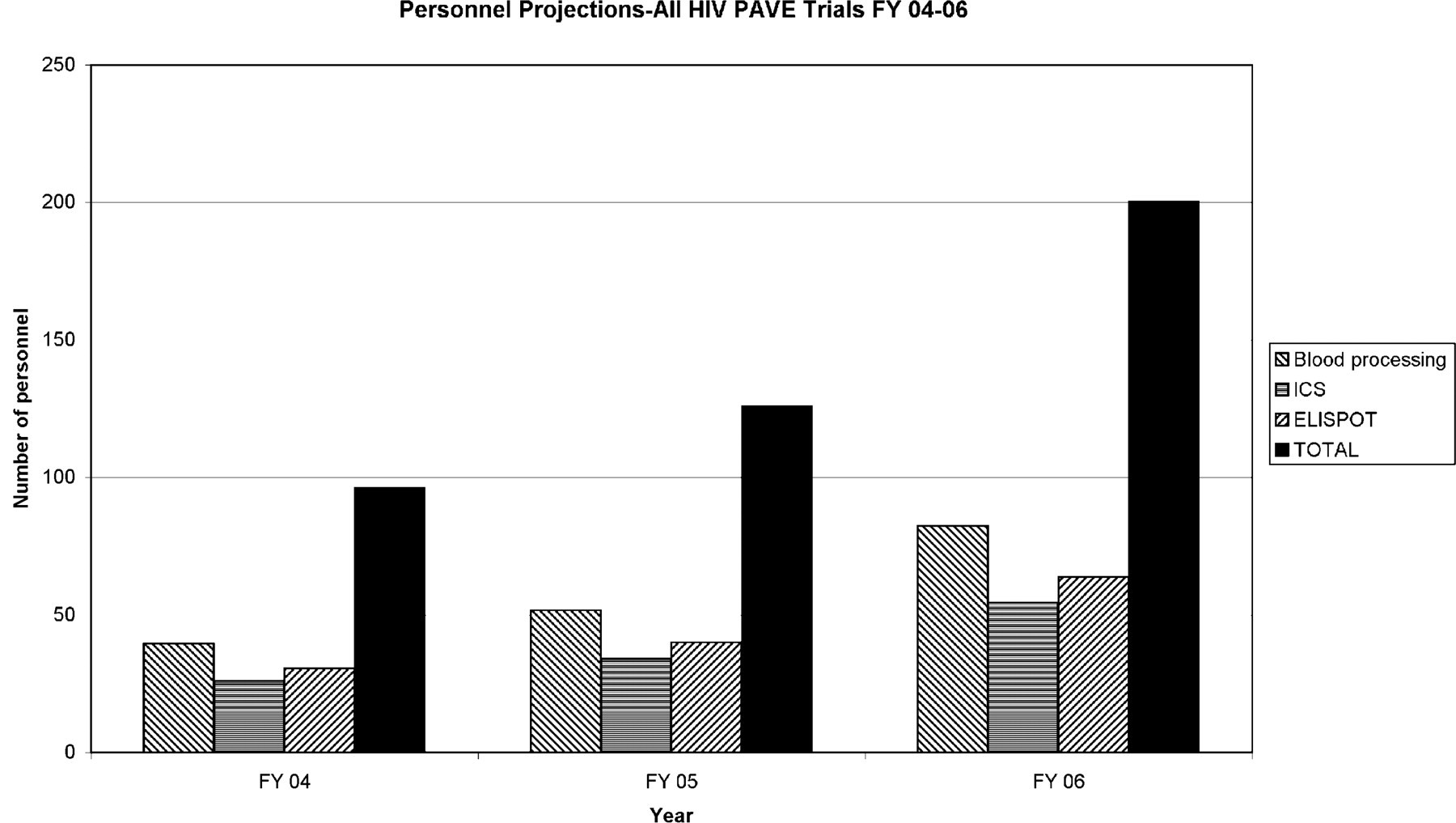
Personnel projections for PAVE Trials FY 2004–2006. Based on current throughput using manual operations, personnel projections are presented for blood processing/cryopreservation, ICS, ELISPOT, and the total number of personnel. Projections are based on the number of samples from all PAVE trials slated for Fiscal (FY) 2004–2006. All projections are for reference purposes and are based on a model applied uniformly to all trials.
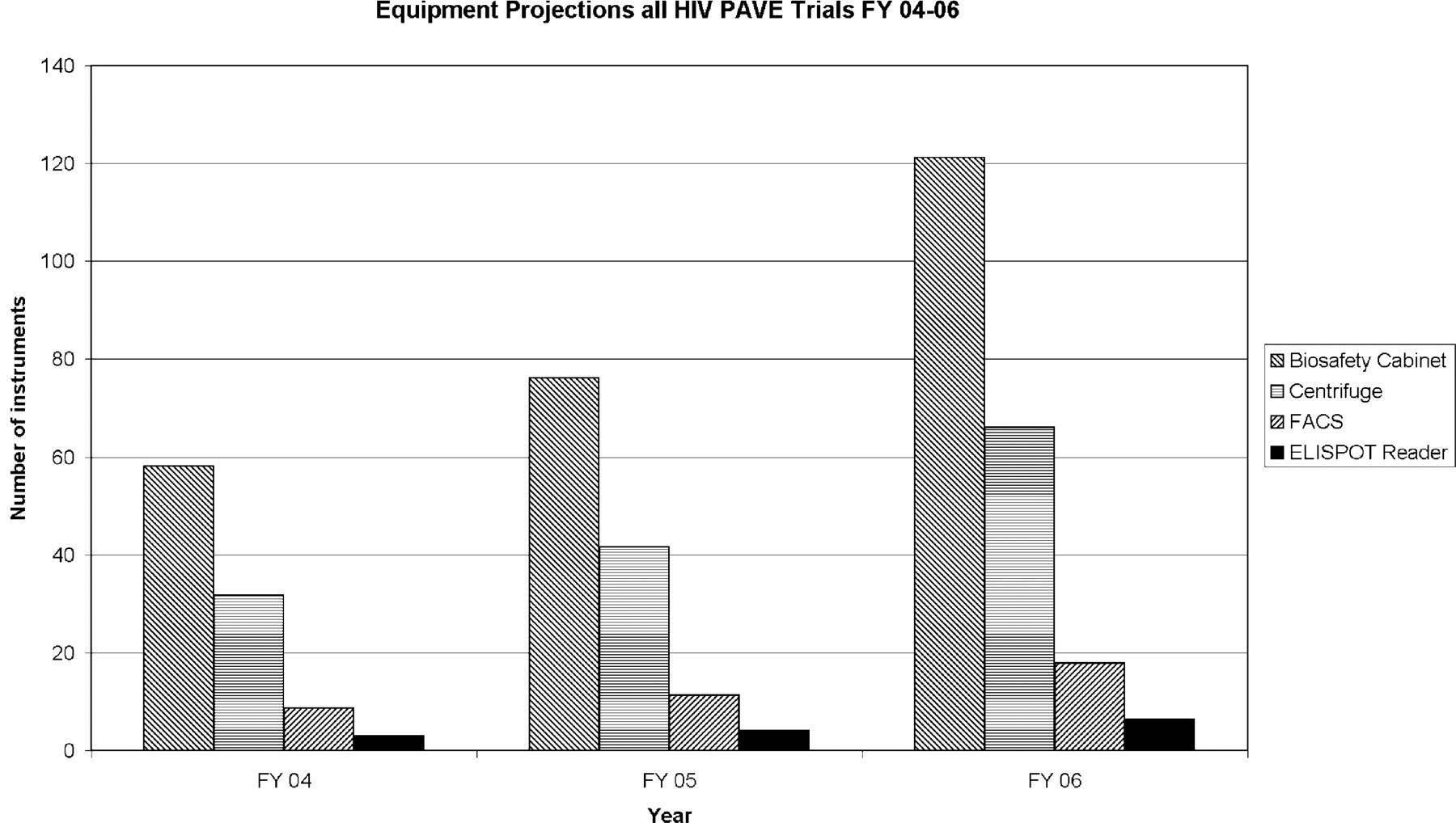
Equipment projections for PAVE Trials FY 2004–2006. Based on current throughput using manual operations, equipment projections are presented for blood processing/cryopreservation, and immunogenicity testing broken down by instrument types (FACS = fluorescence activated cell sorter). Projections are based on the number of samples from all PAVE trials slated for Fiscal (FY) 2004–2006. All projections are for reference purposes and are based upon a model applied uniformly to all trials.
Other opportunities exist requiring large numbers of T cell assays to be conducted in the cancer vaccine field. 7 Assessment of human immune responses is also seen as a critical need for the U.S. government in its fight against bioterrorism. A better understanding of the human immune response to potential agents of bioterror, and rapid development of countermeasures such as vaccines and therapies, are among the objectives of a new $85 million program announced by the National Institute of Allergy and Infectious Diseases (NIAID). A particular emphasis will be to move new findings about immune system function out of the laboratory and into clinical trials. 38 It is more practical and safe for a robot or automated instrumentation to handle potentially lethal biohazardous samples rather than have humans do so. Thus, there are clearly many future opportunities for automation in the cellular immunology laboratory.
Processing and Cryopreserving Peripheral Blood Mononuclear Cells (PBMC)
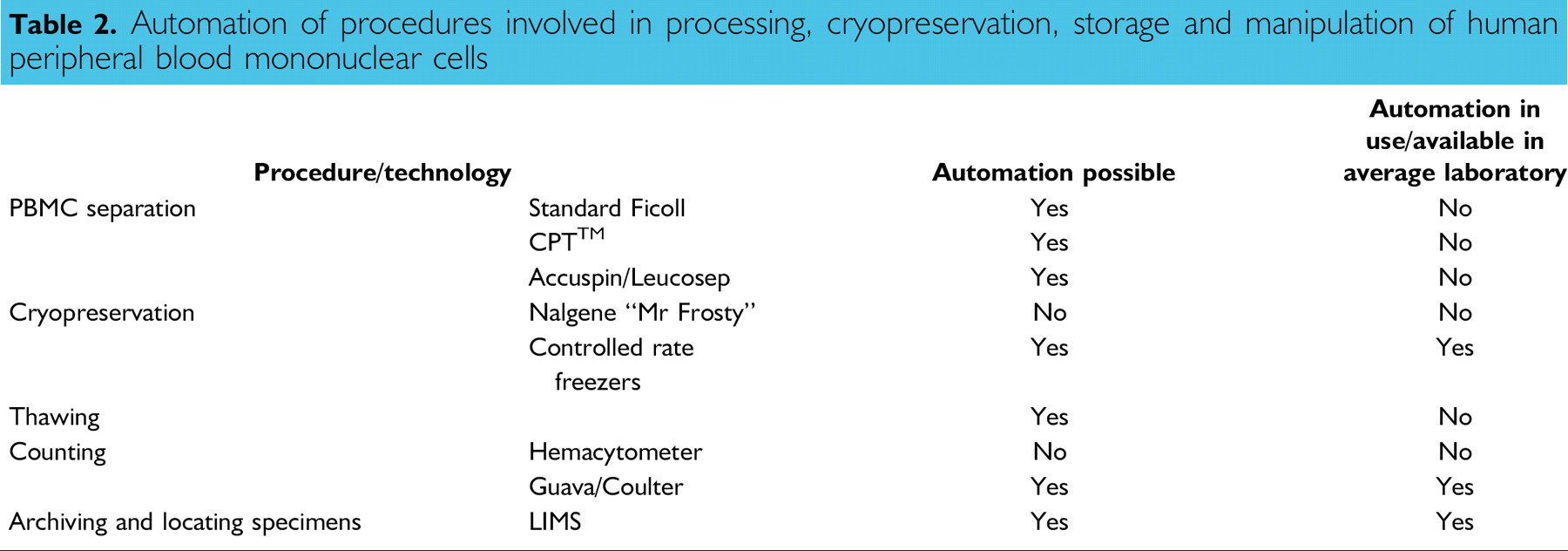
The functionality of T cells used in assays may be affected by all the procedures that follow the collection of the blood from the donor; namely, cell separation, cryopreservation, storage, and thawing. The potential for automation of these procedures is outlined in Table 2, and the technical nuances associated with each of these procedures is described below.
Automation of procedures involved in processing, cryopreservation, storage and manipulation of human peripheral blood mononuclear cells
The most convenient source of T cells for the types of assays described above is from peripheral blood derived from venipuncture (i.e., PBMC). Although it is possible to use whole blood for certain functional T cell assays, in most cases, this is not possible, as red hemolysis and secretion of inhibitors from platelets adversely affect T cell function. Thus in most cases, separation of mononuclear cells from peripheral blood is required. The technique for separating PBMC from blood was first described in the 1960s and has changed very little since. 39 The original technique of layering blood onto a ficoll-hypaque gradient requires some technical expertise and is not amenable to automation. However, layering Ficoll underneath the blood is an acceptable alternative and is more amenable to automation. In addition, two other improvements in the technique in the last few years have substantially reduced the technical burden of the procedure, improved yield, and may be even more amenable to automation.
The Vacutainer® cell preparation tubes (Vacutainer® CPT™; Becton Dickinson, Franklin Lakes, NJ), the Accuspin tubes (Sigma-Aldrich, St. Louis, MO), and the Leucospep (Greiner Bio-One, Longwood, FL) tubes provide a convenient, single-tube system for the collection and separation of mononuclear cells from whole blood. The Vacutainer™ CPT™ Cell Preparation Tube combines a blood collection tube containing anticoagulant with a Ficoll-Hypaque or similar density separation fluid and a polyester gel barrier, which separates the two liquids. 40 The Accuspin and Leucospep tubes use a porous high-density polyethylene membrane frit that separates the blood from the lower chamber containing ficoll. 41,42 Anticoagulated whole blood is added to the top chamber and upon centrifugation, the whole blood descends through the gel barrier or frit to contact the separation medium below, giving a clear separation of the blood components. The erythrocytes aggregate and the granulocytes become slightly hypertonic, increasing their sedimentation rate and resulting in pelleting at the bottom of the tube. Lymphocytes and other mononuclear cells, i.e., monocytes, remain at the plasma interface.
These tubes are convenient to use and result in high viability of the cells after transportation. The blood specimens can be transported at ambient temperature without being removed from the tube, since the gel forms a stable barrier between the anticoagulated blood and ficoll. Cell separation is performed at the processing/storage laboratory during a single centrifugation step, which reduces the risk of sample contamination and eliminates the need for additional tubes and processing reagents. In many instances, and in particular when biological safety cabinets are not available on site, these methods may be useful because the centrifugation step can be done on site and the remaining processing steps can be performed after shipment to a central automated laboratory within the shortest time possible. The central laboratory can then process the cells in a biological safety cabinet and set up functional assays or cryopreserve the samples as needed.
The optimal time frame between collection of blood sample to processing, separation, and cryopreservation of PBMC should be less than 8 h or on the same day as collection. This is not always feasible when samples are being shipped across the country to processing centers. It seems likely that if PBMCs are left too long in the presence of anticoagulants or at non-compatible temperatures, adverse effects on PBMC function and or changes that affect the PBMC separation process may occur. 13,43 In trials and protocols involving longitudinal study designs, it is often advantageous to store cryopreserved specimens so that batch testing of samples can be performed. The minutia of cryopreservation techniques are beyond the scope of this review; however, it should be pointed out that cryopreservation needs to be performed correctly or functional integrity and the recovery of the PBMC can be significantly reduced. The basic principal involves cooling cells at a rate of approximately 1 °C per minute in the presence of a cryoprotectant such as dimethyl sulfoxide. 44 –46
Because of the pharmaceutical, biotechnology, and agricultural needs for large-scale cryopreservation of tissues such as sperm, oocytes, and seed stocks, automated procedures for cryopreservation have been in place for many years. The use of automated controlled rate freezers provides the step-wise cooling that is necessary for optimal cryopreservation of cells. The optimal condition for storing PBMC is in the vapor phase of liquid nitrogen (LN). Currently, there exist mechanisms to automatically transfer specimen tubes of serum, plasma, or reagents from a processing center to long-term storage in standard –80 or –20 °C freezers. 47,48 It is technically much more challenging to automate the procedures for robotically transferring aliquots of cryopreserved PBMC because of the absolute requirement to keep the PBMC at a constant low temperature. PBMC are particularly prone to small fluctuations in temperature. There does not currently exist an automated mechanism for transfer of cryopreserved PBMC to the appropriate LN vapor phase storage conditions.
Once PBMCs are cryopreserved correctly, they can be stored long term in vapor phase LN. If cells are to be shipped to a central laboratory for testing, then a further round of retrieval, tracking, and shipping of specimens will be required. Maintaining good laboratory practices (GLP) is paramount for all phases of clinical trials. As such, there are very definitive requirements for appropriate documentation of sample inventory, data collection, and chain of custody. 49 A practical approach is the implementation of a laboratory information management system (LIMS), which maintains this documentation, and at the same time, it simplifies the need for sample query, retrieval, and reporting. 50 There are numerous commercial off-the-shelf (COTS) LIMS products that can be tailored to the particular laboratory needs while meeting FDA 21 CFR part 11 guidelines. 49
Thawing PBMC and Cell Counting Procedures
Currently, no automated mechanism exists to thaw cells. Like cryopreservation, this is a critical step that requires a great deal of care in order to avoid significant cell loss and viability loss. Thawing procedures can significantly impact recovery, viability, and function of PBMC, and counting procedures can increase assay variability. For example, in a recent study conducted under the auspices of the Division of AIDS (NIH, Bethesda, MD), 11 laboratories that were already conducting HIV vaccine trials were provided with 11 PBMC specimens. The PBMC specimens were thawed and counted manually using a standard hemacytometer and trypan blue dye exclusion for assessment of viability. The median recovery of PBMC immediately after thawing for all samples in all laboratories was 60% of that initially provided, with a range of 4.7 to 114%. This represents a 24-fold difference between the lowest and highest recovery. While some of this was due to certain samples having very poor recovery, overall the data indicated that there were large differences in a laboratory's ability to thaw and recover PBMCs. The median viability immediately after thawing for all samples in all laboratories was 86%, with a range of 24.8 to 100%. This represents a four-fold difference between the lowest and highest viability. A statistical analysis of all the viability and recovery data revealed significant patient and laboratory effects for all parameters using a non-parametric Kruskall Wallis test and a two-way ANOVA. Recovery and viability after thawing were all significantly different when stratified by laboratory or by sample. 31 These data clearly illustrate the need for improvement and standardization of PBMC thawing and cell counting.
Cell Counting and Viability Assessment
It is essential to have correct cell counts and viability determinations for accurate performance of cellular immune assays. For example, in the ELISPOT assay, an exact number of cells needs to be added to each well. If the cell counts are incorrect, then the number of cytokine-secreting cells will also be incorrect. Likewise for CTL assays, where the number of CTL is titrated against a fixed number of target cells, an exact number of cells needs to be added to each well, or the ratio of the CTL to the target will be incorrect. In both the ELISPOT and CTL assay, it is also important to minimize the number of dead cells, since the presence of dead cells can impact the integrity of the assay. Although no guidelines exist for optimum PBMC viability, a cut-off of >70% viability might be recommended as a minimum for specimens in an endpoint assay. For flow cytometric assays, the flow cytometer does the particle counting, and gating provides a way to exclude dead cells. Manual PBMC counting and viability assessment with a hemacytometer have been in use for decades. While hemacytometer counts can be very accurate, the procedure is entirely dependent on operator competency and hence prone to operator–operator inconsistencies. Manual viability counts rely on the operator being able to distinguish overall size and shape of a lymphocyte as well as different hues of blue. Thus, there is a need for accurate and reliable assessments of lymphocyte counts and viability, which do not depend on trypan blue exclusion and manual counting. Within the last year, several automated units that can determine cell counts and viability have appeared on the market. 51 –53 The Guava Personal Cell Analysis (PCA) system uses a combination of two fluorescent nucleic acid dyes; the automated assay identifies cells while excluding debris and distinguishing live cells from dead. Assay results include total count and cell counts for both viable and apoptotic cells. Within just a few years of being on the market, the Guava PCA system has been placed in most PAVE laboratories and has provided a platform for decreasing cell counting variability and increasing throughput. The new Guava PCA-96 provides an automatic, high-throughput, 96-well microplate, and multiple microcentrifuge tube analysis platform. This system can also be readily validated to comply with GLP. 52,54
Discussion
While many of the individual components of a system that could automate blood processing, counting, aliquotting, and cryopreservation do exist, they have yet to come together into a workable system. Many companies sell liquid-handling systems 47 that could, if integrated with indexed centrifuges, perform most of the initial steps in the process of blood separation by Ficoll density gradient centrifugation. The most difficult parts of that process would be the overlaying of the blood onto Ficoll. This problem could be overcome with the Accuspin tubes by underlaying Ficoll instead of overlaying blood or by using the Vacutainer CPT tubes. The other step that will be problematic is the removal of the white blood cell layer from the surface of the Ficoll, a process that may require the integration of a pipettor and an electronic eye in order to detect the level of the interface and accurately and completely remove that layer. Beyond that step, all of the various robotics already exist to dilute, centrifuge, resuspend, aliquot, count, and freeze the samples with a high degree of accuracy and in a system where each sample is treated identically tracked and there is no possibility of sample mix-up or mislabeling.
When one looks at the types of automated systems that have been developed for the complex activities within many hospital clinical laboratories, the pharmaceutical/biotech industry, and the Human Genome Project, one must question why an automated system for blood processing and cryopreservation has not yet been developed. The answer probably lies in economics. The technology exists to build the system, but there has not been, until recently, a market for such a system. The proliferation of vaccine and therapeutic immunologic products whose activities will be measured within accurately processed and cryopreserved T cell samples may generate the economic drive to develop such an automated system. When one considers the number of blood samples that will require PBMC processing and cryopreservation just within the field of HIV vaccines in the next 3 years (Fig. 1), the personnel required to do that processing (Fig. 2), and the laboratory facilities to house those technologists (Fig. 3), and then realize that there are similar programs in cancer, TB, malaria, and biodefense, it becomes clear that it will be virtually impossible to complete all of these trials in the absence of an automated blood processing system. The market exists for any company that is willing to invest in the development of such a system.
Acknowledgments
This article is dedicated to all the laboratory research assistants and their supervisors who have labored over thousands of CTL, ELISPOT, LPA, and ICS assays in support of HIV vaccine development.
