Abstract
Following infection with SARS-CoV-2, cellular components of the adaptive immune system play a crucial role in eliminating the virus. Specifically, virus-specific CD4+ and CD8+ T cells generate effector cytokines and display cytotoxic activity. A number of studies carried out during the COVID-19 pandemic highlighted the importance of CD4+ T cells, CD8+ T cells, and memory cells in this process. T-cell responses emerge early and contribute to protection, but are comparatively impaired in severe cases, often accompanied by intense activation or lymphopenia. Since December 2020, SARS-CoV-2 vaccines have been licensed and administered worldwide. These vaccines induce a targeted T-cell response against SARS-CoV-2. The cellular response after the third dose was strong and superior to that obtained with the second dose. COVID-19 multiple vaccines elicit a robust CD4+ and CD8+ T cell response after the short-term booster. While, the T-cell response induced by COVID-19 vaccines has been shown to decline within 6-12 months of vaccination. In addition, the long-term persistence of cellular immunity may protect against the development of severe disease. In addition, adoptive T-cell therapies have shown considerable potential in the development of COVID-19 traitement. These therapies involve the transfer of T cells with specific antiviral properties into patients to boost their immune response against SARS-CoV-2.
Introduction
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection is causing coronavirus 2019 (COVID-19) disease. As of April, 2023, more than 636 million confirmed cases have been reported and more than 6.88 deaths worldwide. SARS-CoV-2 infection causes a large range of clinical manifestations, from asymptomatic or mild disease to severe disease associated with an inflammatory response. To effectively control the COVID-19 pandemic, a better comprehension of the immune response against SARS-CoV-2 is necessary (M. K. 1 Cellular immune responses have evolved to recognize and control intracellular pathogens. During viral infection, CD4+ and CD8+ T cells contribute to viral control by producing effector cytokines and exerting cytotoxic activity. During the development of COVID-19, numerous studies have reported the phenotype and function of various immune cell subtypes in COVID-19 patients. In addition, several studies have investigated and characterized SARS-CoV-2 specific CD4+ and CD8+ T cells in COVID-19 patients. SARS-CoV-2-specific CD4+ and CD8+ T cells were examined by in vitro stimulation-based functional assays, including the ELISpot interferon (IFN)-γ assay, intracellular cytokine staining (ICS), and activation-induced marker (AIM). Evidence to date suggests that T cells play a key role in preventing SARS-CoV-2, and the current pace of progress bodes well for optimizing future pandemic control 2 This review evaluates published studies on the T cell immune response to SARS-CoV-2. Particular foccus is information on SARS-CoV-2-specific CD4 + and CD8 + T cell phenotypes and functions in COVID-19 patients and convalescents. In addition, we discuss the development of memory T cell responses in recovered SARS-CoV-2 patients,T cell responses against SARS-CoV-2 variants and T cell responses induced by COVID-19 vaccines, and adoptive T cell based therapies.
Implications of T cells in human Anti-Covid-19 responses
The recognition of SARS-CoV-2 by T lymphocytes
The SARS-CoV-2 virus is from the β-coronavirus (β-CoV) lineage B, this family comprises an enveloped, non-segmented, positive-sense, single-stranded RNA virus genome with a 5' cap structure and a 3' poly-A tail. The genomic sequences of SARS-CoV-2 range in length from 29.8 kb to 29.9 kb with 12 open reading frames (ORFs) coding for 27 proteins. The genomic organization includes 5′- leader sequence- ORF1/ab- S- ORF3a- E- M- ORF6a- ORF7a- ORF7b- ORF8- N- ORF10-3′ from left to right. 3 So the replicase gene encompasses 67% of the genome that encodes a large polyprotein that is processed into 16 non-structural proteins (nsps). 4
Interestingly, the T cell response during SARS-CoV-2 infection can trigger a potent immune response, which includes immune activation and antiviral immune responses via T helper (Th) cells and cytotoxic T cells (CTL), and thus induce death of the infected cells. Regarding activation, some studies suggest that CD8+ T cell activation is superior to the CD4+T cell response which has been characterized by higher expression of the activation markers HLA-DR and CD38. 5 However, Grifoni et all, revealed that SARS-CoV-2 specific CD8+ T cells in mildly symptomatic patients were generated against the viral proteins S, M, NP, ORF3a, and ORF1ab, whereas CD4+ T cells were found to be specific for Spike, nsp3, nsp4, nsp12, ORF3, ORF7a, and ORF8. 6 In addition, six major epitopes have been identified as being involved in T cell responses, three epitopes are from Spike and two from M proteins and one from nucleocapsid. 7 Thus it was demonstrated that the CD4 + T cell response against Spike glycoprotein in recovered patients, while the CD8 + T cell response was specifically attributed to SARS-CoV-2 internal proteins. 8 Then a notable observation was the finding that CD8 + T cell responses against the ORF1ab, ORF3a,S, NP, epitope (CTDDNALAYY, FTSDYYQLY, KCYGVSPTK, FTSDYYQLY), restricted by MHC I alleles (A*01:01, A*01:01, A*03:01, A*01:01) demonstrate high antiviral activity (100%) in convalescent patients. 9 In addition, Bert Nina et al, showed that CD4+ and CD8+ T cells that recognized several regions of the N protein in recovered individuals. Since, NSP7 and NSP13 specific T cells are detected and often dominant in unexposed donors, while representing a minor population in individuals who have recovered from SARS or COVID-19. Among the different coronaviruses, structural and nonstructural protein sequences are highly conserved (NSP7 and NSP13 are 100% and 99% identical, respectively, between SARS-CoV-2, SARS-CoV, and bat). 10 Then, more importantly, 54% of the total positive response was associated with Spike-derived epitopes 11% for the receptor binding domain (RBD) and 44% for the non-RBD portion of Spik. 11 During the genome evolution of SARS-CoV-2, a wide range of mutations was observed, whereas the structural proteins Spike (S) and Membrane (M) were highly mutated, while Envelope (E) and Nucleocapsid (N) proteins were highly conserved. 10 This mutation is usually concentrated in RBD of the Spike protein. And so, lead to the development of variants of concern, especially in terms of relative evasion of the immune system. So, it is not likely that these variants can escape a considerable proportion of the SARS-CoV-2 specific T cell response, 12 and may indeed suppress the functional responses of individual T cell clones, 13 de Silva et all, has been shown that mutations in Spike result in loss of T cell recognition in epitopes restricted by common HLA alleles, such as A*03:01, A*11:01 and A*01:01. 14 In addition, mutations in the Spike sequence are thought to have an impact on highly binding epitopes in individuals carrying certain HLA alleles. For example, a 69-70 HV deletion is expected to prevent strong binding in DRB1*1501 individuals while increasing binding in DRB1*0101 individual. 15
Subsequently, some studies have detected that some people who have not been exposed to SARS-CoV-2 have a pre-existing reactivity to SARS-CoV-2 sequence. 16 Due to the existence of homologous sequences between SARS-CoV-2 and human coronaviruses. Pre-existing memory CD4+ T cells were detected that cross-react with comparable affinity to SARS-CoV-2 and the common cold coronaviruses human coronavirus (HCoV)-OC43, HCoV-229E, HCoV-NL63 and HCoV-HKU. 17 In addition, Bert Nina et all was shown that patients who recovered from SARS (the disease associated with SARS-CoV infection) have long-lived memory T cells that respond to the SARS-CoV N protein. These T cells displayed robust cross-reactivity with SARS-CoV-2 N protein. 10
T cell responses against SARS-CoV-2
Humans produce CD4+ T cells and CD8+ T cells and specific antibodies against SARS-CoV-2 infection and can each play a protective role in controlling viral infections, but these roles and the importance of each component of adaptive immunity vary depending on the viral infectio. 18 So in a normal immune environment, SARS-CoV-2 virus recognizes cells expressing the ACE2 receptor, including macrophages and epithelial cells. These macrophages detect SARS-CoV-2 via the ACE2 receptor and present virus-derived peptides to CD4+T cells (Th0) via the TCR-MHC II interaction. 19 Once exposed to SARS-CoV-2 antigen, Th0 cells polarize predominantly to Th1, leading to the release of the effector cytokines IFN-γ, and IL-2 but little or no IL-4, IL-5, IL- 13 or IL-17α to clear the virus, and Th2 to elicit humoral-mediated immune responses including the secretion of neutralizing antibodies against SARS-CoV-2. 20 In addition, infected epithelial cells degrade viral particles and present them to cytotoxic CD8 + T lymphocytes (CTL). 19 Most of these cells produce IFN-g and express granzyme B and perforin, but a fraction also secrete TNF-a (tumor necrosis factor alpha), but not IL-10. 20 In one study, it was revealed that CD4+ T cells appeared as a Th1 phenotype and produced a substantial amount of IFN-γ and expressed lower levels of IL-4, IL-13, IL-5 or IL-17A against the SARS-CoV-2 Spike protein in recovered individuals. Thus CD8 +T cells produced IFN-γ and also expressed granzyme B and tumor necrosis factor alpha (TNF-α). 6 Then, in an incompetent immune environment, as in the case of COVID-19, SARS-CoV-2 recognizes epithelial cells or macrophages via the ACE2 receptor. The viral RNA will replicate by rerouting the host's transcriptional machinery. This viral proliferation will infect multiple cells, resulting in tissue damage and other lethal complications. Under these conditions, CD4+ and CD8+ T cells fail to provide adequate cell/humor-mediated immune responses to eliminate virus-infected cells. 19 Notably, in one study was described patients with moderate disease have higher percentages of IFNγ-producing Th1 cells. In contrast, in patients with mild COVID-19, With regard to CD8+ T cells, CD8 + T cells were shown to express higher levels of cytotoxic molecules, such as granzyme A and FAS ligands, in patients with mild COVID-19, In contrast, in patients with severe COVID-19. These cytotoxic molecules were reduced due to a reduced proportion of CTLs. 21 On the other hand, a number of studies also describe Th0 cells are primed towards the Th17 phenotype, so have been reported a higher number of pro-inflammatory CCR6 + Th17 cells in peripheral blood, in severe cases of COVID-19, and thus indicating a potential role for Th17 cell-mediated immunopathology, including release of key cytokines such as IL-17 and GM-CSF. 22 Boechat et al, also showed that an increased Th17 signature may also be associated with elevated IL-6, an important cytokine in Th17 cell development. And thus, IL-17 produced by Th17 cells mediates the activation of monocytes/macrophages, dendritic cells and neutrophils, and increases the production of cytokines (IL-1, IL-6, IL-8, IL-21, TNF-α and MCP-1) by these cells, thus contributing to the cytokine storm. 23 From another perspective, effector T cell hyperactivation (and resulting exhaution) may be associated with the reported significant reduction in circulating regulatory T cells (Treg) in patients with severe versus moderate COVID-19. In some studies, exhaution of CD4+ and CD8+ T cells was shown. As a result, protein expression of three depletion markers. The CD28 family member programmed cell death marker 1 (PD-1), the receptor mucin domain-containing protein-3 (TIM-3) and the NKG2A receptor carrying ITIM. Were increased in both peripheral CD4+ and CD8+ T cells of patients with severe COVID-1. 24 Conversely, have been described a decrease in immunoregulatory subsets such as Treg and γδ T cells. 25
Impact of SARS-CoV-2 on disease progression
An adequate T cell response is crucial for controlling SARS-CoV-2 infection and reducing disease severity. CD8+ T cells destroy infected cells, limiting viral replication. CD4+ T cells play a key role in regulating the immune response, helping to modulate the inflammatory response and stimulating antibody production by B cells (Zeng et al., 2022). In individuals with a robust T-cell immune response, COVID-19 disease is often less severe. However, in some cases, an excessive immune response can lead to severe pulmonary inflammation (Mallajosyula et al., 2021)
T lymphocytes play an essential role in the immune response against SARS-CoV-2, controlling infection, reducing disease severity and contributing to long-term protection. Their activation and coordination are crucial for an effective immune response against COVID-19 (Niessl et al., 2021)
T cell responses against SARS-CoV-2 variants
T-cell responses against SARS-CoV-2 variants are an essential aspect of the immune response to COVID-19. T cells play a crucial role in the adaptive immune system and can recognize and target specific viral proteins. In the case of SARS-CoV-2 variants, T-cell responses can be influenced by mutations in the virus spike protein and other regions. In particular, variants such as Delta and Beta have mutations in the spike protein, which is the target of most neutralizing antibodies. Although these mutations can affect antibody recognition, T cells can still recognize other parts of the virus and remain effective in establishing immunity. Other, T cells can recognize several parts of the virus, not just the spike protein (Walensky et al., 2021). This broad recognition is advantageous, because even if a variant has mutations in the spike protein, T cells can still target other viral proteins. Tarke et all showed that in subjects 6 months after vaccination, 90% (CD4+) and 87% (CD8+) of memory T cell responses were preserved on average against the variants by the AIM assay, and 84% (CD4+) and 85% (CD8+) were preserved against Omicron (B.1.1.529) (Tarke et al., 2022).
Alterations of lymphocyte subsets
CD4+ T cell subsets, CD8+ T cells, play a central role in immune system function. Several reports have studied the different lymphocyte populations in SARS-CoV-2 infected individuals. There is evidence of functional alteration and increased expression of markers of activation and/or depletion by lymphocyte subsets. 26
CD4+ T-cell subset
First of all, CD4+ T cells regulate the response to acute and chronic viral infections. They coordinate the immune system mainly by secreting cytokines. CD4+ T cells activate many cells, such as B cells, cytotoxic CD8+ T cells and non-immune cells. CD4+ T cells also play a key role in establishing specific long-term cellular and humoral immunity. Naive CD4+ T cells differentiate into several subsets of effector cells, depending on their function and the cytokines released. The main subsets are T helper type 1 (Th1), T helper type 2 (Th2), T helper type 17 (Th17), follicular T helper cells, and T reg cells. Each has a specific function against viral infection. 27 So polyfunctional Th1 helper T cells that have been associated with protective antiviral immune responses. This is characterized by the expression of transcripts encoding polyfunctional-related cytokines such as IFN-γ, IL-2 and TNFα, and several other cytokines and chemokines such as IL-3, CSF2, IL-23A and CCL2. 28 Whereas Th2 cells, which produce IL-4, IL-5, IL-6, IL-9, IL-10, and IL-13, and evoke strong antibody responses (including those of the IgE class) and eosinophil accumulation, but inhibit several functions of phagocytic cells (phagocyte-independent inflammation). So upon viral infection with SARS-CoV-2, Th cells are switsh to the Th17 phenotype, resulting in the inhibition of Th1-mediated immune responses. 29
CD8+ T-cell subsets
After viral infection, CD dendritic cells that have taken up viral antigen stimulate activation of naive CD8+ T cells in the lymph node draining the lungs to induce robust virus-specific CD8+ T cell responses. The activation of these cells leads to their acquisition of two important functions: the production of inflammatory cytokines such as IFN-γ and TNF, and the process of degranulation, which can be assessed by measuring the expression of CD107a. Following respiratory virus infection, CD8+ T cells with specificity for the virus undergo activation and acquire an activated phenotype, along with effector functions. These activated cells show increased expression of effector molecules like granzyme B and perforin. After the infection is resolved, a subset of virus-specific CD8+ T cells persist in the host as long-lasting memory cells, providing protection against future infections. The contraction of CD8+ T cell numbers during this process, leading to the formation of memory populations in the lungs, is partly regulated by signaling through inflammatory chemokines. 30 CD8+ T cells play a crucial role in eliminating viruses by employing multiple effector mechanisms that induce apoptosis in infected cells. One of the ways CD8+ T cells achieve this is through direct cell-to-cell contact, utilizing surface molecules like Fas (CD95) and FasL (CD95L) to eliminate target cells. Furthermore, CD8+ T cells express TRAIL, which can interact with its DR4 and/or DR5 receptors, leading to the destruction of virus-infected cells. CD8+T cells can also secrete perforin and granzymes to cause membrane pore formation and induce apoptosis. Finally, CD8+ T cells produce inflammatory cytokines, such as IFN-γ and TNF, which can directly or indirectly promote cell death in virus-infected cells. 30 In the case of SARS-CoV-2 viral infection, patients in the intensive care unit (ICU) had low total CD8+ T-cell lymphocyte levels. After activation from CD8+ T cells can induce the activation and differentiation of corresponding B cells and subsequently promote the production of virus-specific antibodies. Subsequently, neutralizing antibodies are able to trigger antibody-dependent cell-mediated cytotoxicity to kill virus-infected cells and block virion entry. 31
T helper lymphocyte type 17 (Th17)
Naive CD4+ T cells differentiate into several subsets of effector cells, depending on their function and the cytokines expressed. The main subsets are T helper 17 (Th17) cells and regulatory T cells (Treg). Th17 cells are important for host defense against bacterial, fungal, and extracellular infections. In fact, in the immune system, Th17 cells have both a protective and pathogenic role. Therefore, the Th17 phenotype has been associated with chronic inflammation. These cells are characterized by the production of a wide range of cytokines, including IL-17A, IL-17F, IL-21, IL-22, granulocyte-macrophage colony-stimulating factor (GM-CSF), IL-10, and IFN-γ. In addition, IL-21 and IL-23 contribute to the formation of pathogenic Th17 cell. 28 The Th17 inflammatory response plays an important role in the pathogenesis of COVID-19 pneumonia. Several studies have demonstrated that elevated levels of Th17 cells in the periphery of SARS-CoV-2 infected patients, and subsequent signaling via IL-17 resulted in aggravation of the severe disease, either directly via the action of IL-17A signaling, or indirectly by inhibiting a Th1 or Treg response that would have induced viral clearance. 32
T reg
Treg cells regulate the activation of several lineages of the innate and adaptive immune systems through several effector mechanisms. Tregs are induced by Forkhead Box 3 (Foxp3) protein to maintain immunological homeostasis and produce anti-inflammatory cytokines TGF-β and IL-10 which play an important role in dampening hyperactive immune responses. In severe cases of COVID-19, several studies have been reported that a decrease in Treg numbers, indicates an insufficient regulation of pro-inflammatory immune responses that may further aggravate hyperinflammation and tissue damage. Furthermore, this decrease in Treg cells in COVID-19 cases may change the balance between the regulatory and effector parts of the immune system. This imbalance can lead to massive expansion and activation of neutrophils, macrophages, mast cells, dendritic cells, and Th17 cells, resulting in uncontrolled inflammatory responses mediated by innate immune cells. This imbalance may also contribute to tissue destruction. 32
Follicular helper cells (T FH)
Follicular helper cells (FH T cells) a specialized subset of CD4 + T cells that provide essential "helper" signaling to B cells, promoting the survival and differentiation of B cells into long-lived plasmocytes. Repeated interaction of antigen-specific FH T and B cells in the germinal center of B cell follicles is a critical step in the generation of high-affinity antibody responses. Phenotypically, T FH lymphoid tissue is characterized by chemokine receptor expression (e.g. CXCR5, PD-1, and expression of the transcriptional repressor Bcl-6). These cells provide related assistance to B cells via co-stimulatory molecules such as CD40L and indirectly via ICOS, as well as locally secreted cytokines including IL-21. 33 However, a subpopulation of circulating CD4+ T cells was present in the blood that was remarkably similar to FH T cell lymphoid tissue. These cells are called circulating TFH (cT FH) and, unlike TFH cells, are characterized by low or no Bcl-6 content, therefore expressing c-Maf, which can drive the expression of CXCR5 and IL-21 in the absence of Bcl-6. In addition the (cT FH) has a greater capacity to secrete IL-21 and IL-10. 34 Thus, during viral infection with SARS-CoV2, SARS-CoV-2 antigen in lymph nodes leads to activation of antigen-specific B cells and FH T cells. Their interaction leads to the initiation of the germinal center reaction. This leads to the development of memory B cells with increased affinity, as well as long-lived plasma cells that circulate to the bone marrow and provide a long-term source of neutralizing antibodies. At the same time, a population of activated cT FH (CD38 +, PD-1 +, ICOS +) appears in the circulation. This population contains antigen-specific cT FH cells. Activated cT FH cells also correlate with the development of neutralizing antibodies. These cT FH cells are a potential biomarker of FH T activity in lymphoid tissue. 33 In some cases of severe COVID-19, potential impairment of FH T cells has been reported. Specifically, B cells and FH T cells were reduced, and BCL6 expression in the germinal center of FH T cells was lost in lymphoid tissues in a subset of COVID-19 patients who died. In addition, one study found increased proportions of cytotoxic follicular helper cells in hospitalized patients compared with non-hospitalized patients. These cT FH cells expressed cytotoxicity-associated transcripts such as PRF1 and GZMB (encoding perforin and granzyme B respectively). Thus, several studies have shown that circulating T follicular (cT FH) is a robust correlate of the neutralizing antibody response 34
T γδ cells
γδ T cells are T cells that express a single T cell receptor (TCR) composed of a γ chain and a δ chain. These cells are sparsely distributed in the body, are found in the intestinal mucosa, skin, lungs and uterus, and are implicated in the initiation and propagation of immune responses. They have the ability to regulate the inflammatory response and promote tissue repair through the secretion of anti-inflammatory cytokines such as IL-10 and TGF-b. And play an important role in homeostasis and disease. In addition, γδ cells are highly cytotoxic and can contribute to the immune response against intracellular pathogens by directly lysing infected cells. 31 γδ T cells exhibit cytolytic effector functions on cells infected with a variety of pathogens. This function is associated with the expression of granulysin, granzyme B, and perforin, all of which are known to affect the viability of intracellular and extracellular pathogens. γδ T cells can perform a variety of functions, including the production and secretion of cytokines such as IFN-γ, TNF-α, IL-6, and IL-17.They can also perform cytolysis through the cytotoxic actions of perforin and granulysin, as well as interact with other immune cells such as B cells and dendritic cells. 35 In addition, lymphopenia affects γδ T cells but not αβ T cells. In some studies it was shown that COVID-19 patients characterized by lymphopenia had lower levels of γδ T cells. They appeared exhausted, which was detected by the PD-1 exhaustion marker. They also appeared to be highly activated, as detected by the activation markers CD25, CD69, and IL-18. However, γδ T cells produce large amounts of IFN-γ in response to COVID-19. 36
The development of memory T lymphocytes
Memory T cell in the COVID-19 patient
Immunological memory is mainly provided by cells of the adaptive immune system. In response to most severe viral infections, T cells with the ability to link to viral proteins through their antigenic receptors are activated, grow, differentiate and begin to secrete effector molecules to control the infection. Once the infection is resolved, approximately 90% of these virus-specific effector cells die, while 10% persist as long-lived memory cells.
37
Immune memory cells can produce a continuous number of effector molecules, such as the case of long-lived antibody-secreting plasma cells. On the basis of their localization properties and expression of adhesion molecules and chemokine receptors, memory T cells are classified into central memory (CM) and effector memory (EM) CD4+ and CD8+ T cells.
38
A small population of EM T cells take up CD45RA and are termed terminally differentiated effector T cells (T EMRA). These subsets differ in terms of proliferative response, cytokine production, effector properties and sensitivity to apoptosis.
39
In SARS-CoV-2 viral infection (Figure 1), several studies have reported that SARS-CoV-2-specific memory T cell responses are maintained over the long term. For example, in a study by Memory T cells in the COVID-19 patients. Après une infection par le SARS-CoV-2, certains lymphocytes T spécifiques au virus subissent un processus de différenciation en lymphocytes T mémoire. Ces cellules mémoire T conservent la capacité de reconnaître spécifiquement les peptides viraux présentés par les cellules infectées. Il existe deux principaux types de lymphocytes T mémoire: les lymphocytes T mémoire centraux (Tcm) et les lymphocytes T mémoire effecteurs (Tem). Les Tcm ont tendance à résider dans les ganglions lymphatiques et sont capables de se diviser pour produire de nouvelles cellules T lors d'une réinfection. Les Tem circulent dans la circulation sanguine et dans les tissus périphériques, prêts à attaquer rapidement en cas de réinfection.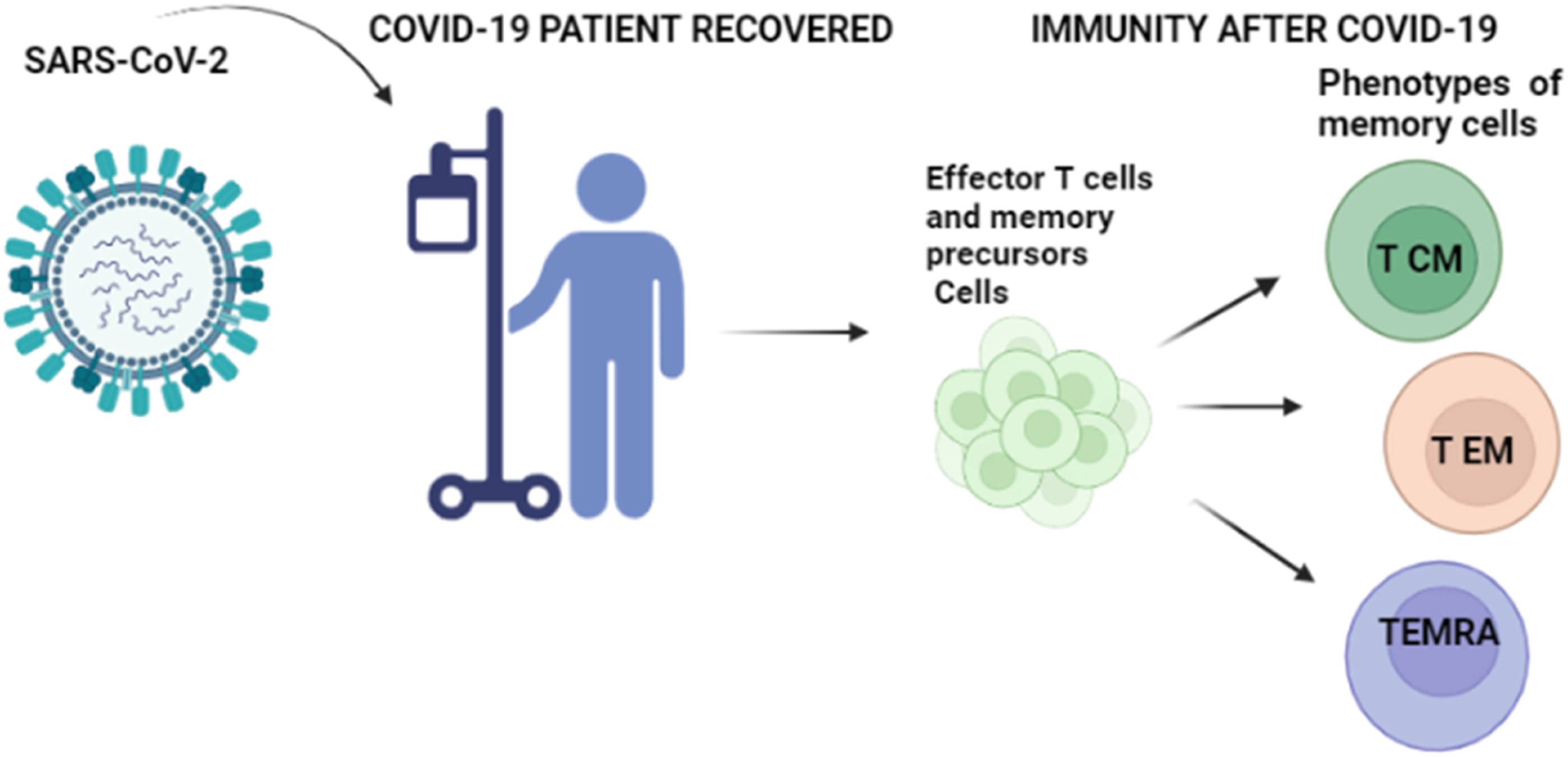
Specific T cells memory CD4+ elicited by different vaccines
Previous studies have reported COVID-19 vaccines can elicit robust T-cell memory after two dose and third dose of vaccination. The antigens-specific CD4+ T-cells memory were identified by activation-induced marker assay AIM (OX40+CD137+,OX40+CD40L+surface (sCD40)) and intracellular cytokine staining ICS for Cytokine (IFNγ, TNFa, IL-2), granzyme B (GzB). After vaccination with Pfizer-BNT162b2, memories CD4+ T-cells were detectable in 85%–100% of individuals followed 6 months after the second dose (Y. Zhang et al., 2022). With regard to the Moderna-mRNA-1273 vaccine, Spike-specific CD4+ T-cell responses peaked in 100% of responders after two doses of the vaccine and were successfully maintained for up to 6 months following post-immunization (Y. Zhang et al., 2022). Additionally, another study found that the third dose of Pfizer-BNT162b2 vaccine-induced increases in the median Interferon-gamma IFN-γ secretion in vaccinated-individuals and induced IFN-γ secretion in a fraction of seronegative patients (Re et al., 2022). Yorsaeng et all, Reported that 84% and 53% of vaccinated individuals to had CD4+ and CD8+ spike-specific memory responses, respectively, before the third dose (9 months after the second dose) (Yorsaeng et al., 2022). Furthermore, 2 weeks after the third dose, the same study found an increase in percentages to 100% for CD4+ and 90% for CD8+. Three months later, after the third dose, 97% of vaccinated individuals still harbored activation induced marker CD4+ T cells. These results suggest that as the immune response stabilizes 3 months to 6 months after the second dose, the third dose is needed to provide a booster response against SARS-CoV-2 (Naaber et al., 2022). It is important to note that the Moderna-mRNA-1273 vaccine induced higher frequencies of peak-specific memory CD4+ T cells than those observed in previously infected individuals, in contrast to the Pfizer-BNT162b2 vaccine, which induced similar frequencies of peak-specific memory CD4+ T cells as those in infected individuals. This shows that, despite the two-mRNA –based vaccines, if there is a difference in CD4+ memory T cells. CD4+ T cells differentiation produces a variety of functional subpopulations with diverse roles, including promoting B cell proliferation, modifying antibody subclasses, assisting CD8+ T cells, attracting innate immune cells, and promoting tissue repair. (Laidlaw et al., 2016). During viral infection, follicular helper cells (Tfh) typically differentiate by producing cytokines to exert antiviral activity (Wu et al., 2015).
According to one study, a low-dose of Moderna mRNA-1273 vaccine induced strong polarization of Tfh cells 6 months after the second immunization (Mateus et al., 2021). However, a recent study reported that 100% of vaccination recipients generated memory CD4+ T cells via conventional Tfh 6 months following the third immunization with Moderna mRNA-1273 and Novavax NVX-CoV2373 vaccines (Z. Zhang et al., 2022).
For Sinopharm/BBIBP-CorV, 5 months after the second dose, Yihao and all reported that vaccinated individuals had SARS-CoV-2-specific memory cell, the phenotypes were T central memory CD45RA−CCR7+ and CD45RA−CCR7- T effector memory, most importantly, decreased T naive cells, with an increase in effector memory and central memory cells in the peripheral blood (Figure 1), This phenotype T cells memory is similar to that from recovered COVID-19 patients (Liu et al., 2022). On the functionally side, the production of IFNγ by CD4+ T cells specific for SARS-CoV-2 shows a decrease over time until the third immunization. Furthermore, Sinopharm/BBIBP-CorV generated Spike-specific CD4+ by 5.9 folds at 1 week after a third dose. Despite the persistence response at 5 months after second dose, the third dose is needed to reinforce a booster response cellular memory (Liu et al., 2022). After receiving the AstraZeneca-ChAdOx1 nCoV-19, one study showed that Spike-specific T-cell responses were reinforced after a third dose and were more important than responses measured after two doses (Flaxman et al., 2021). Notably, T-cell responses boosted after a third dose (median response increased from 200 spot forming units [SFUs] per million peripheral blood mononuclear cells [PBMCs; IQR 127–389] immediately before the third dose to 399 SFUs per million PBMCs [314–662] by day 28 after the third dose (Flaxman et al. 2021). Additionally, heterologous primary immunization against SARS-CoV-2 with Pfizer-BNT162b2 and AstraZeneca-ChAdOx1 nCoV-19 vaccines elicits a stronger initial immune response than homologous vaccination with AstraZeneca-ChAdOx1 nCoV-19, including specific T-cell memory responses as measured on the basis of IFN-γ release after stimulation of fresh whole blood from individuals vaccinated, they found IFN-γ levels were 2.8-fold higher 2 weeks after ChAd/BNT, compared to after ChAd/ChAd vaccination (Marking et al., 2022), Moreover, the larger decline of IFN-γ responses was noted among recipients of heterologous ChAd/BNT at 3 months post-second vaccine dose (Marking et al., 2022). Seven days after the third dose of AstraZeneca vaccine, Schrezenmeier and all detected a significant increase in the frequencies of IL-4, and secreted polyfunctional CD4+ T cells of IFN-γ, TNFα, and IL-2 (Schrezenmeier et al., 2021).
The Ad26.COV2.S vaccine studies examined the immunogenicity of the vaccine as measured by interferon-quantiFERON levels. It was found that patients who received the Ad26.COV2.S booster dose had higher T-cell responses, as measured by interferon levels, than patients in the no booster single dose (Sablerolles et al., 2022). Zili et all demonstrated that at all time points (baseline, 1 week, 1 month, 3.5 months, and 6 months after the initial immunization (single dose), 100% of individuals generated spike-specific CD4+ T cells and were largely stable out to 6 months post-vaccination, as determined by activation-induced marker assays. They also demonstrated that 46% of individuals with spike-specific CD4+ T cells were detected by intracellular cytokine staining at all-time points (Z. Zhang et al., 2022). It is important the noted that peak CD4+ T cell responses were lower to Ad26.COV2.S than either of the mRNA vaccines. Notably, peak CD4+ T-cell AIM+ responses to Ad26.COV2.S were 2.2- to 3.3-fold lower than BNT162b2 and 3.5- to 4.2-fold lower than mRNA-1273 peak responses, similar to intracellular cytokine staining ICS IFN γ+ CD4+ T cell responses. Moreover, cTfh cells were detectable in the majority of individuals, this indicating memory CD4+ T cell frequencies were lower after Ad26.COV2.S immunization compared to mRNA vaccines and assessed as total spike-specific memory (AIM+), cTfh memory, IFNγ+ memory, CD4+ T cells with cytotoxic activity CD4-CTL memory,or memory CD4+ T cell multifunctionality (Z. Zhang et al., 2022).
Specific T CD8+ cells memory elicited by different vaccines
The elimination of intracellular viruses requires lysis of infected cells, T CD8+ cells induce apoptosis to lyse target cells (Wherry & Ahmed, 2004). Compared to others, TCD8+ cells have high-intensity cytotoxic capacity (Takata & Takiguchi, 2006). Otherwise, and they strongly express IFN-γ, perforin, granzyme B and CD107a (Kumagai-Takei et al., 2018). For patients vaccinated against SARS-CoV-2, the COVID-19 multi-platform vaccine can elicit a robust CD8+ T-cell immune response specific for SARS-CoV-2 proteins. To know the immunogenicity of the third "booster" dose on CD8+ T-cell response was determined the T-response by different techniques such as T-ELISPOT assays, quantiFERON (QFT), activation-induced-marks (AIM), and intracellular cytokine staining (ICS). According to several studies, individuals who received different COVID-19 vaccines showed short-term persistence of pre-existing CD8+ T-cell memory after a booster vaccination (Schrezenmeier et al., 2021; Sette & Crotty, 2022). In line with ELISPOT IFNγ, Spike-specific CD4+ and CD8+ T cells increased significantly, by 5.9 and 2.7 folds, respectively, for Sinopharm/BBIBP-CorV vaccines who were followed up 1 week after the third dose compared to vaccines that were followed up 3 months (12 weeks) after the second dose (Liu et al., 2022). Additionally, IFNγ expression in CD8+ T cells was measured by flow cytometry. CD8+IFNγ+ T cells were detected on 3 months after second immunization, and decreased over time till the third vaccination, which elevated IFNγ-expressing CD8+ T cells to a greater extent when compared to that on 3 months after two dose (Liu et al., 2022). CD45RA is predominantly expressed on naive or resting T cells, which have not yet encountered their specific antigen (Appay et al., 2008). Phenotypic markers revealed that the majority of the SARS-CoV-2-specific CD8+ T cells were CD45RA+CCR7– terminally differentiated effector memory cells (TEMRA), whereas CD45RA–CCR7– effector memory T cells (TEM) accounted for only one-fourth of the total antigen-specific CD8+ T cells (Liu et al., 2022),similar to that from recovered COVID-19 patients (Sette & Crotty, 2022). On another side, the expression of 4-1BB on immune cells is often used as a marker to identify activated or antigen-specific T cells (Vinay & Kwon, 2011). SARS-CoV-2-specific CD8+ cells were identified by activation-induced markers (AIM) T cell assay, that indicates CD8+CD69+4-1BB+ T cells were seen on 5 months after the second dose, and further elevated by 2.7 folds following a booster shot, such as ELISPOT test T cells assays, demonstrated the IFNγ-secreting T cell responses enhanced by 2.3 folds after the third dose (Liu et al., 2022). A study examined immune responses spike-specific to SARS-CoV-2 longitudinally for 6 months by Moderna-mRNA-1273, Pfizer/BioNTech -BNT162b2, Janssen Ad26.COV2.S and Novavax NVX-CoV2373 vaccines, utilizing previously described flow cytometry intracellular cytokine staining (ICS) and activation-induced marker (AIM) assays (CD69+CD137+), that indicate memory CD8+ T cells were only detectable in 60%–67% of subjects at 6 months (Z. Zhang et al., 2022). This resulted indicate while memory T cells were comparatively stable over 6 months. Intracellular cytokine staining is used to identify IFNγ, TNFα, and IL-2-producing cells in individuals who have received two doses of the COVID-19 vaccine (30–32). Notably, they demonstrated the increased levels of IFNγ+ TNFα, and IL-2 producing T CD8+ cells at 1 month after two doses of the vaccine. Additionally, the memory CD8+ T cells maintained out to 6 months following BNT162b2 vaccination. This result is similar between mRNA-1273 and Ad26.COV2.S immunizations (Guerrera et al. 2021; Y. Zhang et al. 2021). It is important the noted, among individuals with detectable CD8 T cell memory to Mrna vaccines after immunization, the magnitude of the memory is generally observed to be low up to 6 months (Guerrera et al., 2021). For this reason, was necessary the third dose of vaccines. Also, they found the antigen-specific population of steam cellular memory TSCM, which persisted over time notably, CD8+ TSCM followed, with 94, 87, and 96% of individuals showing these cells at one the day after the first immunization, 2 weeks after the second immunization, and 6 months after the first immunization (Guerrera et al., 2021).
After receiving a single dose of Ad26.COV2.S vaccine, it was observed that CD8+ T-cell responses expressing IFNγ and IL-2 continued to increase from 1 to 8 months after vaccination (Le Gars et al., 2022). This increase was measured using the enzyme-linked immune absorbent spot (ELISPOT) and intracellular cytokine staining (ICS) techniques for IFNγ and IL-2. Additionally, the individuals vaccinated generated CD8+ central memory (CD45RA−CD27+) and effector memory (CD45RA−CD27−) T-cell subpopulations elicited by Ad26.COV2.S was durable through at least 8 months after a single dose of Ad26.COV2.S. For the reason a homologous or heterologous booster dose elicits cellular immune responses that provide durable protection against SARS-CoV-2 infection (Le Gars et al., 2022). The Participants vaccinated with AstraZeneca vaccine showed a significant decrease in the number of S1-specific cytokine-producing CD8+ T cells between 8 and 12 weeks after receiving two doses of inactivated vaccines, compared with the interval 4 to 8 weeks after vaccination (Swanson et al., 2021). Cytokine-secreting CD8+ T-cell responses were also reduced 28 days after booster vaccination with AstraZeneca vaccine in the 8-12 weeks interval compared with the shorter interval. This may be explained by a reduction in effector T-cell responses prior to booster vaccination. Consequently, a third booster dose is required (Munro et al., 2021).
Pathophysiological and immunological characteristics of COVID-19
Strom-Cytokine
Various cytokines are secreted by innate and adaptive immune cells during viral infection which acts as a defense mechanism against the virus.
47
Recent studies in COVID-19 patients have shown that high levels of pro-inflammatory cytokines which include (interleukins, interferon and chemokine) are produced in an unregulated way by innate and adaptive cells. Including macrophages, neutrophils, DCs, and NK, B and T cells. There are numerous reports that individuals with COVID-19 may have syndromes identified as a cytokine storm. This is considered an important contributor to acute respiratory distress syndrome (ARDS) and multiple organ dysfunction syndrome (MODS).
48
Some studies have been shown elevated levels of chemokines and cytokines, such as CCL2/3/5, CXCL8/9/10, and IFN-γ, TNF-α, IL-1β, IL-1RA, IL-6, IL-7, IL-8, IL -12, IL-33, granulocyte colony-stimulating factors/granulocyte-macrophages (G-CSF and GM-CSF), vascular endothelial growth factor A (VEGFA), facilitate the recruitment of other leukocytes to tissues and promote effector functions leading to severe ARDS and tissue damage (Figure 2). Vaccine-induction-memory T cells in humans. Generally, vaccination leads to the activation of dendritic cells DCs and draining to lymph nodes where DC present processed antigens and co-stimulate T naïve CD45RA+, CCR7+, CD62L+, CD27+, CD4+, CD8+ T lymphocytes (Foged et al., n.d.). After a two or three dose COVID-19-vaccines, Ag-SARS-CoV-2-specific T cells become activated, CD45RA+ or CD45RO+, CCR7−, CD62L−,and CD27− effectors that divide rapidly, acquire cytolytic capabilities, secrete IL-2 and the antiviral cytokines IFN- and TNF-, provide B cell help, and traffic to sites of infection. After the invading microbe has been cleared, more than 90% of the effector T lymphocytes die via apoptosis whereas a few effector lymphocytes go on to express high levels of Bcl-2 and become long-lived CCR7− T effector-memory (TEM) or CCR7+ T central-memory (TCM) lymphocytes.
Therefore, the cytokine storm plays a critical role in the disease aggravation process. It has also been reported that increased levels of pro-inflammatory cytokines (e.g., IL-4 and IFN-γ) are also observed in COVID-19 patients. It has been found that COVID-19 patients are characterized by a significant decrease in the number of CD4, CD8+ T cells, but increases in the number of neutrophils. Loss of T cells may result in increased inflammatory responses, whereas restoration of T cells may reduce inflammatory responses during SARS-CoV-2 infection, indicating that cytokine expression may not be the primary cause of impaired lung function in COVID-19 patients. Dynamic cytokine storms and T-cell lymphopenia are associated with the severity of COVID-1. 49
T-Cell exhaustion
T-cell induced adaptive immune responses are essential for viral clearance and long-term antiviral immunity, but they may participate in the cytokine storm and may be impaired in severe COVID-19 patients due to T-cell exhaustion. Thus, sustained expression of inhibitory immune checkpoints in response to persistent antigenic stimulation may result in progressive loss of effector function. A number of studies show evidence suggesting that T cells in COVID-19 patients may have a depleted phenotype. Kusnadi et al, showed that SARS-CoV-2 reactive CD8+ T cells express programmed cell death protein (PD-1), CD38 and human leukocyte antigen DR (HLA-DR) isotype, which are markers related to T cell activation and exhaustion. It was found that the proportion of SARS-CoV-2 reactive CD8 + T cells expressing PD-1 was not significantly different between patients with severe versus mild disease. Furthermore, recent reports from patients with COVID-19 have suggested the presence of depletion-related markers in global CD8+ T cell populations based on the expression of a few co-inhibitory receptors, including PD-1. In other study, it was reported that in COVID-19 patients of severe group characterized by expression of high levels of PD-1, CTLA-4, TIGIT, granzyme B, compared to mild group. While CD4 + T cells with low levels of IFN-γ, IL-2 and TNF-α were higher in severe COVID-19 patients compared to healthy controls and mild patients.
50
This indicates that SARS-CoV-2 infection may result in functional impairment of CD4 + T cells and maintain excessive activation of CD8 T cells.
Potential therapeutic approaches related to T cells
Vaccine-induced T cell responses
Vaccines are an essential new arm in the control of COVID-19. As of December 12, 2020, according to the WHO list, there are more than 200 candidate vaccines against Sars-CoV-2 infection in development, of which 48 have entered clinical phase. For the vaccines selected by Morocco and as for all vaccines in other countries, rare mild to moderate adverse events (pain at the site of infection, skin reaction, headache …). The interim results of the clinical trials confirmed that these adverse events were transient. Interestingly, in clinical studies, COVID-19 vaccines were shown to elicit not only neutralizing antibodies but also SARS-CoV-2 specific T cell responses 1
Immunogenicity of SARS-CoV-2 specific T cells induced by different vaccines.

Adoptive therapies based on T cells
The use of virus-specific T cells against SARS-CoV-2 appears to be a logical therapeutic approach to treat COVID-19. These T cells are able to recognize various viral antigens, including structural antigens such as membrane proteins. It has been previously found that virus-specific T cells can be isolated from convalescent donor PBMCs, cultured in the presence of IL-4 and IL-7, and expanded with SARS-CoV-2-derived peptides and exploited to treat severe cases of COVID-19, and to restore effective antiviral immunity.
Recombinant cytokines
Naturally, IL-7 can enhance the diversity of the T cell receptor repertoire, induce naive and memory T cell proliferation, and increase the circulating pool (CD4 + and CD8 + T cells) that transfer T cells to the site of infection. 52 For these reasons, clinical trials to evaluate the efficacy of recombinant IL-7 administration to restore lymphocyte counts in COVID-19 patients in particular, recombinant IL-7 administration may improve T cell receptor diversity and generation of memory CD8+ T cells, promote T cell transport to the lungs, attenuate lymphopenia. IL-2 is a growth factor. However, its major role is antagonistic, as it induces homeostasis and regulatory T-cell functions that limit the action of effector cells. Therefore, the administration of low doses of recombinant IL-2 in COVID-19 patients should resolve lymphopenia and restore normal T-cell numbers. The safety of low-dose recombinant IL-2 and its efficacy in Treg expansion and activation in patients with autoimmune diseases have been reported. 19
Immune checkpoint inhibitors
Naturally immune checkpoints are expressed throughout the immune system and are primarily responsible for modulating the immune response. As mentioned above, SARS-CoV-2 infection caused T cell exhaustion by increasing the expression of inhibitory immune checkpoints (ICs), such as programmable cell death-1 (PD-1), T cell immunoglobulin and mucin domain-3 (TIM-3) and T cell immune receptor with Ig and ITIM domains (TIGIT) on the surface of T cells. Importantly, the symptomatic COVID-19 patient is characterized by increased expression of PD-1 and TIM-3. Therefore, targeting IC immune checkpoints may have therapeutic potential in COVID-19 patients. 53 There are monoclonal antibodies (mAbs) that are used to recover cell-mediated immunocompetence. In one study monoclonal antibodies were used to recover cell-mediated immunocompetence by targeting T cell exhaustion pathways. Four clinical trials were designed to evaluate the safety and therapeutic efficacy of the anti-PD-1 monoclonal antibody (mAb) in patients with COVID-19 (NCT04268537, NCT04333914, NCT04356508 and NCT04413838). However, results from preclinical models have shown that targeting the PD-1/PD-L1 axis can result in potentially deadly. It has been combined the anti-PD-1 mAb with other treatments, to avoid any potential risk of severe inflammation, especially minimize the immunopathology caused by the cytokine storm, such as anti-IL-6 receptor mAb or anti-IL-1 receptor mAb. For this it is encouraging to examine the effect of combination therapy, anti-PD-1 with anti-IL-6R or anti-IL-1 receptor mAb in COVID-19 patients. Targeting PD-1 in SARS-CoV-2 infected patients induces the slowing of T-cell exhaustion to induce more potent antiviral responses mediated by effector T-cells and cytotoxic CD8+, thus developing functional memory T-cells for long-term immunity. 54
Th17 blockers
IL-17 plays a key role in the cytokine storm seen in ARDS of any cause and is associated with alveolar inflammation and poor prognosis. In severe versus nonsevere COVID-19 cases, different studies have shown increased levels of IL-17-regulated cytokines, including IL-6, monocyte chemoattractant protein-1 (MCP-1), IL-8, granulocyte colony-stimulating factor (G-CSF), macrophage inflammatory protein (MIP)-1-α, and TNF-α; however, IL-17 was increased only in severe cases compared with nonsevere controls. On this basis, monoclonal antibodies directed against IL-17A (secukinumab and ixekizumab) and IL-17r (brodalumab) have been used as a strategy to stimulate antiviral responses and decrease the exacerbated pulmonary inflammation leading to acute respiratory distress syndrome in severe acute respiratory syndrome coronavirus (SARS CoV-2) infections. 55 It has been suggested that severe COVID-19 disease is associated with elevated levels of IL-6, on this basis it was used a monoclonal antibody directed against inteleuine-6 (tocilizumab). When tocilizumab administration was used to control the high levels of interleukin-6, and resulted in a decrease in mortality in COVID-19 patients.56–58,60–64
Conclusion
The relationship between SARS-CoV-2 infection and lymphocyte T cells is an important area of study in understanding the immune response to the virus. Lymphocyte T cells, specifically CD4+ helper T cells and CD8+ cytotoxic T cells, play a crucial role in the adaptive immune response by recognizing and eliminating infected cells. Other hand,It is important to note that the impact of variants on the effectiveness of the immune response is not uniform and depends on the specific features of the variants themselves. Some variants can partially evade the immune response, while others can be effectively neutralized. For this reason, researchers and vaccine manufacturers are closely monitoring the evolution of variants, and adjusting COVID-19 vaccines and control strategies as necessary to take account of these changes. The limitation of this study is Knowledge about COVID-19 is evolving rapidly, and some studies may become obsolete as new information emerges.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
