Abstract
Demands for higher quantity and quality of sequence data during genome sequencing projects have led to a need for completely automated reagent systems designed to isolate, process, and analyze DNA samples. While much attention has been given to methodologies aimed at increasing the throughput of sample preparation and reaction setup, purification of the products of sequencing reactions has received less scrutiny despite the profound influence that purification has on sequence quality. Commonly used and commercially available sequencing reaction cleanup methods are not optimal for purifying sequencing reactions generated from larger templates, including bacterial artificial chromosomes (BACs) and those generated by rolling circle amplification. Theoretically, these methods would not remove the original template since they only exclude small molecules and retain large molecules in the sample. If the large template remains in the purified sample, it could understandably interfere with electrokinetic injection and capillary performance. We demonstrate that the use of MagneSil® paramagnetic particles (PMPs) to purify ABI PRISM® BigDye® sequencing reactions increases the quality and read length of sequences from large templates. The high-quality sequence data obtained by our procedure is independent of the size of template DNA used and can be completely automated on a variety of automated platforms.
Keywords
Introduction
Most DNA sequencing facilities have chosen to automate as many components of their process as possible. Purification after the sequencing reaction is an important step to consider automating, since it is time-consuming and the efficiency of this step is critical in influencing the quality of the sequence trace. Components of sequencing reactions can interfere with data collection, and it is helpful to remove them prior to electrophoretic analyses. Salts can give rise to abnormal migration patterns, and unincorporated dye-labeled terminators can interfere with the collection of accurate sequence information. Therefore, efficient purification of samples prior to electrophoresis can dramatically increase the quality and quantity of sequence information, thus allowing more efficient extraction of data from every sample run. While substantial improvements have been made to increase the throughput and automation of template DNA preparation, sequencing reaction, and analysis of data, many protocols still rely on cumbersome and labor-intensive procedures for purifying samples prior to electrophoresis. 1
Traditional methods of purifying sequencing reactions rely on precipitation with ethanol or gel filtration to remove excess dye-terminator nucleotides, excess salts, and other interfering molecules. 1 These purification methods can result in difficulties during purification and analysis of BAC templates because of the large physical size and, therefore, increased mass of DNA typically required in sequencing reactions. For example, purification by alcohol precipitation removes unincorporated dye terminators and concentrates the large mass of template DNA in BAC sequencing reactions. This can lead to significant amounts of template being retained in the purified product, ultimately affecting sample analysis. The size and mass of BAC DNA in sequencing reactions can also result in physical clogging of limited porosity filter systems during purification. Precipitation and filtration methods are also problematic for automated approaches, often requiring multiple centrifugation or vacuum drying steps during purification. 1 More recently, commercially available paramagnetic particles (PMPs) have been used for purifying sequencing reactions. 2 However, many systems using PMPs either do not offer the throughput or performance desired or require specialized primers for purification. Most importantly, many standard and magnetic systems do not remove the original large template from the purified reaction, an important consideration when using larger sequencing templates such as BACs or rolling circle-generated templates.
Promega's MagneSil® Sequencing Reaction Clean-Up System uses a composition of silica-coated PMPs that has been optimized for dye terminator purification. 3 We demonstrate the use of MagneSil® PMP purification of BigDye® sequencing products generated from BAC and rolling circle amplified templates. Samples are purified in a 96-well format without user intervention in a process that can be adapted to a variety of automated platforms and throughputs. The rapid magnetic response characteristics of MagneSil® as a mobile solid phase generate a purification system in a completely automated and scaleable format that is capable of purifying away the large quantity of template DNA from sequencing reactions containing large templates without using modified sequencing primers.
Materials and Methods
Preparation of DNA Templates
BACs and P1-derived Artificial Chromosomes (PACs) used as templates here were of two varieties. Full-length clones containing human DNA inserts ranging in size from 120 to 160 kb were isolated at ResGen (Invitrogen) or Genome Systems (Incyte). The construction of clones containing nested deletions created in BACs and PACs using a loxP transposon retrofitting procedure has been described elsewhere. 4 Full-length BACs and PACs were sequenced using the standard end-sequencing primers SP6 and T7L, while nested deletion clones were sequenced with primers Seq 1 and either SP6 for BACs or T7L for PACs. 5 BAC template DNA was purified from bacterial cultures using either the Promega Wizard® Plus Midipreps System following the manufacturer's protocol (Promega; Madison, WI) or a modification of the Qiagen Tip plasmid purification method (Qiagen; Valencia, CA). 6 For TempliPhi™ reactions (Amersham Biosciences; Piscataway, NJ), dilutions of Promega pGEM®-3Zf(+) (1 ug/uL to 1 ng/uL) were amplified for 4 h using the manufacturers instructions.
Cycle Sequencing
For BAC template BigDye® Terminator-based fluorescent sequencing, reactions were set up in 96-well MicroAmp reaction plates (PE Applied Biosystems; Foster City, CA). Each sequencing reaction was 30 μL in total volume and contained 16 μL v1.0 BigDye® Terminator Ready Reaction mix (PE Applied Biosystems), 2 to 4 μg of BAC clone DNA template, and 16 pmol of primer. Cycling conditions: Initial denaturation at 95 °C for 5 min, followed by 50 cycles of 95 °C for 30 s, 50 °C for 30 s, and 60 °C for 4 min.
For TempliPhi™ sequencing, 2 μL of amplified DNA was added to 8 μL of Dnase/Rnase-free water and 2 μL (3.56 pmol) of Promega pUC/M13 primer was added. Added to each sample for cycling were 8 uL of v1.0 BigDye® Terminator Ready Reaction Mix. Cycling conditions: Initial denaturation at 95 °C for 2 min, followed by 25 cycles of 95 °C for 30 s, 50 °C for 15 s, and 60 °C for 4 min.
Sequencing Reaction Cleanup
All purifications were performed on a Beckman Biomek® FX (Beckman Coulter; Fullerton, CA). For purification of BAC sequencing reactions, the entire sample from identical sequencing reaction plates was processed using either MagneSil® or an alternative magnetic particle technology following the manufacturers' protocols. For purification of TempliPhi™ reactions, each reaction was divided in half and each half was processed using MagneSil® or a 96-well format using size-exclusion technology following the manufacturers' recommendations.
For MagneSil® purification, sequencing reactions were processed by adding 180 μL of MagneSil® GREEN particles and incubating at room temperature with mixing to bind sequencing products. Bound complexes were washed 2 times with 90% ethanol by sequential capture and resuspension using the MagnaBot® II magnet array (Promega) and dried briefly at room temperature. Purified nucleic acids were eluted by adding 10 μL of water and recovering the eluted material.
For size exclusion purification of TempliPhi™ reactions, 20 μL of water was added to each sample, and samples were mixed by pipetting in a 96-well format. Samples were transferred to a 96-well membrane filter platform, and vacuum was applied (15–18 inches Hg) until total volume passed through the membrane. Wells were washed with 25 μL of water and vacuum filtered for 6 min. Purified products were eluted by adding 25 μL of water.
Analysis
Samples were analyzed on an ABI 3700 with the following conditions: inject, 50 s, injection voltage 1000 V, run, 10,000 s run voltage 5250 V. Sequencing results were analyzed and signal strengths were determined using ABI Sequencing Analysis 3.7 (PE Applied Biosystems), and Phred scores were generated using the Trace Tuner software package (Applera; Norwalk, CT).
Results and Discussion
The MagneSil® Sequencing Reaction Clean-Up System uses paramagnetic particles for automated purification of dye terminator sequencing reactions prior to electrophoresis. This system has been shown to yield high-quality sequence information when used with plasmid templates. 3 It was of interest whether this technology would be able to overcome technical difficulties others and we have encountered with other systems when purifying sequencing reactions generated from large templates.
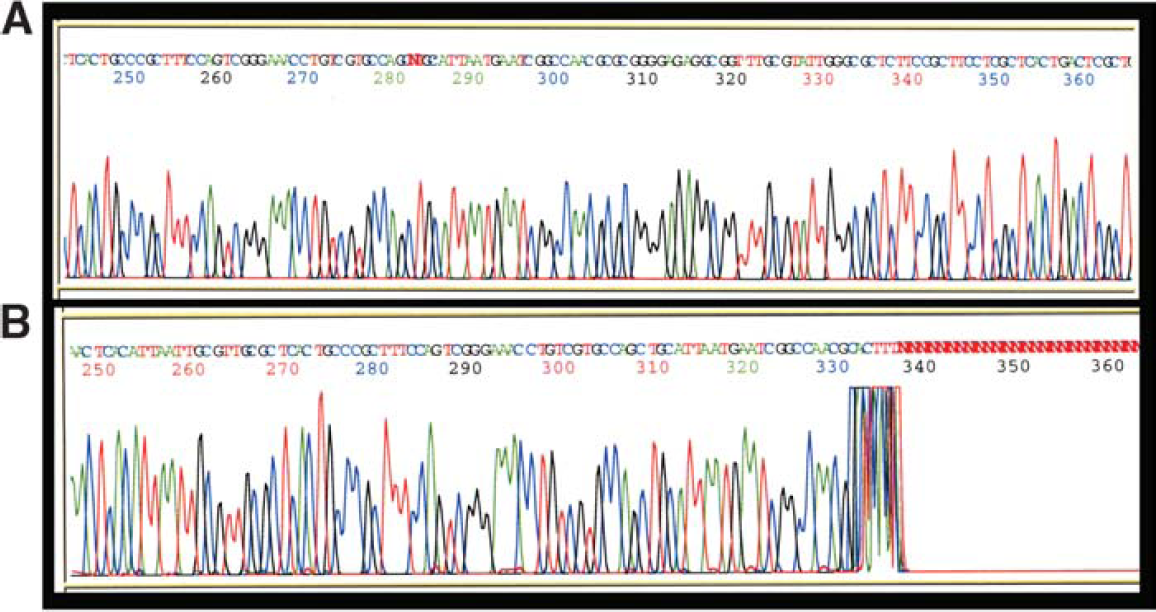
When identical 96-well plates of BigDye® sequencing reactions of BAC templates were subjected to purification using either MagneSil® or an alternative magnetic particle technology, overall sequence quality was good with both methods (Figure 1). However, while one MagneSil®-purified sample generated dropouts and another had abrupt stops in the sequence output (approximately 2%), more than 19% of the samples purified with the alternative magnetic bead technology demonstrated such problems (Figure 2). Average PHRED 20 sequence lengths were also more than 35% longer (average QV > 20 of 704 bases for MagneSil® vs. 452 bases for size exclusion) when using MagneSil® (Table 1). Overall signal strengths were similar using the two purification methods. A summary of our comparisons is presented in Table 1.
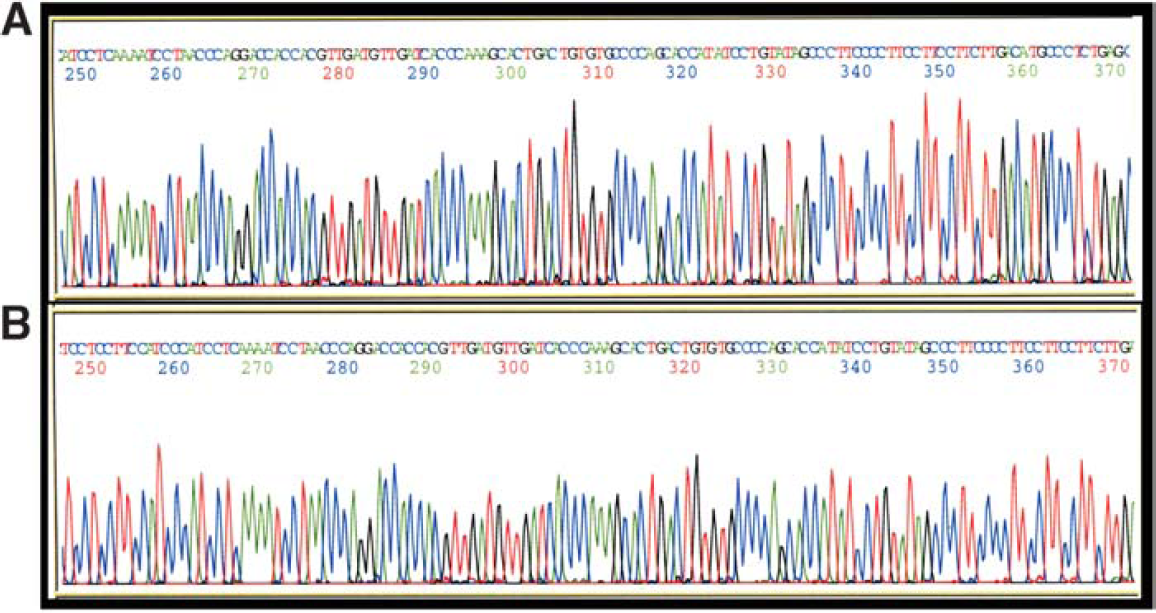
Comparison of sequence data generated from MagneSil® and an alternative magnetic particle purification of BAC sequencing reactions. Sequencing reactions were assembled using a BAC target and BigDye® sequencing chemistries as outlined in “Materials and Methods.” Panel A: sequencing reactions were purified using MagneSil Sequencing Clean-Up System prior to analysis. Panel B: sequencing reactions were purified using an alternative magnetic particle technology.
MagneSil® purified samples had fewer dropouts and abrupt stops than other magnetic particle purified samples with BAC sequencing reactions. Sequencing reactions were assembled using a BAC target and BigDye® sequencing chemistries as outlined in “Materials and Methods.” Panel A: sequencing reactions were purified using MagneSil® Sequencing Clean-Up System prior to analysis. Panel B: sequencing reactions were purified using an alternative magnetic particle technology.
Summary of BAC and TempliPhi™ sequencing purification quality using MagneSil® and commercially available PMP and gel filtration methods
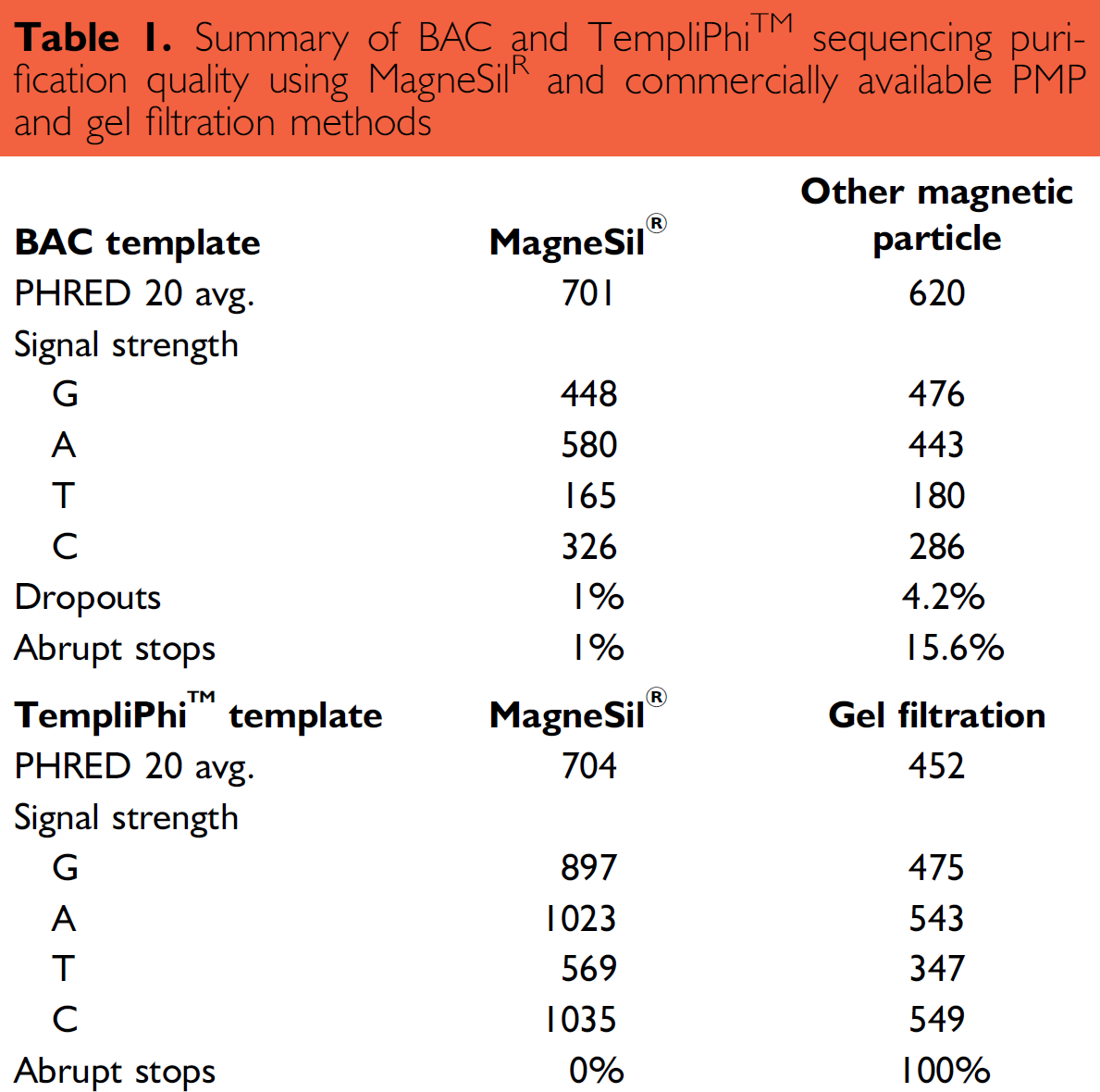
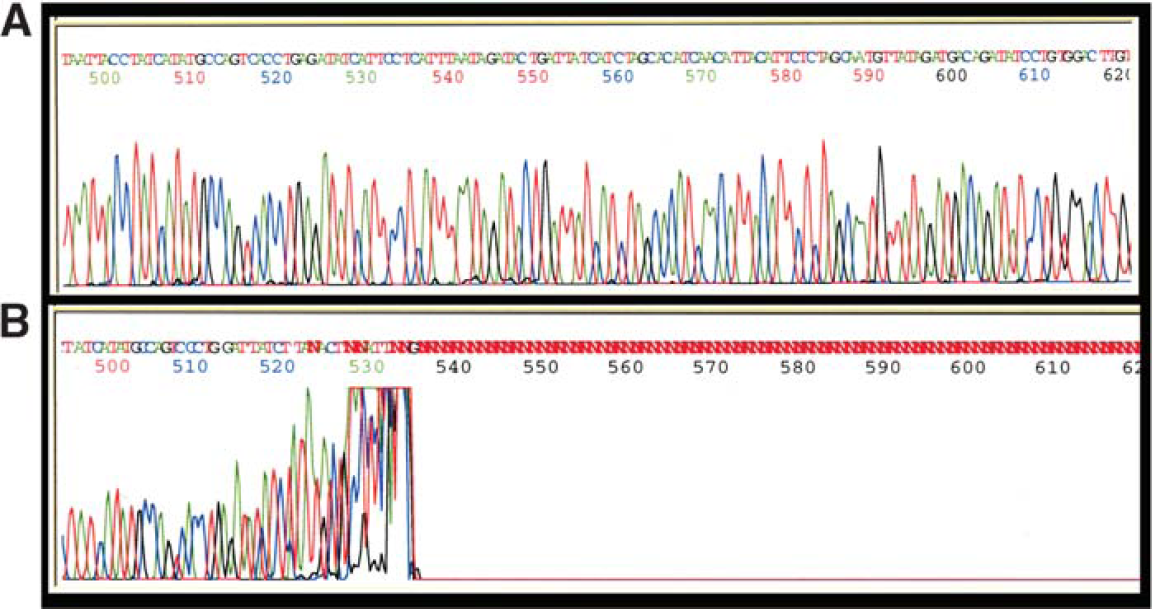
Rolling circle amplification is an alternative to traditional plasmid template preparation for generating sequence-ready DNA. 7 We compared MagneSil® and a plate based filtration method for purification of sequencing reactions prepared from rolling circle templates as outlined in MATERIALS AND METHODS. All samples purified using gel filtration exhibited abrupt stops in a moderately GC rich region of the target, while MagneSil® purified samples gave long reads (Figure 3). Identical samples purified using the filtration plate suffered from decreased signal strengths, shorter read lengths, and poorer sequence quality as measured by a 36% reduction in average PHRED 20 scores during analysis (Table 1).
Comparison of MagneSil® and Gel filtration sequencing purification of TempliPhi™-generated samples. Sequencing reactions were assembled using a TempliPhi™ target and BigDye® sequencing chemistries as outlined in “Materials and Methods.” The region shown is slightly GC-rich. Panel A: sequencing reactions were purified using MagneSil® Sequencing Clean-Up System prior to analysis. Panel B: sequencing reactions were purified using a commercially available gel filtration technology.
A potential concern is the loss of MagneSil® PMPs during automated processing. Careful positioning of the PMPs is important to eliminate loss during processing, to avoid carryover of particles during elution, and to maximize elution efficiencies in small volumes (typically 5–15 μL). Promega has developed a magnet specifically designed for the 96-well dye terminator sequencing purification presented here (the MagnaBot® II). This design uses an array of neodymium magnet rails to maximize field strength and positioning of the PMPs during processing. Magnet size and positioning, coupled with an aluminum base, serves to provide a uniform magnetic field across all samples at an optimal geometry for PMP capture. This magnet is designed to purify sequencing reactions directly in thermocycler plates, and an accessory adapter is available that allows liquid handlers with gripping capabilities to process skirted and unskirted 96-well plates. A newly developed 384-well magnet is also available.
Conclusion
The use of MagneSil® particles allows completely automated purification of dye terminator sequencing reactions prior to electrophoresis. When coupled to the throughput capabilities of the Biomek® FX, this system is capable of processing large numbers of samples per hour. The use of a dual POD Biomek® incorporating two 96-well pipetting heads or a second 384-well pipetting head increases the throughput and flexibility of this technology further by simultaneously processing multiple 96- or 384-well plates or by arraying samples from a 384 sequencing run into individual 96-well plates for data collection and analysis. The use of MagneSil® clearly demonstrates a practical advantage over the use of ethanol precipitation and the labor associated with centrifugation of spin columns.
Large templates derived from BACs/PACs or rolling circle technologies are extremely valuable as templates for direct sequencing. We show here that using MagneSil® in dye terminator sequencing purification leads to higher quality data, generating longer reads and better signal strengths when compared to an alternative magnetic bead method or a 96-well gel filtration plate. The increase in data quality seen when using large templates after purifying with MagneSil® has led us to conclude that this procedure efficiently removes the BAC template during the purification process.
The improved accuracy of sequence reads obtained directly from large templates such as BACs and PACs may render unnecessary the subcloning of fragments from large genomic clones as a means for obtaining good sequence. The modifications described here should greatly facilitate gap-filling and other chromosome walking experiments where direct sequencing of BAC/PAC templates becomes necessary. From the standpoint of a full-service core facility, the use of MagneSil® to purify templates from rolling circle amplification templates decreases template processing time fivefold and increases sequence read quality substantially. The combination of the two methods saves an enormous amount of time and money invested in growing bacterial cultures, harvesting DNA, and resequencing. Since resequencing is, by far, the most overlooked and undercalculated cost in sequencing, the fact that MagneSil® gives optimized post-sequencing purification results from large templates should be seriously considered.
Acknowledgment
W.W., R.C.G., D.J.G., and P.K.C. were supported in part by a Minority Biomedical Research Support—Support of Continuous Research Excellence grant #SO 608049 from the National Institute of General Medical Sciences, grant #1U56 CA92077–01 from the National Cancer Institute, and EXPORT grant #1P20 MD00175–01 from the National Institutes of Health. We thank Drs. Ken Harewood and Nancy Shepherd for encouragement and support.
