Abstract
Microwave-assisted organic chemistry has received considerable attention during the last decade and nowadays, more and more research chemists are applying microwave technology to organic reactions on a small scale successfully. However, efficient application of this technology to cover the specific needs of larger-scale preparations, e.g., in a kilolab, remains to be shown. Therefore, the current study was initiated to investigate the scalability of the microwave technology. Two different microwave systems designed for large-scale operation (Multiwave 3000 and CEM Voyager SF) were evaluated to characterize strengths and weaknesses of each instrument for special use in a kilolab with focus on temperature/pressure limits, handling of suspensions, ability for rapid heating and cooling, robustness, and overall processing time. (JALA 2005;10:412–7)
Keywords
Introduction
Microwave-assisted synthesis
1
offers various advantages. Two of these are the potential for dramatically shorter reaction times and access to reaction conditions, which are not attainable with conventional thermal heating, offering the opportunity to save time in the lab and to increase productivity.
2
Research chemists at Novartis Institutes for BioMedical Research (NIBR) are therefore increasingly using microwave reactors to carry out chemical reactions on a small scale. In recent years, several microwave devices for use in medicinal chemistry have been installed at NIBR and even more will probably be acquired. Therefore, it might be assumed that this new technique is going to become more and more a standard technology in research labs at NIBR because it is very successful for small-scale organic synthesis, in particular for the rapid optimization of reaction conditions. Microwave devices, like the Emrys Optimizer
3
(former Personal Chemistry, Uppsala, Sweden, now Biotage), are commonly used for synthesis on a small scale (1–3 g) and up to 15 mL reaction volume. The application of microwave technology in research labs offers the following advantages compared to conventional heating by an oil bath:
Temperatures up to 250 °C and pressures up to 20 bar are reachable in closed vessels. Boiling point of the solvent is not a limiting factor any more, therefore the reaction time can be reduced significantly. Vials (12 × 20 mL) can be processed sequentially in an automated manner reducing the processing time and resulting in a more efficient workflow.
Examples from the literature 4 and NIBR Global Discovery Chemistry show that in the context of the drug discovery process, there is an increased demand to use this technology also on a larger scale in a kilolab. Nevertheless, up to now published examples of scale-up experiments are rare, in particular those involving complex organic reactions. Besides research institutes like CSIRO (Dickson, Australia) and pharmaceutical companies like Boehringer Ingelheim (Ridgefield, USA) that constructed their own prototypes,5, 6 only a few companies (most of them mentioned in this article) are currently developing commercial microwave devices for scale-up.
Microwave heating technology has some inherent physical limitations (e.g., magnetron power, penetration depth) and two different approaches for microwave-assisted synthesis on a larger scale (>100-mL volume) have emerged. 7 Whereas companies like Anton Paar 8 (Graz, Austria) have developed a batch-type multimode reactor, others like Milestone 9 (Sorisole, Italy) are offering continuous flow systems. CEM 10 (Matthews, USA) in contrast tried to overcome the scale-up problems by developing a stop-flow concept, which can be regarded as a combination of both. The Voyager SF is a monomode reactor with an 80 mL glass vessel, which is automatically filled and drained by a pump system. Reactions are carried out on a 50-mL scale sequentially and fully automated. Therefore, it was of great interest to us to evaluate the potential of commercially available microwave reactors on a large scale (with at least 300–500 mL reaction volume), to speed up the processing time for reactions that require long reaction time under conventional conditions.
Evaluation Approach
Up to now, synthesis work in a kilolab normally is done batchwise using conventional thermal heating techniques under atmospheric pressure, so that the achievable reaction temperature is limited by the boiling point of the solvent. In contrast, microwave heating in a pressurized vessel allows access to reaction temperatures far above the boiling point of the solvent and leads to remarkably shortened reaction times. Furthermore, by choosing the solvent no longer dependent on its boiling point but on more favorable work-up and purification properties, time-consuming work-up procedures can be avoided, which should result in a significantly shorter overall processing time for experiments.
In the light of some model reactions, our intention was to compare conventional heating by an oil bath and heating by a microwave first in a small-scale microwave reactor like the Emrys Optimizer (20 mL glass vials, filling volume about 15 mL). In a second step, the same reactions were carried out on a larger scale using a multimode batch reactor (Anton Paar Multiwave 3000, 8 × 100 mL PTFE vessels in a ceramic vessel jacket, filling volume 60–70 mL for each vessel, see Fig. 1) and a monomode stop-flow reactor (CEM Voyager SF, 80 mL glass vessel, filling volume 50 mL, see Fig. 2). For comparison, some of these reactions were done additionally in an autoclave.
Multiwave 3000 (Anton Paar). Voyager SF (CEM).
Some technical details of the microwave reactors used in this study are shown in Table 1.
Commercially available microwave reactors used in our evaluation

Requirements for use of those devices in our kilolab were defined as follows:
Synthesis of compounds on > 100-g scale in a reasonable timeframe; use of microwave technology should be time saving compared to conventional heating. No additional optimization work for transfer from small scale to large scale. Ability to process heterogeneous mixtures, especially suspensions, without problems. Reproducibility. Ease of handling.
In addition, the possibility to run reactions automatically in a continuous or repetitive manner would also be regarded as an advantage.
On the other hand, it has to be kept in mind that some critical issues are linked with the application of microwave in chemical scale-up. 11 First, penetration depth of microwaves in suitable solvents is limited to 2–5 cm, and therefore the vessel size cannot be expanded unrestrictedly. Secondly, high-microwave power may lead to rapid input of energy into the reaction and to overheating. Further issues are field homogeneity (inhomogeneity may lead to hot spots and degradation of products) and stability of solvents, reagents, and products at temperatures higher than 200 °C. Instability and degradation of the reaction mixture may lead to safety problems. Also the construction of the microwave reactor has to withstand high temperatures and high pressure (e.g., 250 °C, 20 bar). Moreover, measuring the temperature inside the reaction mixture is important because IR-sensors outside the reaction vessel always register a significantly lower temperature due to the inverted heat flow from inside to outside.
Results
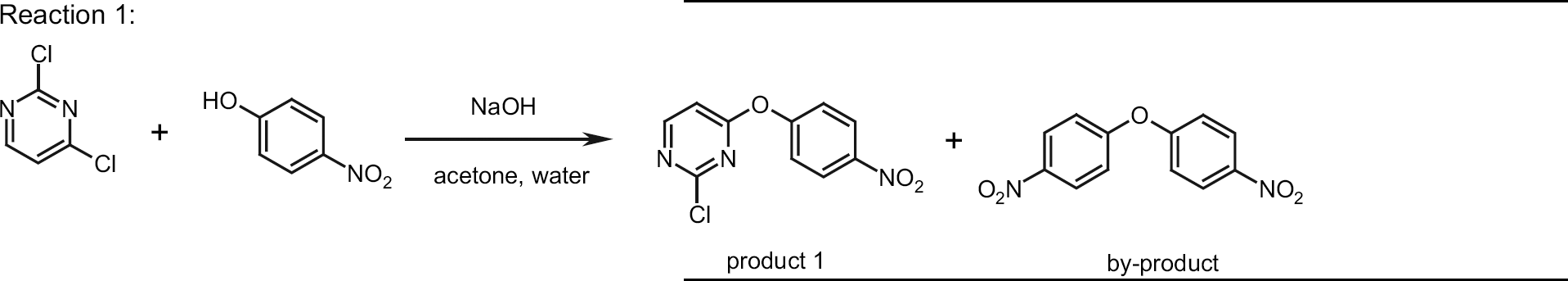
For model reactions, we choose the aromatic substitution of aryl halides with nucleophiles like phenolates or amines, whereas our special interest was focused on reaction time, selectivity, work-up procedure, and overall processing time. Results of reaction 1 are summarized in Table 2.
Results of reaction 1 under different conditions

Measured by area % HPLC.
On a small scale under reflux in acetone/water, 5% of starting material remained after 12 h reaction time and about 20% of the by-product was formed (entry 1). Performing the reaction at the same concentration in a lab-scale microwave device (Emrys Optimizer, entry 2) at 120 °C, the reaction was complete after 5 min and gave a product of significantly higher purity and in higher yield. In the next step, 400 mL of reaction mixture was reacted in an eight-vessel rotor batch microwave (entry 3) at the same temperature for 10 min. After a cooling down period of 18–20 min, the precipitated product was isolated by simple filtration in 76% yield but also 10% of the by-product was formed under these conditions. The overall processing time, which includes heating, maintaining the temperature for 10 min, and cooling was only 30 min.
In contrast to this, conversion of a reaction volume of 400 mL in the Voyager SF took about 4 h (entry 4) because eight cycles on a 50 mL scale were necessary to convert this volume. Each cycle consists of adding the starting material, heating, maintaining the temperature for 15 min, cooling, and removal of the mixture from the reaction vessel, which required 30 min. Furthermore, when the mixture was pumped out of the vessel after the reaction, precipitation of the product blocked some lines and valves, so that the process had to be interrupted manually. 12 For comparison, the reaction was also carried out in an autoclave (1 L volume, entry 5). Due to the long heating and cooling process, processing time in total was 275 min, leading to a product yield of 74% that contained more than 10% of the byproduct.
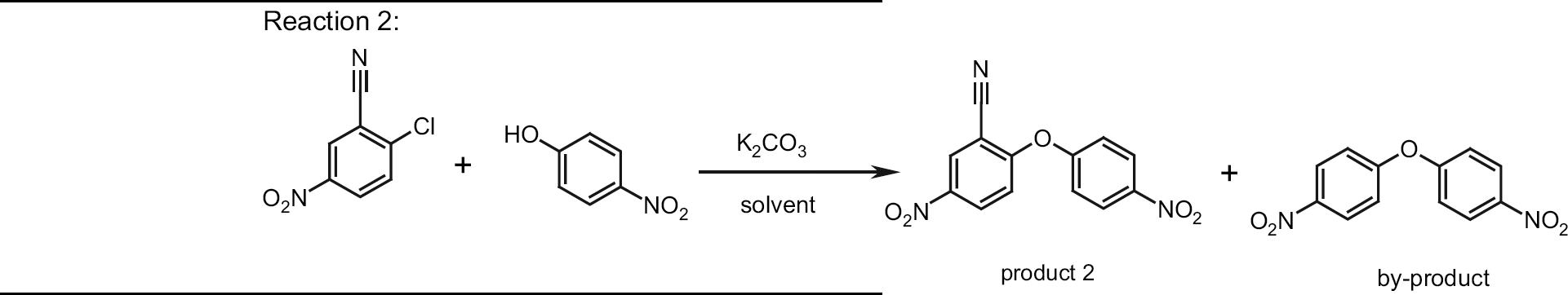
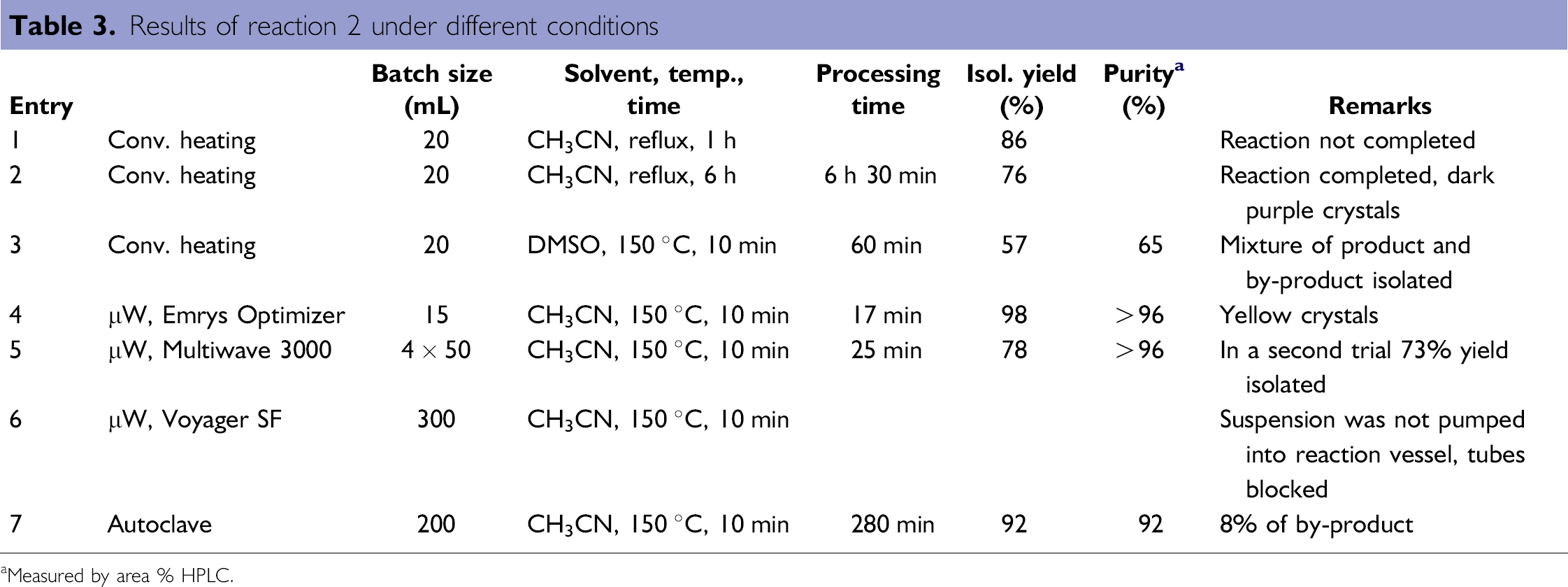
Substitution reactions of this type are often carried out with solid carbonate in suspension, which is easily removed after reaction by a simple filtration. Therefore, this reaction was chosen to test the handling of suspensions and also to investigate the influence of different solvents on selectivity and work-up. Results are shown in Table 3.
Results of reaction 2 under different conditions
Measured by area % HPLC.
Under conventional conditions in the lab (acetonitrile, reflux for 60 min) the product was isolated in 86% yield but the remaining starting material revealed that the reaction was not complete (entry 1). Prolongation of the reaction time to 6 h lead to full conversion of the starting material but also to reduced yield and formation of dark purple crystals (entry 2). Performing the same reaction in DMSO at higher temperature (150 °C for 10 min) lead to a significantly shorter reaction time but also to increased formation of by-product and a significantly lower yield (entry 3). In contrast, when a microwave reactor was used at 150 °C for 10 min an isolated yield of 98% could be achieved (entry 4). Scale-up trials were then done in the Multiwave 3000, where the suspension could be reacted without problems. To check the reproducibility, two identical batches were carried out, the first batch giving 78% yield (entry 5) and the second giving 73% yield after crystallization. When the same reaction was done in an autoclave, a gray product containing 8% of by-product was isolated in 92% yield but with lower quality (entry 7). Using the Voyager SF for this reaction failed because the system was not able to pump the suspension into the reaction vessel (entry 6). Although we used finely ground potassium carbonate, the inlet tube was blocked repeatedly by the solid carbonate. It should also be mentioned that in terms of processing time the reaction in an autoclave required 280 min, whereas in the Multiwave 3000 only 25 min were needed for the conversion.
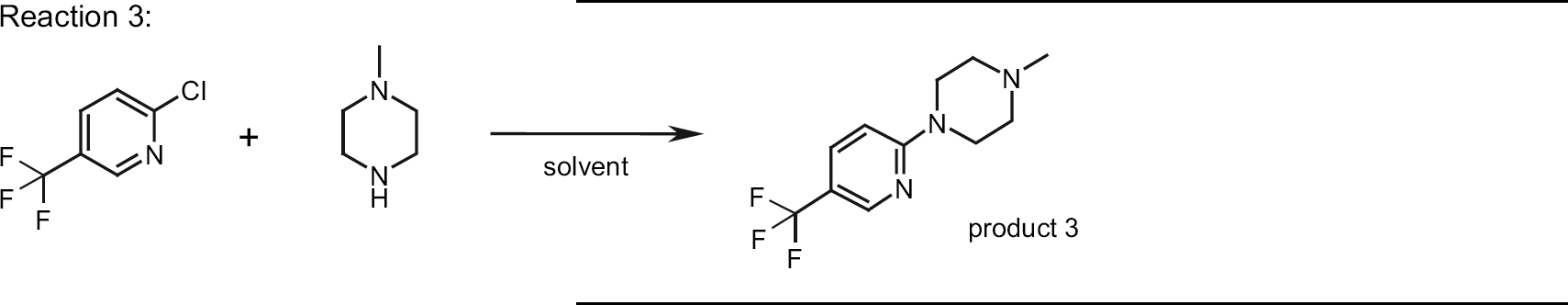
The third model reaction was a homogenous one and again our intention was to compare the influence of different solvents under conventional heating and microwave heating. Results are shown in Table 4.
Results of reaction 3
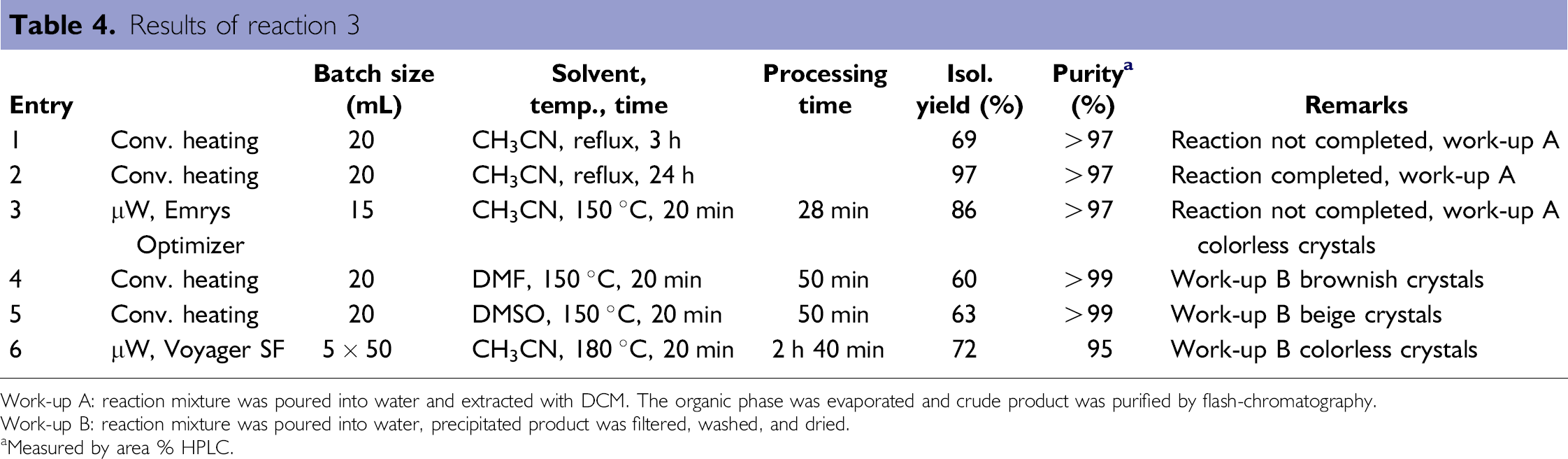
Work-up A: reaction mixture was poured into water and extracted with DCM. The organic phase was evaporated and crude product was purified by flash-chromatography.
Work-up B: reaction mixture was poured into water, precipitated product was filtered, washed, and dried.
Measured by area % HPLC.
Under reflux conditions on a small scale in acetonitrile, the reaction was complete only after 24 h (entry 2). In a microwave reactor at 150 °C, 86% of isolated yield was achieved after 20 min of reaction time (entry 3). Running the same reaction in DMSO or DMF at 150 °C gave complete conversion of starting material but due to losses during work up, isolated yield was lower (entry 4 and 5). For scale-up trials, the Voyager SF was used, and the reaction was done at 180 °C for 20 min in each cycle. To process a 250 mL batch, five cycles of 50 mL each had to be performed, leading to a processing time of 2 h 40 min (entry 6). A typical diagram of temperature, pressure, and time of all five runs is shown in Figure 3. The temperature rises very rapidly to 180 °C and is held for 20 min, and the pressure during the reaction is about 11 bar. Then, the vessel is cooled by air ventilation to about 60 °C within 8–10 min, which results in a cycle time of about 32 min.
Diagram of reaction 3, Voyager SF.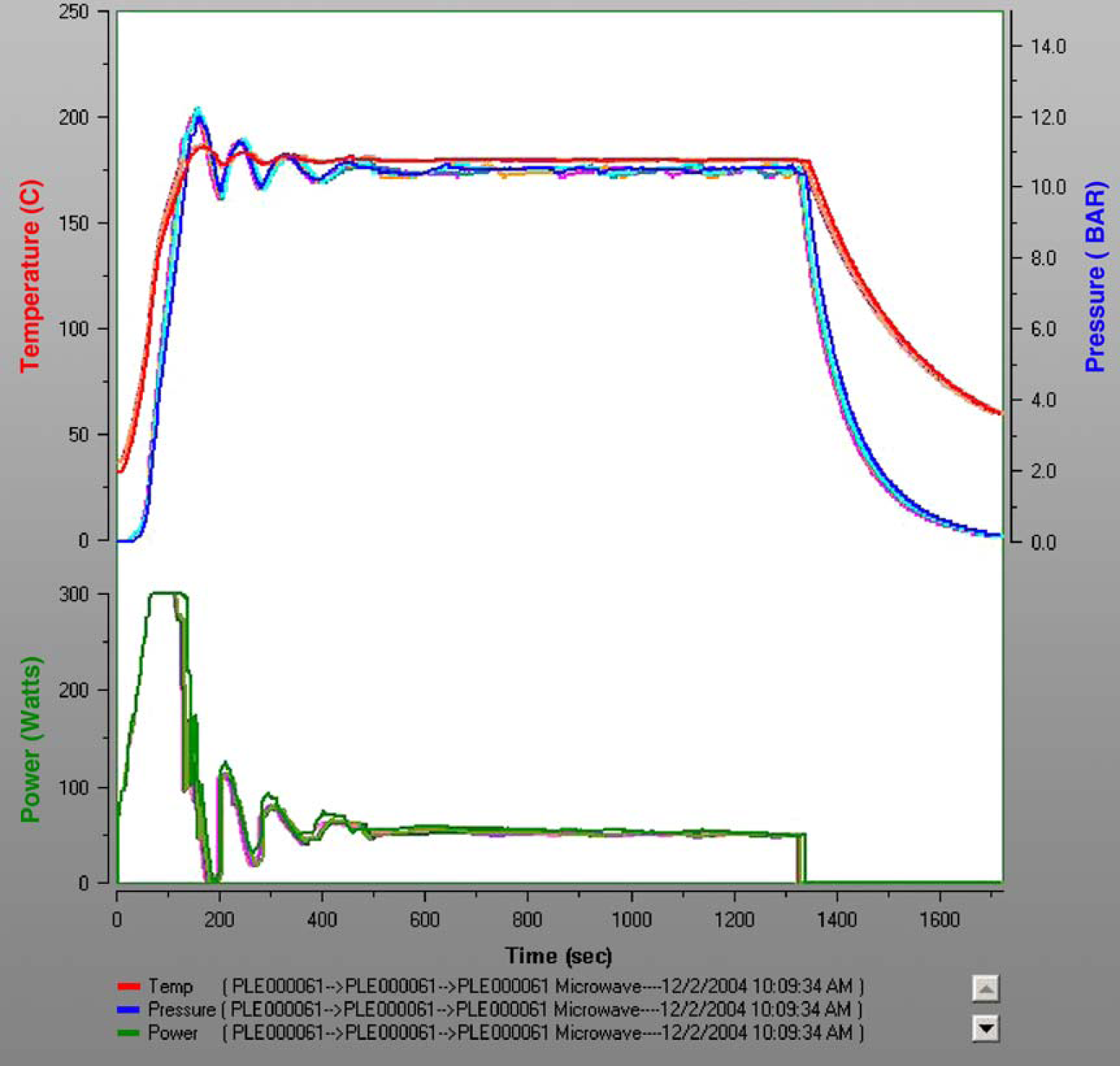
Discussion and Conclusions
Two different microwave reactors for scale-up have been tested with special focus on handling, automation, workflow, and typical scale-up issues. The Multiwave 3000 as a batch mode reactor provides a relatively large reaction volume (16 × 70 mL), allows high temperature and pressure (250 °C/40 bar), and has proved to be very robust. Handling of suspensions revealed no problems, and the system was shown to have a very good temperature control. The software is easy to handle and results from small scale could be reproduced on a larger scale without additional optimization effort. Nevertheless, filling and removal of the vessels is laborious, and the cooling period is comparatively long. Also, the tested reactor provides no possibility to run several reactions sequentially in an automated manner.
The possibility for automation is offered by the Voyager SF with its stop-flow system, which is able to process larger reaction volumes by repeating reactions on a 50-mL scale sequentially. The relatively small reaction volume guarantees a low-safety risk, but leads, on the other hand, to relatively long processing times. Also handling of suspensions proved to be a critical issue and in one case, precipitated product blocked the lines. Therefore, running reactions for a longer period without monitoring, for example overnight, can only be recommended for homogeneous reaction mixtures.
In various experiments it could be shown that the use of microwave technology leads to a significant decrease of the reaction time and in some cases also to less by-products and a higher yield. This technology provides the opportunity to optimize the reaction with focus on work up and purification, independent of reaction temperature and boiling point of the solvent. In most cases, the reaction conditions applied on a 15 mL scale in the Emrys Optimizer could be transferred without further optimization to the tested microwave reactors and lead to comparable results. Additional optimization in a few cases was limited to a moderate adjustment of reaction temperature or reaction time.
In theory, conditions in a microwave reactor, with regard to temperature and pressure, should also be attainable by using an autoclave. Therefore, some reactions were done in an autoclave for comparison. The main difference was the longer processing time, mainly caused by the long heating and cooling period when an autoclave was used. This leads, as shown in example 2 of our study, to a different selectivity and to a significantly higher amount of by-products, which as a consequence had to be separated by additional purification steps. In such a case, using microwave heating is obviously more advantageous than using an autoclave.
In general, it should be remarked that the commercially available microwave devices for scale-up were developed either from older existing systems for digestion (e.g., Multiwave 3000, Anton Paar) or were completely developed new for synthesis on larger scale (e.g., Advancer, Biotage). Some of these tools are more or less in a prototype phase where further development is ongoing. As this study revealed, the first steps of microwave technology toward scale-up in larger than 100-g scale are taken, but for this to become a standard technology for kg-scale synthesis, further improvements with regard to reaction volume and automation will have to follow in future.
Footnotes
Acknowledgement
We thank Personal Chemistry, Anton Paar, and CEM for their support and permitting us to test their microwave reactors in our laboratories.
