Abstract
Abstract
Introduction
This article provides an overview of the management of a displaced hip in children with cerebral palsy and considers surveillance programmes, principles of surgical reconstruction and options for the salvage of an unreconstructable hip in these children.
Conclusion
Hip dislocation in CP is potentially preventable if children are included from an early age in a surveillance programme that includes repeat radiographic and clinical examinations, and preventive treatment for hips that are displacing. A surveillance programme should be based on the child's age, GMFCS level and migration percentage (MP), and surgical prevention may be considered in children with a MP exceeding 33 %.
Introduction
It has been known for more than 50 years that hip dislocation in a child with cerebral palsy (CP) may be preventable [1]. Few countries have established screening programmes for the hip in patients with CP, although in many centres an annual pelvic radiograph is performed in patients at risk of hip displacement. In practice, children often first present to a clinician with established radiological signs of hip displacement.
The cause of hip displacement in CP is not completely clear, but the hip is usually normal at birth. High hip joint reaction forces and abnormal orientations of force vectors are relevant and place an abnormal force on the acetabulum, which may then begin to deform [2]. The role of spasticity remains unclear, but is probably overestimated, because hip dislocations occur in hypotonic as well as hypertonic conditions. Another factor may be positioning [3], and many children with severe CP rest in the same position—particularly at night—in side-lying, where one hip remains in adduction. A further influence may be impaired proprioception, which results in inadequate muscle control [4].
A saucer-shaped deformity develops in the acetabulum, usually in a craniolateral direction [5], and the femoral head gradually migrates proximally and may also then become deformed. As the femoral head migrates proximally, it may be subjected to increasing forces on the medial side which result in medial flattening of the head. The lateral side of the head may be subjected to pressure from the hip capsule and abductors, resulting in lateral flattening. The combination of medial and lateral flattening can produce a triangular or dunce's cap appearance to the head as seen on an anteroposterior radiograph. Finally, direct force on the head from the reflected head of rectus femoris can produce a groove on the superior part of the head. The end result of the femoral displacement is often loss of articular cartilage and early osteoarthosis. Other directions of dislocation include anterior (8 %) and posterior (1 %), and are sometimes only visible on three-dimensional computed tomographic scans [5]. Increased anteversion and/or valgus and muscle contractures may also contribute to the femoral deformity [6]. Although the direction of spastic hip subluxation is generally agreed to be posterolateral, a recent radiographic study has suggested that the location of the acetabular deficiency is global and more apparent anteriorly [7].
Hip surveillance
Preventing hip dislocation in a population of children with CP requires a surveillance programme, including routines for preventive treatment of children with hips at risk [8]. Clinical examination of hip displacement in CP is unreliable, and hip range of motion is also a poor indicator of the risk of displacement, as contractures are common and restrict motion. Thus hip displacement can occur in a hip with a normal range of motion [9] and the diagnosis is made from radiographs. Lateral displacement of the femoral head is common without acetabular dysplasia, and acetabular dysplasia develops at a later stage than femoral head lateralization [10]. An early radiographic indicator is a notch appearing on the superolateral aspect of the femoral head, which is thought to be due to moulding in response to the tendency of the femur to migrate proximally and laterally [11]. Measurement of the lateral displacement is sufficient for hip screening in CP [10, 12, 13]. The Reimers’ migration percentage [14] (MP) (Fig. 1) is a valid and reliable measure [13–15], and probably the most commonly used. Most reports define a hip with an MP of >30–33 % as displaced and a hip with an MP of >90–100 % as dislocated [12, 14, 16]. Some hips with an MP of <40 % do not progress to dislocation, while almost all hips with an MP of >40 % need preventive surgery to prevent dislocation [10].
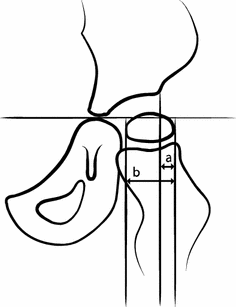
Measurement of the migration percentage (MP). MP = a/b × 100)
The most common age of displacement with an MP of >33 % or >40 % is 3–4 years (Fig. 2), and some children already show displacement at 2 years of age [9]. For an effective surveillance programme, children with CP should be identified early, and those at risk examined radiographically as early as possible.
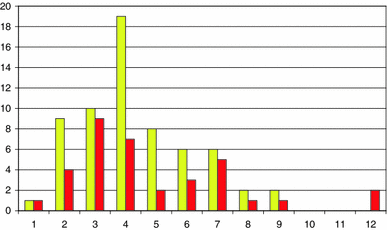
Number of children versus age (years) at first registration of an MP above 33 % (yellow) and 40 % (red) in the total population of children with CP
Hip displacement is related to CP subtype [9]. Children with spastic hemiplegia or ataxic CP have a low risk, and children with spastic tetraplegia or quadriplegia have a high risk. There are, however, several problems with using CP subtype as an indicator of hips at risk in a surveillance programme. There are several different classification systems, and the descriptors for the motor types of CP have not been universally agreed upon; new definitions are under development, and the definitions may be difficult to apply in a reliable manner [17]. It is sometimes impossible to determine the CP subtype before the age of 4 years.
Hip displacement is directly related to the level of gross motor function measured with the gross motor function classification scale (GMFCS) [18]. Similar results have been shown from the Victorian Cerebral Palsy Register [19], Sweden [9] and Norway [20] (Fig. 3). The GMFCS has proven to be a valid and reliable tool [18] and has been reported to remain relatively stable over time [21, 22]. According to the designers of the GMFCS, most children will remain at the same level from the age of two [23]. By using GMFCS instead of CP subtype as an indicator of hips at risk in a surveillance programme, all of the problems described for subtypes seem to be avoided.
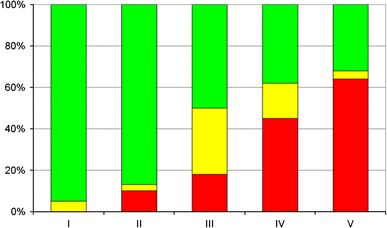
Proportion of children (%) with an MP of <33 % (green), 33–39 % (yellow) and ≥40 % (red) in relation to GMFCS level in the total population of children with CP
In southern Sweden, a follow-up programme for CP was started in 1994 with the specific aim of identifying patients at risk of hip dislocation and preventing this complication. During the subsequent 10 years, the hip dislocation rate dropped from 8 to 0.5 % [16]. Comparable results were reported from an Australian programme introduced in 1997 [12]. Both programmes are based on the child's age and GMFCS level (Fig. 4). The Australian programme contains more frequent radiographic examinations, but in the Swedish programme the children have at least annual clinical examinations by their physiotherapist.
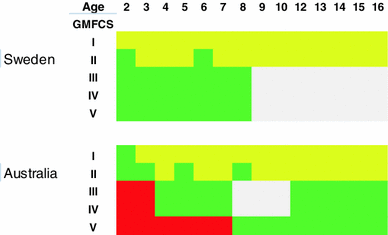
Schematic presentation of the radiographic examinations in the Australian and Swedish surveillance programmes. Green examination yearly, red examination twice a year, yellow no examination, grey individually based examinations
In the Swedish programme, children at GMFCS I are not examined radiographically unless deterioration occurs in the clinical examinations. Children at GMFCS II have radiographic examinations at 2 and 6 years of age, and children at GMFCS III–V have annual examinations from the first suspicion of the diagnosis of CP until 8 years of age. After 8 years of age, the time interval between examinations is assessed individually based on the results of the previous clinical and radiological examinations. In the CPUP programme, all children have clinical examinations twice a year until 6 years of age and once a year thereafter [24].
In the Australian programme, all children have a radiographic examination at 12–24 months of age. Children at GMFCS I are reviewed at 3 and 5 years of age. Children at GMFCS II are examined radiographically once a year until the MP is stable, and all children are examined radiographically at 5 and 8 years of age. Children at GMFCS III–IV are examined radiographically twice a year until the MP is stable, then once a year until 7 years of age. If the MP is <30 %, radiographic examination is discounted until pre-puberty. The children are examined once a year from pre-puberty until skeletal maturity. Children at GMFCS V have radiographic examinations twice a year until 7 years of age, and then annual examinations until skeletal maturity [25].
Management of hip displacement
The common indications for management of a displaced hip are instability that cannot be controlled by the patient, and restriction of motion and pain, which may already be present in mild subluxations [26]. Functionally, this may manifest as difficulties with transfers, perineal access for hygiene purposes, in obtaining stable seating, and problems with pelvic and spinal alignment. There is controversy, however, as to whether a dislocated hip is painful, but Cooperman et al. [27] suggested that 50 % of a small group of institutionalized patients with CP complained of pain in the dislocated hip. Noonan et al. [28] found that the incidence of hip pain was low and not associated with hip displacement or osteoarthritis in their study of 77 adults with CP. They recommended that surgical treatment of the hip in severely affected patients should be based on the presence of pain or contractures and not on radiographic signs of hip displacement or osteoarthritis.
Non-operative prevention consists of positioning the child to prevent prolonged adduction–flexion at the hip. Optimal lying and sitting positioning during long periods [29] and standing treatment with hips in abduction [30] seem to reduce hip displacement. Bracing is not thought to be effective in spastic hip displacement. Graham et al. [31] studied the combined effect of intramuscular injection of botulinum toxin A and abduction hip bracing in the management of spastic hip displacement in a randomized trial, and found that progressive hip displacement continued to occur in the treatment group, so they did not recommend this treatment. Spasticity-reducing treatment with intrathecal baclofen, selective dorsal rhizotomy or botulinum toxin reduces the adductor spasticity but has not, so far, been proven to reduce the risk of hip displacement.
Operative management to prevent dislocation consists of soft tissue release, varus osteotomy of the proximal femur and pelvic osteotomy. There are still no studies that have shown at what age or at what degree of displacement which of these operations is preferable. The muscle imbalance over the hip joint is present postoperatively and, irrespective of the operative method used, there is a risk of re-displacement during the remaining postoperative growth period.
Soft-tissue release consists of myotenotomy of the adductor longus, brevis and often gracilis. The operation should also include iliopsoas tenotomy. It has been questioned whether the iliopsoas tendon should be divided at its insertion at the lesser trochanter or by an intramuscular lengthening at the pelvic brim to prevent flexor weakness. Bialic et al. [32], in a comparative study, found more robust results and no adverse effects related to flexor weakness after division at the lesser trochanter. Additionally, division of the psoas tendon at the lesser trochanter in children who do not walk is unlikely to produce an adverse functional effect on hip flexor function. Several studies have shown that children undergoing unilateral soft-tissue release have a high risk of displacement of the contralateral hip [14, 33, 34], and these authors conclude that soft-tissue release should be carried out bilaterally. Adductor longus intramuscular tenotomy, gracilis myotomy and psoas tenotomy is only likely to be effective in the longer term if hip abduction can be maintained. A review of the effects of surgical adductor releases for hip subluxation in cerebral palsy (CP) [35] concluded that there was weak evidence to support the efficacy of the procedure. Interestingly, a very recent report [36] found that adductor surgery was only successful in 27 % of children at GMFCS level IV and 14 % of children at GMFCS level V. The criteria for “success” in this study [36] was defined as the absence of subsequent surgical procedures during the study period and a migration percentage of <50 % in both hips at the time of follow-up.
Progressive uncovering of the femoral head requires the addition of varus derotation osteotomy, provided the acetabulum is not also deformed. A varus osteotomy of the femur has the same effect as passive hip abduction but has the advantage that its effect is independent of passive movement. The femoral deformity is either excessive anteversion or valgus or both. Robin et al. [37] have shown that the neck–shaft angle and anteversion increases with GMFCS level as a result of delayed walking, limited walking or inability to walk. It should be remembered that coxa valga is often a radiographic artefact due to femoral torsion, and a corrected view of the proximal femur should be obtained. An anteroposterior radiograph with the femur in internal rotation to neutralize anteversion is a prerequisite to show the true femoral pathology and the neck–shaft angle. Significant re-operation rates have been reported after early soft tissue or proximal femoral procedures when performed at an average age of 4 years as acetabular remodelling did not reliably occur postoperatively [38]. Others [39] have reported satisfactory results of femoral osteotomy in patients whose pre-operative MP was >50 %. Varus osteotomy, sometimes combined with derotation to reduce increased anteversion of the proximal femur, is made at the level of the lesser trochanter. A further controversy is whether or not the opposite femur should be osteotomized, and some authors consider that only the displaced hip should be operated on [36, 38, 39], whereas others recommend bilateral osteotomy in all cases [40].
Where there is a combination of acetabular and femoral head deformity, the aim of surgery is to relocate the hip by reducing the neck–shaft angle to about 120°, anteversion to a physiological value, and to cover the femoral head with a pelvic osteotomy such as a Pemberton [41] or Dega type of periacetabular osteotomy [42]. The Dega type, as originally described [42], extended into the sciatic notch, but many surgeons direct the osteotomy down the posterior column towards the posterior part of the triradiate cartilage [43, 44]. It is also possible to perform a Dega type of osteotomy after closure of the triradiate cartilage in patients with severe CP, as the pelvic bone is often osteoporotic [45]. An open reduction of the hip is not always necessary, but when needed should include resection of the ligamentum teres and division of the transverse acetabular ligament. Sufficient shortening of the femur at the time of femoral osteotomy is most important to lengthen, relatively, the contracted muscles and to avoid excessive pressure on the femoral head; additional soft tissue procedures are not usually necessary as part of this procedure. Anterior hip dislocation can pose a challenge and a Pemberton osteotomy, rather than a Dega type, may give a better prospect of anterior coverage. Even severe deformity of the femoral head is not necessarily a contraindication to reduction, since the risk of subsequent symptomatic arthritis appears to be small.
The evaluation of functional outcomes is challenging in patients with severe CP who have undergone surgery for hip displacement. The literature consists mainly of case series of relatively small numbers of patients with short- to medium-term follow-up, and not always conformity of management or outcome measures between case series. However, it seems that a one-stage hip reduction and pelvic and femoral osteotomy is generally successful [46] and gives superior results to femoral osteotomy alone [39, 47]. In experienced hands, reconstructions for lateral or posterior dislocations have given success rates of 95 % for range of motion and hip reduction and 90 % for pain reduction [48].
The unreconstructable hip
The demand on the hip in patients with severe CP is low and osteoarthritic changes seen radiologically can remain asymptomatic for many years. Hip pain does not seem to correlate well with femoral head deformity. If the hip is symptomatic and unreconstructable, there is a choice of salvage procedures to obtain reasonable abduction and relief of pain. These include proximal femoral resection [49–61], prosthetic replacement of the hip [62–70], prosthetic interposition arthroplasty [71], valgus femoral osteotomy [72, 73], a combination of valgus osteotomy and proximal femoral excision [55, 57], and hip fusion [66].
The commonest reported procedure is a proximal femoral resection described by Castle and Schneider [49]. The gluteus medius, minimus and vastus lateralis are used as an interposition between the resected femur and acetabulum. About 80 % of patients are comfortable afterwards but insufficient femoral resection and heterotopic bone formation are two important potential complications of the procedure. A proximal femoral excision is not recommended for patients who have a displaced hip but are capable of walking, or have independent or assisted transfer capability, because of the subsequent loss of hip stability. In a recent report, Egermann et al. [50] have reported a reduction in heterotopic bone formation from about 80 to 60 % after a Castle and Schneider [49] procedure by “capping” the proximal femoral stump with the excised femoral head.
Although the Castle and Scheider [49] procedure is the most commonly reported solution for the painful unreconstructable hip, the literature on salvage procedures consists of mainly case series with small numbers and short-term follow-ups, and, again, outcome measures are difficult to evaluate in patients who have severe CP.
Conclusion
Hip dislocation in CP is potentially preventable if children are included from an early age in a surveillance programme that includes repeat radiographic and clinical examinations and preventive treatment for hips that are displacing. The surveillance programme should be based on the child's age, GMFCS level and the degree of lateral displacement as measured by the MP. Non-operative prevention by proper positioning should be considered in all children with CP, although the evidence base for this is weak. Operative prevention should be considered in children with an MP exceeding 33 %. Children will require rehabilitation after any surgical procedure, and this should be factored into any decision about surgery, which would also take into account the wishes of the child and/or their family/carers. Children should also be followed postoperatively until at least skeletal maturity, and if the child has scoliosis associated with a pelvic tilt they should, ideally, be followed further into adulthood.
