Abstract
Abstract
Purpose
The pathogenesis of unicameral bone cysts (UBCs) remains largely unknown. Osteoclasts have been implicated, but the role of osteoblastic cells has, to date, not been explored. This study investigated the pathophysiology of UBCs by examining the interactions between the cyst fluid and human bone marrow stromal cells (hBMSCs) and the effect of the fluid on osteogenesis.
Methods
Fluid was aspirated from two UBCs and analysed for protein, electrolyte and cytokine levels. Graded concentrations of the fluid were used as culture media for hBMSCs to determine the effects of the fluid on hBMSC proliferation and osteogenic differentiation. The fibrocellular lining was analysed histologically and by electron microscopy.
Results
Alkaline phosphatase (ALP) staining of hBMSCs that were cultured in cyst fluid demonstrated increased cell proliferation and osteogenic differentiation compared to basal media controls. Biochemical analysis of these hBMSCs compared to basal controls confirmed a marked increase in DNA content (as a marker of proliferation) and ALP activity (as a marker of osteogenic differentiation) which was highly significant (p < 0.001). Osteoclasts were demonstrated in abundance in the cyst lining. The cyst fluid cytokine profile revealed levels of the pro-osteoclast cytokines IL-6, MIP-1α and MCP-1 that were 19×, 31× and 35× greater than those in reference serum.
Conclusions
Cyst fluid promoted osteoblastic growth and differentiation. Despite appearing paradoxical that the cyst fluid promoted osteogenesis, osteoblastic cells are required for osteoclastogenesis through RANKL signalling. Three key cytokines in this pathway (IL-6, MIP-1α, MCP-1) were highly elevated in cyst fluid. These findings may hold the key to the pathogenesis of UBCs, with implications for treatment methods.
Introduction
Unicameral bone cysts (UBCs) are benign bone defects that are defined as minimally expansile lucent lesions consisting of a cavity filled with fluid and lined with a fibrocellular membrane [1]. The pathogenesis of UBCs remains largely unknown, but theories presented in the literature include pressure effects due to blocked fluid drainage [2], local venous obstruction [3, 4], increased lysosomal enzyme activity [5], prostaglandins [6], nitric oxide [3], oxygen free radicals [7], disorders of synovial origin [8] and genetic causes [9]. As these hypotheses concerning the pathogenesis of UBCs have evolved, multiple diverse treatment strategies have emerged. However, theories have often been based on the analysis of single reports or small case series, so a unified hypothesis (and hence targeted treatment) remains elusive. Therapeutic aspiration, curettage [10], autologous bone marrow injection [11, 12], steroid injections [6, 13, 14], demineralised bone matrix [15], grafting of synthetic materials [16, 17] and intramedullary fixation [18, 19] are routinely employed to treat UBCs, though all have a high failure and/or recurrence rate.
Much of the literature defining the pathogenesis of UBCs has suggested a role for osteoclasts. Shindell et al. [6] have reported increased levels of PGE2 within UBC fluid. PGE2 is a protein kinase A (PKA)-mediated signalling molecule involved in the stimulation and proliferation of osteoclasts. A reduction in levels following intralesional steroid injection has been associated with cyst resolution [6]. Similarly, osteoclasts require lysosomal enzymes for bone resorption, and increased levels of these, together with collagen degradation products, have been demonstrated in UBCs, indicating ongoing osteoclast-mediated bone destruction [5]. Increased osteoclast activity within UBCs has also been established by detecting elevated acid phosphatase levels [20] and increased osteoclast-related bone resorptive factors within the cyst fluid and lining [3]. Furthermore, the presence of osteoclasts has also been confirmed in the fibrocellular lining of UBCs by histology [3] and electron microscopy [21].
Complex signalling pathways exist between osteoclasts and cells of the osteoblast lineage [human bone marrow stromal cells (hBMSCs), osteoblasts, osteocytes and bone lining cells] [22], abnormalities of which contribute to a wide range of bone pathologies, including osteoporosis, osteopetrosis, Paget's disease, rheumatoid arthritis and periodontal disease [23, 24]. Differentiation, fusion and activation of osteoclasts from macrophage-monocyte precursors are stimulated by factors derived from cells of the osteoblast lineage, notably RANK ligand (RANKL), so-called because it binds to a receptor on osteoclast precursors, receptor activator of NFκB (RANK) [25]. Cells expressing the osteoblast markers Stro-1 and RUNX-2 have been identified in the UBC fibrocellular lining, and these have been shown to induce osteoclastogenesis in vitro [21]. Beyond this single manuscript, the role of cells of the osteoblast lineage in the pathogenesis of UBCs has not been explored. Furthermore, whilst many of the abnormalities that have been identified in UBCs have been discovered by analysing the cyst fluid [3, 5–7, 20], the effect that the fluid itself contributes to UBC pathology has, to date, not been investigated.
Aims
The purpose of this study was to explore the underlying pathophysiology of unicameral bone cysts (UBCs) by investigating the relationship between cells of the osteoblast lineage and the cyst fluid.
Materials and methods
Cyst fluid from UBCs of two patients was analysed for this study. The use of human tissue was prospectively approved by the local regional ethics committee (LREC194/99/1) and informed parental consent was obtained.
Case 1
A nine-year-old healthy male had a one-year history of progressive pain in his right thigh, and radiographic investigation revealed a lesion of the proximal femur characteristic of a UBC (Fig. 1a, b). Due to the risk of impending fracture, the patient was listed for surgical stabilisation. Under general anaesthetic and fluoroscopic guidance, 20 ml of straw-coloured fluid was aspirated percutaneously from the cyst in the proximal femur (Fig. 1c), prior to stabilisation with a flexible intramedullary nail (Fig. 1d).
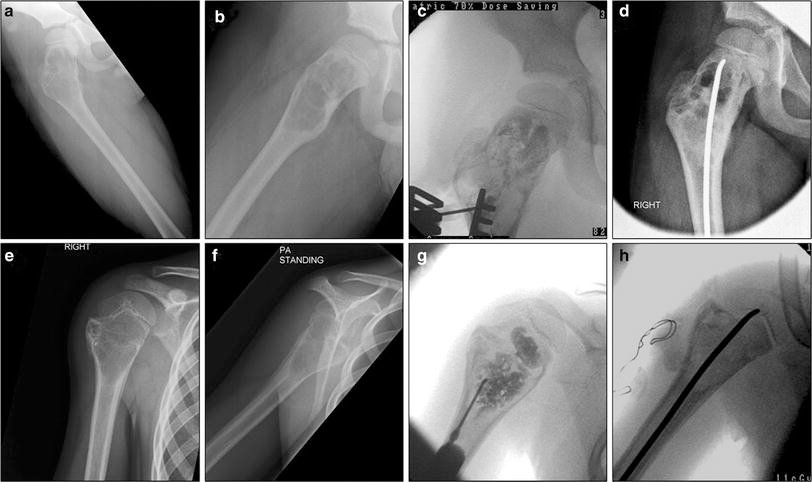
Radiographs of UBCs from cases 1 and 2: preoperative AP and lateral radiographs of case 1 (
Case 2
A six-year-old boy suffered a pathological fracture through a previously asymptomatic unicameral bone cyst of his right proximal humerus. The fractured cortices progressed to bony union, but the cyst remained, and required surgical stabilisation (Fig. 1e, f). Intraoperatively, 4 ml of straw-coloured fluid was aspirated percutaneously (Fig. 1g), and a window of bone and UBC membrane was excised from the medial wall. The proximal humerus was stabilised with a flexible intramedullary nail (Fig. 1h).
Cyst fluid separation
The aspirated samples were centrifuged at 1,100 rpm for 4 min at 21 °C to separate the supernatant, which was removed, providing a pure fluid fraction which was used for further analysis.
Harvesting of human cells of the osteoblast lineage
Bone marrow is a reliable source of human bone marrow stromal cells (hBMSCs), which have the potential for self-renewal and osteogenic differentiation [25]. Human bone marrow was obtained for this study from haematologically normal adults undergoing routine total hip replacement surgery with the approval of the local ethics committee (LREC194/99/1). Adult hBMSCs were harvested by repeatedly washing the marrow in 0.9 % saline, centrifugation, passage through a 70 μm filter and culture expansion in basal media at 37 °C in 4 % humidified CO2. At 30 % confluence, the adult hBMSCs were released using trypsin in EDTA (ethylene diamine tetraacetic acid), centrifuged, re-suspended in basal media and a cell count was performed using a haemocytometer. The hBMSC solution was titrated to a concentration of 5 × 103 hBMSCs per 100 μl of basal media prior to seeding onto culture plates (n = 18).
hBMSC culture in cyst fluid
Graded concentrations of cyst fluid were prepared by mixing with alpha-modified Eagle's medium (α-MEM) as an inert control. One hundred microlitre aliquots of graded cyst fluid/α-MEM mixes were added to 100 μl aliquots of hBMSCs in basal media in culture wells. This provided eighteen 200 μl aliquots of culture media/cyst fluid/α-MEM, each containing 5 × 103 hBMSCs. Cyst fluid concentrations were 0, 25 and 50 %, with six identical wells of each concentration prepared (Table 1). Cultures were run for eight days then fixed in 95 % ethanol for analysis.
The proportions of cyst fluid, α-MEM and basal media in each culture well, illustrating how the % value of cyst fluid was derived
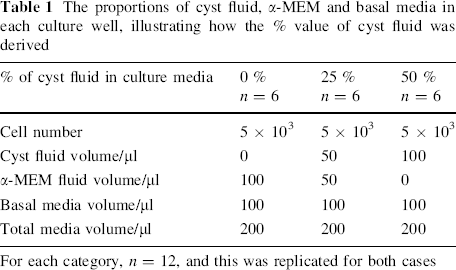
For each category, n = 12, and this was replicated for both cases
Alkaline phosphatase stain
Following fixation in 95 % ethanol for 24 h, hBMSCs were stained with Naphthol AS-MX and Fast Violet B Salts (Sigma-Aldrich, Poole, UK) for alkaline phosphatase (ALP) to measure the osteogenic differentiation of hBMSCs. Microscope images were recorded using Carl Zeiss Axiovision software v.3.0 via an AxioCam HR digital camera on an Axiovert 200 inverted microscope (Carl Zeiss Ltd, Welwyn Garden City, UK).
Biochemical analysis
Culture wells were assessed by biochemical analysis for DNA content (as a measure of total cell number), and ALP activity (as a measure of osteogenic differentiation). Following fixation in 95 % ethanol, cell lysates were obtained via three sequential freeze–thaw cycles in Triton X-100 (BDH Chemical Ltd, Poole, UK). ALP and DNA assays were then performed in a standard manner with 10 μl of lysate run in triplicate for each concentration of cyst fluid on a plate against two standards, and read on a BioTek KC4 ELx800 absorbance microplate reader and FLx800 fluorescence microplate reader (BioTek, Potton, UK), respectively. Statistical analysis was performed by one-way ANOVA using GraphPad InStat v.3.00 (GraphPad Software, San Diego, CA, USA).
Analysis of protein, electrolytes and cytokines of the cyst fluid
The levels of a range of musculoskeletal cytokines in cyst fluid supernatant were assessed using fluorescent bead immunoassay and flow cytometry (Human Chemokine 6plex BMS813FF plate, Bender MedSystems, Vienna, Austria), which tested for levels of granulocyte colony stimulating factor (G-CSF), interferon-gamma (IFN-γ), interleukin (IL)-1β, IL-2, IL-4, IL-5, IL-6, IL-8, IL-10, IL-12p70, monocyte chemotactic protein (MCP)-1, monokine induced by gamma interferon (MIG), macrophage inflammatory protein (MIP)-1α, MIP-1β, tumour necrosis factor (TNF)-α and TNF-β. Protein and electrolyte levels were measured using a Beckman DXC Coulter Counter (Beckman Coulter Ltd, High Wycombe, UK).
Analysis of fibrocellular lining of cyst
Samples were fixed in 4 % paraformaldehyde and freeze dried. SEM analysis was performed using a JSM 6330F scanning electron microscope (JEOL UK Ltd, Welwyn Garden City, UK) and an ISIS 300 system (Oxford Instruments, Abingdon, UK) was used to obtain energy-dispersive X-ray analysis (EDXA) data on the samples. Tissue was dehydrated using increasing concentrations of ethanol prior to wax imbedding, sectioning and staining with toluidine blue for analysis of structural detail.
Results
ALP staining of hBMSC culture with cyst fluid
ALP staining of hBMSCs in 25 and 50 % cyst fluid (n = 3) demonstrated increased cell proliferation and osteogenic differentiation compared to cells cultured in basal media. This was observed to be dose dependent (Fig. 2a–c).
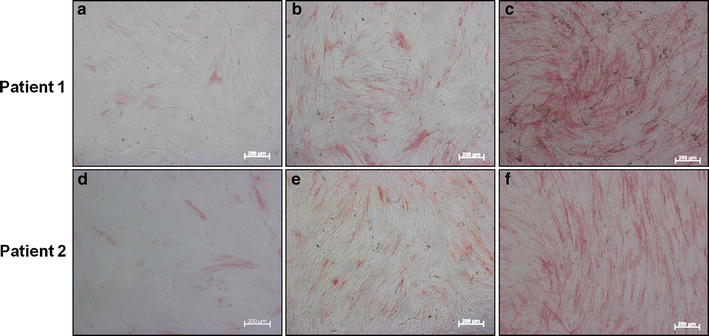
Photomicroscope images of ALP-stained hBMSC cultures in graded cyst fluid concentrations [0 % (
Biochemical analysis of hBMSC culture with cyst fluid
hBMSC culture in graded cyst fluid concentrations from both cysts showed significantly increased ALP activity (p < 0.001), DNA activity (p < 0.001) and ALP-specific activity (p < 0.001) compared to cells exposed to basal media alone. This finding was observed to be dose dependent with increasing cyst fluid concentration from 0 to 50 %, displaying the promotion of osteogenic differentiation of hBMSCs by UBC fluid and providing quantification of the qualitative observations of ALP staining (Fig. 3a–f).
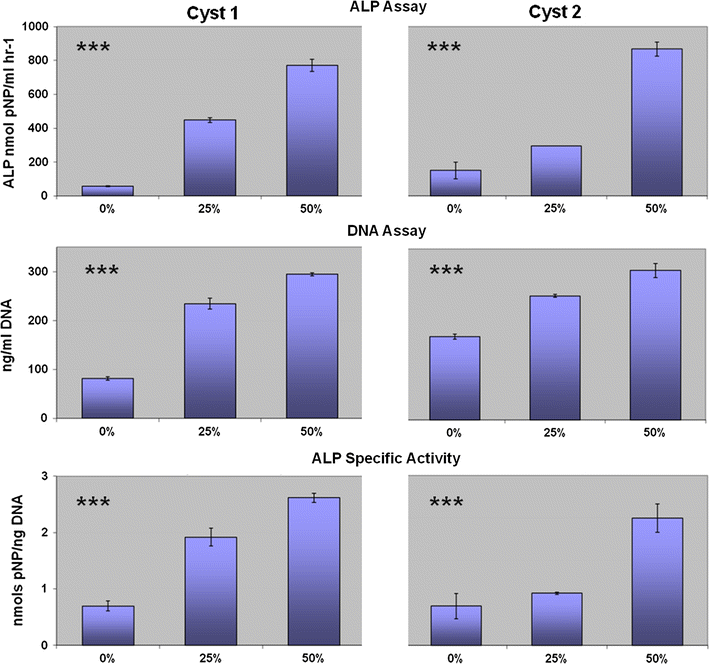
Biochemical assays of hBMSCs cultured with graded concentrations of cyst fluid from cyst 1 (left column) and cyst 2 (right column). Error bars denote the standard deviation, s11832-012-0419-x p < 0.001. Each assay was run in triplicate
Fibrocellular membrane
The fibrocellular membrane that was excised from the cyst of patient 2 showed osteoclast-like giant cells with multiple nuclei and numerous mitochondria on EM (Fig. 4a) and histology (Fig. 4b), in keeping with reports by Komiya et al. [3] and Yu et al. [21]. Interestingly, an abundance of osteoclasts in the paratrabecular region of the cyst lining was demonstrated (Fig. 4b).
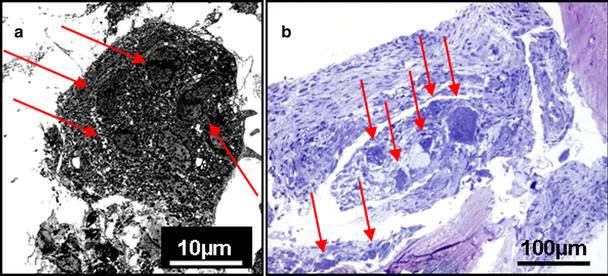
Electron micrograph (EM) and histological section of the fibrocellular lining of the UBC.
Protein and electrolyte levels
Levels of sodium, potassium, bicarbonate and calcium in the cyst fluid were within the normal range for plasma, and protein was 54 g/l, in keeping with previous analyses of cyst fluid [2, 4].
Cytokine levels
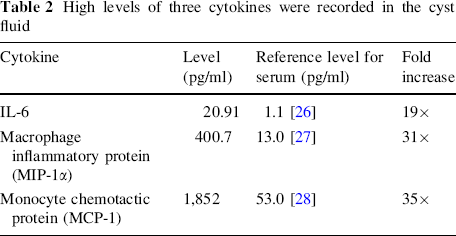
Elevated levels of three out of the panel of 15 musculoskeletal cytokines were recorded (Table 2). IL-6, MIP-1α and MCP-1 levels were increased by 19, 31 and 35 times, respectively, compared to reference values found in the literature [26–28]. This contrasted with all other cytokines tested, which were either below the level of detection or below published reference values. Therefore, for clarity, these are not presented in the table.
High levels of three cytokines were recorded in the cyst fluid
Discussion
The typical radiographic appearance of a UBC is an eroded cyst cavity (Fig. 1), and a role has been proposed for osteoclasts in UBC pathology [3, 5, 6, 20, 21]. The EM and histological sections in this study revealed once again the presence of osteoclasts in high numbers in the cyst lining (Fig. 4). It may appear paradoxical therefore that potentiation of cells of the osteoblast lineage by the cyst fluid was observed; biochemical and histological analysis demonstrated a marked increase in hBMSC proliferation and osteogenic differentiation when cultured in the presence of cyst fluid, which was highly significant (p < 0.001) (Figs. 2, 3).
On the basis of these results, increased osteogenesis could be the expected pathology, as opposed to the eroded bone appearances typically seen on radiographs of UBCs. However, complex signalling pathways exist between cells of the osteoblast lineage and osteoclasts, notably through the release of RANKL, without which osteoclasts neither form nor function [23, 24, 29]. As well as establishing promotion of cells of the osteoblast lineage by the cyst fluid, this study also identified—for the first time—a number of pro-osteoclast cytokines within the cyst fluid, with dramatically increased levels of RANKL-related osteoclast-promoting cytokines: IL-6, MCP-1 and MIP-1α.
IL-6 is a key cytokine in the promotion of bone resorption by osteoclasts [24, 30, 31], acting via Gp130 signalling of cells of the osteoblast lineage to induce RANKL. Normal physiological levels of IL-6 in the circulation are below the level of detection [23], but were grossly elevated in UBC fluid. Notably, IL-6 acts via a different signalling pathway to the PKA-mediated promotion of RANKL by PGE2, which has also been shown to be elevated in cyst fluid [6]. Upregulation of IL-6 has been implicated in osteoclastogenesis causing osteoporosis [32, 33], and with pathological bone resorption in multiple myeloma [33]. Increased IL-6 levels in UBC fluid have been reported once previously, though the authors made negligible comment on this finding [3].
Monocyte chemotactic protein-1 (MCP-1) is induced by RANKL to promote osteoclast differentiation from monocyte precursors. A positive feedback loop exists, as MCP-1 is released by osteoclasts upon differentiation [34]. MCP-1 activity has been linked to osteoclastogenesis and osteoclast activity of bone metastases [28].
Macrophage inflammatory protein-1α (MIP-1α) is potently stimulated by RANKL to promote chemotaxis of human osteoclast precursors and differentiation to mature osteoclasts [34]. MIP-1α-mediated osteoclast formation has been implicated in osteolysis caused by multiple myeloma [35]. An MCP-1/MIP-1α/RANKL autocrine loop has been described and, interestingly, this mechanism was hypothesised to cause pathology when chemokines are abundant [34].
Thus, the increased levels of MCP-1, MIP-1α and IL-6 detected in the cyst fluid provide a mechanism for the enhanced osteoclast activity and the macroscopic appearance of bone erosion seen within a UBC (Fig. 5). The promotion of hBMSC activity by cyst fluid provides an explanation for cytokine upregulation—through the RANKL signalling pathway. Osteoblasts are known to stimulate osteoclast growth and differentiation through the release of RANKL [23], though osteocytes and bone lining cells have also been associated with RANKL release and osteoclast function [22, 36, 37]. Whilst the cyst fluid may be a reflection of the initial pathogenesis of UBCs, the source of the elevated cytokines remains unclear, and further work is necessary to identify this.
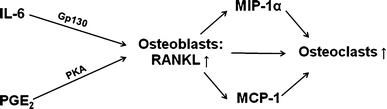
Flow chart of the RANKL signalling pathway, illustrating the dependence of osteoclasts on osteoblasts for their function, and the role of MCP-1, MIP-1α and IL-6
Like most other studies on UBCs, this study consists of a small sample size, and more patients are required. The musculoskeletal cytokine panel used in this study did not include RANKL, the levels of which are important to verify the theory presented here. Furthermore, studies on the effect of cyst fluid when cultured with osteoclasts are planned.
RANKL-mediated upregulation of osteoclasts may hold the key to the pathogenesis of UBCs, and further studies are planned to explore this hypothesis further. Novel avenues for UBC treatment may therefore need to be directed at downregulation of the pro-osteoclast cytokines. Resolution of these defects by targeted medical therapies to restore the normal osteoblast/osteoclast signalling may reduce the high failure rate of current therapies and avoid the requirement for invasive intervention in this group of patients.
Footnotes
None.
Acknowledgments
The authors are grateful to Esther Ralph for technical laboratory support. The assistance of Dr Paul Noakes, for cytokine analysis, and Sue Cox, electron microscopist in the Biomedical Imaging Unit, for the production of the EM images and slide preparation of the cyst lining, is gratefully acknowledged. Surgical costs were funded by the National Health Service (NHS), and laboratory costs were funded by grants awarded from the Biotechnology and Biological Sciences Research Council (BBSRC) and the Medical Research Council (MRC).
