Abstract
Purpose:
Simple bone cysts are among the most prevalent benign cystic tumor-like lesions in children. Proximal femoral simple bone cysts may require specific treatment because of increased fracture risk. With limited literature available on this specific localization, consensus regarding optimal treatment is lacking. We present a large international multicenter retrospective cohort study on proximal femoral simple bone cysts.
Methods:
All consecutive pediatric patients with proximal femoral simple bone cyst from 10 tertiary referral centers for musculoskeletal oncology were included (2000–2021). Demographics, primary treatment, complications, and re-operations were evaluated. Primary outcomes were time until full weight-bearing and failure-free survival.
Results:
Overall, 74 simple bone cyst patients were included (median age 9 years (range = 2–16), 56 (76%) male). Median follow-up was 2.9 years (range = 0.5–21). Index procedure was watchful waiting (n = 6), percutaneous procedure (n = 12), open procedure (n = 50), or osteosynthesis alone (n = 6). Median time until full weight-bearing was 8 weeks (95% confidence interval = 0.1–15.9) for watchful waiting, 9.5 (95% confidence interval = 3.7–15.3) for percutaneous procedure, 11 (95% confidence interval = −0.7 to 13.7) for open procedure, and 6.5 (95% confidence interval = 5.9–16.1) for osteosynthesis alone (p = 0.58). Failure rates were 33%, 58%, 29%, and 0%, respectively (p = 0.069). Overall failure-free survival at 1, 2, and 5 years was 77.8% (95% confidence interval = 68.2–87.4), 69.5% (95% confidence interval = 58.5–80.5), and 62.0% (95% confidence interval = 47.9–76.1), respectively.
Conclusion:
A preferred treatment for proximal femoral simple bone cysts remains unclear, with comparable failure rates and times until full weight-bearing. Watchful waiting may be successful in certain cases. If not feasible, osteosynthesis alone can be considered. Treatment goals should be cyst control, minimizing complications and swift return to normal activities. Therefore, an individualized balance should be made between undertreatment, with potentially higher complication risks versus overtreatment, resulting in possible larger interventions and accompanying complications.
Level of evidence:
Level IV, retrospective multicentre study
Keywords
Introduction
Simple bone cysts (SBCs) are intramedullary, cystic bone lesions which are most often identified in children. 1 SBCs are mostly unicameral, but can be multicameral bone lesions. They are filled with clear to amber fluid, which contains histiocytes, prostaglandins, chronic inflammatory cells, and giant cells.1 –3 The etiology and pathogenesis are unknown, 1 with bone resorption from the cyst lining, increased intra-cystic pressure, inflammation, or traumatic causes all reported as underlying mechanisms.3 –7 About 25% of SBCs are diagnosed in the proximal femur. 1 For this weight-bearing location, there is a significantly increased pathologic fracture risk, and there is no consensus on the treatment of SBCs. Consequently, treatment strategies are highly variable in clinical practice.
Epidemiologically, the peak incidence of SBC is between 3 and 14 years, with a mean age at diagnosis of 9 years,8,9 and a 2:1 to 4:1 male predisposition.4,10 SBCs account for around 3% of all bone lesions;8,11 however, the true incidence remains unknown since SBCs are often diagnosed as incidental finding, and as a consequence, many SBCs may remain undiscovered. 12 However, if symptomatic, 63%–87% present with pathological fracture.10,13 Around 10%–15% of SBCs show spontaneous healing after fracture.9,14,15 Younger age, large cyst size, and fracture are reported risk factors for recurring/persistent symptoms, 16 which occur in 10%–20% of cases. 17
On plain radiographs, SBCs are well-circumscribed lesions with possible mild expansile remodeling and thinning of the cortex (Figure 1). Pathognomonic features such as the fallen fragment sign 18 and rising bubble sign 19 allow for differentiating from aneurysmal bone cyst, cystic fibrous dysplasia, or intraosseous lipoma with cystic degeneration. 1 However, especially after pathological fracture, there is an increased chance of erroneous radiological diagnoses. 20 When in doubt, magnetic resonance imaging (MRI) can clarify the diagnosis (Figure 2), with low signal intensity on T1-weighted images and bright, homogeneous signal intensity on T2-weighted or other fluid-sensitive sequences revealing the fluid-filled cystic aspect of the lesion. 21
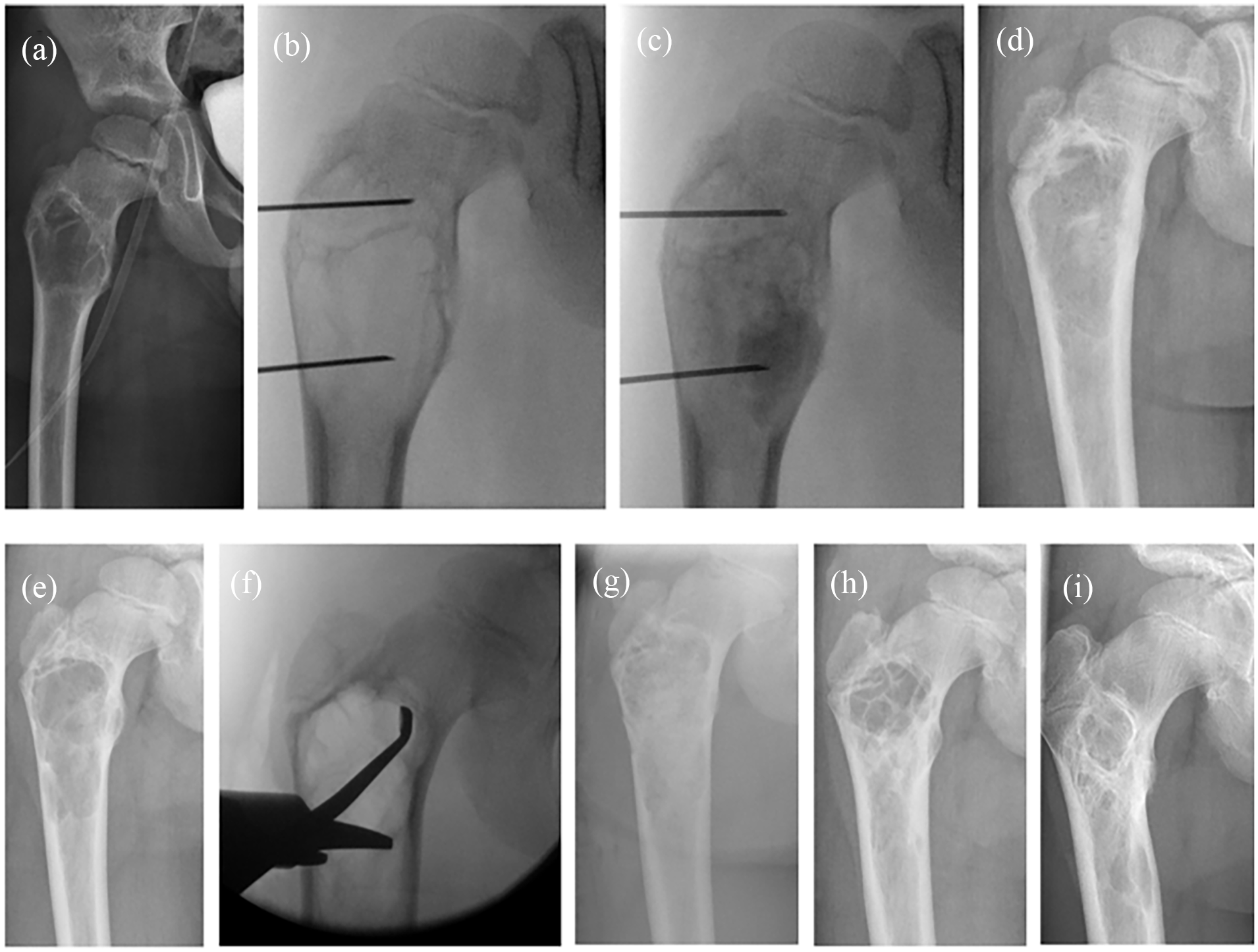
(a) A 6-year-old boy with an SBC in the right proximal femur found accidentally. (b and c) Treated with three ethoxysclerol injections within a period of 7.5 months. (d) Control X-rays 9 months after the last injection showed partial filling of the lesion and cortical thickening. (e) However, 1 year later, cyst progression was observed, which indicated insufficient effect of the injections. (f) Therefore, 3 months later, curettage was performed with ethanolization and allograft bone filling. (g) Post-operative X-ray. (h) Then, 1 year later, cyst residual was observed cranially, for which two more ethoxysclerol injections were administered. (i) The end result, 6 years after the initial diagnosis.
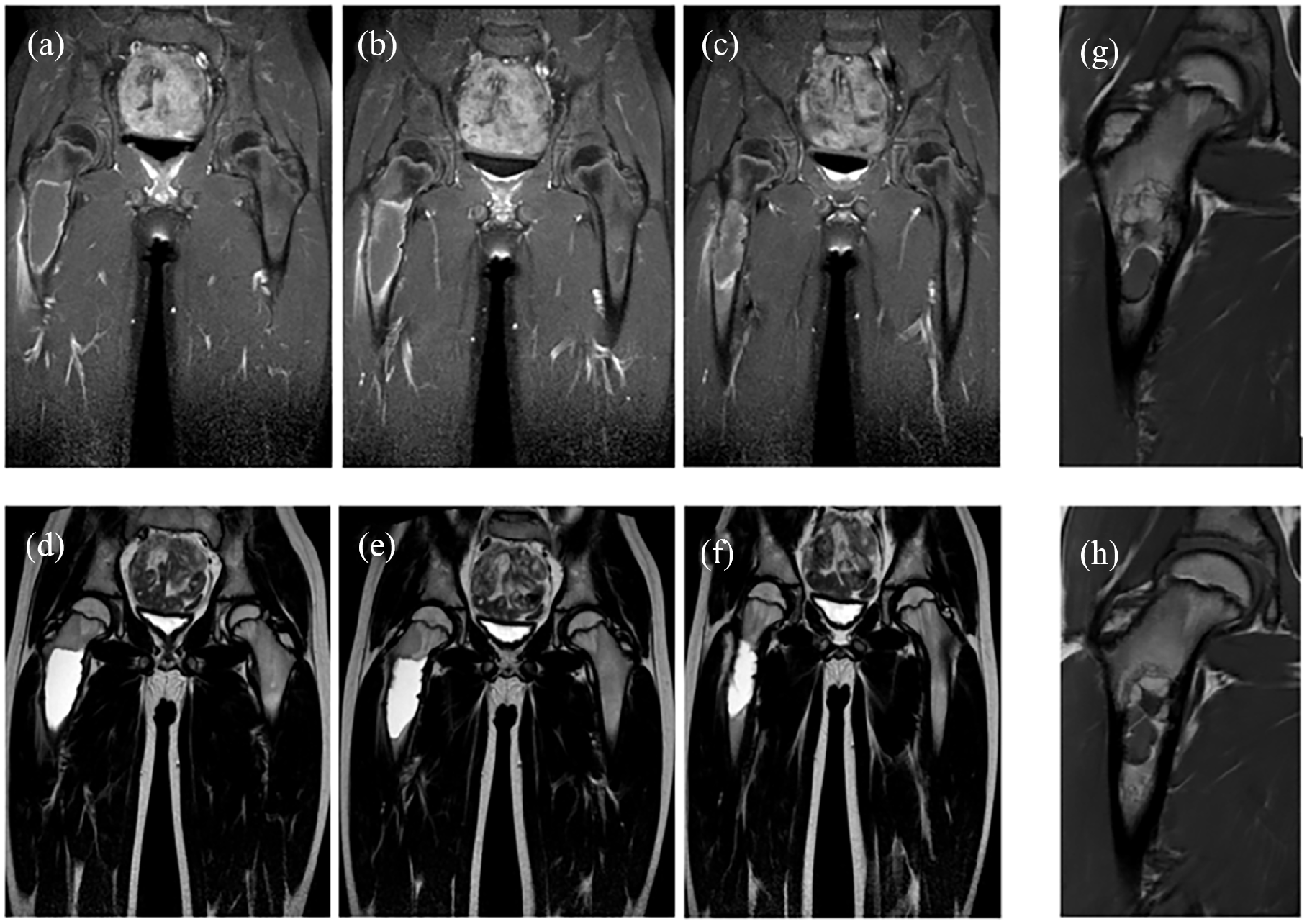
MRI slices of a 9-year-old boy, who came with the complaint of pain in the hip. Images are sorted from dorsal to ventral. (a–c) T1-weighted images show predominantly hypointense signaling within the cyst with an hyperintense inner rim of the cyst. (d–f) T2-weighted images show homogeneous hyperintense signaling. (g and h) T1-weighted images 2 years and 3 months after Depo-Medrol injection showing partial filling of the cyst and multicameral aspect with new septa. After this, follow-up was discontinued since no further treatment was deemed necessary and no further complications were expected.
The objectives of treatment are to prevent the growth of the cyst and optimize bone remodeling, in combination with maintaining structural integrity and function, while minimizing complications, such as fractures, insufficient treatment effect or persistent cysts, and growth disturbances. Treatment strategies include watchful waiting;14,15 percutaneous procedures, for example, with steroids, bone marrow, sclerotherapy, bone morphogenetic protein, or filling with bone substitutes;22 –25 open procedure with curettage, possibly with filling or chemical adjuvants; or a wide range of osteosyntheses, some of which with de-compressive properties, and with or without additional treatment of the SBC.4,11,26 Treatment strategies depend on size, symptoms, localization, (impending) pathological fractures, and local preferences and beliefs in different theories on pathogenesis.
Limited evidence exists regarding the treatment of SBC in the proximal femur, with smaller retrospective studies on this specific localization as the main sources of knowledge.26 –30 These previous studies tend to focus on open surgical interventions, without assessing groups receiving percutaneous procedures alone or watchful waiting. We present a large international multicenter retrospective study on SBCs in the proximal femur, with a wide range of treatment options and their follow-up outcomes. The aim of this study on SBCs in the proximal femur in children was to assess the outcomes and failure-free survival (FFS) of different treatment modalities. As such, the current study may help physicians in making decisions about treatment methods in children and adolescents for this specific weight-bearing location.
Materials and methods
This collaborative study was instigated by [institution] and joined by the members of the European Paediatric Orthopaedic Society (EPOS), the European Musculo-Skeletal Oncology Society (EMSOS), and the International Society of Limb Salvage (ISOLS). The patient population consisted of pediatric patients with proximal femoral SBC, treated in 1 of 10 participating tertiary referral centers worldwide for musculoskeletal oncology between 2000 and 2021.
Inclusion criteria were age ≤ 16 years, primary SBC localized in the proximal femur, diagnosis confirmed by X-ray, MRI, and histology, and a minimal follow-up period of 6 months. Exclusion criteria were involvement distal to the isthmus of the femur, or comorbidities increasing fracture risk, such as rickets, osteogenesis imperfecta, or enchondromatosis/Morbus Ollier.
Data were collected from medical records by all individual centers and added to a pseudo-anonymized database. Demographics, diagnostic features, and treatment characteristics, as well as complications and reinterventions during follow-up were evaluated retrospectively. Index procedures were categorized as watchful waiting, percutaneous procedure (e.g. glucocorticoids, sclerotherapy, and filling with injectable bone substitute), open surgical treatment (e.g. curettage, with or without adjuvants, filling, or osteosynthesis), or osteosynthesis alone (i.e. without specific additional treatment of the SBC).
Cyst volume was approximated on X-ray, computed tomography (CT), or MRI using maximal AP × CC × ML (anteroposterior × craniocaudal × mediolateral distance in mm).
Clinical outcomes were 1-, 2-, and 5-year FFS, time from index procedure until full weight-bearing, re-procedures (either percutaneous or open), (re-)procedures for (impending) fractures, insufficient treatment effect or persistent cysts, time until insufficient treatment effect, or persistent cysts and complications. For watchful waiting, failure was defined as any percutaneous or surgical intervention during follow-up, cyst progression, or pathologic fracture during follow-up. For percutaneous procedure, open surgical treatment, and osteosynthesis alone, failure was defined as pathologic fracture during follow-up, open surgical (re-)intervention, or insufficient treatment effect or persistent cysts. Insufficient treatment effect or persistent cysts were defined as cyst- or symptom progression during watchful waiting or progression or residue after first treatment.
According to our Institutional Review Board and Dutch legislation, this retrospective study was not depending on the Medical Research Involving Human Subjects Act (G19-064) and no informed consent was required.
Statistical analysis
Descriptive analyses were assessed for the complete group and stratified for index procedure in a flowchart and tables. For continuous data, means and standard deviations, or medians and ranges in case of skewed distributions, were presented. Categorical variables were summarized as number of observations and percentages (%).
Comparisons were made between treatment groups using Kruskal–Wallis analysis for age at diagnosis, follow-up, cyst width, cyst volume, and distance to epiphyseal plate. Chi-square tests were applied for sex, localization, pathological fracture at diagnosis, insufficient treatment effect or persistent cysts, whether (re-)procedures were performed (for any reason or for impending fracture), and whether failure occurred. Median time until determination of insufficient treatment effect or persistent cysts was estimated with the reversed Kaplan–Meier method.
For survival analyses, FFS was analyzed using Kaplan–Meier and time until allowed full weight-bearing with one minus Kaplan–Meier graphs, using log-rank tests. Time was determined from index procedure until failure or full weight-bearing. FFS was described for the 1-, 2-, and 5-year time-points in percentages, time until allowed full weight-bearing, and time until determination of insufficient treatment effect or persistent cysts with median and 95% confidence interval.
For the risk factors of survival outcomes, univariate Cox regression analyses were estimated. Covariates included in the model were sex, age at diagnosis below 10 years, fracture at diagnosis, and volume more than 55 cm3, and were chosen based on previous literature. Higher fracture rates have been reported for younger children, males, and patients with larger cyst sizes. 31 Also younger children, large cyst size, and fracture are reported risk factors for recurrences, 16 while fractures have also been described to be associated to spontaneous healing, leading to lower failure rates.14,15 Estimated hazard ratios (HRs) along with their 95% confidence interval (95% CI) were reported.
No imputation methods were used on missing data. IBM Statistical Package for Social Statistics (SPSS) version 25 (Chicago, IL, USA) was used for analysis.
Results
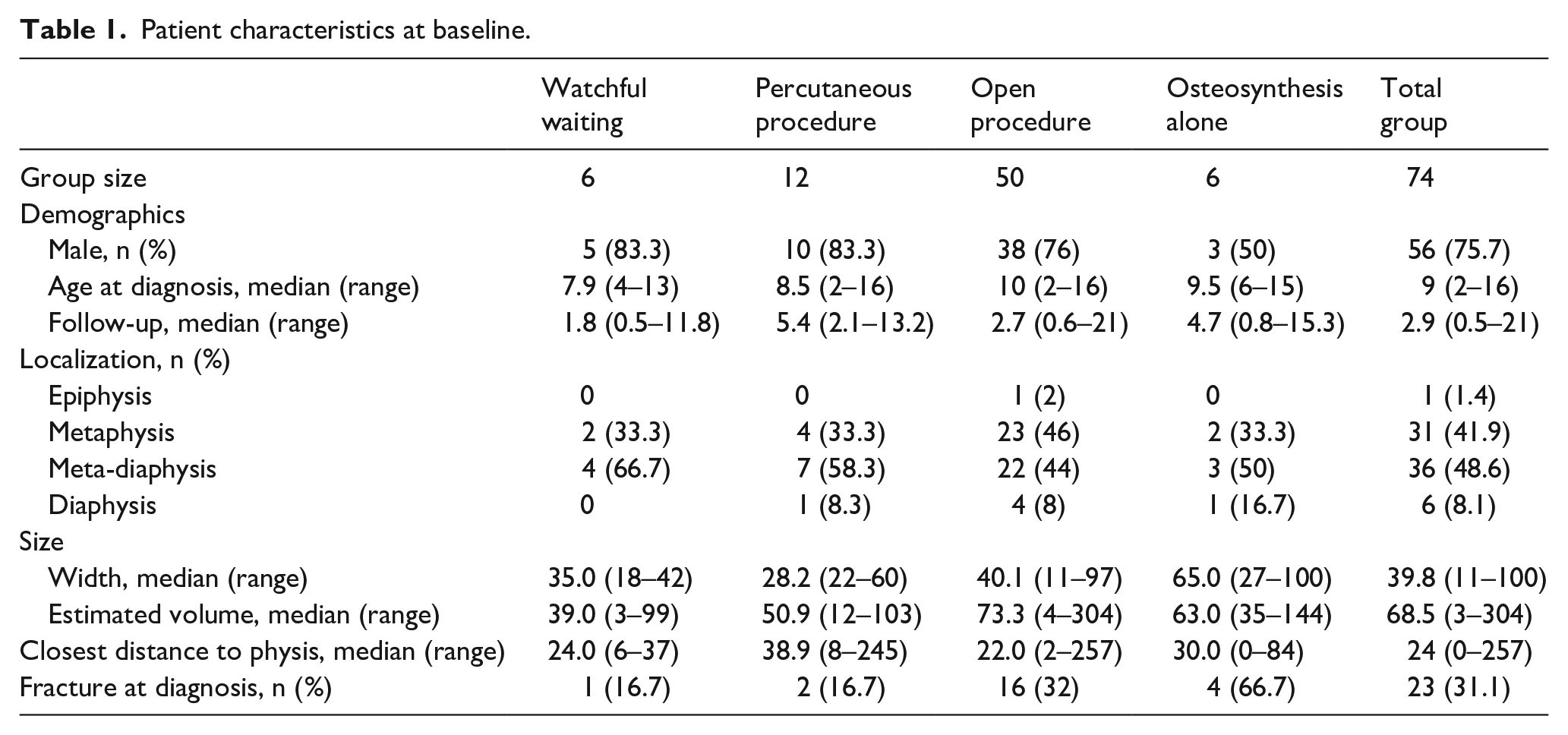
Out of 89 pediatric and adolescent patients with SBC in the proximal femur, 74 were included (Table 1). However, 12 were excluded because of initial presentation (n = 9) after previous treatment with insufficient effect elsewhere or for insufficient follow-up (n = 3; 0, 1, and 4 months). For the included patients, median age at diagnosis was 9 years (range = 2–16) and 76% (n = 56) were male. Median follow-up was 2.9 years (range = 0.5–21).
Patient characteristics at baseline.
In total, 40 patients were diagnosed with radiographs, of whom 3 had additional CT scan, 16 had MRI, and 12 underwent biopsy. However, 16 patients immediately got a CT scan, of whom 13 had additional MRI, 11 biopsy, and 3 bone scintigraphy. Moreover, 21 patients immediately got an MRI, of whom 13 had additional biopsy and 11 bone scintigraphy (Figure 3).
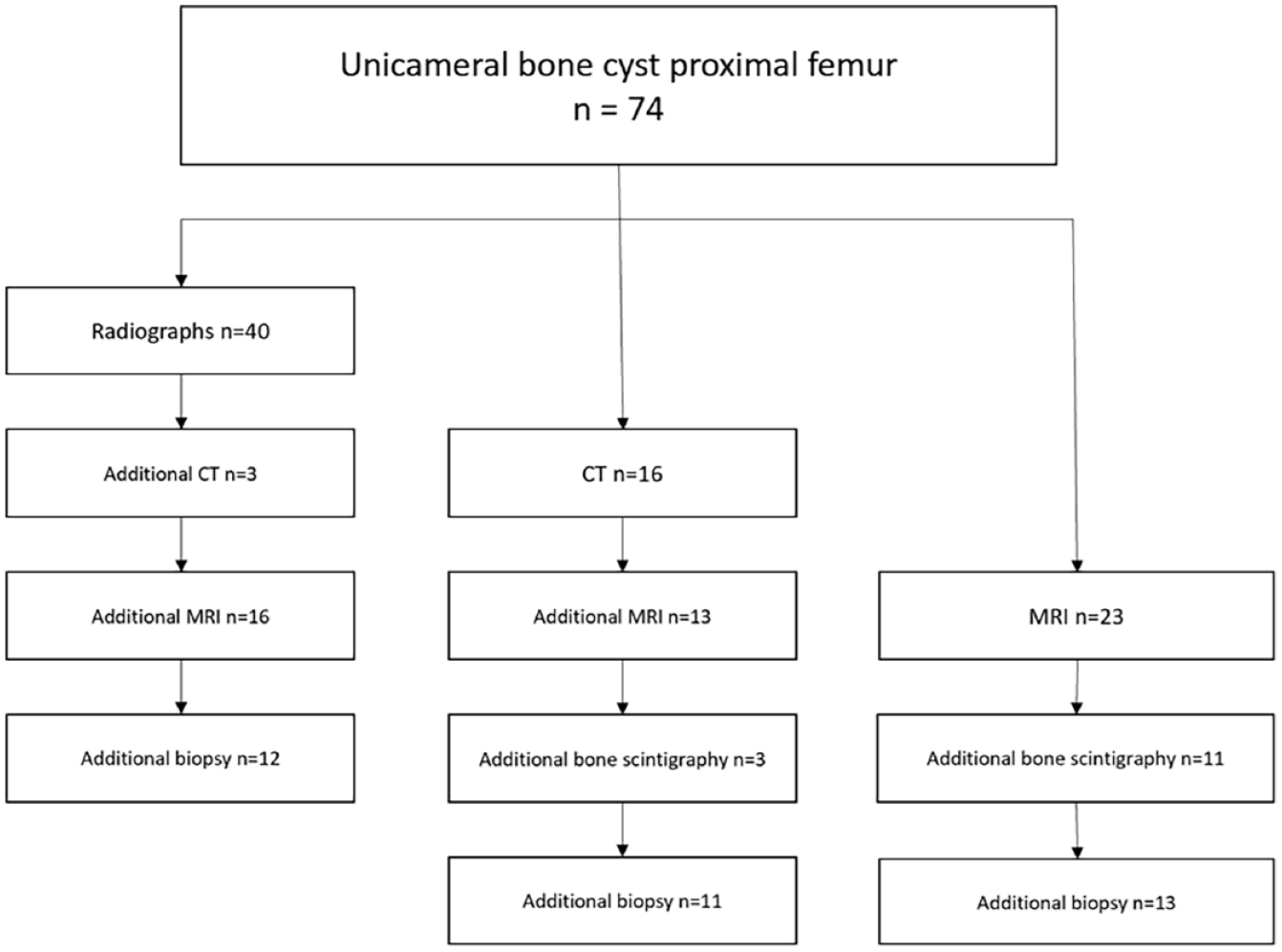
Flowchart of performed diagnostic modalities.
Index procedures were watchful waiting (n = 6), percutaneous procedure (n = 12), open surgery (n = 50), or osteosynthesis alone (n = 6) (Table 2; Figure 4).
Index procedures.

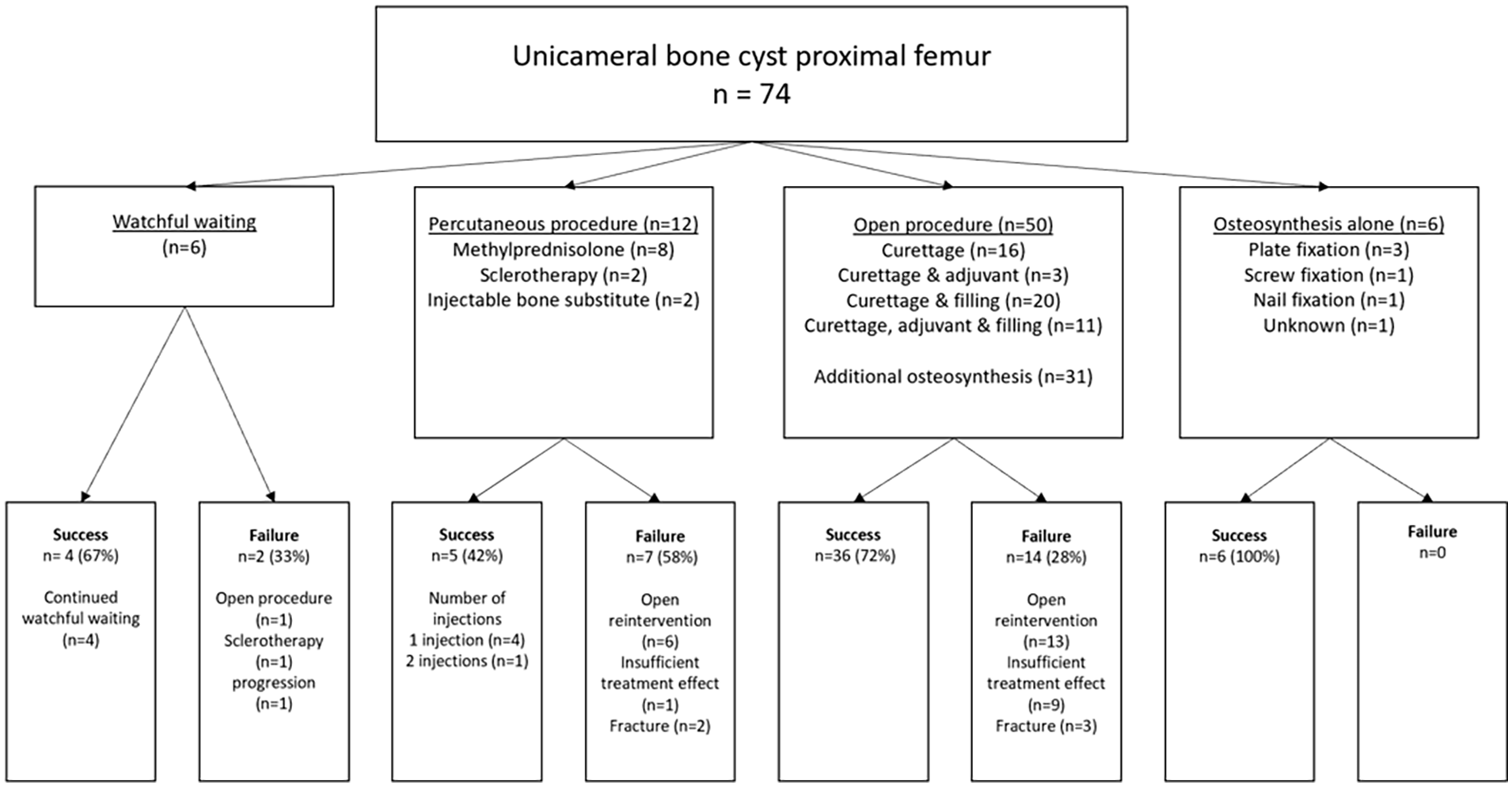
Flowchart of index procedures and their successes and failures.
For the entire group, open (re-)operations were performed 32 times during follow-up. For the different index procedures, the number of patients with open (revision) surgery due to any reason was watchful waiting 1/6 (17%), percutaneous 6/12 (50%), open procedure 13/50 (26%), and osteosynthesis alone 0/6 (p = 0.12). (Re-)operations due to (impending) fractures were performed in 1/6 (16.7%) watchful waiting, 1/12 (8.3%) percutaneous, 7/50 (14%) open procedure, and 0/6 osteosynthesis alone (p = 0.74) (Figure 4), totaling 28% of all open re-procedures overall. In the percutaneous group, nine patients had one injection, two had two injections, and one had three injections during follow-up.
Overall, insufficient treatment effect or persistent cysts were reported in 11 patients (15%), after a median time of 33 months (25.2–40.8). Organized per index procedure: watchful waiting 1/6 (17%) after a median time of 18 months (95% CI = 9.7–26.3), percutaneous procedures 1/12 (8%) after a median time of 86 months (95% CI = 19.1–152.9), and open procedures 9/50 (18%, p = 0.6) after a median time of 30 months (95% CI = 23.7–36.3, p = 0.34).
The main complication of SBCs was fracture during follow-up. Five fractures were observed after the following index procedures: two after injection with methylprednisolone (aged 2 and 7 years), two after curettage with titanium elastic nails (aged 7 and 12 years), and one after curettage and filling with bone graft (aged 9 years). The latter two (aged 12 and 9 years) had diagnosed cyst progression at 3 and 4 months before fracture, respectively. No infections were reported during follow-up in any of the groups.
Overall median time until allowed full weight-bearing was 11 weeks (95% CI = 7.1–14.9): watchful waiting 8 (95% CI = 0.1–15.9), percutaneous 9.5 (95% CI = 3.7–15.3), open procedure 11 (95% CI = −0.7 to 13.7), and osteosynthesis alone 6.5 weeks (95% CI = 5.9–16.1, p = 0.58) (Figure 5).
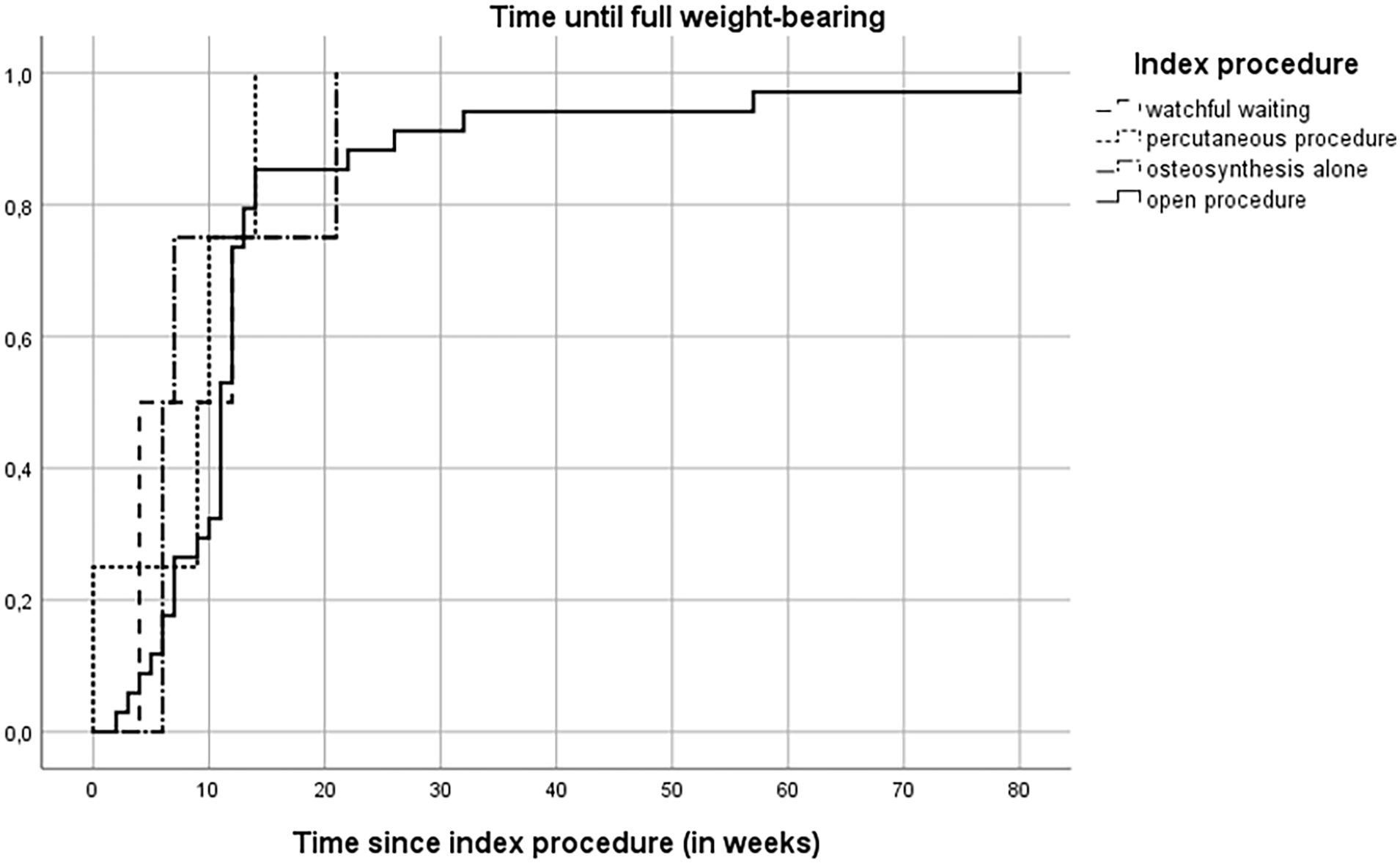
Cumulative incidence of time until full weight-bearing after index procedure.
Failure rates were watchful waiting 2/6 (33%), percutaneous procedures 7/12 (58%), open procedures 14/50 (28%), and osteosynthesis alone 0/6 (p = 0.069; Figure 4). Overall, 1-, 2-, and 5-year FFS was 77.8% (95% CI = 68.2–87.4), 69.5% (95% CI = 58.5–80.5), and 62.0% (95% CI = 47.9–76.1), respectively. For watchful waiting, 1-, 2-, and 5-year FFS was 60% (95% CI = 17.1–102.9) for all three time-points, for percutaneous procedures 50% (95% CI = 21.8–78.2), 41.7% (95% CI = 13.9–69.5), and 41.7% (95% CI = 13.9–69.5), for open procedures 83.8% (95% CI = 73.4–94.2), 74.2% (95% CI = 61.5–86.9), and 62.3% (95% CI = 43.5–81.1), and for osteosynthesis alone 100% until the 5-year mark, respectively (p = 0.049) (Figure 6). For watchful waiting, two patients had failure during follow-up and 4/6 were successful. Among failures in the watchful waiting group, one was treated with three separate injections of ethoxysclerol due to cyst progression, the other patient received titanium elastic nails due to instability; both after 1 year of initial watchful waiting.
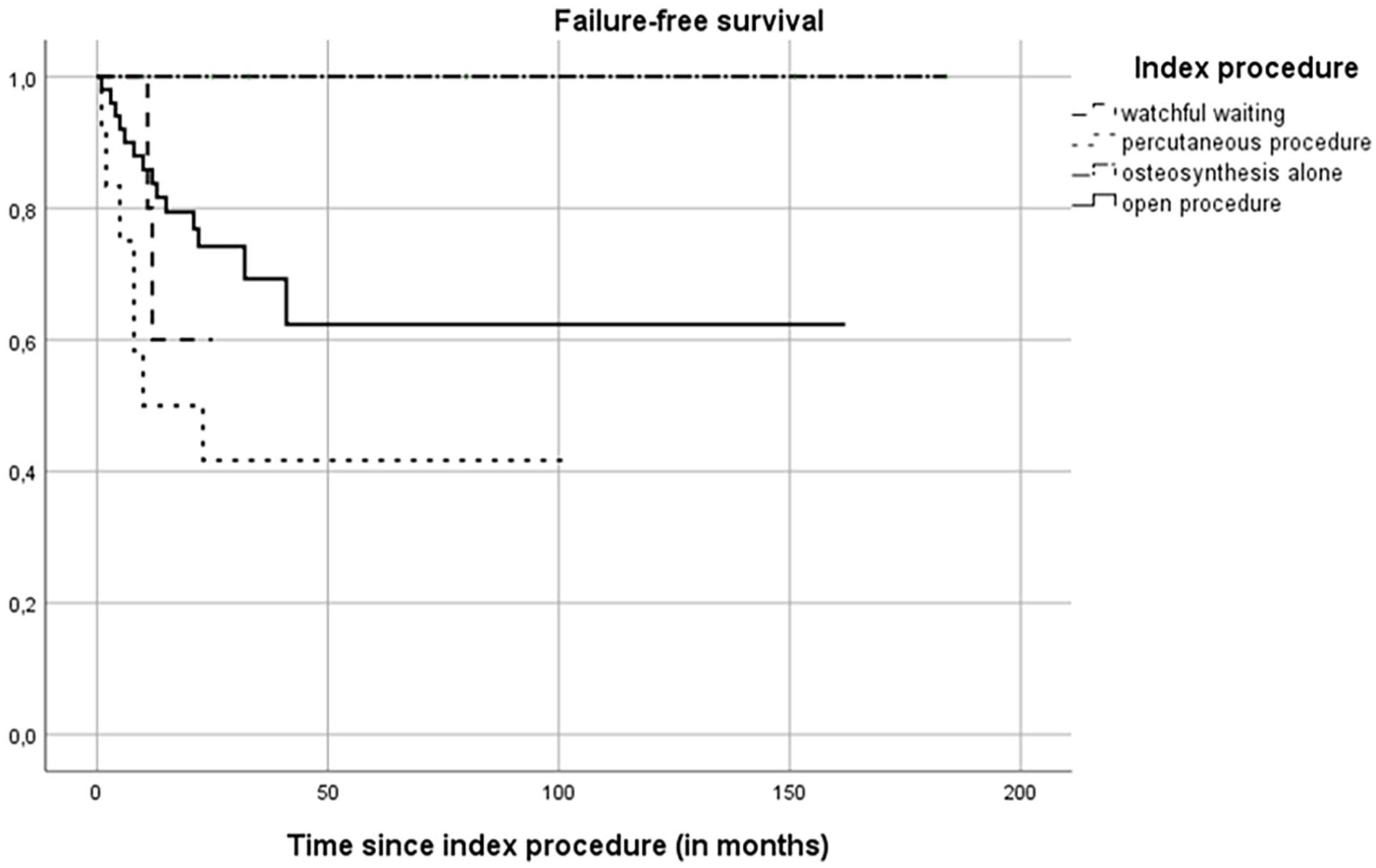
Kaplan–Meier curve of FFS.
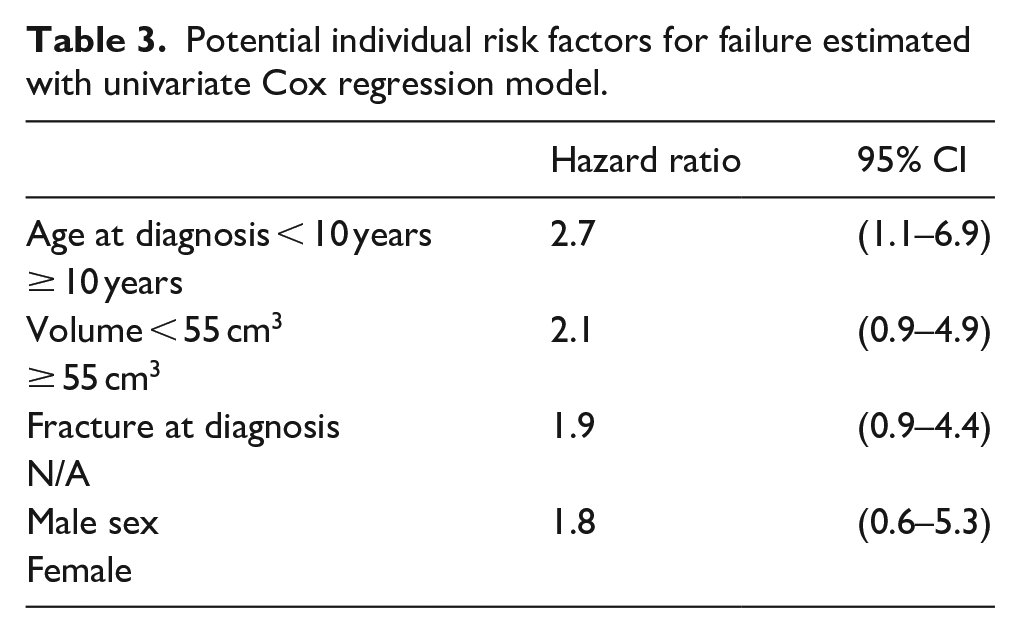
Univariate HRs for failure: age (HR = 2.7, 95% CI = 1.1–6.9), volume (HR = 2.1, 95% CI = 0.9–4.9), pathological fracture at diagnosis (HR = 1.9, 95% CI = 0.9–4.4), and sex (HR = 1.8, 95% CI = 0.6.5.3) (Table 3).
Potential individual risk factors for failure estimated with univariate Cox regression model.
Discussion
This study investigated the outcomes and management of 74 pediatric and adolescent patients with SBC in the proximal femur. The current study may help physicians in making decisions about treatment methods in children and adolescents for this specific weight-bearing location.
Various index procedures were employed, including watchful waiting, percutaneous procedures, open surgery, and osteosynthesis alone. Overall, treatment modalities showed similarity in their outcomes, and at both ends of the treatment paradigm, watchful waiting or osteosynthesis can result in sufficient bone healing and return to normal daily activities.
Failure modes and risk factors for failure
Patient characteristics at baseline showed that most SBCs were located in the metaphysis or meta-diaphysis. Estimated cyst volume varied across groups, with the open procedure group having the largest median cyst volume. In addition, the closest distance to the epiphyseal plate was found to be relatively consistent across the groups. Confounding by indication must be taken into account for relatively smaller cysts more likely undergoing percutaneous procedures with potentially poorer FFS compared to larger cysts more likely undergoing invasive treatments. Differences in FFS might be attributed to the added biomechanical strength in the osteosynthesis group, compared to the percutaneous procedure group where open reinterventions (due to insufficient treatment effect or persistent cysts) and fractures during follow-up were major contributors to the failure rate. On the contrary, a large meta-analysis by Kadhim et al. 32 reported a 81% success rate after percutaneous procedures. However, these results were computed for SBCs in all anatomic locations, many of which are presumably more eligible for percutaneous procedures than the proximal femur (e.g. upper vs lower extremity). 15
More failures were seen in children age < 10 years in this study. An earlier article by Traub et al. 11 described more active cysts in younger patients and consequently, a higher failure rate. The fracture risk may be especially increased in younger children who may be less receptive for fracture prevention instructions of partial or non-weight-bearing. Volume < 55 cm3 and fracture at diagnosis showed a possible association with failure. Increased recurrence rates have been described for younger age and male sex 17 but were not associated with fractures. 33 Cha et al. 33 reported that fractures at diagnosis had no impact on recurrences or healing reliability. In the literature, Traub et al. 11 found a correlation between larger cyst volume in long bones and fracture risk. As mentioned above, the counterintuitive association of smaller volume leading to more failure in this study can be possibly explained by confounding by indication.
Self-healing properties versus persistent cysts
For the osteosynthesis alone group, we hypothesized that penetration of the cyst wall with, for example, screws or intramedullary nails may cause spontaneous healing in SBCs, in concordance with self-healing properties of SBC after fracture. The exact mechanism remains unknown, as does the etiology of SBCs, but we observed good results from osteosynthesis alone. Interestingly, cyst wall disruption without added mechanical stability of osteosynthesis may be insufficient in the proximal femur, as Hardes et al. 34 described that in the proximal femur, cannulated screws performed worse than in the proximal humerus and resulted in long periods of non-weight-bearing.
The overall insufficient treatment effect or persistent cysts rate of 15% in this study was lower compared to recurrence rates reported by Kadhim et al. (23.9% for all available treatments combined), Traub et al. (26.1% for all surgical procedures combined), and Deventer et al. (33.3% after curettage with bone filling).11,32,35
Mechanical stability
Five pathological fractures after index procedure were observed, two after percutaneous procedures (methylprednisolone), two after open surgery with osteosynthesis, and one after open surgery without fixation. Since exactly half of our population received some form of osteosynthesis (n = 37, 50%), it is remarkable that the group without fixation performed so well in this weight-bearing location. This indicates that not all proximal femoral SBCs in pediatric patients need osteosynthesis to reduce fracture risk. 26 Nevertheless, when an open procedure (e.g. curettage) is selected, it might be advisable to use osteosynthesis during the same session.
Watchful waiting
The use of watchful waiting for these pediatric cysts has been rigorously explored and promoted since SBCs are benign lesions with tendencies toward spontaneous healing.11,15,31,32,35 Varying healing percentage is reported in previous reports compared to our 67%: Kadhim et al. reported 63.3% of femoral SBCs healed in a meta-analysis including 712 patients, Urakawa et al. reported 48% spontaneous healing after 6 months in 31 patients, and Green et al. reported 95.7% healing after mean of 3.3 years in 44 patients.15,31,32 Watchful waiting, however, implies an ongoing risk of pathological (re-)fracture or need for surgical intervention during follow-up, which should be mentioned in shared decision-making with patient and their parents or caregivers. Especially in the proximal femur, considering swiftness of return to normal daily activities for these children with SBCs, without constant fear of fracture, it would be our recommendation to choose fixation over watchful waiting, although both can be viable options.
Time until full weight-bearing
Time until full weight-bearing showed no significant differences between index procedures in our series, with a median of 11 weeks. However, in the open procedures group, there were some cases with exceptionally long time until full weight-bearing (Figure 5).
Strengths and limitations
This study’s main strengths were the evaluation of time until full weight-bearing and the large study population of proximal femoral SBCs, with reporting of all traditional treatment strategies. To the authors’ knowledge, we present the largest pediatric proximal femoral SBC study to date. The use of time until full weight-bearing is an especially valuable outcome for informing patients and parents on the anticipated effects of suggested treatment strategies.
Limitations of this study are confounding by indication, short follow-up time, and small subgroup sizes. Choice of treatment strategies has presumably been influenced by cyst size, location, and associated fracture risk, and due to the limited subgroup sizes, multivariate regression models could not be used to correct for this. Also, the minimal follow-up time of 6 months may have led to underestimations in the number of cases with insufficient treatment effect or persistent cysts and failures. Finally, the multicentered, retrospective study design resulted in a diverse array of used treatments, although representative for current common practice.
Conclusion
This study’s findings provide valuable insights into the management and outcomes of pediatric and adolescent patients with SBC in the proximal femur. The results indicate that different index procedures, including watchful waiting, percutaneous procedures, open surgery, and osteosynthesis alone, can be effective in treating SBCs in this population. Age at diagnosis (< 10 years), volume of the cyst (< 55 cm3), and fracture at diagnosis may be associated with an increased risk of failure, but the associations were not statistically significant due to the limited sample size.
The relatively low rate of fractures and infections during follow-up suggests that the chosen treatment approaches were generally successful in managing the cysts and minimizing complications. However, it is essential to monitor patients closely for signs of progression or insufficient treatment effect, as these were reported in 14.9% of cases.
The fairly latent disease course with possibility for self-containment implies that watchful waiting can deliver good results in selected cases. However, due to the high risk of fracture in this weight-bearing location, osteosynthesis alone may be an excellent treatment strategy, avoiding possible overtreatment or multiple anesthesia administrations of open procedures with curettage and percutaneous procedures, while reducing pathological fracture risk. The treatment aim should be maintained for cyst control, while minimizing complications and time until children resume their normal activities. Therefore, an individualized balance should be found between undertreatment, with potentially higher pathological fracture or insufficient treatment effect or persistent cyst risks and overtreatment, with larger interventions and accompanying exposures and longer time until return to full weight-bearing.
Supplemental Material
sj-pdf-1-cho-10.1177_18632521231221553 – Supplemental material for As simple as it sounds? The treatment of simple bone cysts in the proximal femur in children and adolescents: Retrospective multicenter EPOS study of 74 patients
Supplemental material, sj-pdf-1-cho-10.1177_18632521231221553 for As simple as it sounds? The treatment of simple bone cysts in the proximal femur in children and adolescents: Retrospective multicenter EPOS study of 74 patients by Thomas PG van Geloven, Lizz van der Heijden, Minna K Laitinen, Domenico A Campanacci, Kevin Döring, Dietmar Dammerer, Ismail T Badr, Mikko Haara, Giovanni Beltrami, Gerhard M Hobusch, Tanja Kraus, Philipp Scheider, Camilo Soto-Montoya, Masood Umer, Javeria Saeed, Phillipp T Funovics, Marta Fiocco, Michiel AJ van de Sande and Pieter Bas de Witte in Journal of Children’s Orthopaedics
Footnotes
Author Contributions
T.P.G.v.G., L.v.d.H., P.B.d.W., and M.A.J.v.d.S. made substantial contributions to the conception or design of the work. T.P.G.v.G., L.v.d.H., M.K.L., D.A.C., K.D., D.D., I.T.B., M.H., G.B., G.M.H., T.K., P.S., C.S.M., M.U., J.S., P.T.F., M.F., P.B.d.W., and M.A.J.v.d.S. involved in acquisition and analysis. T.P.G.v.G., L.v.d.H., M.F., P.B.d.W., and M.A.J.v.d.S. participated in interpretation of data for the work. T.P.G.v.G., L.v.d.H., M.K.L., D.A.C., K.D., D.D., I.T.B., M.H., G.B., G.M.H., T.K., P.S., C.S.M., M.U., J.S., P.T.F., M.F., P.B.d.W., and M.A.J.v.d.S. drafted the work or revised it critically for important intellectual content. All authors gave final approval of the version to be published. All authors agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Author Note
On behalf of the EPOS study group including: Sebastian Farr1, Manuel R Medellin Rincon2, Kirsten M van der Neut3, Reinhard Windhager4, Mohamed K Mesregah5, Roberto Scanferla6, and Chiara Suardi6
1Pediatric Orthopedics and Foot and Ankle Surgery, Orthopedic Hospital Speising, Vienna, Austria
2Orthopaedic Oncology/Sarcoma Unit, Fundacion CTIC Luis Carlos Sarmiento Angulo, Bogotá, Colombia
3Department of Orthopedic Surgery, Leiden University Medical Center, Leiden, The Netherlands
4Division of Orthopedics, Department of Orthopedics and Trauma Surgery, Medical University of Vienna, Vienna, Austria
5Orthopedic Surgery, Menoufia University, Shebin El-Kom, Egypt
6Orthopedic Oncology and Reconstructive Surgery, Azienda Ospedaliero Universitaria Careggi, Florence, Italy
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval for retrospective,multicenter study
This study was conducted in accordance with the Declaration of Helsinki. Approval was granted on 28 January 2020. The Medical Research Ethics Committee Leiden Den Haag Delft acted as the central Institutional Review Board (IRB), whose review was accepted by all participating institutions’ IRBs (Ref. G19.064). The central IRB determined that this research involved no greater than minimal risk and approved a waiver for informed consent.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
