Abstract
Abstract
Purpose
Retrospective analysis of data to determine if the use of postoperative furosemide therapy decreases the incidence of thoracocentesis in children and adolescents undergoing kyphosis or scoliosis surgery. One of the potential complications seen in patients undergoing scoliosis or kyphosis surgery is the development of a clinically symptomatic postoperative pleural effusion requiring thoracocentesis, which is an invasive procedure that is associated with discomfort to the patient and a minimal risk of an iatrogenic pneumothorax.
Methods
We retrospectively reviewed the medical records and X-rays of 190 patients who had undergone corrective surgery for AIS/kyphosis. All patients underwent an ASF or an APSF or a PSF with a minimum 3 level costoplasty. We compared the rate of thoracocentesis in group 1 (no furosemide; 161 patients) to that in group 2 (furosemide; 29 patients). Follow-up chest radiographs were evaluated for a decrease or resolution of the effusion.
Results
In group 1, 26 out of the 161 patients required thoracocentesis (16%); in group 2, only 1 out of the 29 patients underwent thoracocentesis (3.4%).
Conclusions
Postoperative furosemide in patients who undergo significant pleural manipulation during spinal deformity surgery may decrease the incidence of clinically symptomatic pleural effusion requiring thoracocentesis. This study represents a true trend; however, a high level of significance can only be achieved by performing a larger randomized controlled study.
Introduction
Correction of scoliosis or kyphosis by an anterior and/or posterior spinal fusion involves extensive surgery and may include multiple level rib resections (costoplasty), thoracotomy as well as thoracoplasty. The rib hump deformity, which is aesthetically unsightly to most patients, can be corrected by costoplasties, thereby giving patients a back that is aesthetically acceptable. To improve cosmesis, we have elected to perform multiple level costoplasties with anterior/posterior or posterior only scoliosis correction.
Multiple level costoplasty involves a significant degree of manipulation of the parietal pleura and, most often, of the pleuroperiosteal membrane. The bleeding edge of the cut ribs may damage the pleura, producing an effusion. In addition, the membrane may weep fluid following thoracoscopy/thoracotomy, resulting in more effusion.
Scoliosis or kyphosis surgery may also require an infusion of a large volume of fluid to maintain hemodynamic stability. This can result in a shift of the fluid from the intravascular to the intercellular (interstitial or extracellular or third space) compartment and eventually to the intracellular compartment. Collection of fluid in the third space can manifest itself as pulmonary congestion and a pleural effusion [1].
High-quality anesthesia with close monitoring of the intra-operative and postoperative fluid balance will decrease the likelihood of a pleural effusion secondary to third space loss. We believe that the pleural effusion seen in our patients was due to aggressive manipulation of the pleura or due to multiple level costoplasties. A clinically symptomatic pleural effusion (shortness of breath and room air SpO2 < 90 mmHg) required ultrasound-guided thoracocentesis [2–5]. Though this is a minimally invasive procedure performed by the interventional radiologist, it adds another level of morbidity to the patients.
We were encouraged by reports in the literature describing the effective use of loop diuretics in the treatment of pleural effusion secondary to fluid overload conditions such as congestive heart failure [6, 7] and chronic renal failure. We, therefore, retrospectively reviewed the patient charts and documented the incidence of thoracocentesis, with and without the administration of postoperative furosemide, following scoliosis or kyphosis surgery from 2000 to 2004.
The aim of this study was to determine whether postoperative furosemide decreases the incidence of thoracocentesis following scoliosis or kyphosis surgery.
Materials and methods
We retrospectively reviewed the medical records and the X-rays of 190 patients (ages 10–17 years) with AIS or idiopathic kyphosis (Scheuermann's/juvenile roundback disease). The patients underwent an anterior, posterior or an anterior and posterior spinal fusion in order to achieve scoliosis correction and anterior and posterior spinal fusion to achieve kyphosis correction. Anterior release and spinal fusion was performed by video-assisted thoracoscopic release (VATS) or by conventional thoracotomy. The pleura was not routinely closed in the VATS procedure, but was always closed in the conventional thoracotomies. All patients who underwent a standalone posterior spinal fusion also had a minimum three-level costoplasty for rib hump deformity correction. The abovementioned surgeries involve significant manipulation of the pleura, predisposing the patients to the development of a pleural effusion. The patients were divided into two groups. This was a single surgeon sequential series in both groups. Group 1 consisted of 161 consecutive patients (10–17 years) who underwent the corrective surgery and postoperatively did not receive furosemide. Group 2 consisted of 29 consecutive patients (11–16 years) who underwent the surgery and were administered 20 mg furosemide intravenous (IV) initially and then per oral (PO) when diet was tolerated in the postoperative period until the day of discharge (average 5 days). All patients were administered the same dose in IV as well as per oral form. All 190 patients had a chest tube placed during the surgery, which was removed when the drainage was less than 50 cc in an 8 h nursing shift. This was usually 48–72 h post surgery. Average length of hospital stay for both groups was 5 days. The thoracocentesis, when required, was performed with ultrasound guidance, by a board-certified radiologist. A pigtail catheter was retained if the thoracocentesis drained more than 500 cc of fluid. The pigtail removal criteria were the same as the chest tube removal criteria. The patient's pre-discharge chest radiograph was reviewed to confirm a decrease or a resolution of the pleural effusion. Average length of stay for those who underwent thoracocentesis was 6 days.
Results
In spite of the presence of the chest tube for the first 48–72 h after surgery, 27 (5 male, 22 females, aged 11–16 years) patients out of 190 required a thoracocentesis after the removal of the chest tube due to a clinically symptomatic re-collection of the pleural effusion. Twenty-six patients who underwent thoracocentesis were in group 1, and 1 patient was in group 2 (Fig. 1). The incidence of thoracocentesis in group 1 was 16% (26 out of 161 patients), and 3.4% (1 out of the 29 patients) in group 2 (Fig. 2).
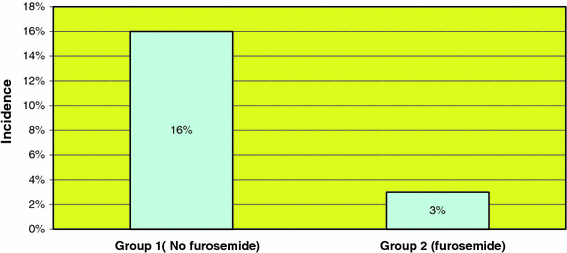
Incidence of thoracocentesis (%)
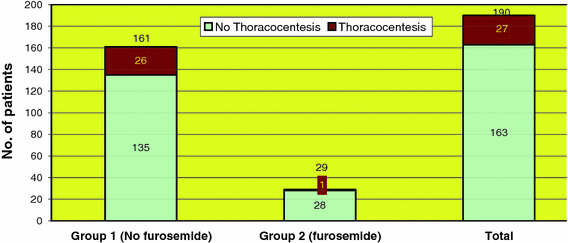
Incidence of thoracocentesis (no. of patients)
Of the 161 patients in group 1, 32 patients underwent a standalone anterior spinal instrumentation and fusion (ASF), 66 patients underwent an anterior release and posterior spinal instrumentation and fusion (APSF), and 63 patients underwent a standalone posterior spinal instrumentation and fusion (PSF) with a minimum three-level costoplasty. Of the 32 patients who had an ASF, 12 patients had a VATS ASF with instrumentation and 20 patients had an open anterior thoracolumbar instrumentation. Of the 66 patients who underwent an APSF, 53 patients underwent VATS anterior release while 13 patients underwent open anterior release via thoracotomy.
Of the 29 patients in group 2, 4 patients underwent a standalone ASF (3 VATS and 1 open), 8 patients underwent an APSF (all were VATS releases), and 17 patients underwent a standalone PSF.
Of the 27 patients who underwent thoracocentesis, 5 patients had a standalone ASF (4 VATS and 1 open thoracolumbar instrumentation), 11 patients had an APSF (7 VATS and 4 open thoracic releases), and 11 patients had a standalone PSF (Fig. 3).
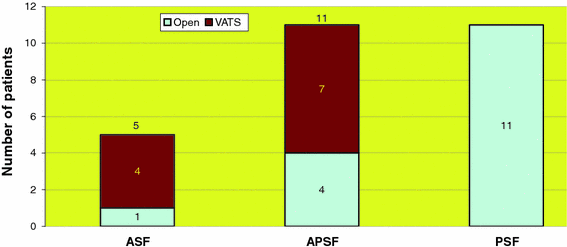
Thoracocentesis
The rate of thoracocentesis in patients who underwent ASF was 16% (5 out of 32 patients) in group 1 and 0% in group 2 (Fig. 4). Similarly, the rate of thoracocentesis in patients who had an APSF was 17% (11 out of 66 patients) in group 1 as compared to 0% in group 2 (Fig. 5). Patients in group 1 who underwent a standalone PSF had an incidence of thoracocentesis of 16% (10 out of 63 patients), as opposed to an incidence of 6% (1 out of 17 patients) in group 2 (Fig. 6). In a further comparison, patients in group 1 who underwent VATS ASF had an incidence of thoracocentesis of 33%, as compared to an incidence of 13% in patients in group 1 who underwent VATS APSF. The use of furosemide postoperatively resulted in a statistically significant (P < 0.05, chi square test) decrease in the rate of thoracocentesis in the ASF, APSF and PSF patients.
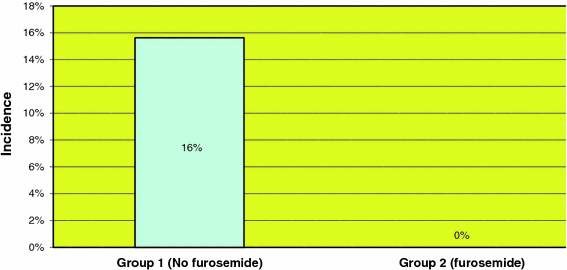
Incidence of thoracocentesis in patients with ASF
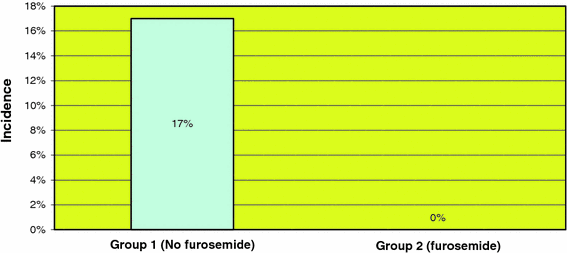
Incidence of thoracocentesis in patients with A/PSF
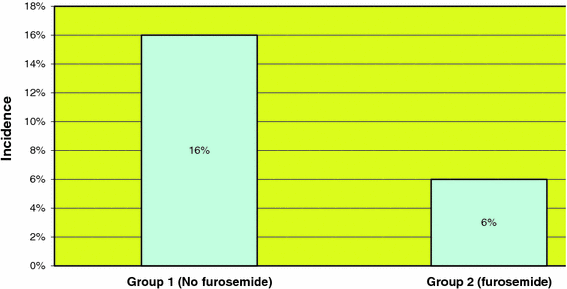
Incidence of thoracocentesis in patients with PSF
Discussion
There is an appreciable incidence of symptomatic pleural effusion after spinal deformity surgery, as the pleura undergoes manipulation as part of the surgical procedure. Though ultrasound-guided thoracocentesis is a minimally invasive procedure performed by the interventional radiologist at our institute, it carries significant risks of infection and iatrogenic pneumothorax [8], even when performed by skilled physicians. We therefore decided to look at a pharmacotherapeutic option to treat pleural effusions. One of the 27 patients who underwent thoracocentesis did in fact develop a small iatrogenic pneumothorax. The patients who underwent thoracocentesis stayed one extra day in the hospital. Though furosemide has been used to treat pleural effusion in other medical conditions, its use had not been described in the postoperative situation after spinal deformity surgery. This prompted its use and investigation at our institute.
Pleural effusion [9] is defined as a collection of at least 10–20 ml of fluid in the pleural space. It can develop due to either to increased filtration (transudative) or due to inadequate absorption of accumulated fluid. The space between the parietal and visceral pleura (pleural cavity) normally has a small amount of fluid. Both the pleural surfaces are supplied with blood via the systemic circulation. They also possess lymphatic vessels. The venous circulation drains approximately 90% of the normal fluid, and the remaining 10% is drained by the lymphatics.
A fine balance exists between the hydrostatic pressure (promoting filtration) and the oncotic pressure (promoting resorption) in the pleural capillary vasculature. Normally the net absorption of the pleural fluid equals the net filtration. The lymphatic drainage also adds to the net absorption of the pleural fluid. Pleural effusion occurs when this fine balance is disrupted by either a manifold increase in the hydrostatic pressure, resulting in an increase in the net filtration forces (called transudative effusion), or a significant decrease in the net absorptive forces as seen in patients with pleural trauma or pleural inflammation. In the latter there is an increase in the permeability of the pleural membrane to proteins and other inflammatory cells, resulting in an effusion.
When multiple level costoplasty is performed, the rib periosteum is not closed. As a result, the rib bed as well as the cut ribs ooze, despite being covered with bone wax. Even when there is no obvious pleural injury from posterior costoplasties, the cut end of the ribs may damage the pleura, resulting in effusion. Similarly, when a thoracotomy or thoracoscopy is performed, there is significant manipulation of the parietal pleural, resulting in pleural damage and ensuing pleural effusion.
The chest wall and diaphragmatic movements also enhance the absorption of pleural fluid. Postoperatively, the chest wall and diaphragmatic movements are often decreased due to pain emanating from the costoplasty site, thus causing a decrease in the absorptive capacity of the pleura and resulting in a pleural effusion.
Not all patients with pleural effusions are symptomatic. Mild pleural effusions are usually asymptomatic and undergo resorption, in due course, without any intervention. These patients do not need a thoracocentesis. Moderate to severe pleural effusions are often clinically symptomatic and need to undergo a thoracocentesis. Ultrasound-guided thoracocentesis is currently the treatment of choice for clinically symptomatic pleural effusions.
Pharmacotherapy, on the other hand, is noninvasive and is unlikely to lead to complications if administered in the correct doses. Furosemide [10, 11] is a powerful loop diuretic. It acts primarily at the loop of Henle; however, it also acts on the proximal and distal collecting tubule in the renal parenchyma. It inhibits the resorption of sodium and chloride from the loop of Henle, leading to a secondary decrease in the absorption of water and thus diuresis. The diuresis causes a direct reduction in the intravascular volume, resulting in the resorption of the transudative and interstitial fluid (third space fluid; e.g., pleural effusion) back into the intravascular compartment and circulation. This brings about a decrease of the pleural effusion, and possibly the rate of thoracocentesis.
In a review of the literature, we could not find any articles evaluating the role of furosemide in the treatment of pleural effusion following kyphosis or scoliosis surgery.
Conclusion
This study suggests that the use of postoperative furosemide in patients who undergo significant pleural manipulation during spinal deformity surgery may decrease the incidence of clinically symptomatic pleural effusion requiring thoracocentesis. This study represents a true trend; however, a high level of significance can only be achieved by performing a larger randomized controlled study.
