Abstract
Two morphologically different porous gold layered on Cu-oxide-based electrochemical sensors were developed for the selective detection of dopamine in the presence of uric acid, ascorbic acid or dextrose. The nanoparticles were prepared by layering Au onto either a cubic or octahedron-shaped Cu-oxide crystal via a galvanic reaction. These were characterized with scanning electron microscopy, energy dispersive X-ray and X-ray photoelectron spectroscopy. The porous structure of the gold over layer was clearly visible on the scanning electron microscopy image while the macro morphology was maintained. X-ray photoelectron spectroscopy confirmed the presence of metallic gold while both CuI (CuO) and CuII (Cu2O) were present in the samples. These two Au/Cu-oxide nanocomposites were used to modify glassy carbon electrodes and were tested for their dopamine sensing ability. Differential pulse voltammetry was used to investigate the selectivity towards dopamine in the presence of different interfering molecules uric acid, ascorbic acid and dextrose). From the differential pulse voltammetry, the lowest limit of detection was found to be 1.1 μM, with a sensitivity of 3.4 μA mM−1 mm−2 in the linear range of 10–250 μM for the porous gold layered covering the octahedron Cu-oxide-modified glassy carbon electrode.

Introduction
Dopamine (DA) is neurochemical, which transmits signals between neurons. 1 DA influences many physiological conditions (such as mood, memory, motor control, cognitive functions, etc.) 2 and organ systems (cardiovascular, central nervous, renal and endocrine systems). 3 Irregular blood levels of DA induce the onset and advancement of a number of neurological diseases, such as Parkinson’s, Alzheimer’s, attention deficit and hyperactivity disorder, Huntington’s, schizophrenia and addiction. 4 Therefore, the detection in a highly sensitive and selective approach is critical for the early detection of the aforementioned diseases.
Numerous reports are available on potential methods to be used for the determination of DA concentrations, these include spectroscopic,5,6 chromatographic,7–9 and electrochemical sensing.6,10–12 Of these various methods, electrochemical sensing offers rapid, effective and efficient testing with low detection limits.13–15 During the electrochemical oxidation of DA, dopaminoquinone is formed through a one-step two-electron transfer process. 16 This process, however, this is not reproducible on bare electrodes. 17 To circumvent this complication, nanoparticle-modified electrode surfaces have been employed due to the improved chemical, thermal and mechanical properties the nanoparticles provide as well as large their surface areas. Nano-sized metal oxides electrochemical sensors resulted in the detection of biomolecules with good sensitivity and stability. Cu2O nanomaterials have been used in the detection of glucose,18,19 tryptophan, 20 and DA.21,22 In addition, it is well known that the physical and chemical properties of nanoparticles are dependent on their shape, enhanced glucose sensing was reported when the morphology and accordingly the exposed facets of Cu2O nanocrystals is controlled.23–25 The octahedral exposing the (111) facet showed superior sensing ability towards glucose over the cubic morphology. 23
Due to the high conductivity of gold nanoparticles, they have been successfully employed for DA sensing,26–28 using nanoporous gold structures results in an even higher electrocatalytic activity due to the increased surface area.29,30 A gold–cuprous oxide nanocomposite resulted in a synergistic effect for the detection of DA and could differentiate it in the presence of interfering biological molecules, such as uric acid (UA) and ascorbic acid (AA).31–33
Based on the background presented above, in this work, porous nano gold layered on a cubic or octahedral Cu2O nanocrystals is proposed for the electrocatalytic detection of DA. The two Cu2O nanocrystals were prepared by colloidal synthesis. The nanoporous gold structure was layered onto the Cu2O nanocrystals via galvanic reaction in a surfactant-free oil-in-water emulsion. The porous Au/Cu-oxide nanoparticles-modified glassy carbon electrode (GCE) was tested for the sensing of DA alone and in the presence of interfering biological molecules.
Results and discussion
Cubic and octahedron shaped Cu-oxide nanocrystals (having an average edge length of 1.14 and 1.31 μm, respectively, see Supplemental Figure S1 for the scanning electron microscopy (SEM) images) were prepared by established colloidal synthesis. 34 A porous gold structure was deposited onto the cube and octahedron morphology of the Cu-oxide nanocrystal templates during a galvanic reaction in a surfactant-free oil-in-water emulsion. 35 Hexadecane were adsorbed onto the surface of the Cu-oxide surface forming interfacial nanodroplets, which protected the surface of the Cu-oxide. Only the uncapped Cu-oxide surface was involved in the galvanic reaction. 36 The progression of nanodroplet adsorption and the galvanic gold layering on the cube and octahedron morphology is shown by the SEM images in Figure 1.

SEM images illustrating nanodroplet adsorption and the galvanic gold deposition on the Cu-oxide cubic morphology after (a) 6 h, (b) 12 h and (c) 24 h and on the Cu-oxide octahedron morphology after (d) 6 h, (e) 12 h and (f) 24 h. The scale bar applies to (a)–(f).
After 6-h reaction time, no visible changes were observed for the cubic morphology. The SEM image after 12-h reaction time showed nanodroplet adsorption with an average size of 190 nm adsorbed onto the surface of the (100) facet. The energy dispersive X-ray (EDS) of the sample after 12 h (Supplemental Figure S3) revealed the presence of approximately 5.5% Au (compared the 63% Cu), indicating that the galvanic reaction has commenced, whereas the EDS after 6 h (Supplemental Figure S2) showed no Au present. After 24 h, etching of the Cu and formation of a porous structure can be observed, with the EDS (Supplemental Figure S4) indicating 13.2% Au (as compared to 58.4% Cu).
The SEM image of the octahedron morphology in Figure 1 illustrates first signs of nanodroplet adsorption and formation of the porous gold structure at 6 h. The EDS (Supplemental Figure S5) confirmed this with 33.2% C (the hexadecane) and 2.8% Au (compared to the 43.4% Cu). The presence of the Cl (3%) implied that some of the Au molecules might still be ionic and has not deposited yet. The adsorption of the nanodroplets is significantly faster than the adsorption on the cubic morphology, which is in correlation with the reported surface energy of the (111) facet being lower than the (100) facet, 0.76 and 1.26 J m−2. 37 The SEM taken at 12 and 24 h (Figure 1) illustrates the advanced etching of the Cu-oxide nanoparticles making way for the porous gold structure to form, with the EDS showing an increase in Au concentration of 15.8% and 35.1% (compared to the 53.2% and 42.1% Cu present). Like the cubic structure, the global octahedron morphology of the nanoparticle was retained.
X-ray photoelectron spectroscopy (XPS) was used to analyse the elemental composition and to confirm the oxidation state of the Au and the Cu in the porous Au layer on the cubic (

XPS spectra: (a) the entire Cu 2p area, (b) the deconvoluted Cu 2p3/2 area and (c) the deconvoluted Au 4f area of cubic (C) and octahedron (O) porous Au/Cu-oxide nanocrystals, respectively.
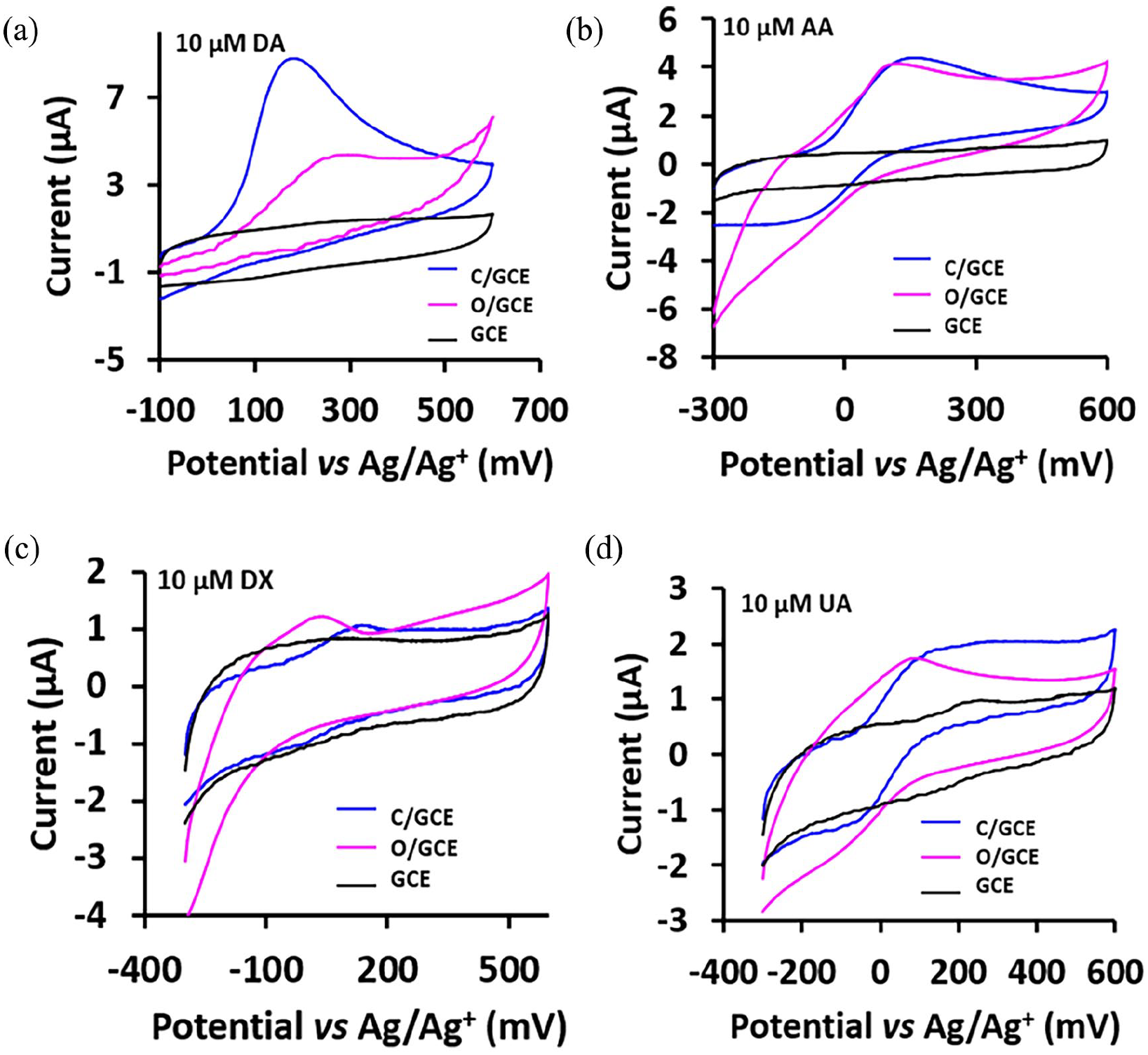
The CV curves of the response of (a) 10 μM DA, (b) 10 μM AA, (c) 10 μM DX and (d) 10 μM UA, over C/GC and O/GC in 0.1 M PBS solution at a scan rate of 50 mVs−1.
The binding energy as measured by XPS for all the simulated peaks for the Cu 2p3/2 as well as the Au 3f7/2 photoelectron lines, all measured in eV. The ratio between the Cu0, CuI and CuII as well as the atomic ratio between the Cu and Au are also reported.

From the atomic ratio between the Cu and Au (which is an indication of the amount of Au deposited on the Cu-oxide nanocrystal), presented in Table 1, it is clear that more Au deposited on
The cyclic voltammetry (CV) curves of the modified electrodes
Cyclic voltametric studies were conducted to investigate the electrochemical response of DA, UA, AA and dextrose (DX) on both the
The AA oxidation was observed at 162 and 105 mV over
The detection of AA and UA over
Differential pulse voltammetry (DPV) was used to obtain better sensitivity, since its results in enhanced signals by removing the non-Faradaic current.
To investigate the sensitivity and limit of detection (LOD) of the
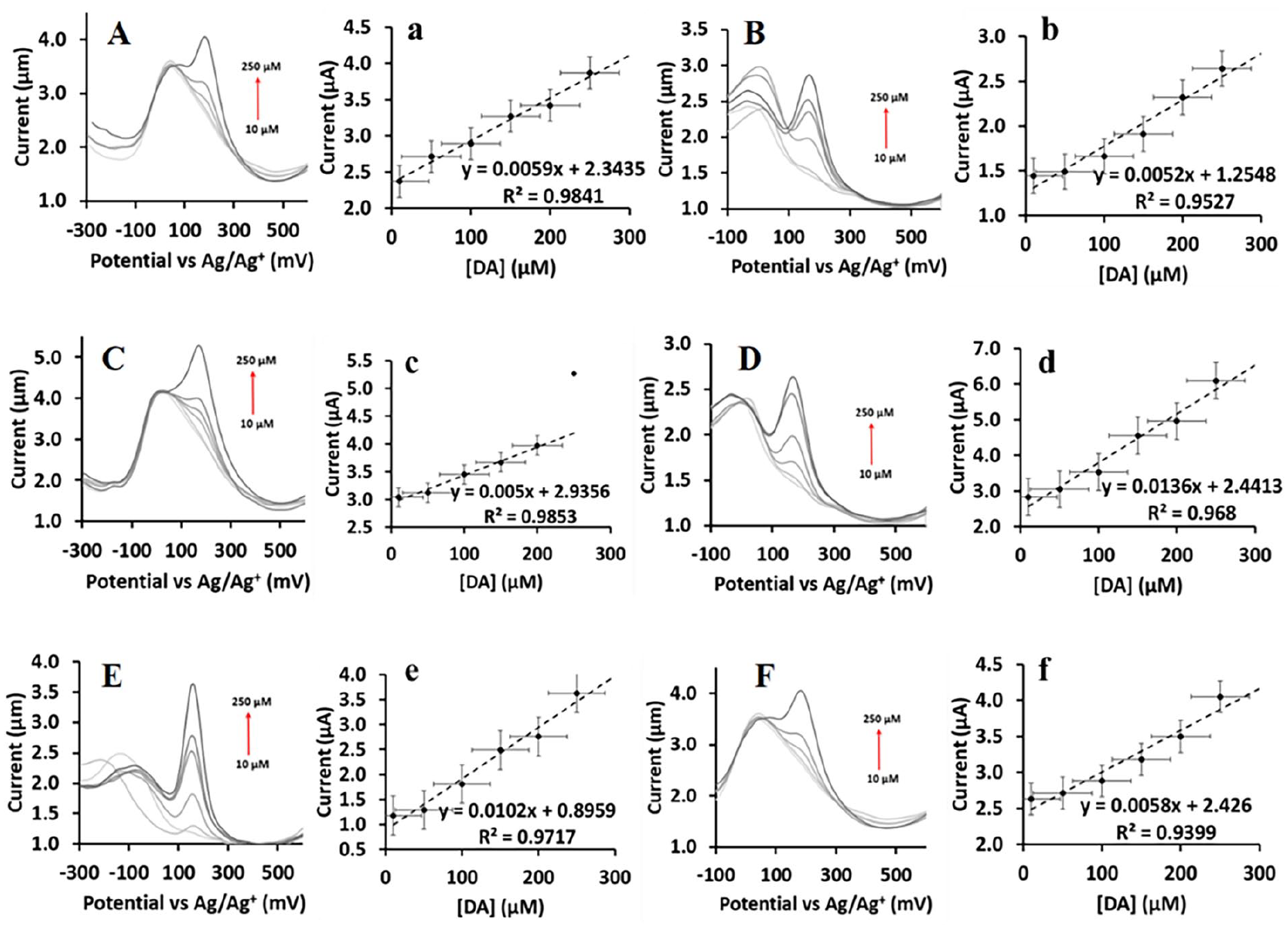
The DPV curves showing the current response of increasing concentration of DA in (A) a 10 μM UA solution over C, (B) a 10 μM UA solution over O, (C) a 10 μM AA solution over C, (D) a 10 μM AA solution over O, (E) a 10 μM DX solution over C, (F) a 10 μM DX solution over O, with the corresponding calibration curves of DA in (a) a 10 μM UA solution over C, (b) a 10 μM UA solution over O, (c) a 10 μM AA solution over C, (d) a 10 μM AA solution over O, (e) a 10 μM DX solution over C and (f) a 10 μM DX solution over O.
The oxidation potential of DA, AA, DX and UA was detected at a lower potential over
The DPV peak separation (see Table 2) between the DA and the other biomolecules, such as UA, AA and DX, is sufficient large to selectively detect DA without interference of the other biomolecules. A linear directly proportional relationship was established between the peak current for the DA and its concentration.
The DPV peak separation, limit of detection, linear range and the sensitivity of C/GC and O/GC. Also presenting comparative data with already reported results.

C/GC:
This paper.
Could be higher.
The LOD was calculated by equation (1): 31

where s represents the standard deviation of the blank voltammogram and m is the slope of the calibration curve. The LOD (measured in μM) of
Sensing of DA over
The effectivity of
Conclusion
In summary, cubic- and octahedron-shaped Cu-oxide nanocrystals were used as well-defined morphological templates for the galvanic etching and deposition of porous Au layers. The SEM images indicated that the surfactant-free hexadecane-in-water nanodroplet adsorption as well as formation of the Au layer occurred faster on the (111) facet of the octahedron morphology. This was affirmed by the % Au present in the samples at different times. The samples contained metallic Au and mixed oxidation states of Cu (0, I and II) were detected by XPS. GCEs modified with the Au/Cu-oxide nanocomposites were tested for their DA detection capability in the presence of interfering molecules (UA, AA and DX). The GC electrode modified with the porous Au layered onto the octahedron shaped Cu-oxide crystals resulted in superior DA detection.
Experimental section
All reagents, both solid and liquid, were reagent grade, purchased from Sigma-Aldrich and used without further purification. Distilled water was used throughout. The cubic and octahedron Cu-oxide nanocrystals were prepared according to published procedures.34,58
Preparation of porous Au layer on the Cu-oxide nanocrystals
The preparation is adapted from the original procedure of Ma et al., 35 for nanoparticles in suspension.
Preparation of Hexadecane-in-H2O emulsion via a solvent-shifting method
Hexadecane (1 ml) was added to acetone (9 ml) resulting in a volume fraction of 0.1%. The above solution was then injected into a 200 ml water to create an emulsion.
Preparation of porous Au nanoparticles
The appropriate Cu-oxide nanocrystal (~20 mg) was dispersed into 20 ml of the hexadecane-in-H2O emulsion for 20 min, allowing time for nanodroplet adsorption onto the nanocrystals. This was followed by dropwise addition of HAuCl4 (2 ml, 2.9 mM) to the mixture and subsequent incubation for 6, 12 and 24 h, respectively. The samples were centrifuged after the addition of ethanol to prevent the freezing of hexadecane while centrifuging. The collected nanoparticles were dried overnight at 60°C.
Characterization
The crystalline phase of the samples was characterized by the X-ray diffractometer (Bruker D8-ADVANCE) with Cu–Kα (λ = 1.54060 Å) operated in a continuous θ–θ scan in locked coupled mode with Cu–K radiation. Measurements ran within a range in 2θ defined by the user with a typical step size of 0.034° in 2θ. A position sensitive detector, Lyn-Eye, was used to record diffraction data at a typical speed of 0.5 s/step, which is equivalent to an effective time of 92 s/step for a scintillation counter.
For SEM, a JEOL-JSM7800 Field Emission Scanning Electron Microscope was employed. The electron beam energies in the range of 5 keV.
XPS data were recorded on a PHI 5000 Versaprobe system with a monochromatic AlK X-ray source. Spectra were obtained using the aluminium anode (Al Kα = 1486.6 eV) operating at 50 μm, 12.5 W and 15 kV energy (97 X-ray beam). The survey scans were recorded at constant pass energy of 187.85 eV and region scans at constant pass energy of 29.35 eV with analyser resolution ⩽ 0.5 eV. The background pressure was approximately 2 × 10−8 mbar. The XPS data sets were analysed utilizing Multipak version 8.2c computer software with Gaussian–Lorentz fits.
DA detection
DA detection was investigated by means of electrochemical measurements. CV and DPV were conducted on a BAS 100 B/W electrochemical workstation. A three-electrode cell was used, utilizing a Pt auxiliary electrode, an Ag/Ag+ reference electrode and a modified glassy carbon working electrode with a surface area of 3.14 mm2. The pre-electrocatalyst suspension is prepared by adding the Au/Cu-oxide nanoparticle (20 mg) in MeOH (methanol) (0.5 ml), followed by sonication. The pre-electrocatalyst suspension (20 µl) is drop-coated onto the electrode surface. After drying, Nafion (20 µl) was dropped over the modified surface and allowed to air-dry. CV was conducted in a solution of 10 µM DA and 0.1 M Dulbecco’s PBS. The potential was scanned from −300 to 600 mV using a scan rate of 50 mV s−1. DPV was used investigate the selectivity towards DA detection in the presence of interfering biomolecules. The DPV was again scanned from −300 to 600 mV, with a pulse width and amplitude of 0.05 s and 50 mV, the pulse period was 200 s. The solution for the DPV was a mixture of 0.1 M PBS and 10 µM of either UA,
Supplemental Material
sj-docx-1-chl-10.1177_17475198241247964 – Supplemental material for Porous gold-layered cubic and octahedral Cu-oxide nanocrystals: Dopamine sensing
Supplemental material, sj-docx-1-chl-10.1177_17475198241247964 for Porous gold-layered cubic and octahedral Cu-oxide nanocrystals: Dopamine sensing by Carla Jacobs and Elizabeth Erasmus in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The Sasol is acknowledged for financial support. The Central Research Fund of the University of the Free State, Bloemfontein, South Africa is also acknowledged for financial support.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
