Abstract
Lipase from
1. Introduction
Biotransformations are already being used industrially to manufacture a wide range of products, including drugs, agricultural chemicals, organics, fine chemicals and plastics. Pharmaceutical companies, as well as chemical and food industries using biocatalysts, are developing, due to the increasing demand for new, efficient and low production costs [1-4].
Lipases (E.C. 3.1.1.3.) are remarkable industrial biocatalysts due to their specificity, thermal stability and tolerance to organic solvents. Lipases are enzymes that catalyse the hydrolysis of triacylglycerols to glycerol and free fatty acids, but they can also perform ester transesterification as well as ester synthesis [5, 6]. Native lipases have limited stability, so immobilization is essential for their usage in an industrial setup. Immobilized lipases show a variety of properties, such as increased catalytic activity, temperature and solvent tolerance [7, 8], reusability and ease of the downstream process [9-12].
The success of an immobilization technique depends on various parameters. Stability is economically important, since a stable enzyme can be used more times. It is also very important to maintain high catalytic activity. A general practice is to compare the activity of the immobilized enzyme with the “free” enzyme [7].
Sol-gel encapsulation is an easy and effective process for enzyme immobilization. The sol-gel approach involves fabrication of inorganic matrices through the formation of a sol, which is a colloidal suspension of solid species in a liquid, followed by the gelation of the sol to yield solid structures (wet gel) with spontaneous drying to remove the organic solvents and leave solid material. The sol-gel methods were particularly effective in the case of lipases. Most of the reported protocols used mixtures of low molecular weight tetraalkoxysilanes (tetramethoxysilane, tetraethoxysilane) or alkyltrimethoxysilanes, based on the recognized stimulation of the interfacial activation of the entrapped lipase by the hydrophobic alkyl groups present in the matrix [13-15].
Lipases immobilized in sol-gel matrices exhibited improved properties such as enhanced activity, enantioselectivity and operational stability, increased conformational rigidity and, in several cases, higher activity compared with other immobilization procedures [7, 8, 13, 16, 17]. Sol–gel immobilized lipases can be supported on inert materials to improve the diffusion of substrates and products to and from the enzyme and thus improve the reaction rate [17-20]. Studies have also shown that sol-gel entrapped lipase adsorbed on Celite 545 exhibited higher activity and thermostability compared with lipase deposited directly on Celite 545, without using the sol–gel method [18, 20].
The recent development in understanding enzyme catalysed processes at molecular level, due to the latest advances in molecular genetics, structural biology and bioinformatics [1], allowed a much closer identification of the complex interactions and influences between the immobilization support and the enzyme molecule [21].
Although several methods can be employed to determine structural characteristics that are associated with the catalytic properties of the immobilized enzymes, the knowledge about the structure of the enzyme protein following the immobilization and the nanoscale level interactions with the surface of the support material is still insufficient. Doubtlessly, an increasing amount of qualitative and quantitative information concerning these interactions, including the morphological visualization of the enzyme following the immobilization, will be essential to design robust and stable solid-state biocatalysts for industrial applications [11, 12].
The aim of the present work was to immobilize a microbial lipase from
2. Materials and Methods
2.1 Materials
Amano AK lipase (from
2.2 General Procedure for Immobilization by Sol-Gel Entrapment
A microbial lipase (lipase from
2.3 General Procedure for Immobilization by Sol-Gel Entrapment and Adsorption
Until the start of gelation, the immobilization protocol was identical to that described for the sol-gel entrapment in the previous section. Next, 0.5 g Celite 545 was blended with the gelling mixture. Subsequently, the obtained solid preparate (SGE-A) was processed as described above.
2.4 Acylation of Secondary Alcohols
Acylations were performed in 4 mL capacity glass vials, charged with a racemic
The mixture was incubated using an orbital shaker (MIR-S100, Sanyo, Japan) at 300 strokes/min and 40 °C (ILW 115 STD incubator, Pol-Eko-Aparatura, Poland). The conversion and enantiomeric excess of the product were assayed by gas-chromatography, on a Varian 450 instrument (Varian Inc., the Netherlands) equipped with flame ionization detector, using a 30 m × 0.25 mm Astec Chiraldex B-PM chiral column with 0.12 μm film thickness (Sigma Aldrich, Supelco). The analysis conditions were: oven temperature from 100 °C to 140 °C with 10 °C/min heating rate, injector temperature 240 °C, detector temperature 280 °C, carrier gas (hydrogen) flow 1.9 mL/min. The reactions were usually run for 24 h. Conversions have been calculated based on the internal standard method.
Transesterification activities were calculated at 24 h reaction time and expressed as the average 2-acetoxyalcohol amount (in micromole) synthesized per hour by 1 mg of free or immobilized enzyme. These are apparent activities used to characterize the catalytic efficiencies of the native and immobilized lipases and do not have any kinetic significance associated with the initial reaction rate. The control reaction without enzyme did not give any product in the same conditions. The enantiomeric excess of the resulting ester product (eeP) was determined from peak areas of enantiomers and the enantiomeric ratio (

All reactions were run in duplicate and sampling was also made in duplicate. As the differences between the data for the same assay were less than 2%, average values have been calculated and presented in the tables and figures.
2.5 Reuse of Immobilized Lipase in a Batch Reaction
The enzyme reuse study was performed at 40 °C. The initial reaction system was set up as described for the acylation study. At the end of every reaction cycle, the product (upper liquid phase of the reaction mixture) was removed with a pipette, the remaining solid phase (native or immobilized lipase) was washed twice with hexane (2 mL), centrifuged at 15 °C and 2,767 × g (RCF) and the supernatant decanted. Subsequently, the same amounts of reagents (2-hexanol, vinyl acetate and hexane) as for the initial reaction were added to the reused enzyme and the reaction was run under the same conditions.
2.6 Temperature Stability
The native and immobilized lipases were incubated in n-hexane at different temperatures (40, 50, 60 and 70 °C) for 24 h. After this, the solvent was removed by filtration and the remaining solid phase (native or immobilized lipase) was washed twice with hexane (2 mL), centrifuged at 15 °C and 2,767 × g (RCF) and the supernatant decanted. The activity of biocatalysts was determined in the acylation reaction of 2-hexanol, 2-heptanol or 2-octanol with vinyl acetate, as described above.
2.7 FTIR Spectroscopy
Fourier Transform Infrared (FTIR) spectra of the samples were obtained in attenuated total reflectance (ATR) mode on a Bruker Vertex 70 (Bruker Daltonik GmbH, Germany) spectrometer equipped with a Platinum ATR, Bruker Diamond Type A225/Q. Spectra were collected in the range 4,000–400 cm−1 with a resolution of 4 cm−1 and with 60 scans/min.
2.8 Specific Surface Area and Porosity
Nitrogen adsorption/desorption isotherms of the sol-gel entrapped preparates were measured at 77 K by using the BET apparatus NOVA 1200-Quantachrome. The samples were previously degassed at 100 °C for 3 h and the analysis was performed at 77 K, using liquid nitrogen. The specific surface area (SBET) was calculated from the nitrogen adsorption isotherms by using the Brunauer-Emmet-Teller (BET) method [23]. The total volume of the pores per gram of solid material (Vtotal) was calculated using the Barrett-Joyner-Halenda (BJH) model [24]. The same BJH method allowed the calculation of pore size mean diameters (Dp) from the adsorption/desorption isotherms, as well as of pore size distribution.
2.9 Laser Scanning Confocal Microscopy (LSCM)
Laser scanning confocal microscopy was performed with a Leika True Confocal Scanner (Leika TCS SPE, Czech Republic) to investigate the distribution of lipase within the sol-gel matrix nanostructures. For this, the lipase was dyed with fluorescein isothiocyanate (FITC) (as described in PIERCE EZ-LabelTM FITC Labeling Kit). The obtained solution (containing the enzyme marked with FITC) was purified on a column and then lyophilized at −55 °C for 34 h. The obtained powder was suspended in TRIS buffer and used for immobilization.
2.10. Scanning Electron Microscopy (SEM)
Scanning electron microscopy was performed with an Inspect S +EDAX Genesis XM 2i system (FEI Company, the Netherlands). The analysis parameters were: pressure 1.5 × 10−2 Pa, resolution <10 nm at 3 kV.
2.11. Atomic Force Microscopy (AFM)
The topography of the sol-gel preparates was performed by the Nanosurf® EasyScan 2 Advanced Research AFM. The samples were crushed in a mortar with n-hexane and then placed onto a silica glass plate and dried at room temperature. The two-dimensional (2D) and three-dimensional (3D) images were recorded in contact mode cantilever, with a scanned surface of 1 μm × 1 μm. AFM data are quantitative on all three dimensions, the method for displaying them being by colour mapping; in a grey scale, dark and light tones represent the low and high features, respectively. The roughness parameters comprised average surface roughness (Sa, nm), maximum peak height (Sp, nm) and maximum valley depth (Sv, nm), calculated using the Nanosurf EasyScan 2 computer software.
2.12. Thermal Analysis (TGA/DTA)
Thermogravimetric measurements (TGA/DTA) were recorded using a TG 209 F1 Libra thermogravimetric analyser (Netzsch, Germany) operating a resolution of 0.1 μg, in nitrogen atmosphere. Curves were recorded from 30 to 1,000 °C with a heating rate of 10 °C min−1. The average sample mass was 7.0 ± 0.15 mg in open alumina crucibles (average mass 190 ±1.0 mg).
3. Results and Discussion
The characterization of biocatalysts is of great importance to explain and predict some of their main properties, such as activity, selectivity and stability. Although characterization supplies different types of information, the properties of the biocatalyst can be correlated with its surface area, texture properties, pore volume, etc. [25]
In order to obtain preparates with high catalytic efficiency, the composition of the silane precursors system must be carefully tuned for the final application. Lipase from
As shown in Table 1, the use of a ternary silane mixture was beneficial to the catalytic activity and enantiomeric excess values, in comparison with the use of TMOS as matrix template (the SGE-1 preparate). The phenyl group content in the matrix was gradually increased and the highest transesterification activity value was obtained at 1.6:0.4:1 PhTMOS:VTMOS:TMOS molar ratio (SGE-2 preparate), for all tested substrates.
Influence of the silane precursors system on the catalytic properties of lipase Amano AK immobilized by sol-gel entrapment in the acylation of sec-alcohols

The activity and enantioselectivity values of the biocatalyst obtained with the combined method (SGE-A preparate) were comparable to those of the sol-gel preparate obtained without adsorption at the same 1.6:0.4:1 molar ratio, except for the 2-hexanol substrate, where a higher activity value was noticed.
For both immobilization techniques, a lower concentration of vinyl groups in the matrix led to higher activity values, probably due to different structural characteristics and hydrophobicity of the gel network. The enantiomeric excess values were high compared with the native enzyme, except for the same SGE-1 preparate, which was also the less active.
The activity decreased after immobilization, being up to 20% of the native enzyme activity. This is a normal result, given that the enzyme was dispersed in a larger amount of silica matrix, particularly when the combined method was used. In fact, the immobilized biocatalyst contained less than 5% enzyme, as will be shown by the thermal decomposition data. In these circumstances, the overall catalytic efficiency of the biocatalyst increased next to the immobilization process, making it economically feasible.
3.1 Structural Characterization by FTIR Spectroscopy
FTIR spectroscopy is one of the more popular methods to characterize the microstructure of the sol-gel materials. FTIR data confirmed the presence of the alkyl or aryl functional groups of the precursors in the xerogel, as shown in Figure 1, but were not able to locate the enzyme, as the protein amide group band was wrapped by other vibrations of the silica matrix structure.

FTIR spectra of the immobilized preparates of
For all tested preparates the characteristic bands of silica compounds are present: 1,055, 1,041, 1,028 and 1,037 cm−1 assigned to Si–O–Si asymmetric stretching (1,090–1,030 cm−1); 786, 777 and 767 cm−1 specific to the presence of Si-CH3 bonds (860–760 cm−1). The intense Si-OH bands between 3,700–3,200 cm−1 are missing, demonstrating that almost all OH groups were involved in the network of the sol-gel matrix, confirming the relationship between shrinkage and condensation reactions of different precursor types. The absence of the residual Si-OH groups explains the matrix hydrophobicity and the availability of these biocatalysts to work in non-polar organic solvents.
The stretching bands of methylene groups in the 2,800–2,900 cm−1 region can be assigned to the alkyl groups of the ionic liquid used as additive. We previously demonstrated that this additive is not completely removed in the washing step, remaining embedded in the structure of the silica matrix [7].
The phenyl groups in the immobilized preparates, as shown in Figure 1(b) and Figure 1(c), are revealed by the presence of the specific bands of the CH group from the benzene ring and the aromatic skeletal at 734–736 cm−1 and 696–698 cm−1, respectively. These bands are particularly well observed in the spectrum of the preparate with high phenyl group concentration (SGE-2). The specific bands at 1,409 cm−1 and 968 cm−1 can be assigned to
3.2 Specific Surface Area and Porosity
Investigation of the porosity of the obtained sol-gel materials was carried out by the BET method, based on nitrogen adsorption. The specific surface area (SSA), the mean pore diameter (from the adsorption branch of the isotherms), as well as the total volume of pores were calculated. The samples were degassed at 100°C for 3 h, presuming that only the humidity is removed, while the enzyme and the pending organic groups remain unchanged. The results presented in Table 2 show that only two of the investigated preparates presented high SSA values: the xerogel obtained solely from TMOS and the preparate from the ternary silane mixture at high vinyl group concentration. As discussed before, the activity data (Table 1) indicate low values for both of these immobilized enzymes. High SSA is associated with lower pore diameter in the case of SGE-1, resulting in possible diffusional limitations and decrease in the measured (apparent) activity of the enzyme.
Specific surface and porosity characteristics of the sol-gel materials obtained by immobilization of
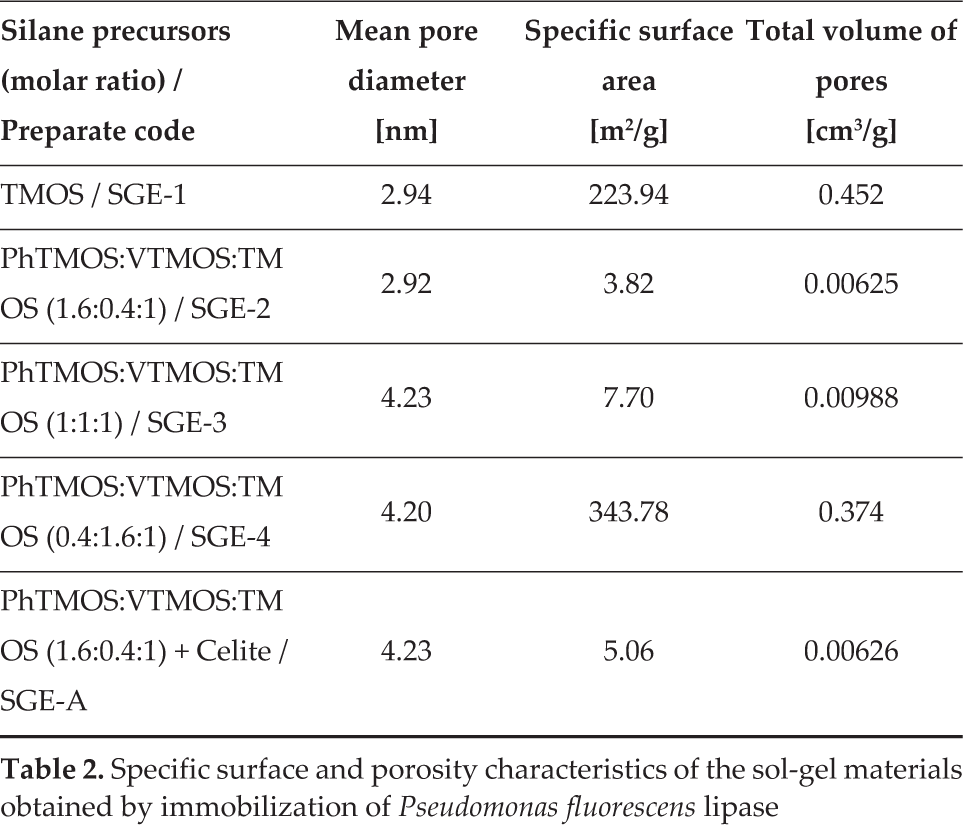
The specific surface area decreased with the increase in the concentration of phenyl groups present in the sol-gel matrix and the highest activity was observed for the xerogel with the lowest SSA and highest phenyl group content. Noureddini and Gao also observed that the SSA values are mostly a function of the silane precursors and the activity decreased with the increase in SSA of the sol-gel immobilized lipase [11].
Although increased porosity of the matrix is commonly associated with higher activity of the immobilized enzyme, structural characteristics most probably play the main role. Our previous results demonstrated that higher content of hydrophobic groups in the silica matrix was propitious for the catalytic efficiency of microbial lipases [26] and such a conclusion is in accordance with the findings of other groups [16, 20]. The hydrophobic phenyl and vinyl groups of the silane precursors that are retrieved in the xerogel are beneficial for the catalytic activity of lipases, particularly in the case of conversion of hydrophobic substrates in non-polar solvents. However, different content of these functional groups obviously leads to different structures of the matrix that can also influence the process. Incorporation of the bulkier phenyl group at a higher content resulted in a xerogel with much lower SSA, but higher activity. PhTMOS may also act as a bioimprinting agent stabilizing the open conformation of the lipase [8], while such an effect might be less prominent in the case of organosilane precursors with smaller size. The main effect of the porous structure is related to the internal diffusion of the substrate to the active site of the encapsulated enzyme and of the product back in the reaction medium, and narrow pores can induce mass transfer limitations in both directions. For this reason, a mesoporous or macroporous structure of the catalyst is preferred, although intuitively a highly porous structure may be considered superior in order to accommodate a higher amount of loaded enzyme and to allow better distribution of the biocatalyst inside the xerogel network. The adsorption-desorption isotherms of the tested preparates were of type IV, according to the IUPAC classification, exhibiting a hysteresis loop [27]. Two of them are presented in Figure 2. Both isotherms show a mainly mesoporous structure, since they presented a steeper section starting from P/P0 values about 4.0 and the gas refilling was almost continuous. The xerogel from TMOS precursor exhibits a H2 hysteresis loop characteristic of materials having so-called “ink-bottle” pores with narrow necks and wide bodies, Figure 2(b), while in the case of the gel from ternary silane precursors (PhTMOS:VTMOS:TMOS at 1.6:0.4:1 molar ratio), Figure 2(a), the hysteresis loop can be considered of H3 type, with aggregates of slit-shaped pores [28].

Adsorption-desorption isotherms of the biocatalysts obtained with
The pore distribution, determined by the BJH, showed a mean distribution of pore diameter in the range 2–50 nm (defined as mesopores according to the IUPAC classification), with values centred around 3–4 nm, as shown by the mean pore diameter values (Table 2).
There is no difference between the calculated pore diameters of the preparates exhibiting high activity and low activity, respectively. However, since in the case of H2 hysteresis the neck size distribution is much narrower than the size distribution of the main cavities, pore blocking phenomena can play an important role [28] and can also explain the low activity of the preparate obtained with TMOS precursor. Even if the enzyme molecules are confined inside the xerogel, the access of the substrate molecules is hindered.
The activity, SSA and pore diameter of the preparate obtained by the combined immobilization technique, sol-gel entrapment and adsorption were very close to the values recorded for the preparate from the same precursors but without adsorption on Celite. Hence, the structural characteristics of the sol-gel matrix are not influenced by the porous structure of this adsorbent, which has no interference in the sol-gel entrapment of lipase, the formed gel being only deposited on this surface.
3.3 Laser Scanning Confocal Microscopy (LSCM)
Confocal microscopy imaging can provide important information concerning the distribution of the enzyme inside the sol-gel preparate. The distribution of the immobilized enzyme within the sol-gel matrix is an important parameter because it relates to the accessibility of the enzyme to the substrate. The aggregation of the enzyme molecules during the gelation process cannot be excluded, since the homogeneity of the mixture cannot be ensured and the gelation time is long enough. The presence of various additives during the immobilization process has the role of preventing such an aggregation, in addition to protecting the enzyme against denaturation by a variety of other molecular species that are present.
Given that the studied lipase from
It must be highlighted that only the compound formed by lipase with FITC presented fluorescence, while the non-labelled native and sol-gel immobilized lipase were not fluorescent.
The fluorescent images of the FITC-labelled SGE-2 and SGE-A were recorded in scanning mode, for a better view of the surface morphology, as shown in Figure 3(a) and Figure 3(b). Figure 3(a) presents the fluorescence image for the SGE-2 sol-gel entrapped Amano AK lipase (ternary mixture of PhTMOS:VTMOS:TMOS precursors at 1.6:0.4:1 molar ratio), while Figure 3(b) shows the image of the biocatalyst obtained by the combined method with the same precursors and Celite 545 as solid support. In the scanning mode, the laser beam scans allow three-dimensional images of the studied particle in a 2–5 μm depth to be obtained. The surface profile of the active immobilized enzyme presents the image of particles having the enzyme distributed on both the surface and in the inner part of the xerogel. In both cases the labelled lipase is distributed in the whole matrix, suggesting a uniform layout of the enzyme in the immobilized preparates.

Fluorescent images (10x magnification) of the sol-gel immobilized Amano AK lipase preparations: (a) FITC-labelled SGE-2 scanning, (b) FITC-labelled SGE-A scanning and (c) FITC-labelled SGE-1
At the same time, the SGE-1 sol-gel matrix synthesized only with TMOS, Figure 3(c), shows a different fluorescence image, with compact blocks that hold the enzyme tightly, a structure that can be correlated with the low transesterification activity of this biocatalyst.
Although the distribution of the enzyme inside the sol-gel matrix did not appear different, the morphology of the material had a strong influence on the catalytic efficiency. Therefore, it was necessary to investigate other characterization methods as well, in order to have a more complete image of this influence.
3.4 Scanning Electron Microscopy (SEM)
The SEM studies were carried out to examine the effect of different silane precursors and immobilization procedures on the morphology of the sol-gel material with entrapped lipase. This analysis reveals important characteristics of the morphology of the immobilized biocatalysts and adsorbent, information that could be correlated with the catalytic efficiency in transesterification reactions. The SEM images are presented in Figure 4, at two different magnifications each, 1,500× and 3,000×, respectively.

SEM micrographs at different magnifications of the sol-gel entrapped lipase from
The SEM images, Figure 4(a) and Figure 4(b), of the SGE-2 preparate with higher phenyl group content (1.6:0.4:1 molar ratio of the silane precursors), show uniform distribution of 1–2 μm microspheres resulting in about 1 μm diameter micropores, but the nanopores are not visible. Such micron-sized particles with nanopores of 3–4 nm (Table 2) indicate a path length of about 500–100 nm for the substrates and products, which is correlated with the best values of the transesterification activity for all tested substrates. In this case, the substrate can easily reach the active site of the lipase, without significant hindrance from the sol-gel matrix.
In the case of the SGE-3 enzymatic preparate obtained at equimolar ratio between the silane precursors, the porosity is almost the same as that of the SGE-2 preparate with higher phenyl and lower vinyl group content, but a more compact structure can be observed (Figure 4(c) and 4(d)), which is correlated with a slight decrease in activity.
Further increase in the vinyl group content in the matrix resulted in significantly lower values of transesterification activity and enantiomeric excess for all tested substrates, as presented in Table 1. The SEM images of the SGE-4 preparate, Figure 4(e) and Figure 4(f), show a more porous structure, with smaller pores, which is consistent with the higher specific surface area of this material. The microporosity of this matrix probably induced a more important mass transfer limitation. As shown in Table 2, the mean pore diameter of the SGE-4 preparate is slightly higher compared with the xerogels SGE-2 and SGE-3, which presented good activity and enantioselectivity. Consequently, the SEM images are in accordance with the specific surface measurements but cannot explain the apparent inconsistency of the activity with the calculated pore diameters.
Among the sol-gel entrapped biocatalysts, the smallest values of transesterification activity (in the range 0.022–0.036 μmole mg−1 h−1) were obtained using only TMOS as precursor for the sol-gel matrix. The micrographs of this preparate (SGE-1), presented in Figure 4(g), are totally different, as the morphology shows non-microstructured large particles of 200–300 μm size, which indicate the presence of more than 200 times longer nanochannels compared with the best preparate (SGE-2). The presence of too-tight nanopores and the major increase in the path length for substrates and products explain the low apparent activity. Therefore, it is essential to use alkyl-silane precursors in addition to the tetra-alkoxy silane in order to obtain a sol-gel material with microporosity suitable for the appropriate mass transfer of reagents and products.
The morphology of the SGE-A biocatalyst obtained by sol-gel entrapment followed by adsorption on a solid support (Celite 545) was specific, as presented in Figure 4(h). It can be easily observed that xerogel particles are formed both on the solid support surface and inside the pores. The development of the sol-gel matrix inside the pores of Celite may be an explanation for the slight decrease in catalytic efficiency [18]. It is possible that a higher amount of lipase was entrapped inside the pores of the adsorbent and the diffusivity of the substrate is also slower in this case.
Once again, it is important to emphasize that the silane precursors, additives and adsorbent must be carefully selected for every application.
3.5 Atomic Force Microscopy (AFM)
The AFM images were recorded to complement the SEM analysis and to investigate the topography of the surfaces, since SEM does not deliver information regarding the conformation of the internal porous structure. AFM provides three-dimensional images of the sample surface and is helpful for the characterization, at nanometric scale, of the surface inhomogeneity and roughness, as well as of the morphology of the porous structure.
The particle size analysis for the studied preparates was performed from the AFM measurements data. The number of nanounits and the mean volume of nanounits were estimated from several images of each sample.
Table 3 summarizes the AFM measurement data. For the sol-gel entrapped biocatalysts, the number of nanounits on the scanned surface of 1 μm2 was between 176 and 680 and the mean volume of nanounits varied from 491 to 37 nm3.
Particle analysis data of the sol-gel immobilized lipases (number of nanounits on 1 μm2 surface, mean volume and surface of nanounits)

Thermal behavior of the sol-gel entrapped biocatalysts

Although it is difficult to directly correlate the activities and enantioselectivities of the studied samples with the AFM data, some valuable information can be revealed. The number of nanounits was the lowest for the SGE-1 preparate obtained with TMOS precursor only and the highest for the SGE-2 preparate that exhibited the best activity. The diameter of nanopores must be smaller than the protein size to prevent leakage, but large enough to overcome the inherent diffusional limitation for small molecules. The number of nanounits of the SGE-A biocatalyst, obtained by the double immobilization method, was lower compared with the same preparate obtained without deposition on Celite. Modification of the relative amount of the silane precursors holding phenyl and vinyl groups led to a different porous nanostructure, but it is not correlated with the activity values. The hydrophobic interactions taking place inside the porous structure between the alkyl or aryl groups and specific components of the secondary structure of lipase called “lid”, which probably play the decisive role in the activity, and the morphology of the porous structure cannot be directly associated with these interactions.
As seen in Figure 5, all studied materials contained nanostructures with well-defined nanounits, uniform in size. The size of the mesopores varied from 2 to 4 nm for all the tested preparates, in accordance with the results of the adsorption isotherms. The surface roughness (Sa) of the sol-gel preparates was between 0.12 and 0.01 nm; the valley depth (Sv) had values of min −1 nm and the max peak height (Sp) reached 2.8 nm for the SGE-A and only 1.1 nm for the SGE preparates. The roughness depends on the nature of the silica precursor, but the surface was uniform and smooth for all preparates, without large aggregates.

3D AFM images of sol-gel entrapped lipase from
In all tested samples the nanoparticles were regularly distributed, hence the AFM images reiterate the SEM and fluorescence microscopy results on the morphology of the structures.
3.6 Thermal Analysis (TGA/DTA)
Thermogravimetric analysis (TGA) provides important information about the temperature range at which a heated sample undergoes a major conformational change, by monitoring the thermal weight loss profile. The weight loss curves of the sol-gel immobilized preparates were divided into three regions, based on the main phenomena that can change the rate of this process.
Region I was associated with the loss of water and some volatile organic molecules, up to a temperature of 280 °C.
Region II, in the range of 280–440 °C, is associated with loss of organic constituents (C, H, O and N) in the form of volatiles either present or formed by the beginning of organic decomposition, including the lipase.
In Region III, the weight loss is associated with final dehydroxylation reactions and carbonization of organic compounds [25]. Above 800 °C, either the thermal stability of the material is reached or it completely breaks down.
In the first interval, the weight loss of 1.7 – 4.3% was due to evaporation of water and possibly small amounts of 2-propanol or other volatiles that remained in the preparates at the end of the drying stage. As expected, the water content of the native lipase was higher (19% weight loss), although the commercial enzyme is a solid material. It was also not surprising that the preparates containing entrapped lipase showed a weight loss approximately 1% greater than that of the references without lipase, since the lipase was introduced as aqueous solution in the immobilization mixture and remained in a hydrated state at the end of the process. This small water amount is linked by hydrogen bonds to the hydrophilic amino acid residues of the enzyme and is essential to maintain the active catalytic conformation, even in nearly anhydrous reaction media [29].
The second and third regions are the most important for the thermal analysis profile of the immobilized preparates. As seen in Figure 6, the thermal decomposition of the organic part of the native lipase preparate is completed until 440 °C, indicating a weight loss of 25.7%. We also measured the protein content by the Bradford method, obtaining 19% (w/w). The 6.7% difference could represent other organic compounds added as adjuvant to the lipase, which also undergo thermal decomposition up to 440 °C. Consequently, the difference of 1.2% between the weight losses of the immobilized preparates (SGE-2 and SGE-A, respectively) and the equivalent reference materials in the 280–440 °C interval can be assigned to the entrapped protein. The DTG curves show that the value of the highest thermal decomposition rate was shifted to more elevated temperatures (for the native lipase the peak maximum was registered at 290 °C, for the immobilized preparates at 350 °C), indicating a better protection against increasing temperatures following immobilization. This finding was later confirmed by the preservation of the lipase activity at higher temperatures, as will be demonstrated by the thermal stability study.

Thermograms showing the weight loss (TG, continuous lines) and derivative weight loss (DTG, dotted lines) for: (a)
The organic functional groups covalently linked in the matrix structure were decomposed almost completely until 750 °C, but according to the thermograms the weight loss rate had two peaks. The first peak, at the same temperature of 350 °C as for the decomposition of the enzyme protein, was smaller (6% weight loss for the sol-gel reference material and 2.2% for the sol-gel adsorbed on Celite) and can be assigned to the loss of the organic functional groups located at the surface of the particles. The second peak at 570 °C was more intense (22% and 11% weight loss, respectively) and indicates the thermal decomposition of the organic groups from the inner pores of the hybrid sol-gel matrix.
As expected, when the combined immobilization method was employed, the total weight loss was significantly lower (around 20%) compared with the sol-gel entrapped lipase (around 37%), this difference being attributed to the inorganic Celite material that undergoes only a small amount of thermal degradation, even at temperatures higher than 800 °C.
The weight loss curve of the native lipase was completely different compared with that of the immobilized biocatalysts and the biggest surprise was the important amount of weight loss around 900 °C (as shown by the peak of the DTG curve), as well as the residual mass of 11%. The explanation may be the presence of an important amount of inorganic conditioning additive, which undergoes thermal decomposition at elevated temperature, leaving a residue of (probably) oxide.
3.7 Operational and Thermal Stability of the Sol-Gel Immobilized Lipase
One of the main objectives of immobilization is the stability in operational conditions shown by the reusability of enzymatic preparates, which leads to reduced production costs. An enzyme can usually be reused until its activity decreases to less than 25% of the initial value. This study was carried out to demonstrate that the most efficient biocatalyst, as resulted from the previous activity and characterization experiments, is robust and stable in operational conditions as well as higher temperatures.
Therefore, the stability of Amano AK lipase, immobilized by sol-gel entrapment and by the combined method using the same precursors (PhTMOS:VTMOS:TMOS at 1.6:0.4:1 molar ratio), was investigated in the acylation reaction of 2-hexanol with vinyl acetate in n-hexane at 40 °C. The results are presented in Figure 7.

Influence of enzyme reuse on the relative activity of native and immobilized lipase from
The immobilized biocatalysts exhibited higher operational stability compared with the native lipase, which showed a noticeable decline from the first reuse cycle. Among the immobilized lipases, the highest relative activity was obtained by the sol-gel entrapment method. The same tendency was observed in the case of enantioselectivity. For the same substrate, 2-hexanol, the enantiomeric excess of the native lipase at 24 h reaction time decreased from 74% to 60% after six reaction cycles, whereas for the immobilized preparates it remained practically unchained from the initial 87–88% value during 11 reuse cycles.
Thermal stability of the enzymes is important because high temperatures lead to the loss of catalytic activity, due to irreversible conformational changes that occur at the level of tertiary structure. Maintaining enzymatic activity over 40–50 °C is an important objective, since loss of activity at high temperatures is one of the main reasons hindering the industrial application of biocatalysts and thus immobilization can be a method of increasing the thermostability of enzymes.
Sol-gel encapsulation is supposed to enhance the temperature stability of enzymes, since the conformational flexibility of the enzymes is reduced and the unfolding of the tertiary structure can be prevented or hindered.
The thermal analysis data also suggested that encapsulation of the enzyme inside the xerogel matrix results in better thermal stability. The temperature stability study of the
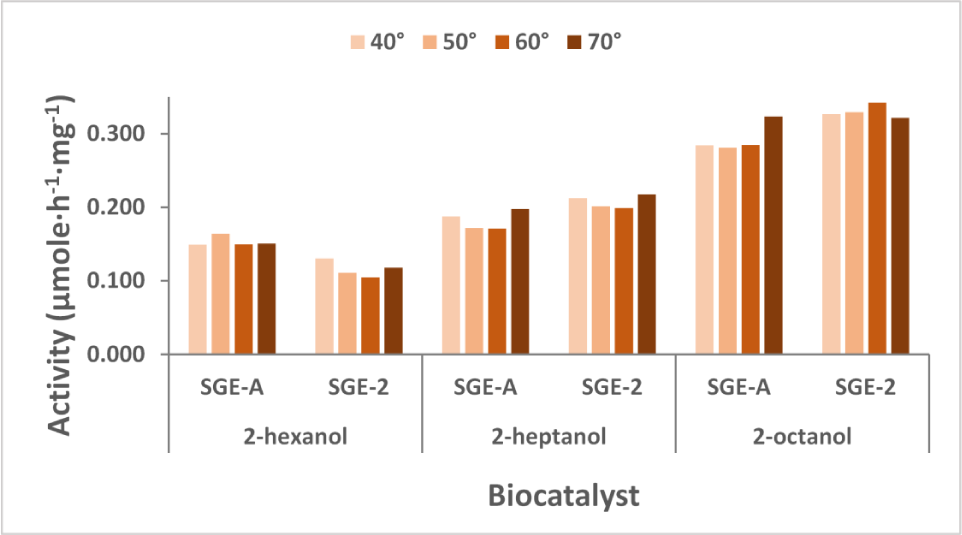
The influence of temperature on the catalytic activity of
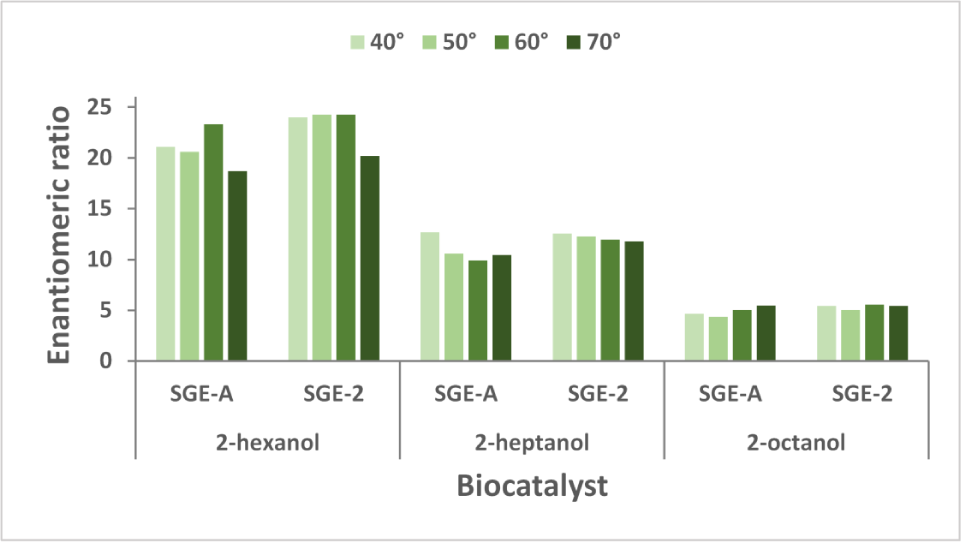
The influence of temperature on enantioselectivity of
The immobilized preparates demonstrated excellent thermal stability in the studied temperature range, for all tested substrates, the activity values increasing with the length of the alkyl chain of the secondary alcohol, as shown in Figure 8. Small variations of the activity can be assigned to the different enzyme distribution in the matrix.
Enantioselectivity is another important parameter for practical applications of enzymatic biocatalysis. Regardless of the immobilization technique used, we obtained good values of the enantiomeric ratio, as presented in Figure 9. The best values (E=24) were noticed in the acylation reaction of 2-hexanol, which could be explained by the higher affinity of the enzyme for this substrate [7].
4. Conclusions
Numerous instrumental techniques are available for the investigation and characterization of hybrid organic-inorganic matrices obtained by the sol-gel method. The presence of a protein entrapped in the silica network makes such an investigation more difficult, particularly when the distribution and sterical arrangement of the embedded molecules are important. Although the correlation between the physico-chemical characteristics of the matrix and the catalytic performance of the immobilized enzyme is obvious, it is difficult to assess the true degree of this influence. Our results demonstrated that FTIR spectroscopy is very useful to confirm the presence of structural elements, while specific surface area measured by nitrogen adsorption cannot be correlated with the biocatalyst activity, providing the surface available for small and relatively nonpolar N2 molecules but not for larger molecules with higher polarity. The enzyme is uniformly distributed in the matrix, as shown by laser confocal microscopy. Based on SEM and AFM data, the activity can be associated with the average length of the nanochannels, which is much shorter for a microstructured material than for a non-microstructured matrix. Thermal analysis gave useful information about the real amount of embedded enzyme and the total content of organic groups, as well as the efficiency of the entrapment. The immobilized
Footnotes
5. Acknowledgements
This work was performed through the Partnerships in priority areas – PN II programme, developed with the support of MEN – UEFISCDI, project no. PN-II-PT-PCCA-2013-4-0734, and was partially supported by the strategic grant POSDRU/159/1.5/S/137070 (2014) of the Ministry of National Education, Romania, co-financed by the European Social Fund – Investing in People, within the Sectoral Operational Programme Human Resources Development 2007–2013.
