Abstract
Nanoparticulate surfaces possessing antimicrobial and fungicidal properties under visible light illumination have found wide applications in a number of fields. In this study, titania nanotubes, as well as titania compact films were designed with pure Cu2O crystals in a mildly acidic copper acetate solution using a simple alternating current (AC) deposition approach. In this way, the thermally oxidized Ti substrate was coated by densely packed pyramidal and bi-pyramidal shaped Cu2O crystals with dominant (111) planes and investigated against several types of fungi and bacteria. For comparison, TiO2 nanotube (TiNT) films were also decorated with similar crystals and tested. The results showed that, compared to bare TiO2 films, both Cu2O-in-TiNT and Cu2O-on-TiO2 heterostructures exhibited remarkably enhanced activity against tested fungi and bacteria. We also demonstrated that the high photoactivity of these crystals remained even after 50 h stability tests under bright light illumination. The results obtained from in vitro tests indicated that Cu2O-in/on-TiO2 heterostructures show promise as visible light driven antimicrobial materials.
1. Introduction
Titanium (Ti), due to its high mechanical hardness, low density, elevated chemical stability, relatively low cost, environmentally-friendly characteristics and photocatalytic activity under UV irradiation, belongs to the category of metals suitable for the production of orthopaedic and dental implants, chirurgical instruments and self-cleaning surfaces. In air, this metal is always covered by a thin TiO2 film, the thickness of which can be increased simply by thermal and chemical oxidation or anodization high up in the sub-μm scale. TiO2 films produced by anodizing/annealing may vary in terms of electronic and phase structure (anatase, brookite, rutile) porosity and activity [1]. Due to a large band gap energy (∼3.2 eV) and a more positive conduction band potential than the potential required for water reduction (1.23 V vs. NHE at pH = 0), pure TiO2 films effectively absorb light, split water and demonstrate self-cleaning properties under UV light illumination [2, 3]. The antibacterial behaviour of UV illuminated TiO2 powders were first reported three decades ago [4]. Recent reports on the bactericidal properties of various TiO2 nanoparticles and films are frequently based on pure TiO2 photocatalytic behaviour under UV irradiation [5]. Many studies have also been devoted to the formation of TiO2-based bactericide particles under visible-light illumination [6–9]. For example, the solvothermal approach has been proposed for the synthesis of various Cu2O crystals, indicating that the obtained Cu2O crystals show good antibacterial effects and that with the morphology of the Cu2O crystals changing from cubic to octahedral, their antibacterial properties change from general bacteriostasis to high selectivity [10]. Dispersed in the tested solutions, these photocatalysts have shown promising bactericidal effects under visible light irradiation. However, in all of these applications, the post-separation of doped TiO2 species from the disinfected solutions are required. This procedure is tedious and cost-intensive, rendering it unsuitable for the cleaning of drinking water.
In the past decade, to avoid the cleaning of tested solutions, immobilization of the TiO2-based photocatalyst species onto various substrates have been proposed [11–14]. For example, CNT-doped TiNT on glass by dip-coating [11], Ag/TiO2 composite films [12], CdS/Pt-TiNT [13] and high-efficient CdS/TiNT films [14] on the Ti substrate have been reported. However, most of these surfaces showed either low-effectiveness under illumination by bright light or were toxic due to the application of CdS.
Currently, several routes have been also reported for TiO2 conduction and valence band re-design, for example, via attachment of non-toxic lower band gap semiconducting nanoparticles, such as Cu2O [14, 15] and CuO [16, 17].
Cuprous oxide (Cu2O) is typically a p-type semiconductor possessing a direct band gap of 2.0–2.2 eV [18]. Owing to visible light absorption, chemical and physical stability, and its low cost, nanostructured Cu2O particle arrays have recently drawn significant interest in terms of the photo conversion of bright light into electrical or chemical energy [15, 19, 20]. Note that solar conversion efficiency is equal to 11% from the theoretically possible [21]. In addition, recent studies have implied that Cu2O crystallites demonstrated distinctive antimicrobial activity against some types of bacteria [22,23]. According to Lu et al. [10], the antimicrobial activity of Cu2O crystallites, including cubic, octahedral, hexa-spindel, polyhedral, etc., depended on their morphology. All of these crystallites demonstrated anti-bactericidal activity, but only octahedral-shaped ones possessed high selectivity. Furthermore, it has recently been reported that polycrystalline Cu2O with four-sided pyramidal shape species that electrodeposited onto the ITO glass substrate, in contrast to single-crystal p-Cu2O ones [24, 25], were quite stable in aqueous solutions under visible light illumination [26, 27]. This stability was linked to the predominant Cu+-termination of (111) planes of electrodeposited p-Cu2O, at which the reduction of Cu2O to Cu0 by reaction Cu2O + H2O + 4e → 2Cu0 + 2OH− did not take place [27]. It was therefore suspected that if well-attached to the titanium oxide substrate, numerous pyramidal Cu2O crystallites with (111) planes could provide stable fungicidal and antimicrobial activity under visible light illumination.
The aim of this study was to design TiO2/Cu2O heterostructured films onto the titanium substrate possessing high stability and effective antimicrobial properties against several types of fungi and bacteria under solar light illumination. To achieve this result, an aqueous solution comprising Cu(II) acetate, Mg acetate and acetic acid at a pH = 5.3, as well as an alternating current approach, are proposed in this study. For comparison, polyhedral- and pyramidal-shaped Cu2O crystals with (111) prevailing surfaces were organized both inside the TiO2 nanotube (TiNT) film and onto the TiO2 substrate. The stability of Cu2O species tethered to the thermally oxidized Ti surface was investigated in a 0.5 mol L−1 Na2SO4 aqueous solution at −0.35 V (vs. Ag/AgCl, sat.) and 100 W tungsten lamp illumination for 50 h. The antimicrobial properties of the obtained Cu2O/TiO2 heterostructures were tested against several types of fungi and bacteria.
2. Experimental
2.1 Reagents
A 98% purity copper acetate, Cu(CH3COO)2•H2O, 99% purity magnesium acetate, Mg(CH3COO)2• 4H2O and 99% purity ammonium hepta molibdate, (NH4)6Mo7O24•4H2O, were purchased from Sigma Aldrich. NaOH (98.8%), aceton (99.8%), ethylene glycol (99%) and acetic acid, CH3COOH (99.8%) were obtained from Eurochemicals, Slovakia. Ammonium fluoride (98% purity) was obtained from Merck.
Saccharomyces cerevisiae, Aspergillus versicolor, Candida parapsilosis and Candida krusei fungi and Pseudomonas aeruginosa, Escherichia coli, and Micrococcus luteus bacteria were obtained from the Liofilchem (Italy).
2.2 Electrochemical formation of TiNT films
Ti foil at 99.7% purity and 0.127 mm thick and purchased from Aldrich was used to prepare disc-shaped specimens 13.8 mm in diameter. The surface of samples was ultrasonically cleaned in acetone, ethanol and water, for 6 min in each, air dried and anodized in the thermostated glass cell containing ethylene glycol (Etg) with 0.3% wt. NH4F and 20 mL L−1 H2O at 20±
2.3 Electrodeposition of Cu2O species
Electrodepositions of Cu2O on TiO2 nanotube walls and TiO2 surface were accomplished in a two-electrode glass cell, in a 200 mL volume and containing an aqueous solution of 0.1 mol L−1 copper acetate, Cu(CH3COO)2 H2O, 0.1 mol L−1 magnesium acetate, Mg(CH3COO)2 4H2O and acetic acid. Based on previous investigations [28], Mg(II) salt was used to prevent the breakdown of the titania barrier layer during AC treatment. Several graphite stripes connected and positioned around the working specimen were used as a counter electrode. All depositions were carried out in a room temperature solution. Milli-Q water (18 MΩ) was used for the preparation of the Cu2O deposition solution. Analytical grade Mg(OOCCH3)2, Cu(OOCCH3)2 and acetic acid were purchased from Eurochemicals Ltd. Owing to the rectifying properties of the titania barrier layer [29], the depositions were carried out by AC at a constant peak-to-peak voltage (Up-to-p) mode using a programmable alternating current source Chroma AC (model 61602, Taiwan). The frequency of AC was varied within the 25 to 500 Hz range. All solutions were prepared with deionized water and reagent grade chemicals. After deposition, each sample was thoroughly rinsed with pure water and air dried.
2.4 Characterization
To ascertain the purity of Cu2O species tethered to the TiNT walls, voltammetric measurements were performed using a potentiostat/galvanostat AUTOLAB 302 and a three-electrode electrochemical cell, in which Pt wire and a hydrogen electrode were used in the working solution (RHE) as a counter and reference electrodes, respectively. Prior to measurements, a working solution composed of 0.1 mol L−1 KOH was de-aerated by Ar gas bubbling for at least 0.5 h.
The stability tests of Cu2O crystals tethered to the TiO2 surface was carried out in 0.5 mol L−1 Na2SO4 solution under ∼10 mW cm−2 irradiation provided by 100 W tungsten arc lamp illumination potentiostatically at −0.35 V (vs. Ag/AgCl, sat.) for up to 50 hrs. The content of Cu2O and Cu deposited (and dissolved during stability tests) was determined by the analysis of solutions, obtained via etching the specimens in a hot HNO3:H2O (1:1) solution followed by careful rinsing and sonication in Milli-Q water.
The inductively coupled plasma emission spectrometer OPTIMA 7000DV (Perkin Elmer, USA) was employed for analysis. Measurements were made on emission peaks at 327.393 and 324.752 nm for Cu, at least five times for the same solution.
To examine the morphology of films on the surface and in cross-sections, a scanning electron microscope, model FEI Quatra 200F and a Cross Beam Workstation Auriga, equipped with a field emission gun and EDX spectrometer were used.
Phase composition of the obtained heterostructures was studied by X-ray powder diffraction (XRD) using a D8 diffractometer (Bruker AXS, Germany), equipped with a Göbel mirror as a primary beam monochromator for CuK α radiation. A step-scan mode was used in the 2Θ range from 18 to 77° with a step-length of 0.02° and a counting time of 8 s per step. The size of crystallites was determined from the broadening of all diffraction peaks using PDXL software and Halder-Wagner (H-W) approximation.
Raman spectra were recorded using an inVia (Renishaw) spectrometer equipped with a thermoelectrically cooled (−70°C) CCD camera and microscope. The 532 nm solid state diode laser beam was used as an excitation source. The laser power at the sample was restricted to 0.06 mW and 0.6 mW for samples Ti oxide/Cu2O and bare Ti oxide, respectively. The Raman scattering wavenumber axis was calibrated by the silicon peak at 520.7 nm. The spectral slit width near 500 cm–1 was 6.7 cm–1. The 50x objective was used during all measurements. The laser line was focused to a ∼ 2 μm diameter spot on the sample. The parameters of bands were determined by fitting the experimental spectra with Gaussian-Lorentzian shape components using GRAMS/A1 8.0 (Thermo Scientific) software.
The antibacterial effects of the fabricated films were assessed using the Saccharomyces cerevisiae, Aspergillus versicolor, Clodosporium cladosporioides, Candida parapsilosis and Candida krusei fungi, cultured in Sabouraud CAF agar at 27°C, and Pseudomonas aeruginosa, Escherichia coli and Micrococcus luteus bacteria cultured in Nutrient agar at 27°C, and the effects on colony-forming units were analysed.
The antimicrobial activity of Cu2O-in/on-TiNT samples against Micrococcus luteus (M. luteus), Escherichia coli (E. coli), Aspergilus fumigatus (A. fumigatus), Geotrichum candidum (G. candidum) and Candida parapsilosis (C. parapsilosis) was investigated using the zone inhibition method. Following this method, the pieces of alumina films were pressed gently onto Sabouraud CAF agar or Nutrient-agar (“Liofilchem”, Italy) plates inoculated with ≅106 CFU mL−1 microorganisms and analysed after 48 h incubation at 27±
It should be noted that unless otherwise indicated, all the above-mentioned experiments were repeated three times and the obtained results were statistically analysed by calculating averages and standard deviations. The resulting data were applied for building corresponding diagrams.
3. Results and discussion
3.1 Composition and structure of heterostructured films
We have reported recently that TiNT walls can be decorated with numerous pure Cu2O species both inside and outside tubes via alternating current deposition from a slightly acidic Cu(II) acetate solution (Fig.1 A, B) [29]. The size of crystallites increased with the deposition bath voltage and AC treatment time attaining 60 nm. In this study, we demonstrate that the same solution can be successfully explored for the formation of densely packed pyramidal- and bi-pyramidal-shaped (octahedral) Cu2O crystals onto the thermally oxidized Ti substrate (Figures 1C and D). The shape of crystallites was found to be dependent on the pH of solution and AC deposition conditions, similar to the case of potentiostatic depositions from the alkaline cupric lactate solution onto the ITO substrate [27]. The pH of solution kept at a pH within 5.2 to 5.35 range was found to be an optimal. In this solution, by variables in the AC treatment conditions (Up-to-p value, AC frequency and deposition time) the surface of the thermally oxidized Ti substrate can be predictably covered with densely packed pyramidal-shaped crystallites (Fig. 1C). The planar dimensions of the pyramidal and bi-pyramidal crystals were found to be dependent mainly on the AC treatment time and Up-to-p value, attaining sub-micrometre size. Most uniform covering of TiO2 surface took place at 100 Hz frequency although some higher deposition rates were observed at 50 Hz frequency (Figure 2). Please note that randomly dispersed crystals can be formed at up to 250 Hz frequency. The shape of the deposited species depends on the pH of the solution although the pyramidal-shaped species can be deposited only from slightly acidic acetate solutions (Figure 3A). In the neutral solutions, e.g. at around pH = 7.0, the coral-shaped species (Figure 3B) were formed, whereas in the alkaline solutions, e.g. at pH = 10.0 (Figure 3C) the nanoparticulate species dominated.

Top-side (A) and cross -sectional (B SEM views of TiNT film fabricated by Ti anodizing in ethylene glycol solution, then annealed at 450 °C for 2 h and AC treated in Cu(II) acetate electrolyte at Up-to-p = 1.1 V for 5 min. In (C) and (D): typical top-side SEM views of crystallites formed onto the thermally oxidized Ti substrate by AC deposition in the same solution at U p-to-p 1.3 V for 10 min
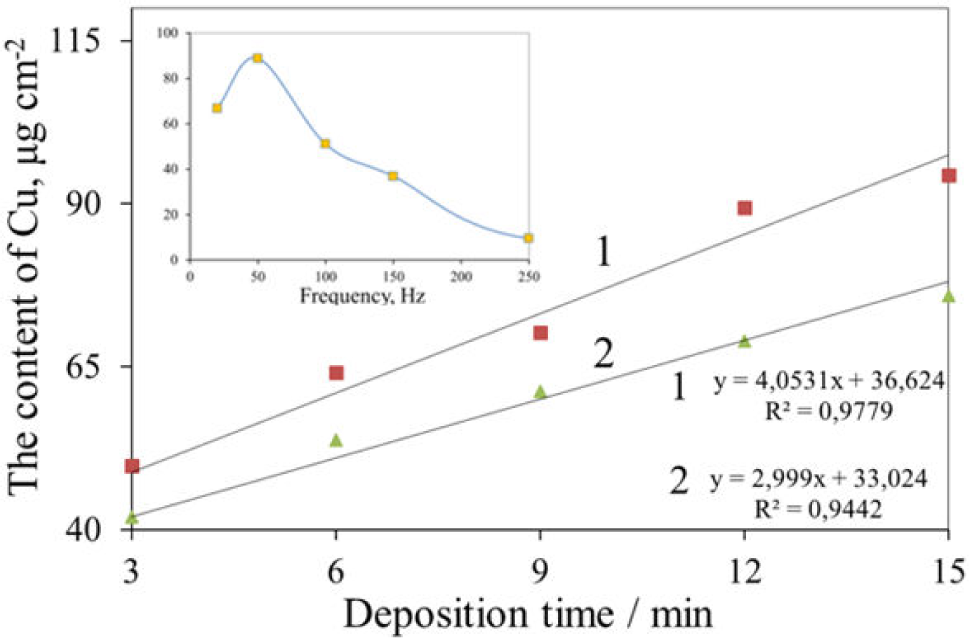
Variations of the content of deposited copper suboxide species onto the thermally oxidized Ti substrate in the solution comprising 0.1 mol L−1 Cu(OOCCH3)2 + 0.1 mol L−1 Mg(OOCCH3)2 + CH3COOH (pH = 5.3) at Up-t-p = 1.3 V and AC frequency: (1) 50, (2) 100 HZ on the deposition time. In the Inset: the dependency of deposited copper at Up-t-p = 1.3 V for 10 min on the AC frequency

Top-side FESEM views of crystallites formed onto the thermally oxidized Ti substrate by AC deposition at Up-to-p = 1.3 V and room temperature for 10 min in the same way as in Figure 2 with the solution kept at the pH: (A) 5.0; (B) 7.0 and (C) 10.0
XRD and Raman investigations were used to characterize the phase structures of the deposited species. Figure 4 shows the XRD patterns of the Cu2O/TiNT heterostructure (A) and Cu2O octahedra obtained by AC deposition from a slightly acidic Cu(II) acetate solution on the thermally oxidized Ti substrate (B). As can be seen, both XRD patterns contain several obvious diffraction peaks at 2Θ positions 29.58, 36.42, 42.32, 61.46 and 73.60 that, according to PDF card no 01-078-2076, can be indexed as (110), (111), (200), (220), and (311) planes of the cubic phase cuprous oxide, allowing us to deduce that the deposited species are polycrystalline Cu2O. It is also obvious that the preferred orientation of Cu2O grains is (111). The size of Cu2O grains calculated from the main diffraction line (111) broadening was found to be dependent on the conditions of electrodeposition varying within 32–45 and 65–85 nm ranges for Cu2O species deposited inside the TiNT and onto the TiO2 substrate, respectively. No peaks that can be ascribed to CuO or Cu(OH)2 were detected, although two small peaks at 2Θ positions around 43.3° and 50.4° (Figure 4B) could be linked with the presence of some Cu0 onto the heat-treated Ti surface. It is also obvious that the pattern of TiNT film demonstrating a set of 10 reflections as shown in Figure 4A by the blue stars, according to PDF card no 00-021-1272, indicated the presence of TiNT in the anatase-TiO2 phase, whereas these peaks are absent in the heat-treated specimen.

Typical XRD patterns (A) (B) of Cu2O species deposited inside the TiNT film (A) and on the heat-treated TiO2 substrate (B) by AC (100 Hz) treatment in the solution as in Figure 2 at Up-to-p = 1.2 V for 15 min
Figure 5A compares the Raman spectra of the TiNT film before and after deposition of copper containing species. Strong Raman bands at 147, 397, 518, and 637 cm–1 are characteristic of crystalline anatase TiO2 structure [30, 31] whereas difference spectrum (c) clearly shows several peaks associated with copper oxygenous compounds. In brief, bands at 219, 533, and 625 cm–1 evidence the presence of Cu2O [32–34]. The Raman spectrum of pure thermally oxidized Ti substrate corresponds to the rutile TiO2 phase with characteristic Eg and A1g modes at 444 and 609 cm–1, respectively (Figure 5B) [31], whereas bands at 98, 215, 531, and 617 cm–1 according to [33–35] evidence the presence of Cu2O.
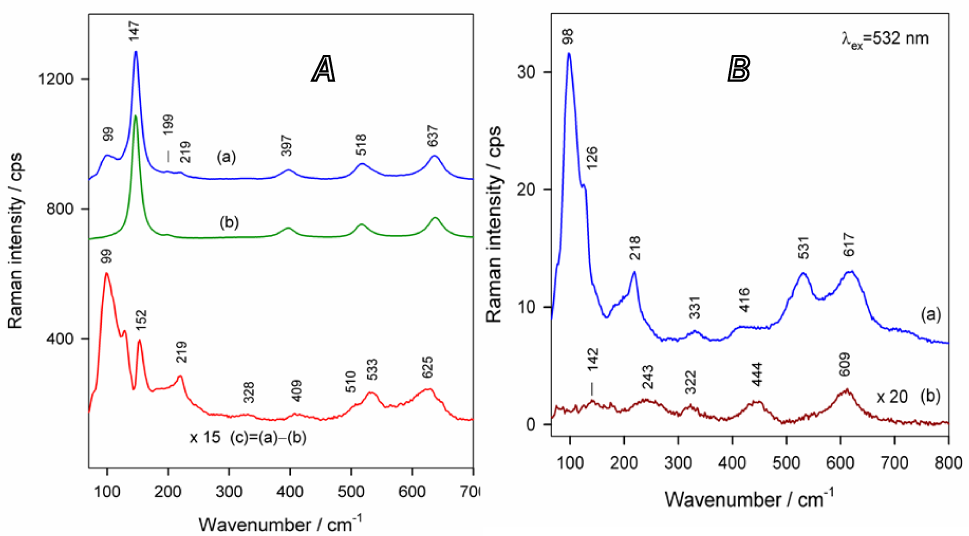
In (A): the Raman spectra of Cu2O-in-TiNT (a), pure TiNT (b) and their difference (c) in wavenumber region from 65 to 700 cm−1. In (B): the same for the Cu2O deposited on the thermally oxidized Ti surface (a) and pure heat-treated Ti (b). Excitation wavelength is 532 nm. The same conditions as in Figure 4 were used for sample preparation.
The purity tests for AC-deposited Cu2O pyramidal crystals were further performed in a 0.1 mol L−1 KOH de-aerated solution by potential sweep from the open circuit potential, e.g. 0.75 V versus RHE, to −0.25 V and backwards. Voltammograms obtained for different contents of deposited species are shown in Figure 6. As seen, only a sharp reduction peak at ∼−0.1 V, attributable to Cu2O reduction is characteristic for all specimens. According to [36], such a reduction plot indicates the presence of pure Cu2O.

Reduction profiles of Cu2O species deposited on the TiO2 substrate by AC (100 Hz) treatment in the solution as in Figure 2, kept at a pH = 5.25 and Up-to-p = 1.2 V for 5 (1), 10 (2) and 15 min (3). Voltammograms were recorded in the O2-free 0.1 mol L−1 KOH solution at ν = 1.0 mV s−1 and 20°C.
3.2. Antimicrobial properties
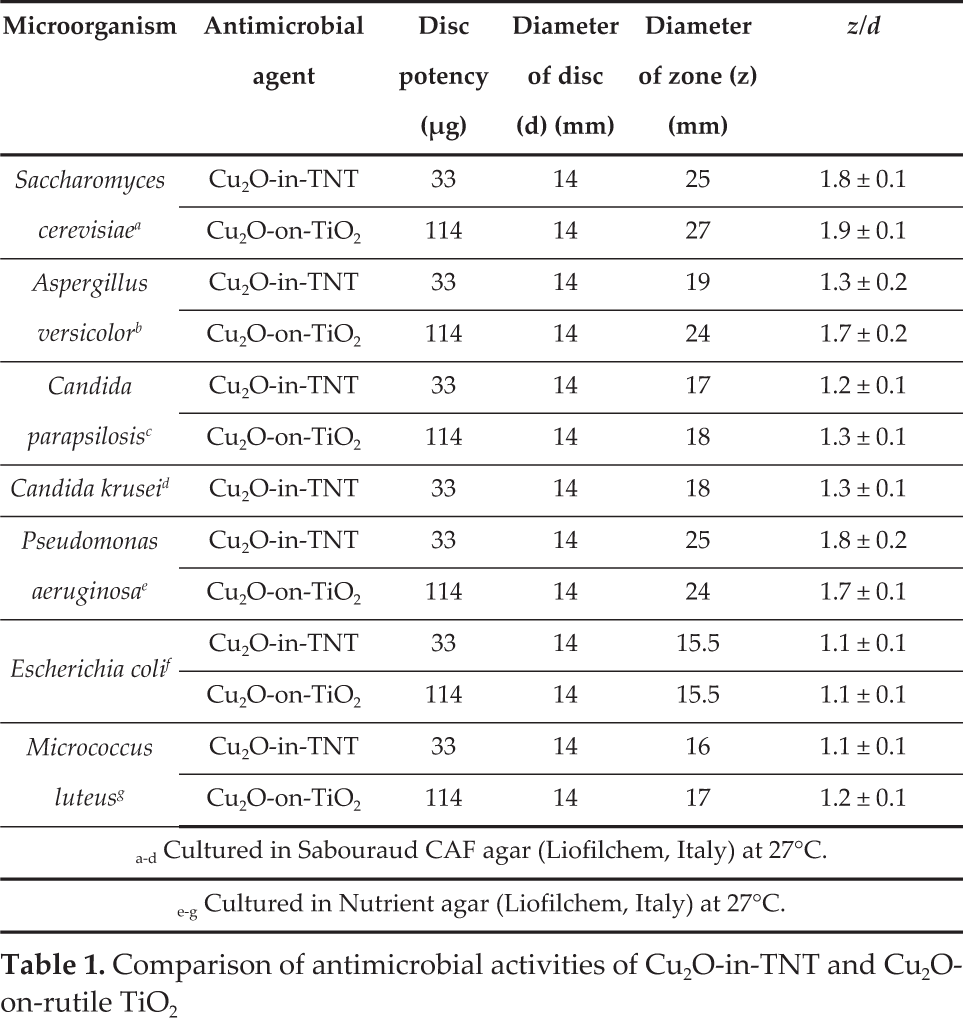
The antibacterial activity results revealed that the pyramidal-shaped Cu2O crystals encasing both the inside of the TiNT film tubes and deposited onto the rutile TiO2 surface acted as excellent antimicrobial agents against all tested fungi as well as Gram-positive and Gram-negative bacterial strains. It should be noted that in the case of Cu2O-on-rutile a fungistatic effect was observed for A versicolor and C. parapsilosis fungi, whereas in the case of Cu2O-in-TiNT the fungistatic effect was obtained in the Petri dishes with A. versicolor, C. parapsilosis and C. krusei strains. Figures 7 and 8 show the zones of inhibition produced by Cu2O-in-TiNT and Cu2O-on-rutile TiO2 against various eukaryotic and prokaryotic microorganism strains, respectively. Based on these results, some stronger fungicidal effects are characteristic for Cu2O-on-rutile TiO2 specimens showing the inhibition zones up to 25–27 mm after 48 h incubation (Table 1).
Comparison of antimicrobial activities of Cu2O-in-TNT and Cu2O-on-rutile TiO2
Cultured in Sabouraud CAF agar (Liofilchem, Italy) at 27°C.
Cultured in Nutrient agar (Liofilchem, Italy) at 27°C.

Zone of inhibition produced by Cu2O-on-rutile (A, B, D) and TiO2 Cu2O-in-TiNT (C) specimens against the lawns of eukaryotic microorganisms: (A, C) A. versicolor, (B) S. cerevisiae, (D) C. krusei. In the left side - control specimen.

Zone of inhibition produced by Cu2O-on-rutile (A, B, D) and Cu2O-in-TiNT (C) specimens against the lawns of prokaryotic microorganisms: (A) M. luteus, (B, C) – P. aeruginosa and (D) E. coli. In the left side - control specimen.
The antimicrobial efficiencies of pyramidal-shaped Cu2O species attached to various TiO2 substrates were also assessed against typical Gram-negative (E. coli and P.aeruginosa) and Gram-positive (M. luteus) bacteria. Note that P. aeruginosa was found to be more sensitive both for Cu2O-on-TiO2 and Cu2O-in-TiNT surfaces: the average inhibition zone in this bacteria lawn was 1.5 to 1.6 times wider than in E. coli and M. luteus bacteria lawns (see Table 1). We note that in contrast to Li et al.'s indication that E. coli is resistant to cubic and spherical Cu2O nanocrystals [37], our Cu2O-TiO2 heterostructures demonstrated a strong antibacterial activity against E. coli. We hypothesized that differences in the antimicrobial efficacy of Cu2OTiO2 heterostructures and Cu2O nanoparticles could be explained by synergetic behaviour of p-n heterojunction between Cu2O and TiO2 possessing a remarkable visible light photoresponse creating OH? and H2O2 oxidative species [15] in the aqueous bacteria lawns. On the other hand, as reported by Ren et al. [23], Cu2O octahedral single crystals bounded by {111} facets, as in our case, exhibited higher activity in killing E. coli than cubic ones. We also note that a similar result has been obtained under visible light illumination for E. coli inactivation by Cu2O loaded on TiNT film via direct deposition from alkaline CuSO4 and lactic acid solution [15].
3.3 Stability tests
To examine deterioration in photosensivity of Cu2O pyramidal-shaped crystals deposited from the slightly acidic Cu(II) acetate solution on the thermally oxidized Ti substrate, the stability tests were performed in a 0.5 mol L−1 Na,2SO4 solution at −0.35 V (vs. Ag/AgCl, sat.) and 100 W tungsten lamp illumination for up to 50 h. In these experiments the tungsten lamp was placed in front of a quartz glass window of an electrochemical cell at a 12 cm distance from the socket of the lamp to the specimen. Figure 9 shows the variations in the open circuit potential of the Cu2O-on-TiO2 specimen after light switching before and after the stability test which lasted for 50 h. As can be seen, the Cu2O-coated-TiO2 specimen remained quite photosensitive to bright light even after prolonged 50 usage. It is noteworthy that the contamination of copper ions was not detected in the tested solution even after 50 h. These results imply the prospective application of the Cu2O-on-TiO2 antimicrobial coatings in food and healthcare industries.
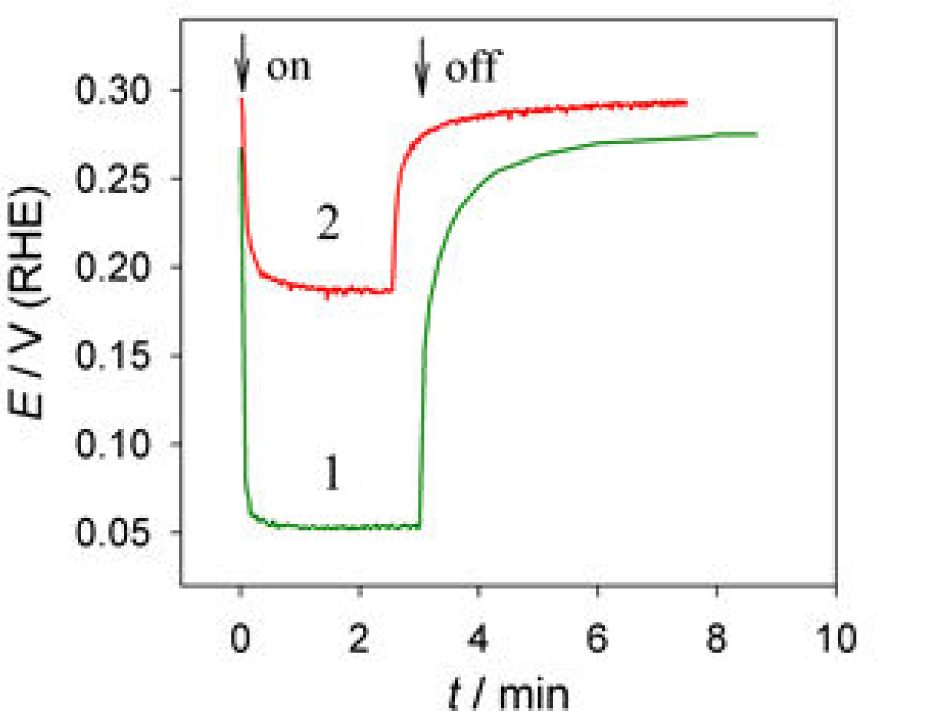
Variation in the open circuit potential for as formed (1) and deteriorate Cu2O-on-TiO2 heterostructure (2) in 0.5 mol L−1 Na2SO4 solution after switching of light. The stability test was performed in the same solution at −0.35 V (vs Ag/AgCl, sat) under 100 W tungsten lamp illumination for 50 h.
4. Conclusions
Pyramidal-shaped pure Cu2O crystallites were successfully deposited both in/on the TiO2 nanotube film and onto the thermally oxidized Ti substrate by using the alternating current deposition approach from the slightly acidic copper acetate solution. Both anodized and thermally oxidized Ti surfaces after covering with pyramidal-shaped Cu2O crystallites possess strong antimicrobial properties against the tested fungi (A. versicolor, C. parapsilosis, C. kruseiandS.cerevisiae)., Gram-negative (E. coli and P.aeruginosa) and Gram-positive (M. luteus) bacteria. In all cases, the Cu2O-on-rutile TiO2 surface demonstrated higher antimicrobial efficiency which could be linked with 2–3 times larger amounts of attached Cu2O species. The antimicrobial effect of the Cu2O species tethered to the TiO2 surface was linked with the semiconducting properties of cuprous oxide capable of absorbing visible light and through synergetic coupling with TiO2 produced oxidative species of OH? and H2O2 destroying the walls of microorganism cells. The Cu2O-coated-TiO2 specimen remained quite photosensitive to bright light even after prolonged 50 h exploitation.
Footnotes
5. Acknowledgements
We would like to thank Dr. V. Pakštas for XRD and Dr. Habil. G. Niaura for Raman spectra collection and comments.
