Abstract
Nanomedicine, as a relatively new offshoot of nanotechnology, has presented vast opportunities in biomedical research for developing novel strategies to treat diseases. In the past decade, there has been a significant increase in in vitro and preclinical studies addressing the benefits of nanomedicines. In this commentary, we focus specifically on the efficacy- and toxicity-related translational challenges of nanocarrier-mediated systems, and briefly discuss possible strategies for addressing such issues at in vitro and preclinical stages. We address questions related specifically to the balance between toxicity and efficacy, a balance that is expected to be substantially different for nanomedicines compared to that for a free drug. Using case studies, we propose a ratiometric assessment tool to quantify the overall benefit of nanomedicine as compared to free drugs in terms of efficacy and toxicity. The overall goal of this commentary is to emphasize the strategies that promote the translation of nanomedicines, especially by learning lessons from previous translational failures of other drugs and devices, and to apply these lessons to critically assess data at the basic stages of nanomedicinal research.
1. Introduction
The last two decades have witnessed the advent of nanomedicines - a powerful new component of nanotechnology that presents new opportunities and challenges in almost all specialties of medicine. There is very little doubt that nanotechnology and its use in medicine is here to stay. It can be predicted with a strong degree of confidence that the next few years will witness a further increase in the number of nanotechnology-based diagnostic and therapeutic applications. Currently, research involving nanomedicines and their potential use for various human conditions is being conducted at a rapid pace. New information relating to the synthesis, characterization and biological response of nanomedicines is being added almost every day.
Nanomedicines generally involve the use of nanoparticles as delivery systems to transport drug to target sites, usually tumours or diseased tissues and cells. Being less than a tenth of a micron in size, nanoparticles acquire certain properties that make them useful in several avenues of medicine, such as medical imaging, drug delivery for enhanced bioavailability, and triggered drug delivery. Drugs can be encapsulated within - or surface conjugated - to nanoparticles, which generally results in enhanced effects of the drug. Drug-loaded nanoparticles in particular have demonstrated enhanced efficacy in killing several types of tumour cells. With nanomedicinal technology growing out of its infancy, a number of nano-therapeutic drugs have emerged from the commercial pipeline. Doxorubicin encapsulated within nanoliposomes (e.g., Caelyx®), PEGylated protein conjugates (e.g., Oncospar® and PegIntron®) and polymeric nanoformulations (e.g., Copaxone®) are a few such examples. Global investment in nanotechnology, specifically in healthcare related areas, has been increasing steadily. The National Nanotechnology Initiative (NNI), a US government initiative for promoting research and development in the area of nanotechnology, received $1.7 billion in the 2014 Presidential budget [1]. Similarly, the European Union, Japan and China are reported to have invested $1.7 billion, $950 million and $430 million, respectively, in nanotechnology initiatives [2]. Global optimism combined with significant international resource allocations to nanotechnology research, especially in healthcare, promises significant positive outcomes and has the potential to provide much-needed breakthroughs in modern medicine.
In the past decade, there has been a significant increase in in vitro and preclinical studies addressing the benefits of complex drug delivery systems [3–5], especially nanoformulations of various drugs (Figure 1.). Thanks to these studies, we now have a much clearer (though incomplete) understanding regarding what it takes for the construction of a biocompatible nano-drug delivery system, its material component and related pharmacokinetic/pharmacodynamic parameters. Even though much fundamental work still remains, we have gathered sufficient material science and cellular and systems biology information to earnestly consider translating nano-drug formulations from bench to bedside. However, this is no ordinary task. While each nanomedicine has its own distinctive set of challenges, there are certain hurdles that remain common to the entire family of nanomedicines. The rapid uptake and clearance of nanoparticles by the mononuclear-phagocyte system, renal and biliary excretion, and cationic charge-dependent toxicity, are a few such examples. Though strategies such as the surface-coating of nanoparticles with protein-resistant polymers [6] have been devised to delay clearance and prolong drug action, challenges concerning tissue accumulation and toxicity still remain.

Publication count for nano-drug-related articles published from 1980 to 2013. A year-wise PubMed search was conducted with the key words ‘nano-drug’, nanomedicine', ‘nano-drug delivery’, ‘nano carrier drug delivery’, and ‘targeted drug delivery’. Both research articles and reviews have been included in the above publication count.
Currently, the primary objective of most nanomedicinal formulations is to enhance the pharmacokinetic characteristics and reduce collateral off-site effects of the loaded drug (Figure 2). While the objective of increasing efficacy and decreasing toxicity seems rather straightforward, the process, however, is fraught with challenges that are technical, biological and financial in nature. In this commentary, we will focus specifically on the efficacy- and toxicity-related translational challenges of nanocarrier-mediated drug delivery systems and briefly discuss possible strategies for addressing these issues at the in vitro and preclinical stages before attempting human translation.

Expected role of nanomedicines: Enhancing efficacy and lowering toxicity
2. Challenges facing the translation of nanomedicines
Translating a nanomedicine formulation from a preclinical stage to a phase trial is a daunting task. There are several examples of nanomedicinal formulations with promising preclinical data showing enhanced efficacy, only to fail in clinical phase trials. The cisplatin encapsulated PEGylated nanoliposome (SPI-077TM) is one such example where in vivo studies showed enhanced anti-tumour activity [7]. Upon SPI-077TM administration in mouse cancer models, cisplatin accumulated within the tumour tissue in larger concentrations and circulated in the blood for much longer without signs of toxicity [7]. An enhanced tumour regression ability and a favourable pharmacokinetic profile meant that SPI-077TM was a promising formulation for human studies. Despite such encouraging results, phase I-II results in patients with locally advanced squamous cell cancer of the head and neck showed negligible activity with SPI-077TM [8]. This lack of efficacy in patients was attributed to lower bioavailability and the altered release profile of cisplatin from SPI-077TM nanoliposomes. Though toxicity was not an issue (both, primate studies [9] and patient data demonstrated no changes in the toxicity profile), minimal gains in efficacy were not sufficient for successful translation and subsequent progression to mainstream clinical practice. This highlights the efficacy expectation that nanomedicines face as they progress through the translational pipeline.
Another challenge in nanomedicinal research is the change in the drug toxicity profile after loading it onto a carrier [10]. Though the desirable effect of nano-encapsulation is to decrease the toxicity profile of the drug, the opposite may also occur. This is especially true for particle-encapsulated cytotoxic drugs, which get sequestered primarily by the kupffer cells in the liver, leading to drug-induced hepatic injury. Increased drug-induced toxicity after encapsulation has also been observed in locally administered particulate delivery systems. Local anaesthetic drugs loaded onto polymeric and lipid-sugar containing microspheres produced local muscle injury that was more intense than the drug alone [11]. This drug-induced muscle injury was attributed to the prolonged exposure of adjacent muscle tissue to the slowly releasing drug from the delivery system. While it may be difficult to control immediate toxic effects locally, more precise control of burst release, smart surface coating, ligand-mediated active targeting and triggered drug release systems are being developed to address drug-induced toxicity challenges.
Another important complicating factor that arises in nanomedicines designed as delivery systems concerns the physiological effects of the nanocarrier itself. Depending on the size, surface charge and surface chemistry of the nanomaterial, there can be independent toxicity effects associated with the particle itself [12]. Some well-known nanoparticle-associated adverse effects include direct tissue injury, selective organ toxicity, long-term tissue retention, potential carcinogenic effects and immunemediated injury, all of which are critical factors to be considered in translation [10,13]. Site-directed targeting is a common approach to circumventing this issue, but non-specific tissue-binding and the accumulation of nanomedicine in the liver, spleen and kidney remain. This report will not discuss the bio-mechanisms of nanomedicinal toxicity, since there have been several exhaustive reports and commentaries written on this topic [14–16]. However, what we will discuss is the need to restructure the bench-side and preclinical research strategies in order to focus on long-term translation while keeping nanomedicinal toxicity in the backdrop. We will address questions related specifically to the balance between toxicity and efficacy, a balance that is expected to be substantially different for nanomedicines compared to that for a free drug.
The current nano-biotechnology approach, of first ‘make it’ and then ‘screen it’ [17] borrowed from traditional pharmaceutical industry (though necessary for the advancement of the field), may not be the most conducive for translational medicine. As an example, platinum is lately being re-examined as a nanoparticle carrier for various biological applications, including cancer [18,19]. It is well-known clinically that platinum is highly nephrotoxic [20,21], and hence one can pre-emptively predict that attempts to translate a platinum-based nanomedicinal formulations from the bench-side to the bed would pose nephro-toxicity challenges. A more prudent approach for clinical translation would be an ‘application-specific design’ methodology. This would require the clear identification of a target profile (a specific disease, its subtype and the cellular target that needs to be modulated), an in-depth understanding of the biology, pathogenesis and, most importantly, shortcomings of the current line of management. This approach is well in line with previous observations that successfully translatable treatment modalities are more likely to arise from methods that are based on ‘quality by design’ [22] rather than starting from scratch. Thus, nano-drug delivery systems designed and developed based on the strong biology and clinical background of a specific target would have a much higher probability of success in translation as compared to the ‘make it and screen it’ approach.
While proof of concept studies demonstrating the advantages of several nano- and micro-sized drug delivery systems have been published, very few of them would in reality have any significant translational value [17,23–25]. Though many nanomedicines demonstrate excellent efficacious effects in vitro, they seldom hold ground in hostile in vivo conditions where factors such as blood flow, turbulence, opsonization and protein coating, complement activation, adhesion, immune reactions, pH changes, enzymatic reactions, and other unknown biological events, may play a major role. Most nanomedicinal researchers recognize the pitfalls of invitro experiments, and hence results established in preclinical animal studies are given much greater prominence. However, despite favourable outcomes in animal studies, a significant number of products fail to enter clinical phase trials [26]. In the majority of such translation failures, disparities in the biology of animal disease models and the harsh subject-selection criteria of phase I trials are seen as the primary contributing factors [26]. While the challenges for translation seem overwhelming, lessons can be learnt from the current list of commercialized nanomedicines, their paths towards translation and by infusing a much more critical view of data obtained from in vitro and in vivo studies. In this commentary, we wish to take up one such basic evaluation at the in vivo and in vitro levels that could possibly add to the overall assessment of a nanomedicine.
3. The Efficacy and Toxicity Dilemma
Despite strong arguments in favour of nanomedicines, considering their relatively recent entry and the limited availability of long-term data, it is only prudent to be extra-rigorous while assessing the benefits of nanomedicine. From a broader perspective, nanomedicines are no different from any other therapeutic options in that they have their advantages and disadvantages; they are efficacious but they also demonstrate adverse effects - what we term as the ‘yin and yang’ of nanomedicines. Efficacy and toxicity are two fundamental parameters that determine the fate of any medicine. In this context, a natural question that arises is whether the nanomedicine is better than its free drug counterpart in terms of efficacy and toxicity. Yet how do we determine whether a specific nanomedicine is better? Should the efficacy of the nanoformulation be better than the free drug? Or should the adverse effect of the former be lower than that of the latter? Or both? In this section, we argue that the benefits of nanomedicines should be considered taking both the efficacy and adverse effects of the nanomedicine and the free drug together. We propose an approach for both in vitro and in vivo data, and provide suggestions for quantifying possible treatment benefits by comparing: 1) toxicity and efficacy in terms of the ratios of nanomedicines to free drugs, and 2) nanomedicine-to-free drug ratios in terms of efficacy and toxicity. This is a rationalistic guideline for assessing the effective profile of a nanomedicine that could be incorporated into the majority of cell culture assays, ex vivo assays and preclinical animal testing. Such assessments are highly relevant in the current scenario, where both in vitro and in vivo research examining the benefits of nanomedicines has only increased (Figure 1.). A robust, objective system that can compare both the positive and negative effects of nanomedicine in comparison to the effects of free drugs would facilitate the better assessment of nanomedicines at the bench and provide a strong checkpoint to help decide whether it would be worth pursuing the nanomedicinal form of the drug.
In this report, we propose two simple mathematical approaches to examine the benefits of a nanomedicine versus its free drug counterpart. The first approach compares efficacy (E) / toxicity (T) ratios for a nanomedicine (E/T)Nanomedicine and a free drug (E/T)Free drug, while the second approach compares the ratio of the toxicity or efficacy of the nanomedicine (N) to that of the free drug (F), namely, (N/F)Efficacy and (N/F)Toxicity. Both ratios could be followed over time and various concentration ranges. Though looking at ratios may seem simplistic, this approach provides convenient yet powerful indices to assess the overall benefit of a nanomedicinal preparation versus that of a free drug.
The (E/T) and (N/F) ratios can be easily utilized in both in vivo and in vitro studies. For effective assessment, it is important that the data in the numerator and denominator depicting efficacy and toxicity be converted into a normalized percentage. The resulting ratio can then be used to assess the overall benefit of a free drug versus its nanomedicinal variant. To further elaborate on these ratios, we have used both hypothetical data and data from previously published studies to create various scenarios in terms of nanomedicine toxicity and efficacy in comparison to free drug effects. In the hypothetical scenarios, we establish possible toxicity and efficacy profiles of nanomedicines and examine the resulting ratios and their interpretation. In the case studies, we summarize published data corresponding to the efficacy and toxicity of nanomedicine and free drug formulations, followed by the conversion of individual data to a percentile format and the calculation of E/T and N/F ratios.
4. Hypothetical scenario depicting possible toxicity and efficacy profiles of nanomedicines.
In Figure 3, we present a few scenarios of varying efficacy and toxicity in the free and nanomedicinal form of the drug. We highlight three typical but important outcomes that we analyse using our proposed ratio metric system. In Figure 3A, we present a nanomedicine-free drug profile, where the efficacy of a nanomedicine and a free drug are similar but the toxicity profile of the nanomedicine is either lower (Scenario 1, Figure 3A) or higher (Scenario 2, Figure 3A) than the toxicity profile of the free drug in all the arbitrary concentration ranges. In scenario 1, where the efficacy of the nanomedicine is similar to that of the free drug and the toxicity of the nanomedicine is lower than that of the free drug, conventional plotting of the data demonstrates an apparent - but moderate - difference in concentration-dependent toxicities between the free drug and the nanomedicine (Figure 3A). However, upon plotting the E/T ratio of the nanomedicine and the free drug (Figure 3B, Scenario 1), we observe that the maximal benefit from the nanomedicine lies within the first two concentration ranges - a fact that is not distinctly apparent in Figure 3A. With a further increase in concentration, the nanomedicine E/T ratio decreases and approaches a ratio value closer to that of the free drug, suggesting a lowered benefit with an increase in concentration. Similarly, plotting the N/F ratio for efficacy and toxicity (Figure 3C, Scenario 1) exhibits values of ∼1 and <1, respectively, for all concentration ranges, suggesting the lowered toxicity of the nanomedicine compared to the free drug. Scenario 2, in Figure 3A, depicts an alternate situation with similar efficacy between the nanomedicine and the free drug but a higher nanomedicine toxicity profile. In this scenario, the nanomedicine E/T ratio is lower than that of the free drug (Figure 3B) while the N/F ratio for toxicity is >1 for all concentration ranges, suggesting a negative benefit with the nanomedicine.

Graphs obtained from hypothetical data showing varying efficacy and toxicity profiles. Toxicity profiles (red line) of nanomedicines and free drugs in a hypothetical system where the efficacy (black line) of the nanomedicine is equal to (A), greater than (D) or less than (G) that of the free drug. The resulting E/T (B, E, H) and N/F (C, F, I) ratios were calculated and plotted as X-Y graphs.
Unlike the E/T ratios, the N/F ratio does not indicate the overall benefit between a nanomedicine and a free drug, but it does provide an idea regarding the magnitude of the difference in efficacy or toxicity between a nanomedicine and a free drug. This information goes hand-in-hand with E/T ratios and needs to be considered in order to acquire an overall picture of how much of a benefit nanomedicine formulations may exert in comparison to free drugs.
In another hypothetical situation, where both the efficacy and toxicity of a nanomedicine is higher than that of a free drug (Figure 3D), the nanomedicine E/T ratio is clearly lower than the free drug E/T ratio (Figure 3E), despite an N/F efficacy ratio of >1 (Figure 3F) at all concentration ranges. This again reiterates the point that, despite the increased efficacy of nanomedicines, it is important to visualize and consider the toxicity profile of both nanomedicines and free drugs together with efficacy in order to get an overall idea regarding the possible benefit or drawback of nanomedicines. Similarly, when the data exhibits decreased efficacy as well as decreased toxicity (Figure 3G), it would be tempting to consider this as a beneficial effect of nanomedicine. However, the nanomedicine E/T ratio shows a clear reduction in benefit, while N/F ratios for both efficacy and toxicity are <1, suggesting that a nanomedicine with such an efficacy-toxicity profile provides no benefit. Overall, if the E/T ratio of the nanomedicine is greater than that of the free drug; and N/FEfficacy is greater than N/FToxicity, it would be reasonable to pursue further testing of the nanomedicine.
5. Case Study One: Comparative effect of vorinostat and the nanomedicinal form of vorinostat on an AML cell line
For our first case study, we chose data from an article by Chandran et al. [27] which includes a typical in vitro cell assay demonstrating cell proliferation activity in the presence of a free cancer drug (vorinostat) or else in the presence of nanocarrier-associated vorinostat. The original published figures have been slightly modified to suite the current article's purpose (Figure 4A,B). In the original article [27], the authors discuss the enhanced in vitro cytotoxic effect of nanoformulated-vorinostat on multiple acute myeloid leukaemia cell lines. For the purpose of this discussion, we focus specifically on the data describing the effect of the free and nanomedicinal forms of vorinostat on the cellular proliferation of Mv4-11 (an AML cell line) and healthy bone marrow-derived mononuclear cells. We chose this data specifically because they are typical for most in vitro studies in this field as well as to highlight the importance of the critical interpretation of in vitro proliferation assays in response to nano-drugs. We utilized the rate of the cellular proliferation of Mv4-11 cells as representative of drug efficacy, and the rate of cellular proliferation of normal human bone marrow-derived mononuclear cells (BMMCs) as representative of drug toxicity. In the original article, the authors observed a significant difference between the nano and free variants of a vorinostat-induced dose-dependent decrease in Mv4-11 proliferation (Figure 4A,B), while with normal BMCCs the authors observed only a non-significant trend with nano-vorinostat being mildly more anti-proliferative than free-vorinostat. We only considered the means for the following data and derived the anti-proliferation activity by taking the inverse of percent cell viability (Figure 4C,D). The E/T ratios for nano- and free-vorinostat and the N/F ratios for efficacy and toxicity were derived using values obtained for the treatment-induced anti-proliferation activity for each group.

Case Study One: Comparative analysis of a nanomedicine versus a free drug in an in vitro cancer cell line study. The cellular viability of the Mv4-11 cancer cell line (A) and normal bone marrow myeloid cells in response to increasing concentrations of nano- and free- vorinostat. The highlighted figures (A and B) have been adapted from a previously published article by Chandran et al. (2013), Figure 3H,I of the original manuscript. The E/T (C) and N/F (D) ratios were calculated from the cell viability data shown in (A) and (B).
The E/TNanovorinostat and E/TFree drug ratios showed that there was not much difference on the overall effect, and hence no major benefit between the nano and free drug formulations (Figure 4C). The N/FEfficacy ratio was >1 at lower concentrations, while the ratio was almost equal to 1 at higher concentrations, suggesting positive in vitro effects in terms of efficacy at lower concentrations (but seemingly no difference between nano- and free-vorinostat at higher concentrations). The N/FToxicity ratio hovered a little over one, which was in line with the slightly lower proliferation activity in normal BMCCs with nano-vorinostat (Figure 4D). Interestingly, the minimal toxicity trend with nano-vorinostat in BMCCs offset the ‘efficacy gains’ seen in Mv4-11 cells. It is worth mentioning that, even though the E/T and N/F ratios give a critical assessment regarding the combined benefit of nanomedicines, it is paramount that conclusions be drawn in relation to the whole context of the study. For example, in the same study, despite a seemingly benign effect of nano-vorinostat compared to free-vorinostat, under similar in vitro conditions the E/T ratio of the nano-vorinostat was 16.6 (at a 0.5 μM concentration) as compared to the E/T ratio of 1.2 of cytarabine and daunorubicin (a free drug combination that is currently in clinical use). This confirms the advantage of vorinostat against existing drugs, whereas the specific advantage of its nano-version may be evident only in the actual in vivo scenario where the free drug (having low aqueous solubility or sub-optimal pharmacokinetics) would face challenges in circulation. Therefore, more realistic in vivo models are essential for evaluating the efficacy of free drugs versus nanoformulations.
6. Case Study Two: Comparative effect of cisplatin the nanomedicinal form of in tumour-bearing mice
In a recent study by Kim et al. [28], the authors tested the anti-tumour efficacy of cisplatin-loaded nanomedicine (glycol-chitosan nanoparticles) in tumour-bearing mice. Among other groups, mice administered with free-cisplatin were used as a comparison group. Tumour size reduction was used as a measure of drug activity, while body weight and mortality were used to determine any adverse reaction to the treatment groups. We used published in vivo data from this study to calculate the N/FEfficacy & Toxicity ratio and the E/TNano & Free drug ratio and examined the comparative effect of the nanomedicine and free-cisplatin. We used plot digitizer software (The Free Software Foundation, MA, USA) to extract data from published figures and re-plotted the extracted data to calculate and display approximate efficacy and toxicity ratios. Since the data extracted from digitized graphs are approximate, we only considered the means of the extracted data while ignoring the standard deviation of each data point. We then converted the actual tumour volume sizes into percentiles (considering the maximum tumour size at the end of study in untreated controls as 100%) and calculated the anti-tumour growth activity of cisplatin-loaded nanomedicine and free-cisplatin. This was considered as a measure of efficacy. We followed similar steps for calculating the E/T and N/F ratios for relative body-weight change, which was considered as an indirect measure of toxicity. The cisplatin-loaded nanoparticulate system exhibited nanomedicine E/T ratios that were similar to the free-drug E/T ratios for the first five-to-six days, after which the nanomedicine shows a clear benefit compared to free-cisplatin (Figure 5A). Similarly, the cisplatin-loaded nanoparticulate system demonstrates an N/F efficacy ratio of >1 over a period of three weeks (Figure 5B), suggesting a clear benefit in efficacy (anti-tumour growth activity). Relative body-weight gain during the duration of treatment demonstrates a N/F ratio of one for the first two weeks of treatment, suggesting no beneficial effect for this duration. However, a clear benefit in relative weight-gain is observed with the nanomedicine after two weeks. Overall, this comparative analysis demonstrates a clear benefit for the use of the nanocarrier-associated cisplatin over free-cisplatin. Though a long-term experimental design would have provided additional translational value, the extrapolation of trends from this three-week study shows promise. The authors of this study reached similar conclusions, but what we intend to emphasize in this commentary is the need for a comparative examination of both efficacy and toxicity, together for free and nanocarrier associated drugs.
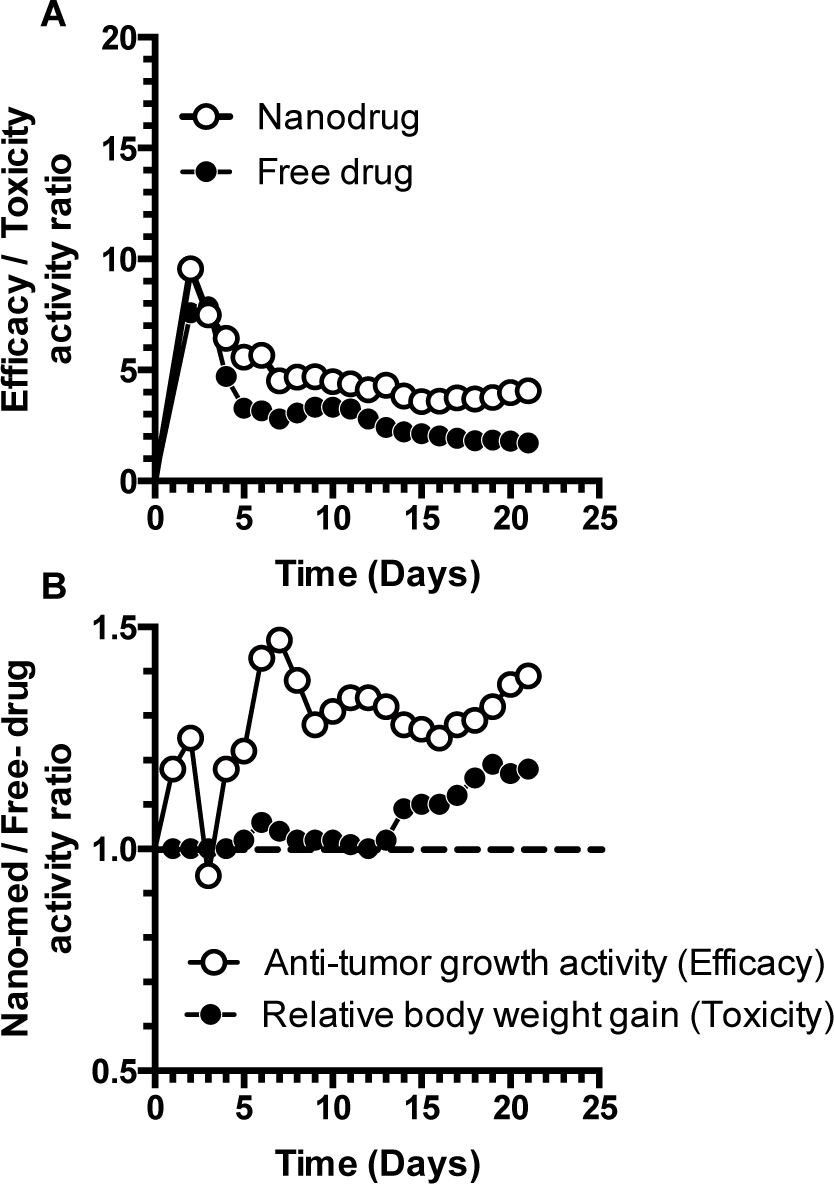
Case Study Two: Time-dependent beneficial effect of a nanomedicine over a free drug in a three-week in vivo study. The E/T (A) and N/F (B) ratios in tumour-bearing mice treated with cisplatin-loaded glycol chitosan nanomedicine and free-cisplatin. The data shown are approximate means, digitally extracted from Kim Jong-Ho et al. (2008), Figures 5A and 6 of the original manuscript.
7. Conclusion
Efficacy and toxicity form the two fundamental factors that determine the role of nanomedicines in clinical applications. With ever-increasing basic research in nanomedicines, one should only expect to see a few nanomedicines at the doorstep of clinical translation in coming years. While the translation of nanomedicines would provide an important alternative in the physician's arsenal, the process of translation faces many challenges. In this commentary, we have tried to highlight these challenges and we have provided examples of nanomedicines that showed promise in preclinical studies but eventually failed during clinical trials. We have underlined the lessons that can be learnt from such examples and stressed the concept of ‘quality by design’ in the development of nanomedicines. Using case studies, we have thus highlighted a simple but critical assessment tool to get a more comparative idea regarding the ‘benefit’ of using a nanomedicinal form of a drug. We would like to reiterate that what we have put forth is supplementary to current modes of assessment. Learning lessons from previous translational failures and adopting a strict and critical data assessment strategy at the most basic nanomedicinal research stage would go a long way towards successful human translation.
