Abstract
Iron oxide nanoparticles (IONPs) occupy a privileged position among magnetic nanomaterials with potential applications in medicine and biology. They have been widely used in preclinical experiments for imaging contrast enhancement, magnetic resonance, immunoassays, cell tracking, tissue repair, magnetic hyperthermia and drug delivery. Despite these promising results, their successful translation into a clinical setting is strongly dependent upon their physicochemical properties, toxicity and functionalization possibilities. Currently, IONPs-based medical applications are limited to the use of non-functionalized IONPs smaller than 100 nm, with overall narrow particle size distribution, so that the particles have uniform physical and chemical properties. However, the main entry of IONPs into the scene of medical application will surely arise from their functionalization possibilities that will provide them with the capacity to target specific cells within the body, and hence to play a role in the development of specific therapies. In this review, we offer an overview of their basic physicochemical design parameters, giving an account of the progress made in their functionalization and current clinical applications. We place special emphasis on past and present clinical trials.
Keywords
1. Introduction
Over the last decade, nanotechnology has become more relevant in many industrial sectors, including medicine. In general, the future projection and promotion of iron oxide nanoparticles (IONPs) as a point of reference among other magnetic nanomaterials for clinical applications relies chiefly on their biocompatibility in moderate doses, their relatively well-known iron metabolic pathways and their ability to be produced in a wide range of sizes and shapes with biofunctionalization potential. Additionally, the added advantage of magnetic actuation makes IONPs stand out from many other nanotechnology-based therapeutic and diagnostic approaches. In any case, IONPs have already passed through the preclinical stage to become a reality in clinical practice. In particular, they improve imaging-based diagnostics of immunological diseases, cardiovascular and cerebrovascular pathologies and cancer. They show great promise to serve as a cell tracking system in cell-based therapies, and to generate local temperature increases in the magnetic thermotherapy of solid tumours.
The purpose of this work is to review the basic parameters involved in the design of IONPs and their functionalization possibilities which, in the next few years, will bring into being a completely new generation of IONPs with selective targeting properties. Functionalized and multifunctionalized IONPs will become nanocarriers that will improve the selectivity of specific cells and, therefore, allow for the development of specific therapies. We describe their biochemical features, their cell internalization and
2. Basic physicochemical characteristics of IONPs
2.1 Size
This is arguably the most important parameter upon which any IONPs system must be designed. First, it profoundly affects the dynamics of the magnetic moments in magnetic NPs—also regarded as the magnetic
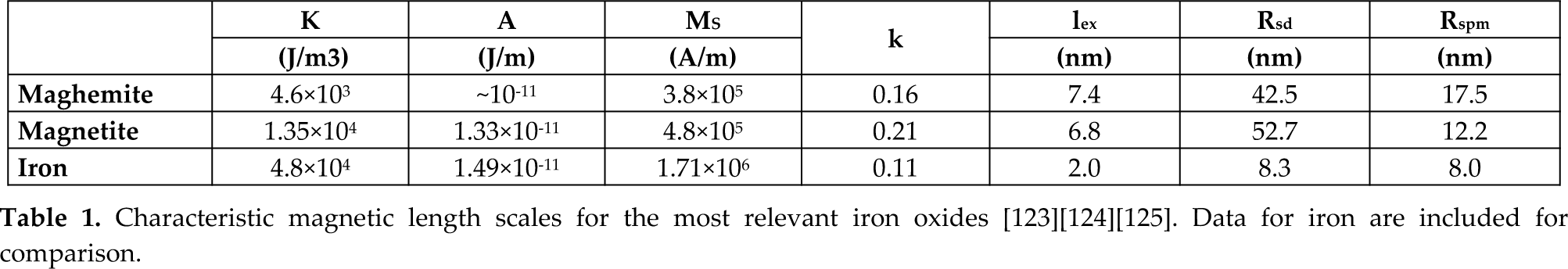
Two of the most relevant magnetic scale lengths that characterize magnetic NP systems are the superparamagnetic radius (
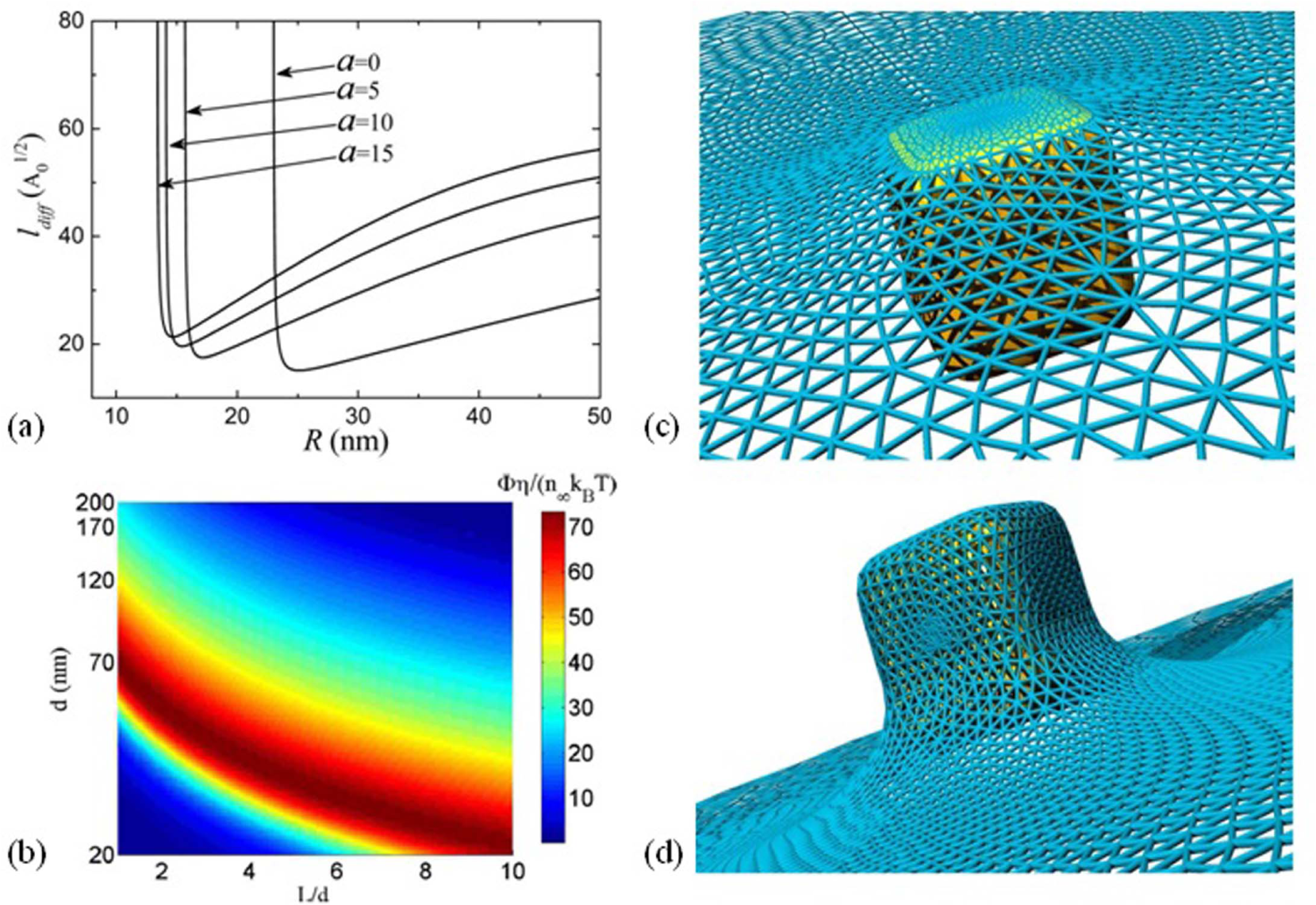
IONPs may be produced by a number of methods that allow for precise control over their size, shape and surface chemistry [3][4][5][6]. These parameters, along with others, can be used to classify IONPs for different purposes [7]. With reference to particle size, the researchers working on IONPs in medicine and biology—especially in the MRI community—use an informal classification divided into distinct ranges as follows: below 10 nm, very small superparamagnetic iron oxide NPs (VSPIONs); between 10 and 50 nm, ultrasmall superparamagnetic iron oxide NPs (USPIONs); and between 50 and 180 nm, superparamagnetic iron oxide NPs (SPIONs).
Although this classification might be useful only for the purpose of gathering some size ranges showing similar circulation and relaxational properties for specific applications, IONPs are persistently referred to in the literature as SPIONs, regardless of their true magnetic properties. This misuse of the term SPION is both inaccurate and confusing, as the lower-end value of the SPIONs' size range lies at the theoretical transition between superparamagnetic and single domain states for iron oxides (Table 1). Thus, most of the particle sizes encompassed within this range would in fact be single domain, not superparamagnetic. Interparticle interactions and surface effects further complicate this picture by shifting
From a biological point of view, IONPs can travel anywhere through the circulatory system of the human body, since the smallest capillaries are 4–6 μm wide [8]. IONPs under 100 nm are considered to be suitable for any application requiring tissue penetration, but those around 5 nm are more effective for tumour penetration [9]. Nonetheless, at the cellular level, NP size also influences the binding affinity of molecules, the uptake of additional particles into the cell, and the actual location within the cell. For a given NP shape, there is an optimal size for NP diffusion through the potential barrier characterizing the particle–cell interaction, as found by theoretical models [10][11]. Numerical calculations of these models indicate that the optimal NP size for a reasonably quick endocytosis is somewhere around 25 to 35 nm. Regardless of the particular considerations in each of the proposed models, the aforementioned optimum size range is given by the interplay between the kinetics and thermodynamics of the diffusion–absorption process of NPs relative to the thermal energy involved. Of particular note is that within the framework of the model proposed by Shi
Membrane deformation for (a) shallow wrapping and (b) deep wrapping of a cubic-shaped nanoparticle. The network of edges and triangles describes the membrane shape and has been used for the numerical calculation of the curvature energy. [Adapted with permission from S. Dasgupta,
2.2 Shape
Consider a magnetic particle as composed of many positive and negative poles; these will cancel each other, but there will still be free poles at the surface of the particle. These free poles create a magnetic field—the demagnetizing field (
Besides its contribution to the total anisotropy energy of magnetic particles, the role of the particle shape in the magnetic properties of NPs is linked to the stability of the single domain configuration. For example, disc-like NPs with relatively higher aspect ratios may show a closed spiral arrangement of their magnetic moments [13], called a
On the biological side, the shape of an NP determines the extent to which the NP will interact with membrane receptors and hence the degree of internalization. Shi
Schematic representation of the theoretical magnetic regimes (superparamagnetic, single domain, multidomain) expected for both magnetite and maghaemite, along with some relevant applications as a function of the particle size. (*) Magnetofection is a trademark of Christian Bergemann and Dr Christian Plank. (**) Refers to uncoated, single nanoparticles. The size ranges represented are approximate and comprise the most common cases.
2.3 Surface charge density
An additional degree of complexity linked to the biological activity of NPs is constituted by their effective superficial charge, which determines the type of active or passive functionalization through specific molecules and the relevant particle–cell interaction mechanisms, which is revisited in the following sections. In this regard, the magnetic properties of NPs would remain virtually unaffected by their surface charge, even though the coating used to tune the surface charge or the affinity for a particular target may introduce sizeable modifications, as discussed later. As a first approximation, bare IONPs possess a surface charge strongly influenced by the environment, involving the pH and the ionic strength of the medium. The point of zero charge (PZC), that is, the pH values for which the surface charge density is zero, for bare IONPs may span several pH units depending on the type and concentration of charged species in the medium. For magnetite, it has been reported to range between 3.9 and 9.9 [20], and for maghaemite the reported range is around 3.3–7.5 depending on the reaction conditions [21]. Besides the PZC, another surface charge parameter of interest is the zeta-potential (ζ) [22][23], whose sign and value give an idea of the colloidal stability of NP suspensions rather than focusing exclusively on the isolated particles. It is commonly accepted that a stable colloidal suspension of IONPs has approximately ζ < −30 mV or ζ > +30 mV. Nevertheless, bare IONPs as such are of very limited interest for biomedical applications, due to their non-specificity, tendency to aggregation and short circulation times, as well as their toxicity, since they may induce vacuole formation and other cell damage leading to cell death [24]. In order to manipulate the surface charge, improve their biocompatibility and/or add a specific functionality (see section 2), IONPs can be coated with a large number of molecules by virtue of their relatively wide PZC range discussed above.
The surface charge of both bare and coated IONPs influences the binding to cell membranes as the limiting step in their internalization process [25], irrespective of the shape. It is rather the sign of the charge that determines the internalization mechanism [26]. Membrane interaction can occur regardless of the charge of NPs, but it is well known that positively- or negatively-charged IONPs are internalized more rapidly than neutral ones. For instance,
A good example of how surface charge may affect the fate of similar IONPs formulations inside mammalians arises from the comparison of ferumoxides (ζ = −32 mV), ferumoxytol (ζ = −49 mV) and ferumoxtran (ζ = −2 to 0 mV). The latter has a thicker dextran layer [29] to avoid opsonization (protein adsorption) typically seen in very small particles and increase its circulation time, but in exchange is internalized to a lesser extent due to its almost neutral ζ. On the other hand, ferumoxides and ferumoxytol do not adsorb proteins and do not readily become opsonized, but are removed from circulation by the reticuloendothelial (RES) system due to their negative ζ.
3. Functionalization of IONPs
Functionalization of IONPs with biomolecules is a subject of great interest for two aspects related to their clinical applications. First, IONPs can be used as nanocarriers for drug delivery to tackle drug resistance of cancer cells and to increase local drug concentrations. Second, IONPs can be functionalized with specific targeting agents in order to improve the selectivity of specific cells, such as cancer cells, and therefore improve the selectivity, reduce side effects and increase local concentrations of drugs and/or IONPs in the targeted tissue. All these strategies are involved in the development of new generations of functionalized and multifunctionalized NPs for biomedical applications that introduce selective targeting properties to such nanocarriers.
The effect of targeting is strongly related to the enhancement of cellular uptake. Indeed, targeting strategies involve the recognition and binding to membrane receptors overexpressed on targeted cells, which changes the cellular uptake pathway and its efficiency. Several functionalization and targeting strategies have been developed in parallel with the latest advances in the discovery of new biomarkers specific to the different types of cells considered (e.g., cancer cells, cancer stem cells, etc.).
Currently, no clinical trials are in progress concerning the use of functionalized IONPs in humans. However, almost all the scientific publications in the field mention the potential biomedical and clinical applications of functionalized IONPs considering the promising
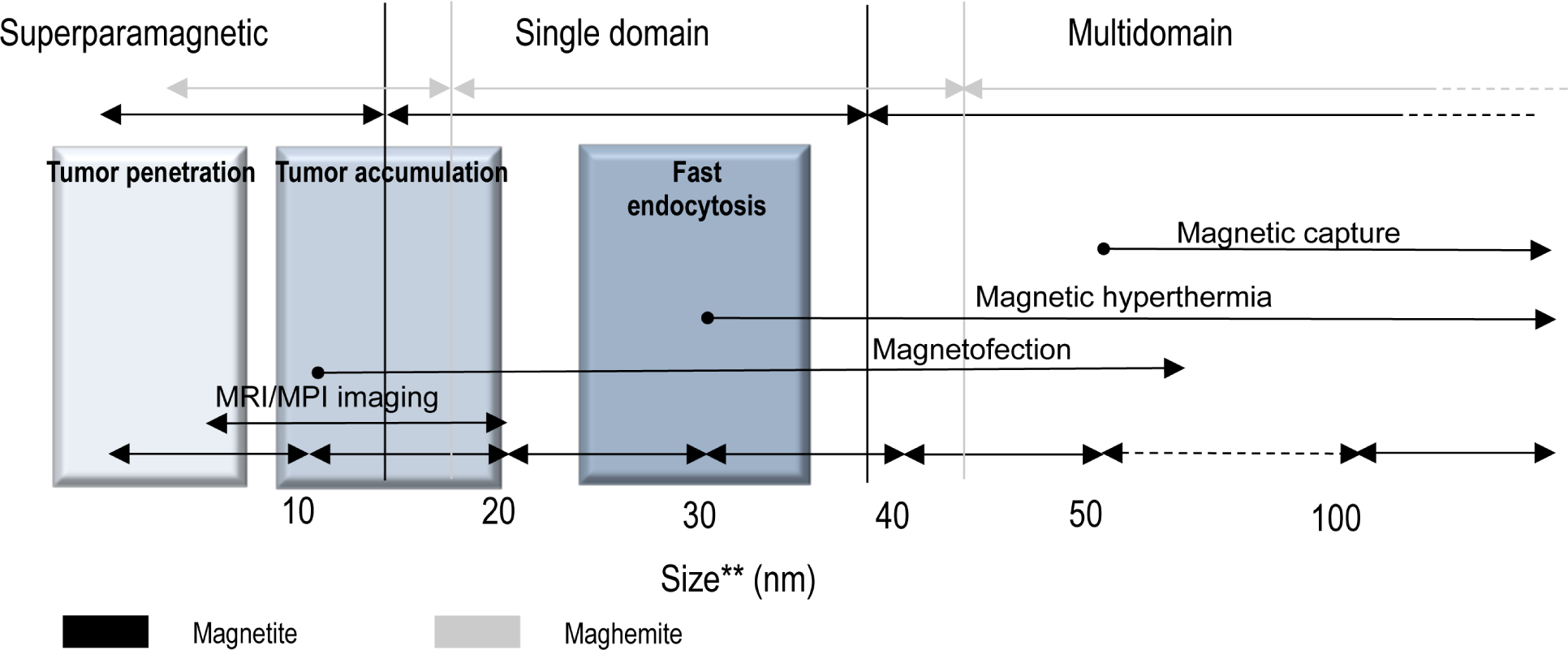
Here, we present an overview of the different molecules that are currently used for the functionalization of IONPs, highlighting some relevant examples and their potential in clinics (Table 2 and Figure 3).
Examples of biomolecules approved for clinical applications or already in clinical trials that are used in IONPs functionalization research studies
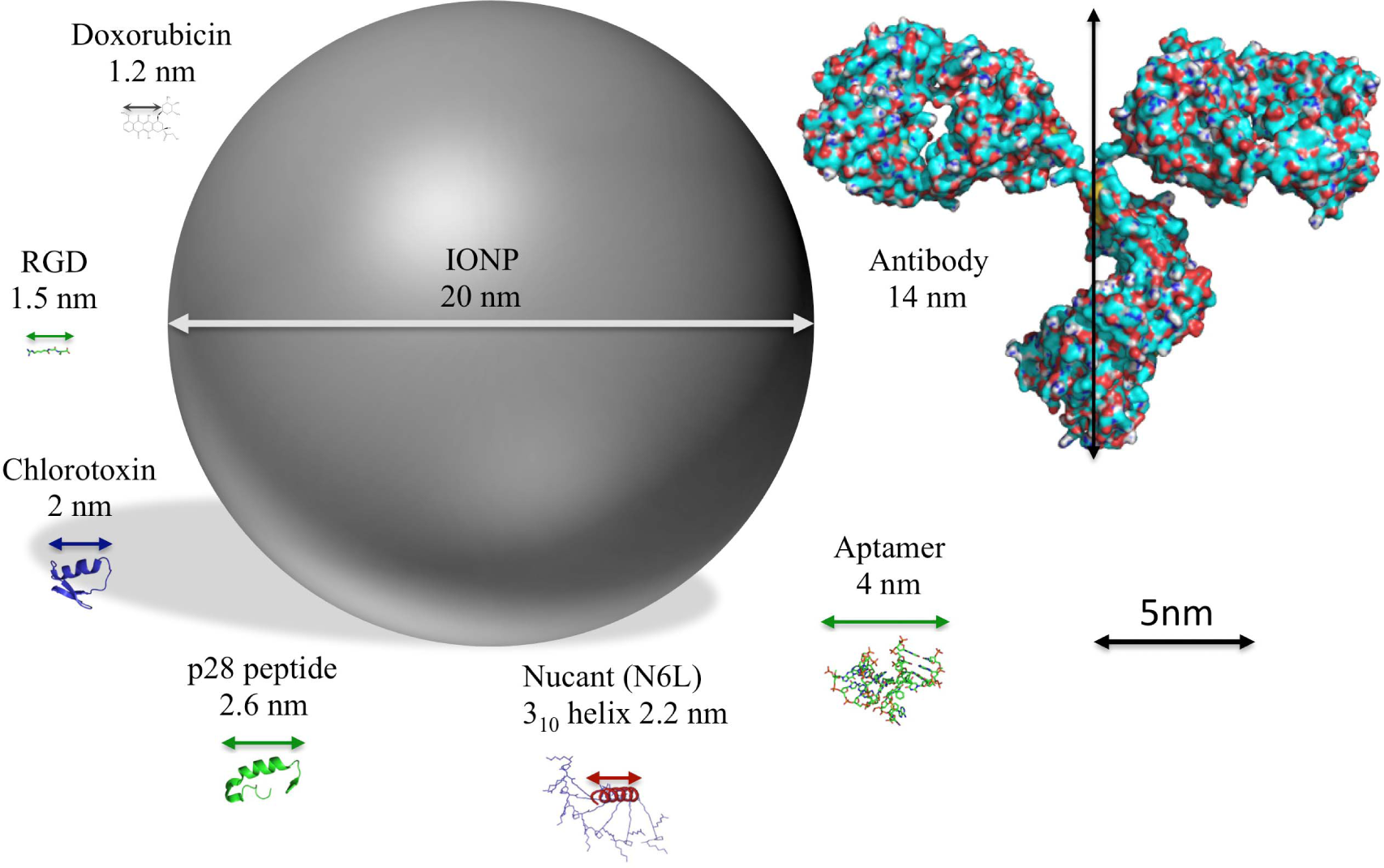
Functionalization of IONPs Schematic representation to scale of IONPs and the structure of different molecules used for their functionalization Structures represented: IONPs doxorubicin RGD peptide (PDB ID: 3VI4) chlorotoxin (PDB ID: 1CHL) azurin p28 peptide (PDB ID: 4AZU) Nucant (N6L) [52] aptamer (PDB ID 4HQU) and antibody (PDB ID: 1IGT).
3.1 Cell-penetrating peptides (CPPs)
Cell-penetrating peptides (CPPs), also known as protein transduction domains (PTDs) are up to 30-amino acid amphiphilic peptides that can be internalized by cells using mechanisms that require no energy and that can be receptor-mediated or not. Among the large variety of CPPs sequences, the presence of positively-charged amino acids and the amphiphilic character are the two common characteristics [30]. First used for intracellular delivery of macromolecules [31], CPPs are very interesting as drug and NPs intracellular delivery carriers. IONPs functionalized with different amounts of Tat peptide showed an exponential increase of cell uptake by increasing the number of CPPs grafted onto IONPs, concluding in a multivalent effect on the internalization [32]. The important parameters for the entrance of CPPs are positive charge and amphiphilic character. Martin
Additionally, newly identified tumour-homing CPPs have been discovered, expanding the use of CPPs for selective tumour targeting [35][36]. The only cell-penetrating peptide that has been tested in clinical trials is the so-called p28 peptide, which is derived from azurin protein and induces an increase of p53 and consequently cell death by apoptosis [37]. A completed phase I study (NCT00914914) and an ongoing phase I study (NCT01975116) have been carried out with the p28 peptide used as an anticancer drug for different types of tumours [38]. As far as we know, this p28 peptide has never been tested for IONP functionalization or for targeting strategies.
3.2 Antibodies and other proteins
Antibodies are complex protein molecules that bind to specific antigens with high affinity. Antibodies can be developed to bind to almost any given antigen, for example, specific receptors expressed on the surface of cells, and are therefore very useful tools to target disease biomarkers. Apart from their use as anticancer agents [39], antibodies have been used in IONP functionalization for their targeting properties toward cancer cells for a decade. As shown in the preliminary and encouraging study of Kang
Numerous research groups are interested in the functionalization of IONPs with trastuzumab, a commercial and approved antibody for breast cancer treatment [41], for the development of targeted therapeutic strategies. Most of the studies confirmed the selectivity of the antibody-functionalized IONPs toward cell lines overexpressing the Her2 receptor, depending on receptor expression level [42][43]. Vigor
Additionally, other proteins are being used to functionalize IONPs and improve the cellular uptake and cell targeting. The example of transferrin-functionalized IONPs to target cancer's overexpression of transferring receptors has been recently published by Piraux
3.3 Targeting peptides
Apart from macromolecules such as antibodies and CPPs, smaller peptides also interact with specific receptors in the cell membrane and increase the cellular uptake of the functionalized IONPs.
Arginine-glycine-aspartic acid, also known as RGD peptide, binds to the integrin receptor (αvβ3) which is overexpressed in several cancer cell lines and is commonly used as a targeting agent. Several works show that the functionalization of IONPs with RGD peptide (in linear or cyclic conformation) induces a significant increase in the cell uptake, compared to bare IONPs, specifically for αvβ3 positive cell lines as in some cancer cell lines (BT-20 and HeLa) and no significant changes in αvβ3 negative cells (9L, MCF-7, or U87MG cells) [46][47][48]. Similarly, chlorotoxin (CTX) is a 36-amino acid peptide that has a high selectivity and binding affinity for gliomas and neuroectodermal tumours [49]. Sun
3.4. Aptamers
Aptamers are single-stranded oligonucleotides that bind to their specific targets with high efficiency. Aptamers have been used for the targeting and specific cellular uptake of IONPs, as they present several advantages, such as their easy and reproducible synthesis, their good stability and their lack of immune response. Chen
3.5 Carbohydrates
Functionalization of IONPs with carbohydrates can improve cellular uptake by two different strategies: (1) providing higher hydrophilicity and biocompatibility and thus increasing lifetime in the bloodstream; and (2) the interaction with specific sugar receptors on the membrane of certain cell lines, including cancer cells [54]. Moros
3.6 Folic acid
Folic acid, also known as Vitamin B9, is a natural product with an important role in DNA and RNA synthesis. Over 20 years, folate receptors have been shown to be overexpressed on several cancer cell lines and can therefore be used for targeting cancer cells and activated macrophages [57][58]. Folic acid has already been approved for several clinical applications. Clinical trials are in progress for drug-folate conjugate use in cancer diagnosis (NCT00003763) and therapy (NCT00291785, NCT00485563, NCT00441870). Folic acid has been widely used for 15 years in the development of NP targeting strategies for biomedical applications, including IONPs. We highlight the work of Fan
3.7 Drugs
The functionalization of IONPs with drugs for their application as nanocarriers is a wide and much studied topic [60]. Indeed, the major portion of the drugs used in combination with IONPs at the research level are already available in clinics, such as doxorubicin, gemcitabine and SN38, and are also involved in several clinical trials of drug-targeting agent conjugates. Currently, there are some promising examples of doxorubicin conjugated with antibodies now in clinical trials (NCT00051584), and the use of liposomal formulations for drug delivery (NCT01227941).
The multifunctionalization of IONPs by targeting agents and drugs, and its acceptance in clinical processes, might be a breakthrough in the development of specific therapies. The presence of targeting moieties such as those discussed above will increase the localization of IONPs in the diseased tissue, therefore augmenting drug delivery and local drug concentration. Thus, research is currently making efforts towards the multifunctionalization of IONPs. Different methodologies need to be developed to ensure controlled multifunctionalization, and preserve the stability and biological activities of active molecules attached to the particles. Additionally, with respect to the clinical application of these nanostructures, functionalization efforts are required to increase the blood half-life of the IONPs, minimizing off-target accumulation and immunological response.
4. Toxicological profiles and potential interference with biological functions of cells
The above-mentioned parameters have a tremendous influence on cell internalization as well as
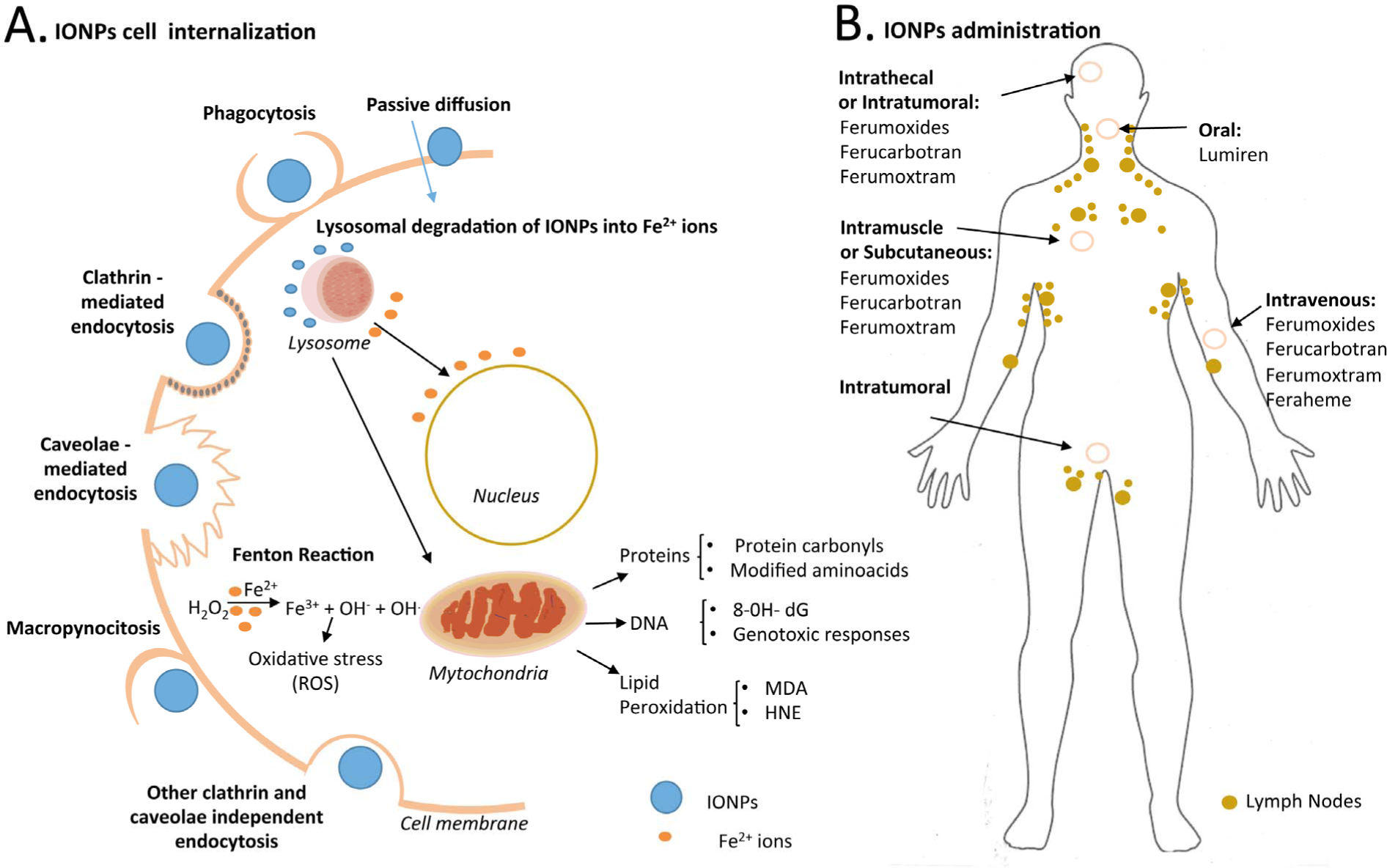
A. Different internalization pathways of IONPs in mammalian cells. Possible mechanisms of uptake including macropinocytosis, caveolae- and clathrin-mediated endocytosis, phagocytosis, passive diffusion and other endocytosis pathways. After internalization, IONPs can produce cytotoxicity effects via a Fenton reaction. Hydroxyl radicals generated could damage DNA, proteins or lipids (8-OH-dG = 8 hydroxydeoxyguanosine, MDA = malondialdehyde, HNE = 4-hydroxy-2-nonenal), triggering genotoxicity. B. IONP administration in the human body, such as intrathecal, intratumoural, intravenous and intramuscular or subcutaneous methods.
4.1 Effects on cellular physiology
4.1.1 Mitochondrial stress
One factor that can contribute to nanotoxicity is the size of the NPs. Smaller NPs have a greater reactive surface area than larger ones, are more chemically reactive, and produce greater numbers of reactive oxygen species (ROS) that include free radicals [63]. SPIONs also can induce (geno-)toxicity via generation of ROS. SPIONs are presumably degraded into iron ions within the lysosomes by hydrolysing enzymes effective at low pH [64]. This “free iron” can potentially cross the nuclear or mitochondrial membrane and, in the latter case, the free iron in the form of ferrous ions (Fe2+) can react with hydrogen peroxide and oxygen produced by the mitochondria to produce highly reactive hydroxyl radicals and ferric ions (Fe3+) via the Fenton reaction (Figure 3a). Therefore, hydroxyl radicals generated by the free iron could damage DNA, proteins, polysaccharides and lipids
4.1.2 Effects on actin cytoskeleton architecture
Intracellular accumulation of NPs can have profound effects on cell morphology. High levels of internalized particles can provoke cellular stress, inducing changes in the architecture of the actin cytoskeleton. An altered actin network displaying actin stress fibres can lead to a reduced proliferative capacity and cell spreading. It can also influence the migration and differentiation of stem cells.
In the case of IONPs, it has been reported to interfere with actin and tubulin structures, inducing cell retraction, rounding and deposition of massive dense filament matters adjacent to the nucleus and vacuoles in the cytoplasm [68].
4.2 Factors that can contribute to non-toxicity
Size is clearly a key factor in determining the potential toxicity of a particle. However, it is not the only important factor. Other properties of nanomaterials that influence toxicity include chemical composition, shape, surface structure, surface charge, aggregation and solubility [63]. A study investigating the effect of different surface coatings on cell behaviour and morphology has shown that dextranmagnetite (Fe3O4) NPs result in cell death and reduced proliferation similar to that caused by uncoated IONPs [24]. Other authors have reported the formation of gas vesicles after exposure to the uncoated NPs, resulting in altered protein functions and changes in ionic equilibrium within the cells, which also promotes toxicity [69].
4.3 In vitro and in vivo toxicity of nanoparticles in 2D vs. 3D cell culture
Common 2D cell cultures do not adequately represent the functions of 3D tissues that have extensive cell–cell and cell–matrix interactions, as well as markedly different diffusion/transport conditions. Hence, testing cytotoxicity in 2D cultures may not accurately reflect the actual toxicity of NPs and other nanostructures in the body [70]. For instance, recent studies on toxicity testing of magnetic NPs, using
When IONPs enter the body, absorption can occur through interaction with biological components such as proteins and cells; afterwards, they can distribute into various organs where they may remain in the same nanostructure or become metabolized [73]. A systematic and thorough quantitative analysis of the pharmacokinetics (i.e., absorption, distribution, metabolism and excretion) of IONPs can lead to improvements in design of biocompatible IONPs, a better understanding of NPs' non-specificity toward tissues and cell types, and assessments of basic distribution and clearance that serve as the basis to understand their activity and potential toxicity [74].
The surface of IONPs is rapidly covered by selective sets of blood plasma proteins after injection. Small IONPs (< 10 nm) are usually rapidly removed through extravasation and renal clearance, whereas large IONPs (> 200 nm) are sequestered by the spleen via mechanical filtration [75]. The typical final biodistribution of IONPs is 80–90 in liver, 5–8% in spleen and 1–2% in bone marrow, due to the high number of macrophages contained in these organs [61]. IONPs were also found to be distributed in the brain, liver, spleen and lungs after their inhalation, demonstrating their ability to cross the blood–brain barrier [76] (Figure 4A).
5. Current clinical applications
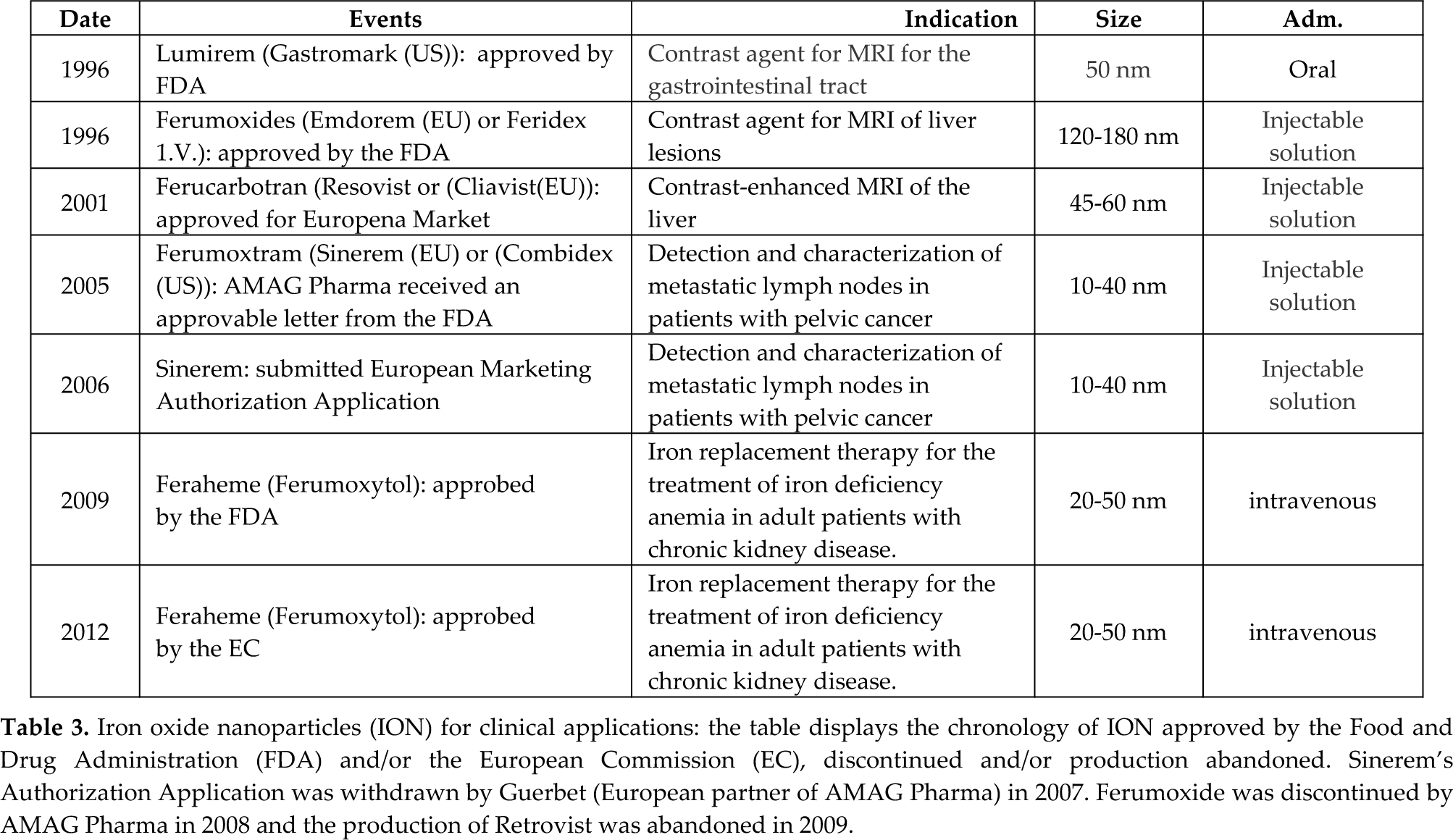
In December 1996, the United States Food and Drug Administration (FDA) approved GastroMARK (AMAG Pharmaceuticals), an aqueous suspension of silicone coated, superparamagnetic iron oxide NPs, intended for oral administration, as a magnetic resonance (MR) imaging contrast medium to enhance the delineation of the bowel to distinguish it from organs and tissues that are adjacent to it in the upper regions of the gastrointestinal tract (www.fda.gov). The approval of GastroMARK, together with a number of preclinical studies supporting the potential use of SPIONs in different medical applications, contributed significantly to expanding clinical research in nanomedicine. A number of new SPION-derived products were developed, some of which were also approved by the FDA and the European Commission (EC) for clinical use. Additionally, the increasing number of clinical trials using these new NPs expanded their potential medical applications. Sixteen years later, just two SPION-derived products are approved by the FDA and commercially available: GastroMARK and ferumoxytol (Table 3). In the following paragraphs, we will describe the current clinical applications of SPION-derived products, as well as the clinical trials testing them for promising applications such as cell tracking and hyperthermia.
Iron oxide nanoparticles (ION) for clinical applications: the table displays the chronology of ION approved by the Food and Drug Administration (FDA) and/or the European Commission (EC), discontinued and/or production abandoned. Sinerem's Authorization Application was withdrawn by Guerbet (European partner of AMAG Pharma) in 2007. Ferumoxide was discontinued by AMAG Pharma in 2008 and the production of Retrovist was abandoned in 2009.
5.1 Imaging
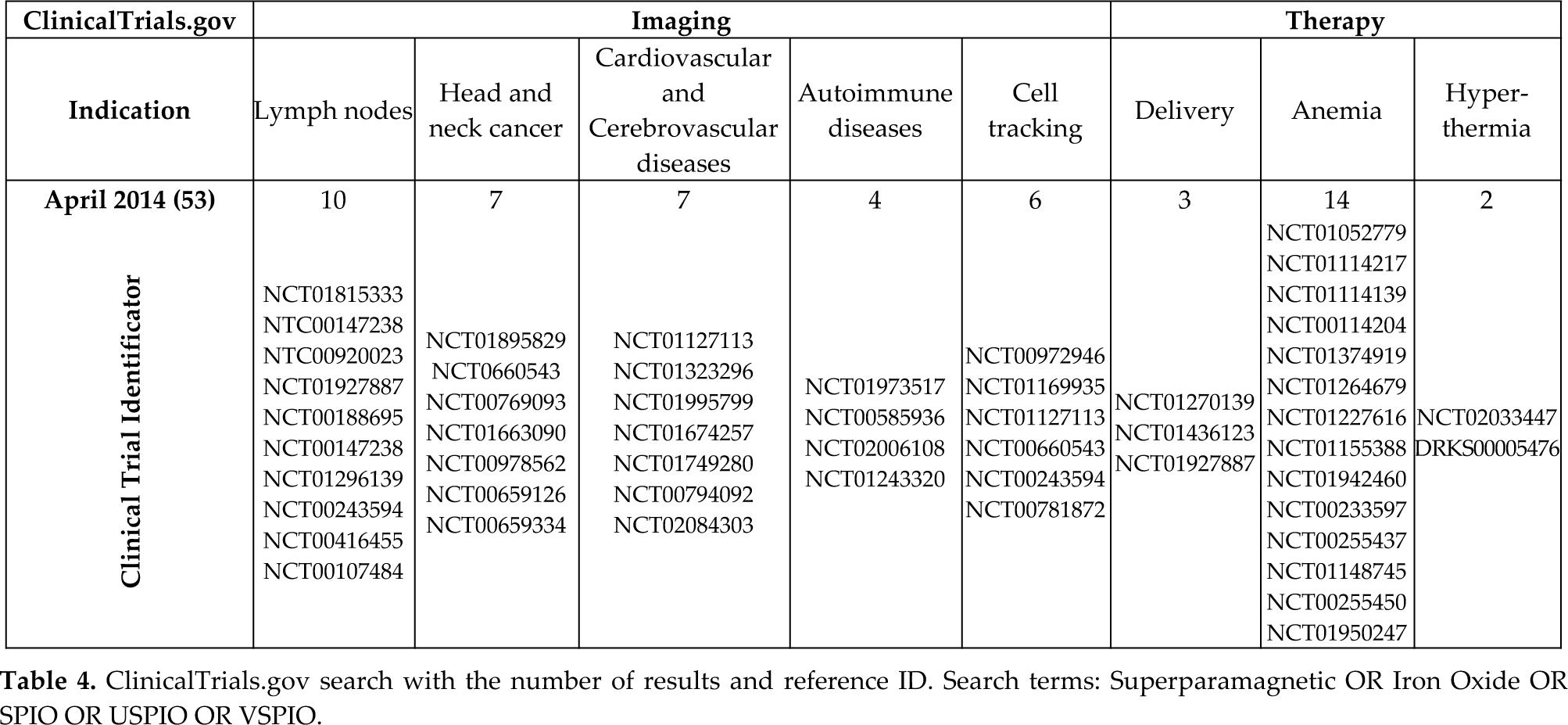
Clinic imaging technologies rely on the use in MRI, optical imaging/MRI and MRI/PEP. SPIONs function as a negative contrast agent, decreasing T2 signals and thus the signal intensity. They cause a magnetic field gradient that affects the surrounding protons of water molecules, disrupting the homogeneity of the magnetic field, which can be observed by MRI [77]. Additionally, SPIONs display lymphotropic properties, as phagocytes of the RES internalize them, causing local changes in magnetic properties. These features make the SPIONs an excellent alternative to complement current contrast agents in MRI, providing higher accuracy in some conditions such as autoimmune diseases, cardiovascular and cerebrovascular pathologies, and cancer (Table 4). Additionally, the low toxicity both
ClinicalTrials.gov search with the number of results and reference ID. Search terms: Superparamagnetic OR Iron Oxide OR SPIO OR USPIO OR VSPIO.
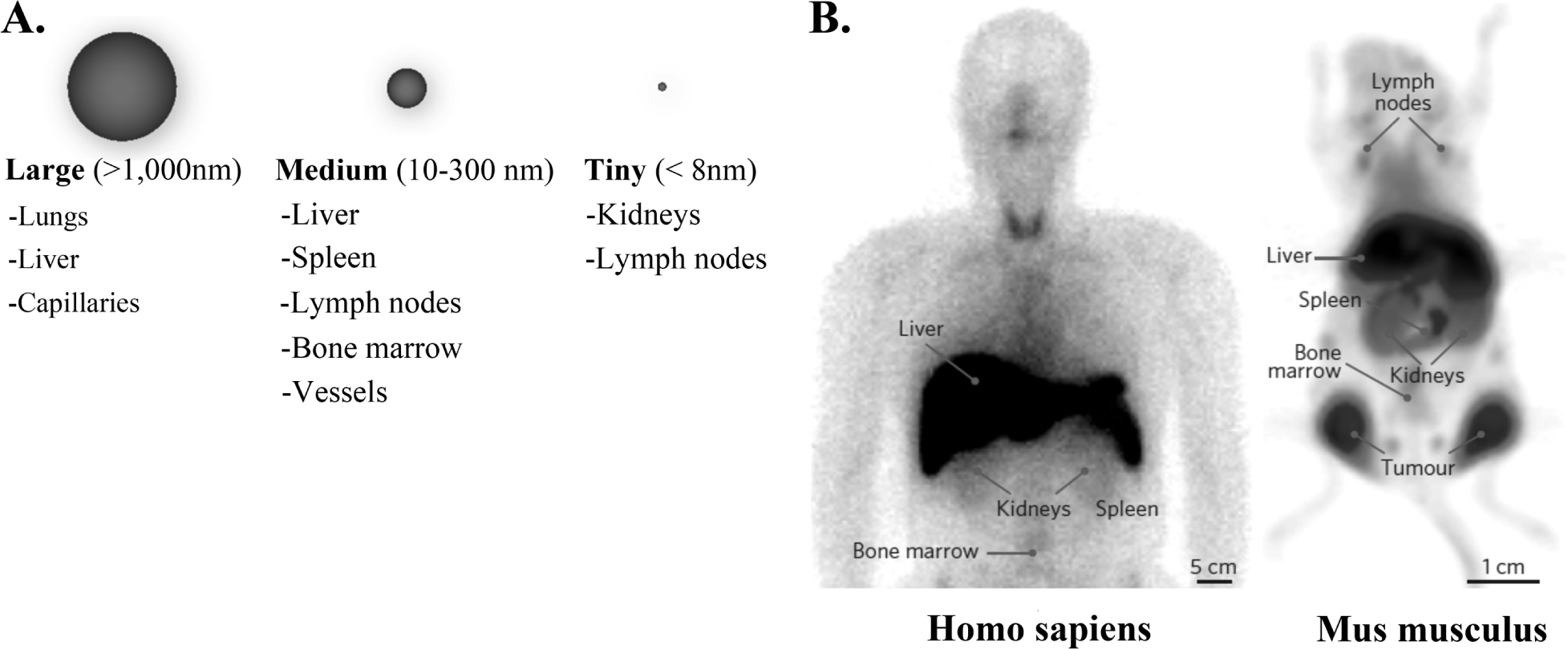
Organ distribution of systemically injected nanoparticles. a. Common organ distribution is shown as a function of particle size. b. Example of the 99mTc-labeled graft copolymer used in a human patient and 89Zr-labeled cross-linked dextran nanoparticles used in a mouse model. This figure was reproduced with permission from
5.1.1 Diagnostic tools
5.1.1.1 Diagnosis in immunological diseases
Autoimmune disease and inflammation processes are characterized by the activation of the endothelium that eventually allows for the extravasation of macrophages from the bloodstream into the inflamed tissue. SPIONs significantly enhance the signal of these events when analysed by MRI, as they are quickly captured by macrophages. In the last few years, a number of authors have taken advantage of this observation and carried out different clinical assays to test the value of SPIONs as a negative contrast in a variety of autoimmune and inflammatory conditions. Oral USPION (ferumoxytol) improved the detection of Crohn's disease [79] and the prognosis of active ulcerative colitis [80][81]. Using ferumoxtran, a significant improvement was observed in a small cohort of Type 1A diabetes patients (NCT00585936) in the diagnosis of pancreatic islet inflammation when compared to T1-gadolinium [82]. In patients with multiple sclerosis, USPION-based MRI showed that iron oxide NPs labelled larger and better defined areas, allowing the detection of what has been referred to as normal-appearing white matter that is histologically affected (NCT01973517) [83][84]. These outstanding results have encouraged physicians and researchers to continue testing the SPION-based MRI in both the previous and new immunological and inflammatory conditions. At least two more clinical trials were running at the time of writing this review (Table 4).
5.1.1.2 Diagnosis in cardiovascular and cerebrovascular pathologies
The use of SPIONs for T2 and T2-weighted cardiovascular magnetic resonance imaging (CMR) has been successfully validated to detect myocardial oedema and myocardial haemorrhage [85][86]. In a more recent trial, Vilmaz
5.1.1.3 Diagnosis in cancer
In healthy liver, spleen and lymph node tissue, IONPs decrease the MR signal intensity as phagocytic cells uptake the nanoparticles, while malignant tissue generally fails to uptake IONPs and appears bright relative to the surrounding tissue [90][91]. This observation emphasizes the IONP-enhanced MRI as an extremely promising imaging technique for the preoperative detection of metastatic disease in lymph nodes. In 2003, Harisinghani
5.1.2 Cell tracking in cell transplantation-mediated therapies
Cell therapy relies on the delivery of cells to the target site. Monitoring and tracking these cells to ensure tissue delivery and engraftment become a key issue in stabilizing clinical safety and therapeutic efficacy. In this sense, detection by MRI of IONP-labelled cells may be one of the most promising approaches for a short-term evaluation [100]. Although the FDA and EC have not yet approved their use for
Cancer immunotherapy approaches can also benefit from SPION labelling technology. Dendritic cell (DC) vaccines have been used to induce tumour-specific cytotoxic T cells. To be successful, injected DCs need to migrate to the lymph nodes (LNs) where they can stimulate effector T cells. DCs have previously been labelled with radionuclides for scintigraphic imaging, which is the only clinical cellular imaging modality approved by the FDA [102][103]. deVris
5.2 Therapy
5.2.1 Delivery
Iron oxide NPs have been successfully used as a carrier for bioactive molecules in a number of conditions both
5.2.2 Hyperthermia
Thermotherapy of solid tumours is one of the most promising applications of SPIONs, either alone or in combination with adjuvant treatments, such as chemotherapy or radiation. The principle of applying IONPs to hyperthermia therapy involves the administration of an IONPs fluid within the tumour, followed by the application of an alternating magnetic field. These nanoparticles achieve high temperatures, promoting warming of the region. Temperature elevation in the range of 41–46
Clinical studies for the application of thermotherapy using IONPs in humans were initiated in 2007 on prostate cancer and glioblastoma patients (NCT02033447 and DRKS00005476). Johannsen
In parallel, Maier-Hauff
More recently, Matsumine
Although these results are promising, the development of therapies based on magnetic hyperthermia is still in its very early stages. In the next few years, many biological and technical advances need to be achieved before this new therapy becomes part of the standard of care for some cancers. Questions such as how to reach homogenous distribution of IONPs and therapeutic temperature, and how to minimize peritumoural tissue damage, need to be addressed. In this regard, a new phase 0 clinical trial to investigate the magnetic nanoparticle thermo-ablation-retention and maintenance in the prostate is being performed by the University College London Hospitals (NCT 02033447). In this trial, the authors will test whether the magnetic nanoparticles actually stay where they are injected or move to sensitive structures around the prostate, which may lead to undesirable side effects. Finally, MagForce is conducting a new open-label, randomized clinical trial employing magnetic hyperthermia to treat glioblastoma multiforme patients (DRKS00005476). The purpose of this trial, expected to enrol up to 285 patients, is to test the feasibility and safety of using magnetic hyperthermia as a stand-alone therapy and in combination with radiotherapy using the commercial IONPs NanoTherm®. The results will help magnetic hyperthermia advance in the clinical setting.
6. Perspectives
Bare IONPs are currently employed in an increasing number of clinical trials. In the next decade, we will see them settle into clinical practice for imaging-based diagnosis for a wide range of diseases. One of the main entries of IONPs into the scene of medical application will surely come from their functionalization possibilities. These will provide them with the capacity to target specific cells within the body, and hence develop specific therapies and diagnostic tools. The main advantages of using nanocarriers for specific drug delivery are the reduction of side effects, potential drug resistance and increased drug payload. As unique targeted nanocarriers can deliver hundreds of small drug molecules, clinical practices shall be adapted to incorporate these new strategies. They should include treatment protocols personalized by choosing appropriate formulations with defined targeting moieties and drug loads. The multifunctionalization of IONPs opens the door to multimodal therapeutic approaches by combining the dual effects of magnetic hyperthermia and chemotherapy, using multiple drugs in the same IONP formulation. Additionally, functionalization of IONPs will allow researchers to tune and optimize IONPs' biocompatibility, blood half-life and immunogenicity, key parameters required to successfully bring these innovative nanotechnology-based tools from research laboratories to clinics. Overall, the multifunctionalization of IONPs is a critical step. It is most likely that it will be broadly implemented in the near future for new nanoformulations for both diagnosis and therapy.
In the particular case of IONPs for magnetic hyperthermia, a number of fundamental and practical aspects still require more research. One of these concerns the magnetic and mechanical behaviour of IONPs inside cells: specifically, we refer to lysosomes under dynamic external fields. Although a few theoretical studies addressing this matter have appeared [118], we require specific experimental approaches to confirm their predictions. Additionally, more work needs to be done to determine the actual heating mechanisms under common frequency and field amplitude conditions, and to evaluate the involvement of any other physical phenomena. In this sense, the results from a very recent publication exploring possible damage to the lysosomal membrane by a remotely induced torque over IONPs are particularly encouraging [119]. The role of phonon-mediated heating and its coupling to those biomolecules in the surroundings, or directly linked to IONPs, is now receiving more attention [120]. These studies will enable us to discern whether or not a sizeable macroscopic temperature change is required to have a therapeutic effect. If not, temperature changes on the micro- or nano-scale could be enough to induce cell death or, at least, to sensitize cancer cells to other treatments (chemo/radiotherapy). Finally, the possibility of having magnetic hyperthermia in an MRI machine has been proposed [121–122], but no clear steps have been taken to further develop this appealing idea.
7. Acknowledgments
We are especially grateful to the Health Research Fund (Fondo de Investigaciones Sanitarias, FIS) from the Carlos III Health Institute (PI10/01069 and CP11/00147 (AAS) and PI11/00537 (CBI)). AAS is a recipient of the Miguel Servet Program from Carlos III Health Institute (MS11/00147). DO is grateful for the support received through the AMAROUT-II Marie Curie Action under the European Commission's FP7 PEOPLE-COFUND program and the Research Fund grant from the Royal Society of Chemistry. ALC is grateful for the financial support received through the EU-FP7 MULTIFUN project (no. 262943), the Spanish Ministerio de Economía y Competitividad (BIO2012–34835), the European Commission International Reintegration Grant (IRG-246688) and the Marie Curie COFUND “AMAROUT-Europe” Programme.
8. Compliance with ethical research standards
Authors have no conflict of interest to declare. No part of this study was performed on any human or animal subject.