Abstract
Nature has had millions of years to optimize photonic crystals - an endeavour mankind only really began in the 1980s. Often, we attempt to mimic and expand upon nature's designs in creating photonic structures that meet our technology-driven needs. While this strategy can be fruitful in fabricating novel architectures, one has to keep in mind that nature designed and optimized these structures for specific applications (e.g., colouration, camouflaging, signalling), but certainly not for use in photonic chips and optical circuits. To take full advantage of biological structures as blueprints for nanotechnology, it is important to understand the purpose and development of natural structural colours. In this review, we will discuss important aspects of the design, formation and evolution of the structures embedded in beetle exoskeletons that are responsible for their striking colouration. In particular, we will focus on the purpose of structural colours for camouflaging, mimicry and signalling. We will discuss their evolutionary and ecological development and compare the development of beetles with and without structural colours. Examples of non-colour-related structural functionalities will also be introduced and briefly discussed. Finally, a brief overview of nature's synthesis strategies for these highly evolved structures will be given, with particular focus on membrane assembly.
1. Introduction
The origins of most man-made colours are based on molecular pigments, while nature has developed a different type of colouration - one not based on molecules but on supramolecular structures. Such structural colours can be found in insects, birds and marine animals.
1
For example, the origin of the deep blue wings of the

a) Photograph of the butterfly
Such structural colours are interesting examples of nature's ability to create function by moulding and shaping
While there are many examples of materials with interesting structure-function relations, this review article will focus on structural colours - or ‘photonic crystals’, as their synthetic counterparts are termed. The aim of this article is to provide insights into structural colours in biology and - by comparing biological photonics with currently available man-made photonics - to provide inspiration for the design of novel optical components and devices.
2. Photonic Structures in Biology and Technology
The purpose and use of photonic structures varies strongly for biological and technological applications, making it sometimes difficult to compare and adapt the structure-function properties of biological photonic structures for technological applications. Photonic structures in biology are mainly produced to create colouration for camouflage, signalling or disguise.1,12 Since light conditions can vary strongly for different habitats, biological photonic structures were optimized to create specific reflection colours under various illumination conditions, from glaring sunlight to the diffuse and dim lighting on forest floors. In addition, these structures are in general designed to fulfil given functions in an organism and are thus integrated into larger body parts, such as feathers, wings, hair, skin, bones, exoskeletons, etc. (Figure 2). In contrast, to create functional devices for technology applications, photonic structures must be integrated into optical circuits and photonic chips - environmental conditions very different from biological ones. Moreover, the purpose of photonic structures in modern devices is not to produce colours but to guide, store and amplify light.13–15 These structures thus need to be optimized in very different ways than is the case with their biological counterparts.

Examples of structural colours in nature. Colours arise from photonic structures in a) the wings of the butterfly
A central goal with many technological photonic crystals is to create a complete - or omni-directional - photonic band gap (i.e., a frequency range for which the propagation of light is classically forbidden in all directions). The concept of photonic crystals and band gaps were independently proposed by Yablonovitch 16 and John 17 in 1987 with the goal of controlling the radiative properties of materials and induce photon localization, respectively. The basic idea of a photonic crystal is to fabricate periodic dielectric composites with periodicities on the order of optical wavelengths. In such structures, the behaviour of light is governed by Bragg diffraction, resulting in band structure properties similar to electronic semiconductors. Unfortunately, calculations and band structure modelling have revealed a complete photonic band gap that forms only under very strict structural and dielectric conditions.18–20 Regardless of the type of lattice structure, the high-dielectric component of the photonic crystal needs to have a refractive index exceeding 2 (assuming that air is the low-dielectric component), while being optically transparent in and around the band gap wavelength region; for example, for an optimized diamond lattice the minimum refractive index is 2.0 in order to open a complete band gap, whereas for a close-packed face-centred cubic lattice of air spheres (inverse opal) the surrounding high-dielectric needs to have a refractive index of at least 2.8.19,20
Interestingly, complete band gaps are absent in biological photonic crystals, despite millions of years of optimization. There has been no evolutionary advantage in selecting for such a phenomenon. For the purpose of producing colouration, incomplete band gaps (also referred to as ‘stopgaps’) are sufficient. In fact, stopgaps exhibit strong direction-dependent reflection behaviour and, therefore, are ideal for producing iridescent properties with different colours being reflected at different angles. Moreover, even for those species that require angle-independent colouration (i.e., with the appearance of a single reflective colour no matter what the angle of observation is), nature has found elegant solutions. For example, the photonic crystal community has known for more than two decades that crystal structures based on a diamond lattice are by far the most promising for achieving near angle-independent reflection behaviour. While diamond-based lattices - the champion structures 21 - have been achieved in photonic crystals operating at infrared wavelengths,22–27 fabricating these lattices for use in the visible part of the electromagnetic spectrum has proven extremely difficult.28,29 In contrast to our difficulties in fabricating diamond-based photonic lattices, beetles overcame this problem millions of years ago. Excellent examples are weevils, whose striking colouration is the result of light interacting with a diamond-based photonic crystal structure built into their exoskeleton scales made of cuticle.2,30
Almost 20 years after theoretical modelling identified diamond as the most effective lattice, the architectures discovered in blue, green and red coloured weevils in 2008 represent the first examples of diamond-based photonic crystals operating at visible wavelengths. 2 It is important to point out that biological diamond-structured photonic crystals do not possess a complete band gap due to the low refractive index of cuticle (around 1.5). Nevertheless, these weevils produce near angle-independent colouration by organizing the diamond lattice into an array of differently-oriented single-crystalline domains (pixels), each with a size of 2–10 micrometres - too small to be resolved by typical animal or human eyes (Figure 3). 2 Besides highly-ordered diamond-based structures, this lattice geometry can also produce angle-independent colouration with low-index compounds in an ‘amorphous’ form. Amorphous diamond photonic structures were theoretically predicted in 2008, 31 and their characteristic feature is the presence of some degree of short-range diamond-order, while the long-range order is lost. Interestingly, four years after their theoretical prediction, amorphous diamond-structured photonic crystals were discovered in bird feathers. 32 Moreover, it was shown that this structure can produce angle-independent colouration in a low refractive index keratin matrix.
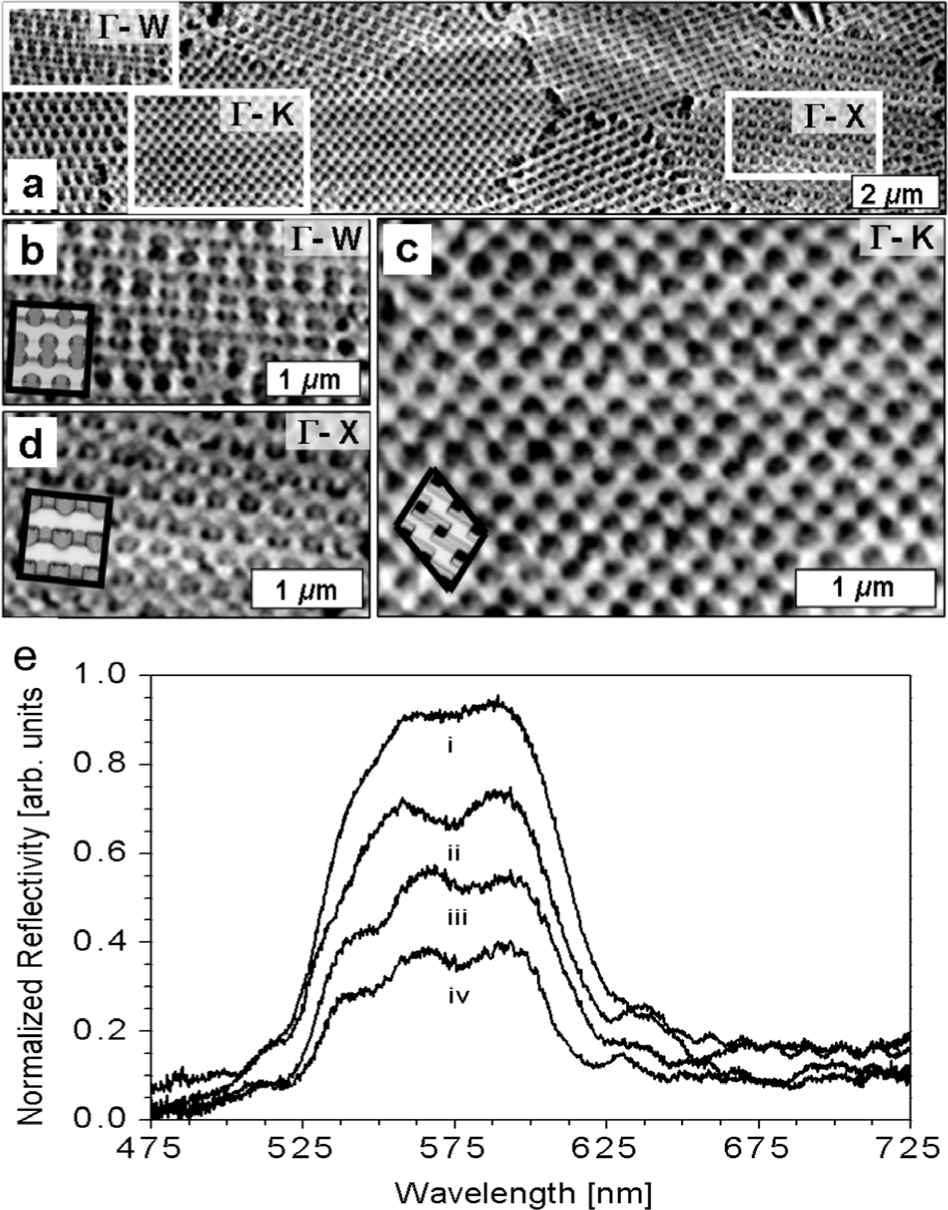
a) Scanning electron micrographs of the top-surface of a coloured scale of weevil
3. Evolutionary Development of Biological Photonic Structures
Iridescence is of great importance in the world of insects, which often rely on structural colours for defence, camouflage and reproduction.
12
A key advantage of structural colours over pigmented colours is that they produce more intense colouration than pigments, especially in low light conditions.
33
For example, the multilayer and diffractive structures in the scales of
While the evolutionary history of structural colour is difficult to reconstruct, some of this information can be drawn from the fossil records and the evolutionary lineages (Figure 4). The development of photonic structures involved a gradual increase in complexity and colouration, from scutes (patterned, hardened external parts of insect wing scales) to multilayer structures, diffraction gratings and, finally, to the complex cubic structures we find in some insects today.41–43 The earliest multilayer systems and gratings in the fossil record were discovered within the hairs and spines of beetles (Figure 5) found in the Burgess shale fossils from the Cambrian, 515 million years ago (mya). 41 Within the spines, the diffraction gratings were aligned in the same direction regardless of the turns and twists of the spines, indicating that these periodic structures were not simply a product of fossilization. However, directly relating these structural features to particular optical properties is very difficult, because the fossil record often does not perfectly preserve the lattice spacing and feature sizes of the periodic structures. 44 Since changes in the periodic lengths in photonic structures are directly related to the wavelength of reflected/diffracted light, the colours we find in fossilized periodic structures are not necessarily the same as they were in the living organisms. Nevertheless, these early examples of periodic structures are of tremendous value in studying the evolution of structure formation and the development of more sophisticated photonic structures. For example, morphology studies by Wickham et al. have revealed that the initially planar mutlilayers have evolved into tilted structures to compensate for the angle dependence of their reflection. 43

Timetable showing the evolutionary history and development of structural colours in biology

Micrographs of the Burgess stem-group polychaete
The largest finds of early fossils of beetles are from the early Triassic (∼250 mya) and were found in Transbaikal, Mongolia. The earliest species appear to be confined to conifers, as determined by being localized together in the fossil record and the presence of characteristic bite marks on the plants. 45 However, with the first appearance of the angiosperm (around the beginning of the Jurassic, 206 mya) and their rapid diversification at the beginning of the Cretaceous, 145 mya, host switching of the beetles from gymnosperms to angiosperms occurred. This was assisted by the formation of the elytra and other body modifications, such as the enlargement of ventral sternites and a rigid connection to the head. 46 An important fossil find of beetles are the Messel - Germany -oil shales (Figure 6). These shales are up to 40 percent water and are able to preserve the optical properties of some of the fossilized photonic structures of beetles from 49 mya. 42 These findings give important insights into early structural colours, especially their development, diversification and increase in structural complexity.

a) Photograph of a fossilized beetle specimen embedded in rock (Messel oil shales). The length of the specimen is 4 mm. b) Transmission electron micrograph of the outer surface of the elytron showing fine lamination. Adapted from reference [42] and reproduced by permission of The Royal Society.
4. Ecological Aspects of Structural Colours
Identifying the most promising biological structures as blueprints for creating particular functions in technological applications can be very challenging, and knowledge about ecological aspects becomes very important. In the following, we will give some insights into the origins and roles of biological photonic structures using the example of the insect order Coleoptera - the beetles. Photonic structures are widespread in beetles and have undergone several adaptive radiations.31,47–49 With this in mind, these structures must convey some evolutionary advantage to have produced the diversity seen today. These complex structures are not restricted to any specific lineage in either the beetles or weevils, and the genera with iridescent species are varied. One feature they have in common, however, is that those insect species and genetic families that possess photonic structures have thrived and proliferated widely.
A widespread purpose of structural colours in insect species - and Coleoptera in particular - is camouflage. 50 Insect species that are exposed during the day (or become active late in the day) are usually camouflaged to some extent; they typically try to resemble flowers, lichens or moss. For example, some beetles are marked with black stripes, which match the colour of prevalent flowers (presumably a strategy of camouflage while feeding). Other examples are metallic iridescent beetles, which fly during sunny hours and rest on bushes and trees where their metallic colours might provide some camouflage. In general, the classically iridescent beetles are primarily diurnally active; however, there are examples of brilliantly metallic species of longhorn beetles (the Cerambycidae) that are nocturnal. Besides longhorn beetles, another beetle clade has evolved to create some of the most interesting, three-dimensional photonic structures - the monophyletic weevils (the Curculionidae). These beetles tend to live and mate upon the same host plants on which they were hatched. 51 The selection of a new host plant and the niche provided by selecting a new host plant provides essential prezygotic barriers, enabling sympatric speciation. For weevils, much like the longhorn beetles, host plants dominate the lifecycle. The close association between beetles and plants would indicate that they diversify together, and possibly that diversifications within the plants cause diversifications within the beetles. The herbivore beetles are significantly more diverse than non-herbivore beetles, indicating that comingling with plants may be a contributing factor to beetle diversification. 52 Host plant restriction and evolution associated with the rapidly diversifying angiosperms likely helped spread the beetles, which were developing photonic structures.
Besides camouflage, structural colours are also often used for signalling and deceptive mimicry.12,50 The two main forms of mimicry in the insect world are Müllerian and Batesian mimicry. 53 In the former, two or more poisonous species - which share common predators - mimic each other's appearance and warning signs, whereas in Batesian mimicry a harmless species uses the appearance and signals of a poisonous or distasteful species to deceive a common predator. The lineages of Cerambycidae are known to heavily use Batesian mimicry. For example, they have adapted dark-coloured elytra and flattened body forms to match the appearance of ants. 54 Other insects that Cerambycidae mimic by this strategy include bees and wasps.
There is also a substantial amount of Batesian mimicry used by Cerambycidae to mimic the appearance of inedible weevils.
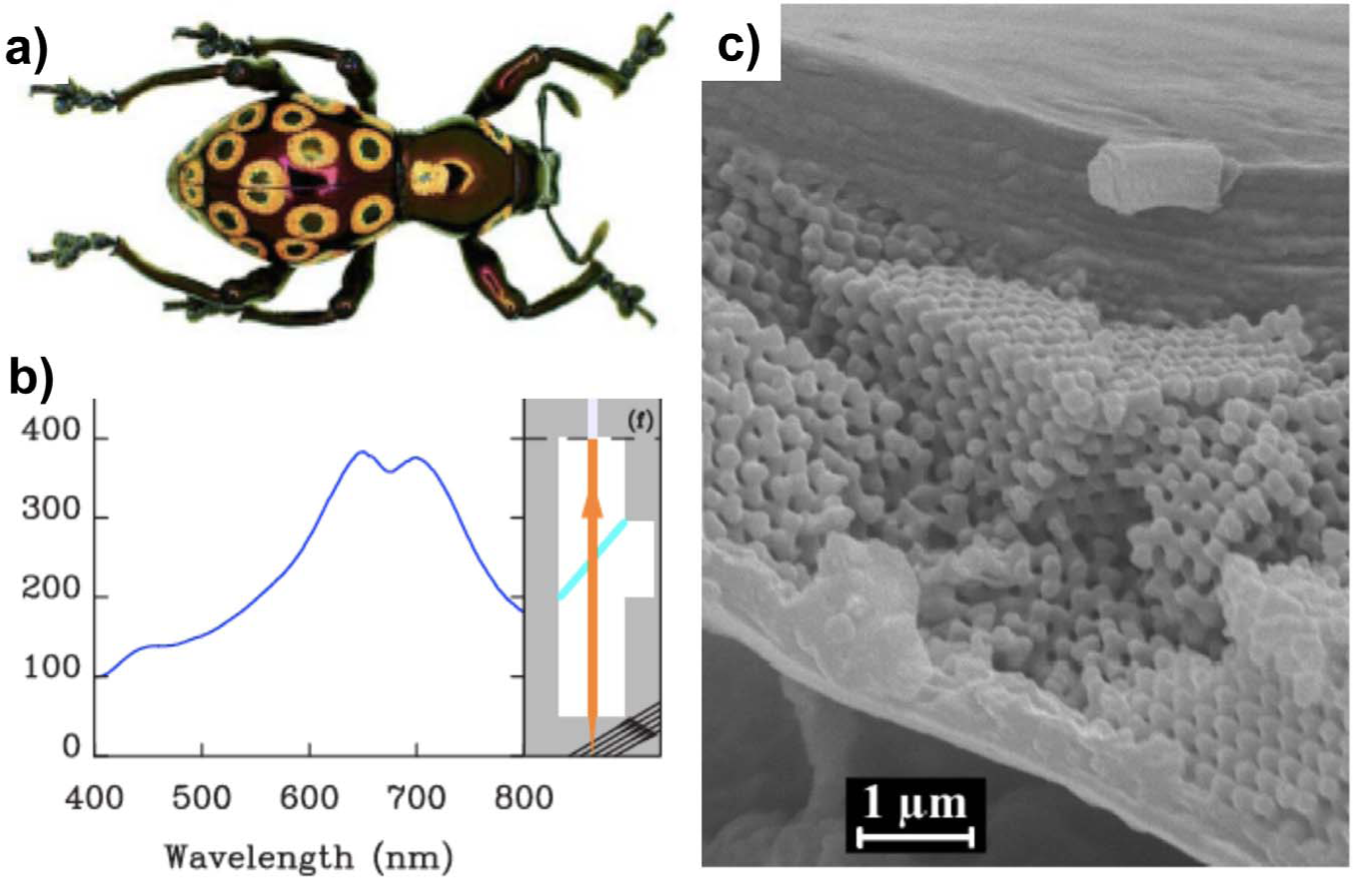
a) Photograph of the weevil
There are several cases, however, in which the advantage of carrying particular structural colouration is not as obvious as in the examples given above. For example, comparison of the similar weevils
Incidental colours are produced as the side products of structures with other, more important purposes. Excellent examples are the Abalone shell and the notoseta, spines from the sea mouse,
Additional examples of structural functionalities in biology other than colouration include the reduction of water adhesion and reduced friction in the wet and sticky environments often encountered when burrowing and plant-boring (beetles), 47 thermal regulation, aerodynamic drag, frictional forces (crustaceans),43,58 superhydrophobicity and self-cleaning (locust and butterflies). 59
5. Photonic Structure-formation Pathways in Biology
Over the past few decades, many synthetic procedures have been developed to produce photonic crystals.8,9,22–30 A basic design rule for a photonic crystal is that band gaps form at wavelength ranges of the electromagnetic spectrum that are of the same order of the periodic length (or lattice constant) of the photonic crystal.13–15 For example, for a band gap within the microwave range, a periodic structure with millimetre lattice constants is needed, while band gaps in the infrared and visible portion of the spectrum form in periodic structures with lattice constants within the micrometre and nanometre ranges, respectively. Naturally, to cover such a wide range of feature sizes different fabrication methods are needed, from mechanical drilling (microwave) to light-based patterning techniques (infrared) and self-assembly methods (visible and ultraviolet).14,15
Generally, man-made photonic crystals are fabricated by either an engineering or a self-assembly approach. The latter can be divided into supramolecular and colloidal assembly methods,8,9,60–62 whereas examples of engineering approaches are holographic lithography, 25 direct laser writing, 28 direct ink writing, 24 and electron-beam lithography. 27 The advantage of engineering methods is the wide range of photonic crystal structures that can be formed, including the champion diamond-based lattices. 21 While these methods are excellent tools for fabricating photonic crystals operating in the infrared and higher wavelengths (Figure 8), creating diamond-based lattices with feature sizes small enough to achieve band gaps within the visible range of the electromagnetic spectrum has proven extremely difficult. Only very recently have engineering methods started to successfully extend into the visible range by fabricating diamond-based woodpile structures.29,30 Self-assembly methods, on the other hand, have long been used to create photonic crystals with lattice constants spanning the infrared, visible and ultraviolet spectral ranges.60,63 Nonetheless, except for some interesting examples in the ultraviolet, 60 self-assembly techniques have unfortunately been incapable of fabricating the most-desired diamond-based lattices.

Scanning electron microscopy views of a germanium photonic crystal with a diamond-based woodpile structure. The sample was fabricated by the direct ink writing of a template followed by chemical vapour deposition of germanium. This structure has a complete photonic band gap in the infrared region of the electromagnetic spectrum. Adapted from reference [24]. Copyright Wiley-VCH Verlag GmbH & Co. KGaA. Reproduced with permission.
In contrast to our limited structure formation capabilities at visible wavelengths, nature is again a step ahead of us. Biological systems have developed photonic crystals with both the champion diamond-based structures and lattice constants of a few hundred nanometres - photonic crystals optimized to strongly interact with visible light.1,34–40 Moreover, nature has managed to synthesize these photonic structures under rather simple environmental conditions and with limited resources. In the following, we will give a summary of some recent insights into photonic structure formation in butterflies and beetles, focusing on the most desirable gyroid and diamond-based lattices.
In early work on butterfly wing scales conducted by Ghiradella et al., cross-sectional scanning electron microscopy imaging was performed at various stages of structure formation in scales.64,65 The authors concluded that in some butterflies iridescent chitin structures are formed by the cellular membrane of the smooth endoplasmic reticulum. Later, Michielsen and Stavenga found gyroid-structured photonic lattices, 39 and Galusha et al. discovered that certain weevils create their iridescent colouration by diamond-based photonic crystal lattices (Figure 3). 2 These discoveries were enabled by various imaging and scattering methods, including three-dimensional transmission electron microscopy and sequential focused ion beam/scanning electron microscopy imaging.
Recently, Prum et al. gained deeper insights into the formation of gyroid lattices in iridescent scales through a combination of small angle X-ray scattering and photonic band gap structure calculations. 66 They concluded that these structures are formed by a multiple-step mechanism involving both the cellular plasma membrane and the smooth endoplasmic reticulum membrane to initially form a double-gyroid structure. The biopolymer chitin is deposited and polymerized within the space that forms the core of one of the gyroid networks (Figure 9). Subsequent to cell death (and hardening of the chitin), the membranes and other cellular components decay and are replaced by air, leaving behind a single gyroid lattice composed of chitin. 66 This connection of gyroid structures and cellular cubic membrane folding is an important step in understanding photonic crystal formation in biological systems. However, it leaves the central question unanswered: How do cubic membranes form from phospholipid building blocks?
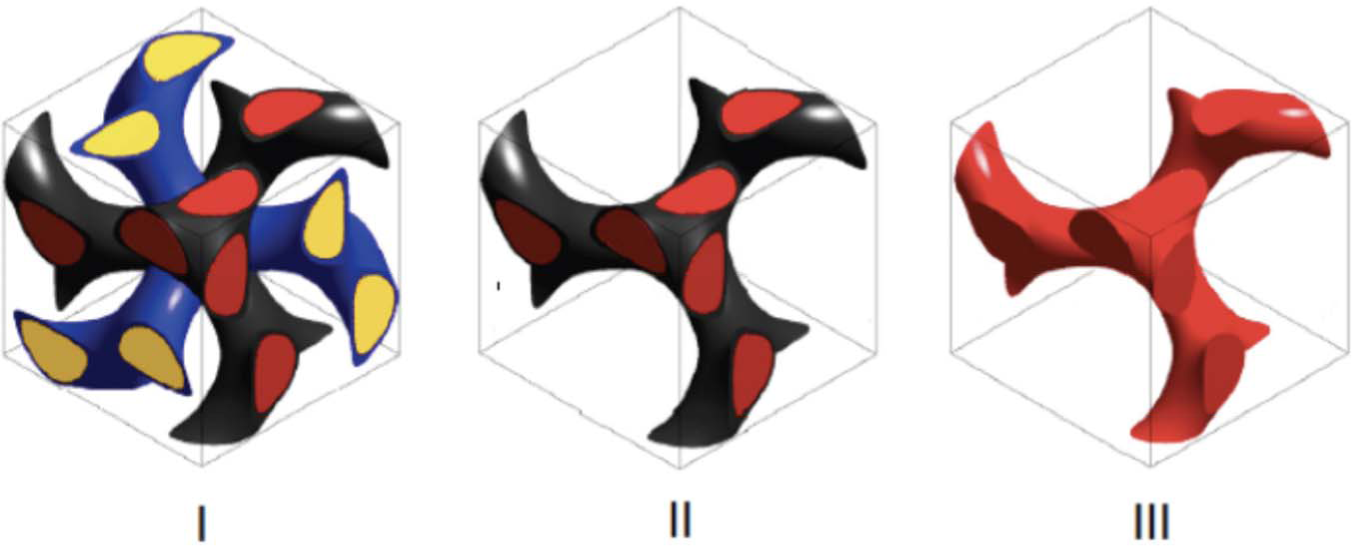
Structural models of a developing butterfly wing scale structure with double gyroid (I) and single gyroid (II and III) lattices. Model III is that of the final chitin lattice found in butterfly wing scales. Reproduced with permission from reference [66], National Academy of Sciences, U.S.A.
While it has been shown that phospholipid systems self-assemble into bicontinuous cubic crystal structures, the lattice constants of such assemblies are typically within the range of a few tens of nanometres. 67 With the help of different additives and modifying the atomic structure of the lipids, the lattice parameters could be increased to the 100 nanometre range;68–70 however, these values are still far below the ultra-high lattice constants of cellular cubic membranes (200–500 nanometres) found in mitochondria and ordered smooth endoplasmic reticulum, also referred to as OSER, as shown in Figure 10.71–73 While the exact mechanism of cubic membrane formation is still not fully resolved, recent studies have identified several key components, such as conical phospholipids,74,75 cholesterol molecules 76 and membrane proteins.71,77–81

TEM images of ordered smooth endoplasmic reticulum (OSER) membranes with bicontinuous cubic symmetry at a) low and b) high magnification. Reprinted from reference [71]; copyright (2006), with permission from Elsevier.
Conical lipids have packing parameters greater than one and induce negative curvature within the lipid bilayer. The lipid's head and splayed chains form a wedge in the bilayer, 82 facilitating the formation of gyroid and diamond-structured lattices. The effect of the presence of cholesterol is thought to be similar to conical lipids. Its planar shape makes cholesterol fit between phospholipid tails, inducing tail separation. 76 Thus, the presence of cholesterol can facilitate a convoluted endoplasmic reticulum, which is an important initial step of cubic membrane formation. Among the many membrane proteins present in cells,77–81 those with the ability to span the lipid-bilayer membrane through homotypic interactions are of particular importance.79,80 Such membrane proteins (e.g., cytochrome b5) can induce a high curvature in lamellar bilayers and the formation of cubic phases.
These recent insights into the formation mechanism of gyroid and diamond-structure photonic crystals in biological systems are a tremendous step towards the bench-top fabrication of these most sought-after photonic structures through directed and self-assembly methods. While there are still many hurdles to overcome in finding the holy grail of photonic crystals - large-scale diamond-based photonic crystals with low defect densities and tunable lattice parameters - it seems that it was again a hint from nature that opened the door to a major technological breakthrough.
6. Conclusions
In our attempts to produce photonic structures for use in modern-world applications, taking a look at nature for ideas and inspiration can be very beneficial. Nature has produced a wide variety of photonic structures throughout millions of years of development. Of course, these techniques taken from nature require modification to efficiently interface with modern technology and devices. However, we can benefit tremendously from studying the variety of biological architectures, the clever design solutions developed to overcome inherent limitations such as low refractive index components, and the effectiveness and elegance of how the most complex structures are formed in simple environmental conditions and with limited resources.
It seems apparent that when nature provides examples and protocols for the production of these complex photonic structures, piggybacking on her work can save time and money.83–85 However, nature did not develop or optimize structures for technological applications, but rather for camouflaging, signalling and mimicking. In this review article, we attempted to provide some insights into the purpose and the evolutionary and ecological development of photonic structures in biology. The comparison of photonic structures in biology with idealized technological structures reveals significantly different design strategies - most likely due to different evolutionary and innovation ‘pressures’, respectively. When evolution selected for certain structures, colouration was often not the original function and the variety of simple photonic structure predecessors most likely had roles related to temperature regulation, reduced drag and mechanical reinforcement. Therefore, the term ‘structural colour’ does not describe the ancestry or diversity of these structures, such that ‘structural function’ may be a more appropriate term. The hope is that as materials scientists become increasingly appreciative of the work achieved by evolution, we will progressively learn from and be inspired by these fascinating biological examples of structural function.
Footnotes
7. Acknowledgments
We would like to thank Bryce Turner, Michael Dahlby, Julie Chapman and Dr. James Casanova for their valuable assistance. This work was supported by the NSF MRSEC programme at the University of Utah under grant DMR-1121252, and in part by the NSF under award DMR-1005382.
