Abstract
Selective synthesis of titanium dioxide (TiO2) polymorphs including anatase, rutile, brookite and TiO2(B) by solvothermal treatment of water-soluble titanium complexes is described with a special focus on their morphological control. The utilization of water-soluble titanium complexes as a raw material allowed us to employ various additives in the synthesis of TiO2. As a result, the selective synthesis of the polymorphs, as well as diverse morphological control, was achieved.
Keywords
1. Introduction
Titanium dioxide (TiO2) has been used in various fields as white pigments, dielectric materials and photocatalysts, etc. Under ambient conditions, titania exists in eight polymorphs, of which rutile, anatase and brookite are in natural polymorphs and TiO2(B) (bronze) [1,2], TiO2(H) (hollandite) [3] and the other three polymorphs [4–6] can only be obtained synthetically in the laboratory. Anatase and rutile have wide practical applications, because they can be easily synthesized. On the other hand, there are only a limited number of methods for the single-phase synthesis of the other polymorphs, including brookite [7–16] and TiO2(B) [1, 2, 17, 18]. Consequently, their functional abilities have not been precisely examined, though they are believed to have high potential as functional materials [19]. Therefore, the development of a method to selectively synthesize rutile, anatase, brookite and TiO2(B) polymorphs is important.
The functional properties of titania as well as other materials are strongly dependent on its morphology. In general in the inorganic material field, particles show two main types of morphology. The first is the shape of the primary particle, which can be regarded as a single crystal; the second refers to the shape of the secondary particle, which is an agglomeration of primary particles. Some examples of the secondary particle shape include hollow, porous and solid spheres. The agglomeration of titania showed additional functional abilities such as high absorption rate [20], photonic character [21] and incorporation of organic compounds, gases and ions [22–24]. The primary particles show four types of structures: zero-, one-, two- and three-dimensional structures. Nano-sized particles are generally categorized as zero-dimensional structures. Whisker- and needle-like particles are examples of one-dimensional structures; sheet-like particles are examples of two-dimensional materials and the rest are classified as three-dimensional structures.
For evaluating the morphological control of a primary particle, it is important to know which of its facets are exposed, as well as the area ratio of the exposed crystal facets; this is because each crystal facet exhibits different properties. To control which crystal facets are exposed, the equilibrium shape of each polymorph should be understood by taking the Wulff construction [25–27] into account. The equilibrium shape of anatase has been reported as a truncated bipyramidal shape constructed by eight {101} and two {001} (Fig. 1(c)) [28–30].

Morphology predicted for anatase (top) with (a) hydrogenated surfaces; (b) with hydrogen-rich surface adsorbates; (c) hydrated surfaces; (d) hydrogen-poor adsorbates; (e) oxygenated surfaces and rutile (bottom) with (f) hydrogenated surfaces; (g) with hydrogen rich surface adsorbates; (h) hydrated surfaces; (i) hydrogen-poor adsorbates; (j) oxygenated surfaces (Reprinted with permission from Reference 30. Copyright 2005 American Chemical Society).
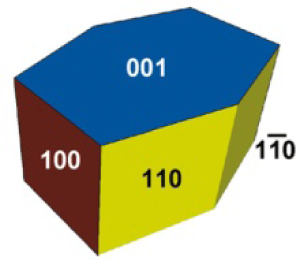
Equilibrium shape of a macroscopic TiO2(B) crystal, using the Wulff construction and the surface energies calculated by Vittadini [32] (Reprinted with permission from Reference 32. Copyright 2009 American Chemical Society).
A tetragonal prism bounded by {110} and terminated by a pair of tetragonal pyramids bounded by {011} was the equilibrium shape of rutile when considered for the Wulff construction. The most stable plane among the rutile crystal facets was {110} (Fig. 1(h)) [28–30]. The equilibrium crystal of brookite possessed seven different facets (Fig. 2) [31]. A flat pseudohexagonal prism constructed from {001}, {100}, {110} and {1–10} is the equilibrium shape of TiO2(B) (Fig. 3) [32].

Equilibrium crystal shape of brookite TiO2, as obtained through the Wulff construction. Different facets have different colours and the orientations are given (Reprinted with permission from Reference 31 as follows: X. Q. Gong, A. Selloni, Phys. Rev. B, 76, 235307 (2007). Copyright 2007 by the American Physical Society).
It is also known that the equilibrium shape varies slightly according to conditions under which the crystals are formed, because the surface energy and tension are dependent on environment [30, 33–36]. For example, under basic conditions, rod-like particles surrounded by {101}, {001} and {010} is the equilibrium shape of anatase, while decahedral shape is described from the Wulff construction without any adsorbates (Fig. 1).
A number of studies report that each crystal facet shows different properties [37, 38]. Nakajima et al. [39] evaluated superhydrophilicity on {110} and {001} of rutile single crystals under UV irradiation and found that the hydrophilicity rate on {001} was faster than that on {110}. It was also reported that when the measurement was repeated, the superhydrophilic property on {001} increased, while that on {110} decreased. Ohno et al. [40] concluded that the photocatalytic oxidation and reduction sites of rutile were present on {011} and {110}, respectively. They also found that the oxidation site of anatase was mainly present on {001} and that {011} was more reductive than {001}. However, Kawahara and co-workers reported low activity on the {110} of rutile for the photocatalytic Ag reduction [41]. Moreover, not only empirical results, but also theoretical calculations indicated the differences in the properties of each crystal facet. Based on density functional theory (DFT) and first-principles molecular dynamics simulations, Gong et al. reported that anatase {101} had higher dissociation-adsorption activity for water and ethanol than that of {001} [42]. Based on a modified DFT calculation, Li and co-workers revealed that water and formic acid were adsorbed on each facet of titania in different modes [43].
Some studies have reported the improvement and control of the functional ability of titania by synthesizing titania particles with different morphologies from the particles with equilibrium shapes via chemical synthesis methods. Yang et al. [44, 45] synthesized anatase single crystal dominated by {001}, which has higher energy than {011}. As a result, the obtained anatase with the anisotropic shape had a sheet-like nature and showed higher photocatalytic activity than that of anatase with equilibrium shape. Similar anisotropic anatase crystals were obtained by other research groups, who also found that the anisotropic anatase exhibited high photocatalytic activity [46, 47]. Amano et al. synthesized octahedral bipyramid-shaped anatase terminated by {101} with less area of {001} and implied that {101} showed low photocatalytic activity for hydrogen evolution without oxygen [48]. Kanie and Sugimoto fabricated a liquid crystal using c-axis-oriented anatase in combination with organic compounds. Their findings showed that the formation of a liquid crystal was dependent on the degree of crystal anisotropy [49]. These results indicate that developing a methodology for enabling the synthesis of well-faceted and anisotropic titania particles is important for making titania more attractive as a functional material.
Conventionally, in the synthesis of titania, toxic solvents such as HCl (aq.), H2SO4 (aq.) and organic solvents are required, because TiCl3, TiCl4, Ti(SO4)2 and titanium alkoxide are the commonly used titanium sources. In order to meet the rising demand for environmental protection, water should be used as a solvent. However, because the conventional titanium sources are sensitive to water and thus moisture, it is not easy to use water as a solvent in this case. In addition, when using the conventional titanium sources, additives that can work as a morphological control agent are difficult to employ. In 2001, Kakihana et al. developed an ammonium citratoperoxotitanate, (NH4)8[Ti4(C6H4O7)4(O2)4]·8H2O [50]. The compound can be readily dissolved in neutral water and the pH of the prepared aqueous solution is approximately 5–6. Moreover, it is stable for up to several years under ambient conditions as a solid and as an aqueous solution, and its ligand (citric acid) is only slightly toxic. In addition, the complex does not undergo hydrolysis in a wide range of pH values (0–14), allowing for the use of various additives. It has been shown that various compounds such as hydroxycarboxylic acid, amino acid and amine can be used as ligands to prepare a stable water-soluble titanium complex [51–57].
The authors believe that the water-soluble titanium complex is one of the valuable raw materials for the synthesis of titania with controlled crystal- and microstructures. We have succeeded in selectively synthesizing titania polymorphs, including anatase, rutile, brookite and TiO2(B) by hydrothermal treatment of water-soluble titanium complexes using additives [52, 54, 55, 57–60]. By adjusting the synthesis conditions, the single phase of each titania polymorph can be obtained. In addition, the morphological control of titania crystals was achieved. The reason for achieving selective synthesis might be related to the individual intrinsic characteristics of a series of water-soluble titanium complexes.
In this paper, we summarize our results on the morphological control of anatase, rutile, brookite and TiO2(B) by hydrothermal and in part solvothermal treatment of water-soluble titanium complexes using additives. On the other hand, we do not deal here with details and discussion of selective synthesis of polymorphs, because the points have been already mentioned in a previous paper [55].
2. General Synthesis Procedure
2.1 Preparation of water-soluble titanium complexes
Water-soluble titanium complexes can be prepared from the titanium-peroxo complex [61] and the appropriate ligands. There are many methods for the preparation of the titanium-peroxo complex [62, 63]. Here, we briefly describe the typical preparation procedure used by our group [55]. Titanium metal powder (5 mmol) was dissolved in a mixture of 30.0 % hydrogen peroxide (20 cm3) and 28.0 % ammonia (5 cm3) in a water bath at room temperature. After two hours, a yellow transparent solution containing the titanium-peroxo complex was obtained. A complexant such as glycolic acid or citric acid was added to the solution. The amount of ligand required was dependent on the compound used, which is described in detail elsewhere [55, 57]. To remove excess hydrogen peroxide and ammonia, the prepared solution was left at room temperature for 12 h and then heated at 353 K until a gel-like solid substance was formed. The gel was dissolved in distilled water and an aqueous solution of a titanium complex was then obtained.
2.2 Hydrothermal synthesis of titania nanocrystals
The prepared aqueous solution of a water-soluble titanium complex was heated in a Teflon-lined stainless steel autoclave at a given temperature for a specified amount of time. After the autoclave was cooled to room temperature, the precipitate was separated using a centrifuge and was washed with distilled water three times. The final product was obtained after drying. In this paper, unless stated otherwise, the titanium concentration of the treated solution was 0.25 mol dm−3.
3. Results and Discussion
3.1 Morphological control of rutile-type titania nanocrystals
Single-phase rutile titania powder was obtained by the hydrothermal treatment of a titanium complex prepared using glycolic acid (titanium-glycolato complex) at 473 K for 24 h [52]. The resulting powder consisted of 50 × 150 nm2 rod-like particles (Fig. 4(a)).

TEM images of the rutile particles synthesized by hydrothermal treatment of titanium-glycolato complex without additives at 473 K for (a) 24 h (b) 100 h and (c) 6 h.
The shape is similar to the equilibrium shape of rutile [28–30]. When the treatment time was extended to 100 h, the crystalline phase, size and shape of the particles were maintained (Fig. 4(b)). However, the hydrothermal treatment at 473 K for 6 h produced anatase in the secondary phase. The transmission electron microscopy (TEM) image shows that anatase particles (5 nm) attached to rod-like rutile particles (40 × 120 nm2) were obtained (Fig. 4(c)). These results indicate that the growth of rutile particles in the present system followed the Ostwald ripening law, i.e., dissolution of nano-sized anatase and subsequent recrystallization as the rutile polymorph [64]. Elongated rod-like rutile particles could be synthesized using glycolic acid as an additive in the hydrothermal treatment of the titanium-glycolato complex aqueous solution. The aspect ratio of the obtained rutile particles increased dramatically with an increase in the additional amount of glycolic acid (Fig. 5).

TEM images of the rutile particles synthesized by hydrothermal treatment of titanium-glycolato complex in the presence of (a) 5 mmol; (b) 10 mmol; (c) 15 mmol and (d) 25 mmol of glycolic acid as additives at 473 K for 100 h.
High resolution TEM and selected area electron diffraction (SAED) analysis revealed that the obtained rutile grew along the c-axis; facets that are perpendicular to the growth direction are {110} and the tips terminated by {111} [64, 65]. The exposed facets are the same as those of the equilibrium shape deduced from Wulff construction [28–30]. However, the aspect ratio of rutile crystals synthesized in the presence of glycolic acid was greater than 10, while that of the equilibrium shape of rutile was approximately 2. Therefore, the method provided us anisotropic rutile particles with a high area ratio of {110}. The anisotropic growth of rutile might be caused by selective adsorption of glycolic acid on {110} [29, 33]. Facets chelated with glycolic acid had a lower surface energy than those of bare surfaces. In order to reduce the total energy of the particles, the chelated surfaces were predominantly developed during further particle growth. At the same time, titanium ions were difficult to adsorb on the chelated facets and were therefore preferentially adsorbed on the non-chelated facets (the capping effect). As a result, the obtained rutile particles had a high aspect ratio and the lowest possible fraction of nonchelated facets. When the pH of the titanium-glycolato complex aqueous solution containing excess glycolic acid as the additive was adjusted from approximately ∼3 to 5–8 by the addition of ammonia, the formation of the whisker-like particles was prevented, resulting in the formation of the usual rod-like rutile particles. The isoelectric point of TiO2 has been reported as pH 4.7–6.7 [66–68], which means that the surface of the rutile particles had a negative charge at pH 5–8. Consequently, glycolic acid anions cannot be adsorbed effectively on the rutile surface and hence, cannot promote whisker growth.
When lactic acid was added to an aqueous solution of titanium-glycolato complex as the additive instead of glycolic acid, rod-like rutile nanocrystals with a high aspect ratio compared to that of the equilibrium shaped rutile were obtained (Fig. 6(a)). When oxalic acid was used as an additive, the solution pH became less than 1 and single-phase anatase was formed. Using ammonia to adjust the pH to ∼3, single-phase rutile was obtained from the solution employing oxalic acid as the additive. However, these rutile particles were rod-like with low aspect ratio and rounded ends (Fig. 6(b)). The reason why rutile particles with a high aspect ratio were not formed might be the high chelating ability of oxalic acid. Oxalic acid can be adsorbed on every facet. As a result, anisotropic growth was not promoted. It is important to note that the hydrothermal treatment of the titanium complex coordinated with leucic acid led to the formation of whisker-like particles with a much higher aspect ratio [57]. This indicates that the aspect ratio of the rutile crystals formed may be controlled by the length of the carbon chain in the hydroxycarboxylic acid used as an additive, though further examination should be performed. We also found that the rutile obtained with a high aspect ratio showed high photocatalytic activity and a robust dielectric constant [65, 69]. These results indicate that the hydrothermal treatment of titanium complexes combined with suitable additives forms anisotropic rutile particles, which showed improved properties.

TEM images of rutile particles synthesized by hydrothermal method at 473 K for 24 h using titanium-glycolato complex in the presence of (a) lactic acid and (b) oxalic acid with ammonia at pH 3.3.
3.2 Morphological control of anatase-type titania nanocrystals
In general, anatase-type titania can be synthesized as nanosized crystals by solution methods without post-firing. The reason for the formation of nano-sized anatase crystals might be explained in terms of the critical size of titania polymorphs [70]. Anatase nanocrystals can also be obtained by the hydrothermal treatment of water-soluble titanium complexes. Moreover, synthesis of spindle-shaped and square bipyramid-shaped anatase particles has been achieved via a solvothermal treatment using ethylenediamine (en) as a key solvent in the presence of a given amount of water. The hydrothermal treatment of a citratoperoxotitanate (titanium-citrato) complex in distilled water without en at 473 K for 24 h led to the formation of an agglomeration of 10-nm-sized anatase nanocrystals (Fig. 7(a)). When the en concentration in the en/H2O solvent was low, i.e., less than ∼75.0 wt%, the powder obtained after the solvothermal treatment consisted of spindle-shaped anatase particles (Fig. 7(b)). A SAED pattern of a spindle-shaped particle revealed that the major axis was parallel to the c-axis [71]. By increasing the en concentration in the solvent, the shape of the particles changed from spindle to square bipyramid (Fig. 7(c)).

TEM and SEM images of anatase particles synthesized by solvothermal treatment of titanium-citrato complex in (a) distilled water; (b) 61.8 wt% en aqueous solutions and (c) 82.7 wt% en aqueous solution at 473 K for 24 h.
These results indicate that the morphology of anatase particles can be modified from a nanoparticle to spindle-shaped by adjusting the concentration of en used in the solvothermal system using the en/H2O solution of the titanium-citrato complex. When the titanium-glycolato complex, or TiOCl2 (which can be prepared from TiCl4 and ice water) was used as initial titanium source, anatase particles with a similar shape could be obtained by carefully choosing the synthetic conditions [71]. Using a mixed en/H2O solvent, sheet-like titanic acid was produced in an intermediate phase, regardless of the en concentration. Therefore, the formation of anatase in the present system may occur through the dissolution-recrystallization of titanic acid. Theoretical calculations revealed that while truncated square bipyramid-shaped anatase should be generated under acidic conditions, spindle-shaped anatase was expected under basic conditions [33, 35]. Nevertheless, square bipyramid-shaped anatase nanocrystals were obtained under relatively high basic conditions used in the present study. Therefore, both the adsorption of en on the particle surface and the solvent pH may contribute to achieving the morphological control of anatase nanocrystals.
3.3 Morphological control of brookite-type titania nanocrystals
As mentioned in the introduction, the synthesis of brookite as a single phase is relatively difficult compared with the synthesis of single-phase anatase and rutile. Therefore, the morphological control of brookite is challenging. In fact, there are only a few reports on the morphological control of brookite particles [13–16]. By hydrothermal treatment of the water-soluble titanium complex, high-quality single-phase brookite particles can be easily synthesized with good reproducibility, as described below. When an ammonia solution was utilized as an additive in the hydrothermal treatment of the titanium-glycolato complex at 473 K for 24 h, rod-like brookite nanocrystals, with dimensions of 20 × 50 nm2, were formed (Fig. 8(a))

TEM images of brookite nanocrystals synthesized by hydrothermal treatment of titanium-glycolato complex in (a) 0.6 wt% and (b) 28.0 wt% NH3 aqueous solutions at 473 K for 24 h.
The size of the obtained brookite particles increased with an increase in the concentration of ammonia in the solvent, while other conditions such as treatment time remained the same. As a result, 80 × 250 nm2 rod-like brookite particles were synthesized from a 28.0 wt% ammonia aqueous solution (Fig. 8(b)). The results show that changing the concentration of ammonia in the solvent can control the size of the brookite generated.
When the titanium-citrato complex was used as a titanium source instead of the titanium-glycolato complex, brookite particles with aspect ratios higher than 10 were obtained. Hydrothermal treatment of the titanium-citrato complex in a 28.0 wt% ammonia solution resulted in the formation of 150 × 2180 nm2 rod-like brookite particles (Fig. 9(a)). The TEM and SAED measurements provided evidence that growth of the brookite particles occurred along the c-axis [72]. The equilibrium shape of brookite is a polyhedral construction [31]. Therefore, the present brookite particle was an anisotropically grown crystal along the c-axis. At high ammonium concentration, agglomerated brookite particles with unusual shapes, where many rod-like particles were branched around a somewhat longer central particle, also appeared (Fig. 9(b)). The side view of the agglomerated particles revealed two-dimensional crystal growth within a given restricted plane (Fig. 9(c)). The multineedle agglomerate of brookite particles was snowflake shaped and could not be obtained using the titanium-glycolato complex. Brookite with this unique morphology was likely formed because of the intrinsic characteristics of the titanium-citrato complex itself, although the mechanism still remains unclear.

TEM and SEM images of brookite particles obtained by hydrothermal treatment of a 28 wt% ammonia aqueous solution of titanium-citrato complex at 473 K for 24 h. (a) [Ti] = 0.5 M, (b, c) [Ti] = 0.25 M
When a suitable amount of amine such as dimethylamine was used as an additive in the hydrothermal treatment of titanium-glycolato complex at 473 K for 24 h, single-phase brookite samples were obtained. The resulting powder consisted of nanocrystals with a low aspect ratio of ∼1 (Fig. 10(a)).

(a) TEM and (b) SEM images of brookite particles with a low aspect ratio synthesized by hydrothermal treatment of titanium-glycolato complex in a 3.2 wt% dimethylamine aqueous solution at 473 K for 24 K.
When other amines such as monomethylamine and trimethylamine were used as additives, the same effect, i.e., formation of brookite with a low aspect ratio, was observed. The Scanning Electron Microscopy (SEM) measurement revealed that the resulting brookite nanocrystals were polyhedral (Fig. 10(b)). Recently, synthesis of pseudo-cube-shaped brookite nanocrystals by hydrothermal treatment of the titanium-glycolato complex in the presence of oleate salt was reported [73]. The shape of the brookite particles formed was similar to those synthesized by the present method, except for the number of exposed facets. The TEM and SAED measurements indicated that the obtained nanocrystal had high crystallinity and that the added amine either retarded, or, at the minimum, did not promote crystal growth along the c-axis (Fig. 11). These results would indicate that the basic conditions of the solvents were not related to growth along the c-axis; instead, ammonium ion and amine crystal affected the growth habit of the brookite crystal.

High resolution TEM and SAED images of the polyhedral-shaped brookite nanocrystals.
3.4 Synthesis of TiO2(B) nanocrystals
In 1998, Kasuga et al. reported the synthesis of TiO2(B) nanotubes via a multistep method [17, 18]. To date, almost all of the reported methods for the synthesis of TiO2(B) have been based on the methods developed by Kasuga et al. We have succeeded in the direct (one-step) synthesis of TiO2(B) nanoparticles by the hydrothermal treatment of titanium-glycolato complex in the presence of H2SO4 (Fig. 12) [58].

TEM image of TiO2(B) sample obtained by hydrothermal treatment of titanium-glycolato complex in a 5.5 wt% H2SO4 aqueous solution.
The samples obtained consisted of single-phase TiO2(B) and an agglomeration of 10-nm-sized nanocrystals. The shape looks similar to the equilibrium shape of TiO2(B) (Fig. 3) [32], though detailed structural analysis is under further study. The conditions under which TiO2(B) can be synthesized as a single phase are narrow. Therefore, we have not yet succeeded in morphological control of TiO2(B).
4. Conclusion
In the present paper, we summarized our results on the morphological control of four titania polymorphs: anatase, rutile, brookite and TiO2(B). Morphological control was achieved by the utilization of a series of water-soluble titanium complexes, as they allowed us to use various additives that acted as morphological control agents. The intrinsic characteristics of water-soluble titanium complexes also seemed to be important for the synthesis of titania particles with unique morphologies, because there is almost no compatibility between different titanium complexes. Therefore, it can be safely inferred that the development of novel sources, especially stable sources, is an effective method for synthesizing materials with controlled structures. The final goal of this type of research is to develop a tailor-made synthesis for a given crystal system. To realize this goal, accumulation of relevant knowledge is the first important step. Recently, development of theoretical study [74], computer simulation [75] and observation techniques [76] have yielded particularly impressive results in the crystal growth field and many new theories are being suggested. Material synthesis should be performed in reference to such chemistries to achieve simultaneous control of nano-and micro-structures. We hope that the present paper provides valuable insights into further elucidation of the morphological control of given crystals, not only for titania, but also for other crystal systems.
Footnotes
5. Acknowledgments
The authors thank Dr V. Petrykin (SuperOx Japan) and Dr K. Tomita (Tokai University, Japan) for fruitful discussions. Dr T. Miyazaki (Tohoku University) and Dr S. Ito (Institute for Materials Research, Tohoku University) made enormous contributions to the TEM and SAED measurements and analysis. This work was supported in part by the Grant-in-Aid for Scientific Research (No. 22107002) on the Innovative Areas: “Fusion Materials” (Area no. 2206) from the Japanese Government's Ministry of Education, Culture, Sports, Science and Technology (MEXT).
