Abstract
Carboxymethyl-hexanoyl chitosan (CHC) has the ability to self-assemble into nanocapsules in an aqueous solution and it has recently shown potential in numerous biomedical applications. Here we investigate the protein loading efficiency and release, as well as the structural properties of CHC protein nanocarriers. Bovine serum albumin (BSA) or its chromophore labelled version, fluorescein-BSA, was used as a model protein and the loading was performed with a simple mixing of pre-formed nanocapsules and protein. Dynamic light scattering and zeta potential analysis revealed that protein loaded nanocarriers with high positive zeta potential were formed. The protein loaded nanocarriers displayed a loading efficiency of 75% and a very slow protein release. In summary, our results highlight the potential of CHC as a protein nanocarrier, but also indicate that protein-CHC interactions need to be considered in protein containing CHC formulations where protein release is not the main function.
1. Introduction
Here we report on protein loading and release from carboxymethyl-hexanoyl chitosan (CHC) nanocarriers, which are of importance since this polymer has demonstrated potential in numerous biomedical applications. CHC, which contains both proton donating carboxylic acid groups and proton accepting amino groups of the native chitosan, was developed in 2006 by Liu et al. [1]. Since then it has been well established in several studies that it can self-assemble into nanocapsules or nanoparticles and that it has excellent drug loading and controlled release properties when used as a drug nanocarrier [2, 3]. Furthermore, cell studies have shown that CHC nanocarriers are effectively internalized into cells [4, 5]. Recently CHC has also been used in macroscopic nanostructured gels, e.g., for injections, with a sustained release of hydrophobic [6] and hydrophilic drugs [7] and as stem cell scaffolds [8]. Based on these successful reports together with reports on chitosan and chitosan derivatives as protein carriers [9–14] it was considered highly interesting to investigate the protein loading and release properties of CHC. The findings were expected to contribute to the important topic of therapeutic protein delivery [15], and to aid in the development of new protein-containing CHC-based systems for biomedical applications, such as stem cell scaffolds loaded with growth factors.
In this study CHC was loaded with bovine serum albumin (BSA) or bovine serum albumin fluorescein isothiocyanate (FITC-BSA) as a model protein. The formed nanoparticles were characterized with regard to loading efficiency, drug release, size and surface charge.
2. Materials and methods
2.1 Materials
Carboxymethyl-hexanoyl Chitosan (CHC), M.W. = 1.5·105 g/mol and viscosity = 2500–3500 cP (4% solution), was bought from Advanced Delivery Technology Inc., Hsinchu, Taiwan under the name AC-SAC (nanocarrier), its chemical identity was confirmed to be similar to previously reported CHC [1, 7] by 1H-NMR (supplementary material Figure S1). Bovine serum albumin (≥98% electrophoresis) and phosphate buffered saline (PBS) tablets were purchased from Sigma Aldrich (USA). Albumin Fluorescein isothiocyanate conjugate bovine was purchased from Sigma Aldrich (USA). A MilliPore purification system was used to obtain Milli-Q water (MQ-water) for the experiments.
2.2 CHC nanoparticle preparation
CHC nanocapsules were prepared by mixing CHC (2.0% w/w) and MQ-water using a magnetic stirrer for 72 hours at 25 °C, followed by sonication in an ultrasonic bath (TRANSSONIC 700 Elma®, Germany) for 5 hours and 30 minutes, resulting in a nano-suspension (pH 5–6, as determined with litmus paper). For dynamic light scattering (DLS), zeta potential and atomic force microscopy (AFM) analyses the nanocapsules were filtered through a 1 μm Acrodisc syringe filter (Pall Life Sciences, USA). Subsequently, BSA or FITC-BSA was added to a concentration of 0.25 mg/ml, which was then mixed for 48 hours using a magnetic stirrer. The process yield was investigated by drying it in a vacuum oven at a temperature of 40 °C for about 3 days (constant weight achieved) to ensure that only a small fraction of the sample was lost during the filtration step.
2.3 Size and surface charge characterization
DLS and zeta potential analyses were performed in MQ-water with a Brookhaven Instruments Corporation ZetaPALS instrument and FOQELS Particle Size Analyzer (USA). The DLS- analysis was carried out on three different batches, with triplicates within each batch. All samples were filtered through a 0.8 μm syringe filter (CA membrane, ADVANTEC MFS Inc, Japan) prior to analysis. AFM was performed using a Dimension 3100 (Digital Instruments, USA). 2 μl of the sample, diluted 100-fold, were placed on freshly cleaved mica and left to dry at ambient temperature overnight before analysis.
2.4 Protein loading and release
FITC-BSA containing nanoparticle dispersions and FITC-BSA reference solutions were diluted 1:10 with PBS (pH 7.4). Release studies were conducted in triplicates under shaking (50 rpm) in the dark at 37 °C using a UBR heating block (Grant, UK) positioned on a SM-25 shaker (Edmund Bühler, Germany). At pre-defined time points the amount of free FITC-BSA was determined as follows: Samples were centrifuged at 15000 rpm for 1 h at 4 °C using an Optima XL Ultracentrifuge with a type 90Ti rotor 90,000 rpm (Beckman Coulter, USA). Subsequently, supernatant was extracted and the UV-Vis absorbance was determined between 300–900 nm using a Lambda 900 UV/VIS/NIR spectrophotometer (Perkin Elmer, USA). The fraction free FITC-BSA was determined as the ratio between absorbance at 495 nm for samples and reference FITC-BSA solutions of the same total FITC-BSA concentration (Asample / Aref). Loading efficiency was calculated as 1-“fraction free FITC-BSA” at time zero. Any effects on the results originating from degradation of FITC-BSA chromophores during the release study were excluded by analysing reference samples at time zero and at the final time point.
3. Results and discussion
3.1 Nanoparticle preparation and formation
In accordance with previous reports [2, 3] CHC did self-assemble into nanostructures, as confirmed by DLS and AFM. The DLS analysis revealed scattering intensities corresponding to diameters starting at around 300 nm. However, larger structures that could not be eliminated were also detected, making exact size determination difficult (Figure 1a, b). These larger structures are probably CHC assemblies caused by variations in the substitution between polymer chains and by heterogeneities in the substitution pattern so that a fraction of CHC on occasion may form larger aggregates. Nonetheless, since large particles have more weight in DLS analysis compared to smaller ones, the presence of narrow distributions with small diameters in the size range of hundreds of nanometres lead us to conclude that CHC mainly existed as nanostructures. AFM analysis further supported this conclusion as it clearly revealed nanoparticles of about 100 nm in size (Figure 1c). The smaller sizes observed by AFM as compared to DLS are to be expected as the samples are dry, while the nanostructures should be swollen during the DLS analysis. With regards to BSA loaded samples, the DLS results were similar to pure CHC (Figure 1d, e), whereas the AFM did reveal a more heterogeneous particle size distribution, containing both smaller and larger particles as compared to pure CHC (Figure 1f). It is also worth mentioning that CHC was passed through a 1 μm filter during preparation, with a process yield of 96% ± 1.8 (S.D., n=3), indicating that major parts of the prepared samples were in the nanoscale range and that the analyses are largely representative also of non-filtrated samples, which would be more relevant for applications.
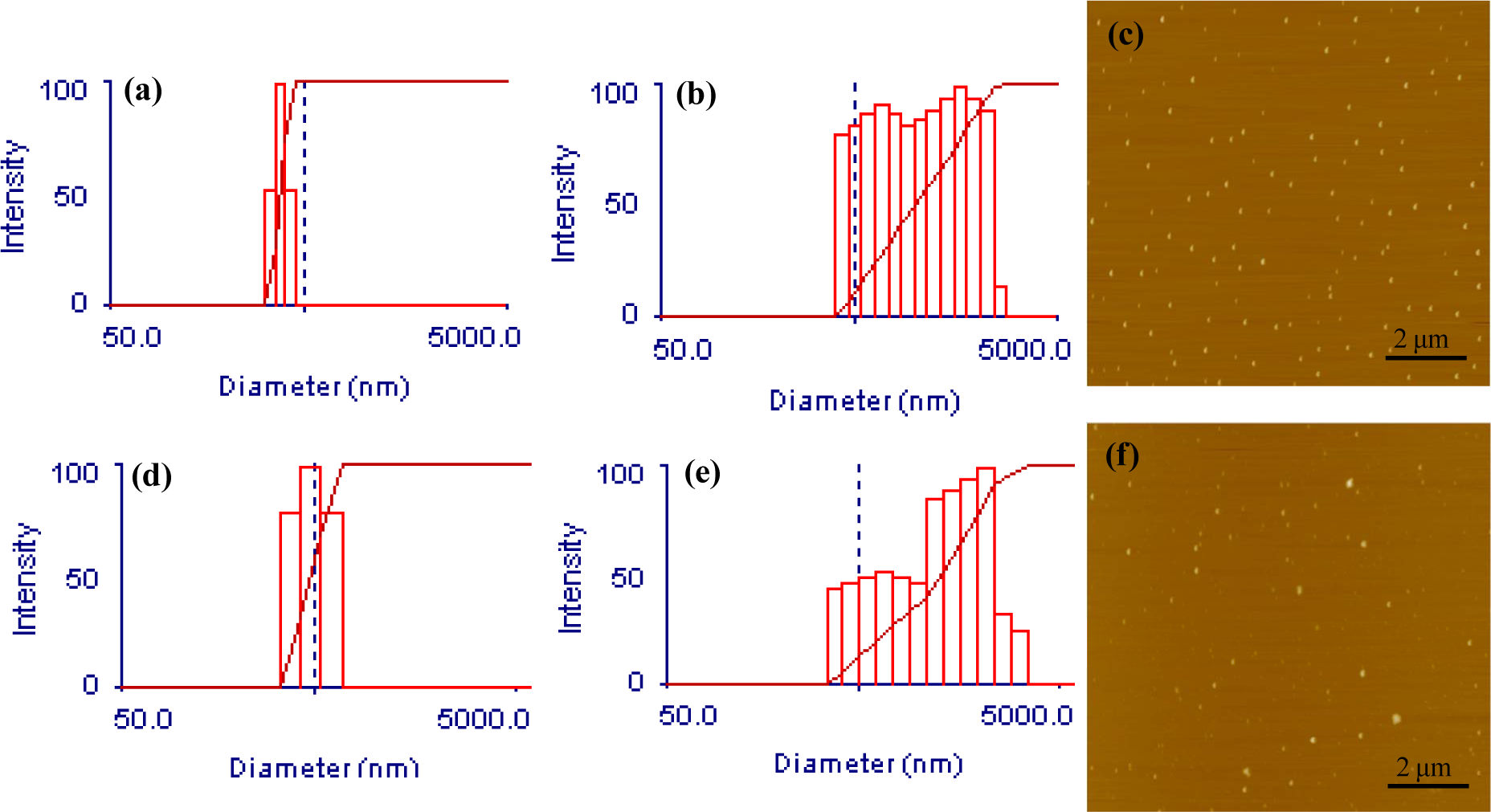
Exemplifying size characterization results for CHC nanocarriers with and without BSA. (a) DLS of CHC with narrow size distribution. (b) DLS of CHC with broad size distribution. (c) AFM of CHC. (d) DLS of BSA loaded CHC with narrow size distribution. (e) DLS of BSA loaded CHC with broad size distribution. (f) AFM of BSA loaded CHC
Zeta potential analysis of CHC nanocapsules revealed highly positive zeta potentials of 42 mV ± 6 (min/max, n=2). For BSA loaded nanocarriers the zeta potential was slightly lower with 33 mV ± 1, but remained highly positive. The zeta potential results thus indicate good colloidal stability of major parts of the formed suspensions and that BSA is loaded in the CHC nanocarriers partly through electrostatic interaction which neutralizes parts of the positive charge of the CHC.
3.2 Protein loading and release
Analysis of the protein loading using FITC-BSA revealed a loading efficiency of 75% ± 1.8 (S.D., n=3) and a very slow release of protein in PBS, pH 7.4 (see Figure 2). After two weeks, only as little as 10% of the actually loaded protein had been released. During the release study it was observed that some aggregation had occurred after one week as some larger dust-like particles were visible in the samples, which might be explained by the fact that CHC possesses an isoelectric point around pH 7. However, such behaviour at physiological pH should not limit biomedical applications. If the CHC nanocarriers are for example injected intravenously or administered orally, the concentration will be much lower and aggregation should thus be circumvented. In the case of macroscopic depot gels, where the nanocarriers could be used to obtain a sustained release, the carriers will be connected or fixated in a matrix so that aggregation would not be expected to influence the formulation performance. The high loading efficiency, achieved by a simple mixing of carrier and protein, in combination with the very slow release clearly suggest that protein-CHC interactions are a factor that should be considered both for further investigations into its utilization in applications but also when designing protein containing CHC formulations where protein release is not the main function.

Free FITC-BSA over time when CHC nanocarriers loaded with FITC-BSA were diluted 10-fold in PBS. Final concentrations: CHC = 0.20%, FITC-BSA 0.025 mg/ml. Error bars indicate one S.D (n = 3).
4. Conclusion
CHC nanocarriers, self-assembled into nanocapsules, could be loaded with a major fraction of the added model protein FITC-BSA through simple mixing of the two. In combination with a very slow release of protein in PBS at pH 7.4 the results clearly indicate the potential of CHC as a protein carrier which may protect protein from degradation and provide a sustained release. However, as chitosan is known to be biodegradable by a range of enzymes where the rate of degradation depends on molecular weight, molecular structure and location in the body [16], the actual release rate in different indications should vary. One particularly interesting area for the use of CHC in protein therapeutics is transmucosal protein delivery, especially in the area of mucosal vaccination using protein-antigens, since hydrophobically modified chitosan has shown strong mucoadhesive properties [17] and chitosan is known to open tight junctions and the ability to increase the permeability of intestinal epithelial cells [18, 19]. In addition to the potential of using CHC as a protein carrier, the results also indicate that the CHC-protein interaction needs to be taken into account when designing protein containing CHC formulations where sustained protein release is not the primary objective, such as the growth factor containing cell scaffolds, since the available amount of protein may otherwise be lower than expected.
Footnotes
5. Acknowledgments
This project is part of the VINN Excellence Centre SuMo Biomaterials (Supermolecular Biomaterials - Structure dynamics and properties). The financial support from the Centre is gratefully acknowledged. Further financial support was acquired from the Swedish Research Council and from the Chalmers Bioscience Program, Chalmers University of Technology. We want to thank Anders Mårtensson at Polymer Technology, Chalmers University of Technology, for his assistance in the AFM analyses and Professor Jan-Erik Löfroth at Surface Chemistry, Chalmers University of Technology, for his help in the DLS and zeta-potential analysis.
