Abstract
Amyloids constitute a class of protein and protein fragments believed to be involved in the pathologies associated with Alzheimer's, Parkinson's and Creutzfeldt-Jakob diseases. These proteins can self-assemble into unique fibrillar structures that are resistant to normal protein degradation. Interesting recent developments in the study of amyloid fibrils demonstrate that they bind carbon allotropes. In this study using single-walled carbon nanotube field-effect transistors (SWCNT-FETs), we show that the fibrillar form of Alzheimer's amyloid β (1–40) and (1–42) peptides specifically bind non-functionalized SWCNT in a saturable manner. Both peptides exhibited near identical binding curves with half-maximal binding concentrations of approximately 12 μg/ml. Binding of the peptides to SWCNTs was diminished by including dimethyl sulphoxide (DMSO) at concentrations that inhibits fibril formation. Lastly a monoclonal antibody (BAM-10), which binds to the N-terminal region of Alzheimer's amyloid fibrils, recognizes the amyloid peptides adhering to SWCNTs in the absence of DMSO, but not in the presence of 75% DMSO. Taken together, these results suggest that the fibrillar form of the Alzheimer's amyloid peptides are specifically binding to SWCNTs.
1. Introduction
Amyloidosis includes neuropathologies such as Alzheimer's, Parkinson's, Huntington's and Creutzfeldt–Jakob disease [1, 2]. Characteristic pathologies of these diseases are tissue deposits of proteinaceous aggregates with fibrillar structures that are resistant to normal proteolysis. These fibrils are often highly ordered β sheet polymers of specific proteins or protein fragments. Although the proteins that form these fibrils do not share significant amino acid sequence homology, the fibrillar structures themselves appear to share similar morphology and mechanisms of toxicity [3]. X-ray diffraction, electron microscopy and solid-state nuclear magnetic resonance (NMR) have shown that most amyloids are long and unbranched fibrils with an organized core structure composed of either parallel or anti-parallel β-sheets [4–6].
Most
Interestingly, many nanomaterials, such as quantum dots, fluorinated nanoparticles and TiO2 nanoparticles, have been shown to bind Aβ amyloids [17, 18]. Manufactured carbon allotropes, such as fullerenes, graphite and multi-walled carbon nanotubes, have also been shown to interact with some Aβ peptides [19–21]. These studies prompted us to examine whether the fibrillar or monomeric form of Aβ40 and Aβ42 peptides bind to single-walled carbon nanotubes using field-effect transistors (SWCNT-FET).
SWCNTs are long, hollow carbon structures with walls formed by one-atom-thick sheets of graphene. SWCNTs show great promise in a wide variety of applications due to their unique physical and chemical characteristics. These one-dimensional wires are the strongest known material and exhibit near ballistic electric transport [22–27] and electronic properties ranging from metallic to semiconducting [28–30]. Therefore, SWCNTs show great potential as the heart of nano- and micro-electronic devices of the near future [31–35].
However, the hydrophobicity and inertness of SWCNTs is an obstacle to functionalizing SWCNT-FET with biological molecules. The conditions required for covalent modification of SWCNTs can require harsh conditions such as sulphuric and nitric acid treatments. Thus, additions to the nanotubes can disrupt the sp2 bonds and compromise their electrical properties [36]. To overcome this barrier, functionalization of the SWCNTs can utilize their hydrophobicity by treating them with reactive hydrophobic linker molecules such as pyrenebutyric acid N-hydroxysuccinimide ester (pyrene-NHS). Pyrenes are hydrophobic polycyclic aromatics that bind avidly to the hydrophobic SWCNTs and they do not adversely affect the electrical properties of their sp2 bonds [37]. The hydrophobic pyrene group anchors the reactive NHS ester to the nanotubes and allows it to react with solution phase molecules. This linker has emerged as the reagent of choice for attaching a wide spectrum of molecules, including antibodies and aptamers to SWCNT-FETs [37–40]. Other hydrophobic molecules that bind SWCNTs include polycyclic drugs and lipids [41, 42].
Previously we have shown that SWCNT-FET can be utilized to measure many biological molecules including IGF-1 [43], glycated human serum albumin [44] and hemorrhagic
2. Materials and Methods
2.1 Materials
Sorted semiconducting single-walled carbon nanotubes (SWCNTs) were obtained from Nano-C, Inc. (Westwood, MA). The SWCNTs were applied to 92 circuits printed on standard 4″ silica semiconductor wafers. Briefly, the semiconducting SWCNTs were made 5% with SDS and applied to functionalized gold electrodes. Peptides were obtained from Peptide Technologies Corp. (Gaithersburg, MD [46]). Aβ amyloid peptides used in this study were CDAEFRHDSGYEVHHQKLVFFAEDVGSNKGAIIGLMV GGVV (Aβ40) and DAEFRHDSGYEVHHQKLVFFAE DVGSNKGAIIGLMVGGVVIA (Aβ42). These peptides correspond to the amino acids 672–710 and 672–712 of the full length amyloid beta A4 protein, respectively (numbering from Uniprot entry P05067). The Aβ40 has a cysteine at the N-terminus, which does not affect the fibril formation [47, 48]. The control peptide used for these studies was TPDAVDKYLETPGDENEHAHFQKAKE RLEAKHRERMSQVM, which corresponds to amino acids 371–410 of the full length amyloid beta A4 protein (numbering from Uniprot entry P05067). The monoclonal antibody BAM-10, which binds to the first 12 amino acids of Aβ (DAEFRHDSGYEV, Sigma Product Information) was purchased from Sigma (St Louis, MO).
2.2 Impedance measurements
The control peptide and Aβ42 were solubilized in 100% DMSO at greater than 10 mg/ml prior to dilution into 50 mM MOPS, pH 7.4. Stock solutions of Aβ40 were made in 50 mM MOPS at 10 mg/ml and kept at RT for at least 24 hr before use to ensure optimal fibril formation [49]. Binding of peptides to the SWCNT-FET was carried out for 60–180 sec. A baseline impedance value for each circuit was first obtained by applying 4 μl of the buffer (or DMSO mixtures) to circuits and recording the initial impedance for approximately 30 sec. Then 4 μl of amyloid peptides in buffer (or DMSO mixtures) were carefully admixed with the buffer on the circuits and changes in impedance were measured for another 90–150 sec. Half-maximal binding concentrations were determined online using BioDataFit.
For antibody recognition of peptides bound to SWCNT-FET circuits, circuits were first incubated with Aβ40 or Aβ42 peptides in either 25 mM MOPS, 150 mM NaCl, pH 7.4 or 75% DMSO, 25 mM MOPS, 150 mM NaCl pH 7.4 for 1 hr at RT in a closed container. Following incubation, the circuits were washed with deionized water (diH2O) and dried. Bound Aβ40 and Aβ42 peptides were detected with 4 μl BAM-10 at 1:250 dilution in 25 mM MOPS, 150 mM NaCl, pH 7.4 for 150 sec following 30 sec baseline determination with 4 μl of the buffer. The impedance value for each measurement was normalized to the corresponding baseline impedance. The impedance of each concentration of amyloid peptide was measured at least in quadruplicate using a fresh circuit for each measurement.
A source/drain bias of approximately 100 mV was maintained throughout the measurements of the electrical signal and the pulse width was 1 sec. The reference electrode is the back (bottom) side of the grounded wafer. The device uniformity was not optimized for entire wafers, but individual circuits used for the assays were carefully evaluated before experiments. The selected SWCNT-FET circuits ranged typically around ten in on-off ratio. The electrical properties of the samples binding the SWCNT-FET were measured using a low current measurement system (MediSourcePlus Inc, S. Korea) that makes electrical contact to the source and drains electrodes of the SWCNT-FET. The transfer characteristics of this circuit design were previously characterized for detection of prostate specific antigen and IGF-1 [50, 43]. The response in the electrical signal is typically in the range of 5 to 40% in the normalized units.
3. Results and Discussion
The six-member ring structure of the SWCNTs carbon-carbon bonds are thought to be similar to aromatic sp2 bonds [37]. Apart from making the nanotubes highly resistant to chemical modification, these bonds also make the nanotubes very hydrophobic. The SWCNTs are not soluble in aqueous media and aggregate readily through extensive van der Waals interactions. These aggregates can be partially brought into suspension by using sonication in the presence of strong amphiphilic detergents, such as sodium dodecyl sulphate (SDS) or cholates [51]. Similarly, hydrophobic interactions are thought to play a major role in the formation of β-sheet polymers from individual Aβ peptides [52–54]. These hydrophobic interactions are also thought to be a vital component for the interactions of fullerenes with Aβ peptides [21].
Fullerene is a carbon allotrope that has the same carbon-carbon bond organization as SWCNTs but is arranged in a spherical structure [55]. The chemical similarities of the fullerenes to the SWCNTs prompted us to examine whether the nanotubes also bound to Aβ peptides - and we chose to examine this using SWCNT-FET.
Assessing specific binding of any charged molecule to SWCNT-FET is a challenge. In general, the current carried by SWCNT-FET is influenced by charged species in its immediate environment and this is the basis for molecular detection by field-effect transistors. However, the presence of any charged species in the media surrounding the SWCNTs can generate a signal on the SWCNT-FET. However, if the charged species is not binding specifically to the SWCNTs, the distribution of the charged species around the SWCNT-FET would be random. The signal will be entirely dependent on the concentration of the added charges. Saturation of the signal then would not be reached until the entire solution surrounding the SWCNT-FET was saturated with the charged species. Conversely, a charged species that binds specifically to the SWCNT-FETs will likely saturate binding sites on the nanotubes before saturating the surrounding solution.
Figure 1 shows our results of adding increasing amounts of Alzheimer's peptides to the SWCNT-FET and monitoring the change in the impedance. In all cases the impedance increases with increasing concentrations of peptides. Both Aβ40 and Aβ42 show near identical saturating binding curves with half-maximal saturation at approximately 12 μg/ml (or 2.6 ×106 M of peptide). The control used for these experiments is a 40-mer peptide from a region of the amyloid precursor protein (APP), N-terminal to the amyloid forming sequence. The control peptide did not demonstrate signal saturation until at a concentration exceeding 300 μg/ml (data not shown). Therefore, the control peptide is either exhibiting weaker binding to the SWCNT-FET than the Aβ40 and Aβ42 peptides, or it is saturating the SWCNT surroundings at those high concentrations.

Binding of Aβ340, Aβ342 and control peptides to SWCNT-FET. Peptides Aβ340 (•, solid line) Aβ342 (▪, solid line) and control peptide (□, dotted line) were solubilized as described in Materials and Methods and applied to the circuits. Measurements were carried out for 120 sec. Each condition was measured in at least quadruplicate. Values are the average +/- SEM).
To further characterize the binding of these peptides to SWCNTs, we performed binding studies in dimethyl sulphoxide (DMSO). DMSO is commonly used in amyloid β research because it influences the extent of fibril formation by these peptides. At high concentrations of DMSO, the Aβ40 and Aβ42 peptides are predominantly monomers or oligomers, and decreasing the DMSO concentration increases the fibrillar form [56, 57].
The changes in impedance of Aβ40, Aβ42 and control peptides with increasing DMSO concentration are shown in Figure 2. The control peptide showed a minimal decrease in impedance with increasing DMSO concentrations, indicating that it does not have a defined structure like the Aβ40 and Aβ42 peptides. At less than 10% DMSO, the Aβ40 and Aβ42 showed maximum impedance, which suggests that the fibrillar form of Aβ40 and Aβ42 are binding to the SWCNTs. This interpretation is consistent with previous studies showing that low DMSO concentrations (≤10%) have a minimal effect of the fibril formations of both Aβ40 and Aβ42 [58–61]. The impedance of Aβ40 and Aβ42 then decrease with increasing DMSO concentrations until reaching a minimum at 50% DMSO. Garzon-Rodriguez et al. [59] demonstrated an increase in solvent exposure of an internal tyrosine and a fluorescent label on the N-terminus of Aβ40 in the presence of increasing concentrations of DMSO. This is consistent with a DMSO concentration dependent loss of Aβ40 fibrils with a concomitant increase in monomeric peptide. Garzon-Rodriguez et al. [59], however, noted that the midpoint of the transition was reached at 50% DMSO, whereas 50% and higher concentrations of DMSO result in the weakest binding of Aβ40 and Aβ42 peptides to SWCNT-FET. These results could indicate that the SWCNTs are binding to more mature fibrils, which are less stable in high concentrations of DMSO [56, 57, 59].

Effect of increasing DMSO concentrations on the binding of Aβ340, Aβ342 and control peptides at 75 μg/ml to SWCNT-FET. Peptides Aβ340 (•, solid line) Aβ342 (Δ, solid line) and control peptide (□, dotted line) were solubilized as described in Materials and Methods and applied to the circuits at the indicated DMSO/diH2O concentrations. Measurements were carried out for 180 sec. Each condition was measured in at least quadruplicate. Values are the average +/- SEM.
To further analyse the interactions of the Aβ fibrils to SWCNTs, we determined whether BAM-10, a monoclonal antibody that recognizes the N-terminal amino acids of Aβ [62], was able to detect the Aβ40 and Aβ42 peptides bound to SWCNTs. Interestingly, this antibody detects amyloid plaque burden in brain tissues [63] and can reverse memory loss in a transgenic mouse model of Alzheimer's disease, possibly by neutralizing Aβ assemblies that impair cognitive function in the brain [62].
Figure 3 shows the reactivity of the antibody for Aβ40 and Aβ42 bound to SWCNT-FET in the presence and absence of 75% DMSO. In these experiments we were attempting to detect antibody binding to Aβ peptides already adhering to the SWCNTs. Any change in signal on the SWCNT-FET would therefore be due to the antibody being in close proximity to the SWCNT current, i.e., binding to the Aβ peptides that are bound to the SWCNTs. BAM-10 recognizes both Aβ40 and Aβ42 bound to SWCNT, but poorly detects the same peptides loaded on to SWCNT-FET circuits in 75% DMSO. These results further support the conclusion that the fibrillar form of Aβ40 and Aβ42 are specifically binding to the SWCNTs, and might include pathogenic Aβ fibrils as well. Furthermore, direct antibody recognition of Aβ fibrils bound to SWCNT-FET slightly increased their detection levels and opens up possibilities of using primary and secondary antibody pairs to further amplify the signal, which can lead to more sensitive detection of Aβ fibrils on SWCNT-FET.
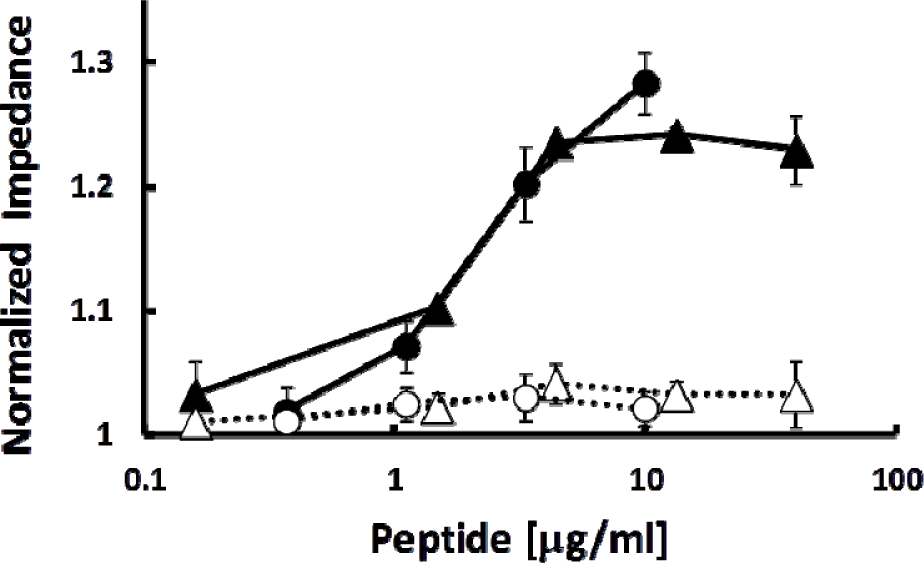
Binding of BAM-10 to Aβ340 and Aβ342 in the presence or absence of 75% DMSO. Peptides Aβ340 (•, solid line) Aβ342 (Δ, solid line) in 25 mM MOPS, 150 mM NaCl, pH 7.4 and Aβ340 (○ dotted line) Aβ342 (Δ dotted line) in 75% DMSO, 25 mM MOPS, 150 mM NaCl were applied to the circuits at the indicated concentrations. After incubation and washing BAM-10 antibody was applied to the circuits as described in Material and Methods. Measurements were carried out for 180 sec. Each condition was measured in at least quadruplicate. Values are the average +/- SEM.
In this study we were not able to determine the specific sequence(s) in the Aβ peptides that are involved in the binding or whether the SWCNTs affected the formation of amyloid fibrils. Fullerenes were shown to inhibit fibril formation by binding to the hydrophobic KLVFF sequence [21], while multiwalled carbon nanotubes and graphite were shown to promote their formation [19, 64]. However, computer simulations have suggested that SWCNTs can inhibit the formation of amyloid from Aβ peptide fragments 16–22 and 25–35 [65, 66], demonstrating that further studies are required to address this issue.
Footnotes
4. Acknowledgments
This paper was supported in part by the Maryland Industrial Partnership (MIPS) programme, no. 4319 and by the Maryland Technology Development Corporation's Maryland Technology Transfer and Commercialization Fund.
