Abstract
The effect of carbon nanotubes on the catalytic properties of Rh-Mn-Li/SiO2 catalysts was investigated for CO hydrogenation. The catalysts were comprehensively characterized by means of X-ray power diffraction, N2 sorption, transmission electron microscope, H2–temperature-programmed reduction, CO–temperature-programmed desorption, temperature-programmed surface reaction, and X-ray photoelectron spectroscopy. The results showed that an appropriate amount of carbon nanotubes can be attached to the surface of the SiO2 sphere and can improve the Rh dispersion. Moderate Rh-Mn interaction can be obtained by doping with the appropriate amount of carbon nanotubes, which promotes the formation of strongly adsorbed CO and facilitates the progress of CO insertion, resulting in the increase in the selectivity of C2+ oxygenate synthesis.
Keywords
• The effect of carbon nanotubes on the catalytic properties of Rh-Mn-Li/SiO2 catalysts was investigated.
• The doping of carbon nanotubes can improve the Rh dispersion and Rh-Mn interaction.
• The moderate Rh-Mn interaction can promote the formation of strong adsorbed CO.
• The moderate Rh-Mn interaction can facilitate the progress of CO insertion.
• The carbon nanotube-promoted Rh-Mn-Li/SiO2 catalysts can improve the selectivity of C2+ oxygenates.

Introduction
In recent years, one of the largest social challenges is the demand for alternative sources of liquid fuels, which would reduce the depletion of fossil fuel resources and greenhouse gas emission. Ethanol has attracted increasing attention as a clean fuel or as an additive to gasoline.1,2 Coal (syngas) ethanol synthesis is one of the most promising technologies, since syngas can be derived from biomass, coal, or natural gas.3,4
Rh-based catalysts have been found to exhibit excellent performance in the catalytic synthesis of C2 oxygenates (e.g. ethanol, acetaldehyde) from syngas. The influence of various supports and auxiliaries on the catalytic activity of Rh-based catalysts has been widely reported.5-15 Following many dedicated efforts, SiO2-supported Rh-Mn-Li catalysts have been found to give excellent activity and C2 oxygenate selectivity,16-18 with the promoters Mn and Li playing an important role in this behavior. 19 Nevertheless, the pace of catalyst optimization continues and catalysts with higher catalytic performance are still pursued by research workers.
Nowadays, more and more attention has been directed to the application of carbon nanotubes (CNTs). 20 CNTs have a series of special properties as a new type of carbon material with large specific surface areas, uniform pore structures, excellent electronic conductivity, and a high ability for hydrogen adsorption. These characteristics make CNTs widely used in the field of catalysis as promising carriers and auxiliaries.21,22 Zhang et al. 23 reported a highly active CNT-supported Cu-based catalyst for methanol synthesis by CO/CO2 hydrogenation. Their results showed that, compared with active carbon and γ-Al2O3 supports, the Cu-based catalysts supported on CNTs can obtain maximum space time yield at a lower temperature. Dong et al. 24 investigated the effects of doping Cu-based catalysts using CNTs on the catalytic performance for CO/CO2 hydrogenation. It was found that the doping using CNTs can significantly improve the specific surface area of the catalyst, especially the specific surface of Cu. The CNTs are the adsorbent, activator, and repository of the H2, which is conducive to the formation of a hydrogen-rich atmosphere around the active site, thus improving the reaction performance of the CO/CO2 hydrogenation.
Here, aiming to further improve the catalytic performance of the Rh-Mn-Li/SiO2 catalyst, various amounts of CNTs were added and the modified catalysts tested for C2 oxygenate synthesis from syngas. Moreover, insights into the effect of the CNTs on the Rh-Mn-Li/SiO2 catalyzed CO hydrogenation were demonstrated.
Results and discussion
The support of SiO2 was prepared by the Stöber method, and the CNTs were bought from Aldrich Chemical Reagent Co., Ltd. The Rh-Mn-Li catalysts were prepared by co-impregnation to incipient wetness of SiO2 with an aqueous solution of the metal precursors, and the CNT was added as an additive into the solvent. The weight percent of CNTs in the catalysts was 2.5%, 5.0%, and 10.0%, respectively. The obtained catalysts were denoted as RML/SiO2, RML-2.5CNT/SiO2, RML-5CNT/SiO2, and RML-10CNT/SiO2, respectively. For comparison, the Rh-Mn-Li catalyst supported on CNTs was also prepared by co-impregnation, which was denoted as RML/CNTs.
Catalytic activities
Table 1 compares the catalytic performance of the catalysts for CO hydrogenation at 300 °C. It can be seen that the addition of different amounts of CNTs promoter modified the catalytic properties of the Rh-Mn-Li/SiO2 catalyst. Compared with the Rh-Mn-Li catalyst supported on CNTs (RML/CNTs), the catalyst supported on SiO2 (RML/SiO2) showed higher catalytic activity, along with lower selectivity for C2+ oxygenates. An appropriate amount of CNTs doping can partially reduce the catalytic activity of RML/SiO2, but it can improve significantly the formation of C2+ oxygenates. As observed, the catalysts RML-2.5CNT/SiO2 and RML-5CNT/SiO2 provided similar CO conversion and higher selectivity for C2+ oxygenates. Correspondingly, the yields of the C2+ oxygenates increased to the higher value range of 266.3–287.6 g kg−1 h−1. However, when the amount of CNTs reached 10 wt%, the CO conversion and selectivity for C2+ oxygenates of the RML-10CNT/SiO2 catalyst clearly decreased. These results show that the amount of CNTs doping can partially inhibit the activity of CO hydrogenation, but it can improve the selectivity toward C2+ oxygenates and increase their yields.
CO hydrogenation performance of different catalysts.

Reaction conditions:
C2+ HC denotes hydrocarbons containing two and more carbon atoms.
C2+ Oxy denotes oxygenates containing two and more carbon atoms.
Typical time-dependent changes of the CO conversion and selectivity for C2+ oxygenates using representative RML-5CNT/SiO2 catalyst are shown in Figure 1. It can be seen that the CO conversion and C2+ oxygenates’ selectivity decreased distinctly in the early stages of the reaction. However, the catalyst was stable after 10 h and the catalytic activity was steady until 150 h.

The catalytic performance versus time-on-stream for the RML-5CNT/SiO2.
Structural and textural properties
The X-ray power diffraction (XRD) patterns of the catalysts are shown in Figure 2. The XRD patterns of RML/SiO2, RML-5CNT/SiO2, and RML-10CNT/SiO2 showed a broadened peak at 2

XRD profiles of different catalysts: (a) RML/SiO2, (b) RML-5CNT/SiO2, (c) RML-10CNT/SiO2, and (d) RML/CNTs.
N2 sorption was carried out to further characterize the textural properties of the supports and the supported catalysts. As shown in Table 2, the Brunauer–Emmett–Teller (BET) surface area of SiO2 was 8.9 m2 g−1, similar to the surface areas of silica prepared by the Stöber method reported by Szekeres et al. 26 and Hsu et al. 27 A relatively large BET surface area (288.2 m2 g−1) was obtained on the CNT carrier. Upon being loaded with metal components, there was a decrease in the surface area and the pore volume for the supported catalysts RML/SiO2 and RML/CNTs. In addition, compared with the catalyst of RML/SiO2, the surface areas of the CNT-promoted Rh-Mn-Li/SiO2 catalysts increased because of the high surface area of CNTs.
Specific surface areas (
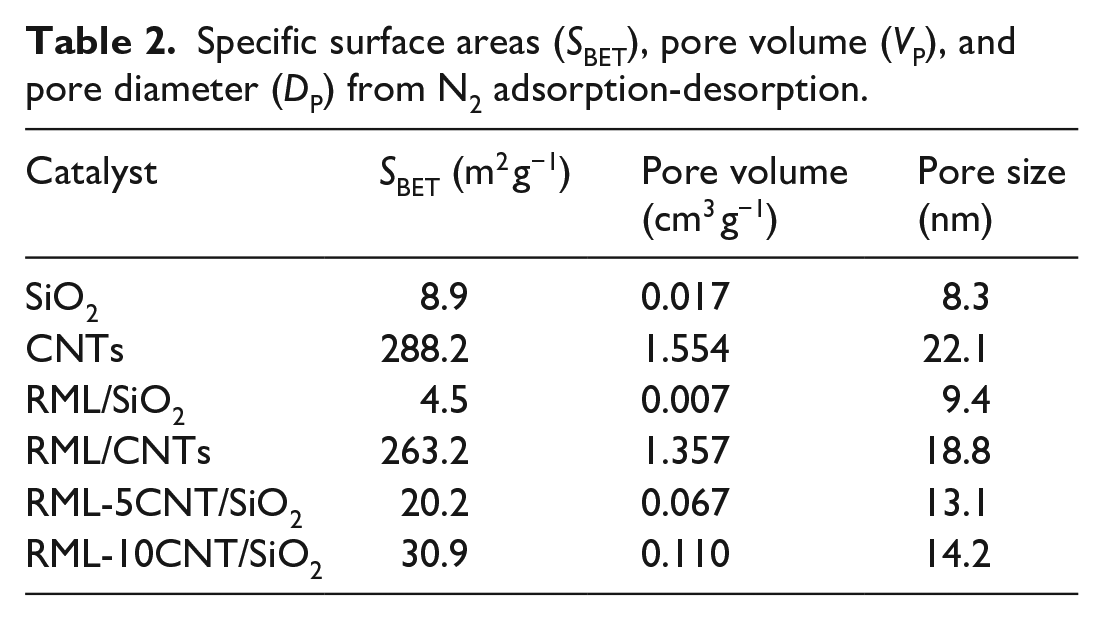
Generally, most research suggests that supports with a high surface area favor the dispersion of metal species, further improving the catalytic conversion of syngas to oxygenates. 28 However, the Rh-based catalyst supported on the CNT carrier with a high surface area did not show the higher activity compared with that supported on SiO2. In fact, Fan et al. 29 found that the high surface area is not the decisive factor for the dispersion of metal species in the carbon-supported Rh-based catalyst. It is suggested that the surface chemical properties of the supports play an important role in promoting CO hydrogenation in addition to the metal dispersion in these multi-component promoted Rh catalysts. In our previous work, we have also inferred that there are some other properties rather than the surface area, such as the role of surface hydroxy groups on the silica, which affect the catalytic performance of the Rh-Mn-Li/SiO2 catalysts. 30
Figure 3 shows the transmission electron microscope (TEM) micrographs and the corresponding Rh particle size distributions of the catalysts. First, as shown in Figure 3(a), the particles of SiO2 were monodispersed and spherical with a mean size of ~500 nm. The Rh nanoparticles were highly dispersed on the surface of the spheres, and the particle size fell in a wide range from 1 to 4 nm. If the pure CNT was used as the carrier (Figure 3(d)), the Rh particles were distributed inside and outside the carbon nanotubes with a mean size of 0.4 nm. For the CNT-promoted Rh-Mn-Li/SiO2 catalysts, it can be seen that partial carbon nanotubes can be attached to the surface of the SiO2 sphere, and the Rh particles can be distributed homogeneously in the interface of the SiO2 and CNTs. The results also showed that the Rh particle distribution on the surface of CNT-promoted catalysts was more dispersed compared to that on the RML/SiO2, and the average Rh particle sizes of RML-5CNT/SiO2 and RML-10CNT/SiO2 were 1.1 and 0.5 nm, respectively. Furthermore, the TEM micrograph of RML-10CNT/SiO2 also indicated that the CNT and SiO2 spheres would be more separate when the doping amount of CNTs reached 10 wt%, and more Rh particles are supported on the CNTs. This indicated that the synergistic effect of CNTs should be weakened when the CNT amount exceeded the optimum amount (5 wt%).

TEM micrographs and the corresponding Rh particle size distributions of (a) RML/SiO2, (b) RML-5CNT/SiO2, (c) RML-10CNT/SiO2, and (d) RML/CNTs.
H2–temperature-programmed reduction
Figure 4 shows the temperature-programmed reduction (TPR) profiles of the catalysts. There are three distinct peaks showing H2 consumption in the TPR profiles. According to the previous results, the high temperature peak is ascribed to the reduction of MnO2.31,32 The two peaks at low temperature are ascribed to the reduction of Rh2O3 weakly contacting with Mn species (denoted as Rh(І)) and of Rh2O3 strongly contacting with Mn species (denoted as Rh(II)), respectively.17,33

TPR profiles of the catalysts: (a) RML/SiO2, (b) RML-5CNT/SiO2, (c) RML-10CNT/SiO2, and (d) RML/CNTs.
For the catalyst RML-5CNT/SiO2, the peak area ratio of Rh(II)/Rh(I) was higher than that of RML/SiO2 and RML-10CNT/SiO2, indicating that the appropriate content of CNTs can enhance the interaction of Rh and Mn. For the catalyst RML/CNTs, the reduction peaks of the Rh2O3 and MnO2 overlapped, which pointed to a strong interaction between the Rh2O3 and Mn species on the CNT support. According to the viewpoint proposed by Ding and co-workers, 34 it is suggested that a moderate Rh-Mn interaction is favorable for the production of C2+ oxygenates.
CO–temperature-programmed desorption
The temperature-programmed desorption (TPD) profiles for CO after the CO is adsorbed on the catalyst are shown in Figure 5. As shown in the patterns, there are two main desorption peaks in the ranges 60 °–150 °C and 500 °–700 °C, respectively. It is generally accepted that the low temperature peak is attributed to the desorption of weakly adsorbed CO, and the high temperature peak should be assigned to the desorption of strongly adsorbed CO. 35 There was a large desorption peak of weakly adsorbed CO for the catalyst supported on pure CNTs. However, the intensity of the low temperature peak became very small for the SiO2-supported or CNT-promoted catalysts. However, when compared with the catalyst RML/SiO2, the intensity of the high temperature peak increased remarkably for the CNT-promoted catalysts.

CO-TPD profiles of different catalysts: (a) RML/SiO2, (b) RML-5CNT/SiO2, (c) RML-10CNT/SiO2, and (d) RML/CNTs.
It has been reported in the literature that strongly adsorbed CO species are precursors for the CO insertion reaction. 36 According to the mechanism for the synthesis of C2+ oxygenates from syngas, the adsorbed CO dissociation and hydrogenation to produce CHx species is likely to be the first step. Then, the CHx species can be converted to the C2+ oxygenates precursor CHxCO by CO insertion. Thus, considering that the largest desorption peak of strongly adsorbed CO was obtained for the catalyst RML-5CNT/SiO2, this is consistent with the higher C2+ oxygenates’ selectivity.
Temperature-programmed surface reaction
The temperature-programmed surface reaction (TPSR) experiments over Rh-based catalysts with different CNT loadings are shown in Figure 6. It can be seen that a sharp CH4 peak centered at ~265 °C appeared in the profiles, and the location of this for all the catalysts was almost the same. With the doping of CNTs, the peak area initially decreased and then increased when the amount of CNTs reached 10 wt%.

TPSR profiles of different catalysts: (a) RML/SiO2, (b) RML-5CNT/SiO2, and (c) RML-10CNT/SiO2.
As mentioned above, the adsorbed CO will first dissociate into C* species which is then hydrogenated to form CHx intermediates. The CHx species then undergo formation of C2+ oxygenates precursor CHxCO by CO insertion, or hydrogenation to form CH4. The amount of CH4 produced on the RML-5CNT/SiO2 catalyst was the least, indicating that less adsorbed CO dissociated and could then participate in the insertion reaction, which in turn promotes the formation of C2+ oxygenates.
X-ray photoelectron spectroscopy
X-ray photoelectron spectroscopy (XPS) studies have been performed to gain information about the chemical states of the Rh on the catalyst surfaces. It can be seen from Figure 7(A) that two peaks are observed at 309.8 and 314.6 eV. These can be attributed to Rh 3d5/2 and Rh 3d3/2 in all the fresh catalysts, suggesting that the Rh species exist as Rh2O3. The corresponding spectra after reduction are shown in Figure 7(B). For all the catalysts, the binding energy of the Rh 3d5/2 peak was located at 306.7–307.8 eV, which can be attributed mainly to Rh0 species. It is obvious that the dominant Rh 3d5/2 peak of the RML-5CNT/SiO2 and RML-10CNT/SiO2 shifted toward higher values when compared with that of RML/SiO2, indicating that a lower electronic density of Rh particles was present in the catalysts RML-5CNT/SiO2 and RML-10CNT/SiO2. 37 That is to say, the Rh0 species was accompanied by some partially positively charged Rhδ+ species. Probably the presence of such oxidized Rh species is indicative of an interaction of some Rh oxide particles with Mn species since the electrons of rhodium atoms can be attracted by the electron-withdrawing effect of Mn, consistent with the result of H2-TPR.

Rh 3d XPS patterns of fresh (A) and reduced (B) catalysts: (a) RML/SiO2, (b) RML-5CNT/SiO2, (c) RML-10CNT/SiO2, and (d) RML/CNTs.
The doping using CNTs enhanced the interaction of Rh and Mn, which promoted the formation of Rhδ+ species. Since Rh+ ions are the active sites of CO insertion, the doping of CNTs facilitates the progress of CO insertion, resulting in the increase in the selectivity of C2+ oxygenate formation.
Conclusion
The effects of the loading of the CNT promoter on the catalytic performance of the Rh-Mn-Li/SiO2 catalyst used for the synthesis of C2+ oxygenates from syngas were investigated. The results showed that the CNTs promoter greatly affected the catalytic performance: CNTs partially inhibited the hydrogenation activity of CO, but it improved the selectivity of C2+ oxygenate formation and so increased the yield of the C2+ oxygenates. The optimized results were obtained by adding 5 wt% CNTs to the catalyst for CO hydrogenation and achieved the best catalytic performance with a maximum yield of C2+ oxygenates of 287.6 g kg−1 h−1.
The texture characterization results showed that the appropriate amount of CNTs can be attached to the surface of the SiO2 sphere so improving the dispersion of Rh particles at the interface of SiO2 and CNTs. Combined with the other characterization results, it was shown that moderate Rh-Mn interaction can be obtained by doping with the appropriate amount of CNTs so promoting the formation of strongly adsorbed CO, and facilitating the progress of CO insertion, resulting in an increase in the selectivity for C2+ oxygenate formation.
Experimental section
Catalyst preparation
SiO2 was prepared by the Stöber method as reported in Szekeres et al. 26 In a typical synthesis, the solution A was prepared by mixing 21 mL tetraethyl orthosilicate (TEOS) (99.5%, SCRC) with 50 mL anhydrous ethanol (99.7%, SCRC); the solution B was a mixed solution of 76 mL NH3·H2O (26 vol.%, SCRC) and 200 mL anhydrous ethanol. Second, the solution A was added slowly into the solution B in a flask under rapid stirring at 25 °C and the mixture allowed for 4 h. Then, this mixture was separated centrifugally at 7000 rpm. Finally, the collected product was washed with de-ionized water three times and dried at 70 °C for 12 h. Before use, it was calcined in static air at 550 °C for 4 h. The CNTs were bought from Aldrich Chemical Reagent Co., Ltd.
RhCl3 hydrate (Rh ~39 wt%, Fluka), Mn(NO3)2·6H2O (99.99%, SCRC), LiNO3 (99.95%, SCRC), CNTs, and the SiO2 mentioned above were used in the catalyst preparation. Catalysts were prepared by co-impregnation to incipient wetness of SiO2 (1.0 g) with an aqueous solution of the precursors of the promoters, followed by drying at 90 °C for 4 h, and then at 120 °C overnight before being calcined in air at 350 °C for 4 h. The weight percent of CNTs in the catalysts was 2.5%, 5.0%, and 10.0%, respectively. The obtained catalysts were denoted as RML/SiO2, RML-2.5CNT/SiO2, RML-5CNT/SiO2, and RML-10CNT/SiO2, respectively. For comparison, the Rh-Mn-Li catalyst supported on CNTs was also prepared by co-impregnation, which was denoted as RML/CNTs. For all the catalysts, Rh loading was 1.5 wt% and the weight ratio of Rh:Mn:Li = 1.5:1.5:0.07.
Testing of the catalytic activity
CO hydrogenation was performed in a fixed-bed micro-reactor with length ~350 mm and internal diameter ~5 mm. The catalyst (0.3 g) diluted with inert α-alumina (1.2 g) was loaded between quartz wool and axially centered in the reactor tube, with the temperature monitored by a thermocouple close to the catalyst bed. Prior to reaction, the catalyst was heated to 400 °C (heating rate ~3 °C min−1) and reduced with H2/N2 (molar ratio of H2/N2 = 1/9, total flow rate = 50 mL min−1) for 2 h at atmospheric pressure. The catalyst was then cooled down to 300 °C and the reaction started as the gas flow was switched to a H2/CO mixture (molar ratio of H2/CO = 2, total flow rate = 50 mL min−1) at 3 MPa. All post-reactor lines and valves were heated to 150 °C to prevent product condensation. The products were analyzed for both hydrocarbons and oxygenates on-line (FL GC 9720) using a HP-PLOT/Q column (30 m, 0.32 mm ID) with detection with a flame ionization detector (FID) and a TDX-01 column with a thermal conductivity detector (TCD). The CO conversion was calculated based on the fraction of CO that formed carbon-containing products according to: %Conversion = (∑
Catalyst characterization
The XRD experiments were carried out with a PANalytical X’Pert diffractometer operated at 40 kV and 40 mA using Ni β-filtered Cu–Kα (
H2 TPR of the catalysts were carried out in a quartz micro-reactor. 0.1 g of the sample was first pretreated at 350 °C in O2/N2 (molar ratio of O2/N2 = 1/4) for 1 h prior to a TPR measurement. During the TPR experiment, H2/N2 (molar ratio of H2/N2 = 1/9) mixture was used at 50 mL min−1 and the reduction temperature ramped from 50 °C to 500 °C (10 °C min−1) while the effluent gas was analyzed with TCD.
CO temperature-programmed desorption (CO-TPD) was carried out in a quartz microreactor. The catalyst (0.1 g) was reduced in situ for 2 h at 400 °C in H2/N2 (molar ratio of H2/N2 = 1/9), followed by flushing with a He flow for 30 min at the same temperature before cooling down to room temperature. The next step was CO adsorption at room temperature for 30 min and then the gas was swept again with He for 3 h. Subsequently, the sample was heated in a flowing He stream (50 mL min−1) up to 750 °C at a rate of 10 °C min−1 with a quadrupole mass spectrometer (QMS, Balzers OmniStar 200) as the detector to monitor the desorbed species.
The TPSR experiments were carried out as follows: after the catalyst was reduced at 400 °C in H2/N2 (molar ratio of H2/N2 = 1/9) for 2 h, it was cooled down to room temperature and CO was introduced for adsorption for 0.5 h; afterward, the H2/N2 mixture was swept again, and the temperature was increased at the rate of 10 °C min−1 with a QMS as the detector to monitor the signals of CH4 (
XPS measurements were performed using a Kratos Axis Ultra DLD spectrometer equipped with an Al-Kα (1486.6 eV) X-ray source. In these experiments, samples were reduced in situ at 400 °C for 1 h under 200 mbar H2 pressure. The binding energies were calibrated relative to the C 1s peak from carbon contamination of the samples at 284.9 eV to correct for contact potential differences between the sample and the spectrometer.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was financially supported by the National Natural Science Foundation of China (21808142) and Shanghai Institute of Technology (ZQ2018-3 and CHJJ-7).
