Abstract
Purpose
We evaluated intradetrusorial OnabotulinumtoxinA (Onabot/A) treatment protocols in patients with idiopathic overactive bladder (OAB), in order to assess the care of patients before, during and after treatment.
Methods
In 64 OAB patients injected with Onabot/A, we reviewed the length of the hospital stay, frequency of catheterization, frequency of intraoperative and postoperative complications, and patients’ satisfaction to the proposed treatment protocol (as assessed by VAS). We also compared the results of the 3-day voiding diary, uroflowmetry with postvoid residual urine (PVR) and VAS to score the bother of urinary symptoms on quality of life (QoL) before and after treatment.
Results
Twenty-one patients were firstly treated in an ‘inpatient’ setting. The mean ± SD duration of hospitalization and catheterization was 39.4 ± 12.6 and 37.8 ± 10.6 h, respectively. The mean ± SD VAS values of treatment satisfaction and of bother of urinary symptoms on QoL were 6.3 ± 1.1 and 8.2 ± 1.3, respectively. The mean ± SD PVR value was 74.3 ± 15.2 ml. Frequency of UTIs was 2.4 ± 1.6. Forty-three patients were treated on an outpatient basis; the mean ± SD duration of catheterization, the ‘outpatient’ stay and the mean ± SD frequency of UTIs were lower than those of patients treated in an inpatient setting. The mean ± SD VAS value to score QoL was high.
Conclusions
Intradetrusorial Onabot/A injection is a simple and fast procedure that can be easily carried on in an outpatient setting under local anesthesia, with low rates of intraoperative and postoperative complications.
Introduction
Overactive bladder syndrome (OAB) is a symptom syndrome characterized by urinary urgency, with or without urgency incontinence, usually with urinary frequency and nocturia, in the absence of an underlying metabolic or pathologic condition (1). The worldwide prevalence is about 10.8% and 49 million people are estimated to suffer from this condition in Europe alone. Sixty percent of patients are represented by females (2). OAB strongly affects the quality of life (QoL) of patients and often is accompanied by alterations of the psychological status, such as anxiety and depression (3). First-line treatment of OAB is constituted by anticholinergics per os, particularly tolterodine, solifenacin and fesoterodine, associated with bladder re-training and fluids restriction (4). Indeed, it has been recently observed that treatment with anticholinergics shows a limited efficacy and can be affected by intolerable side effects, which both reduce adherence and persistence to treatment in the medium and long-term follow-up (5).
Recently, intradetrusorial injections of Onabotulinumtoxin A (Onabot/A-Botox®, Allergan Ltd, Irvine, California, USA) has been licensed in both USA and Europe as second-line treatment for adults affected by idiopathic, refractory OAB with urinary incontinence (6). This has been due to the positive results obtained in multiple randomized trials with the use of Onabot/A in idiopathic OAB patients, which has definitely confirmed the high efficacy and tolerability of this kind of minimally invasive treatment (7–9). Nevertheless, to date, we still need to establish what is the best Onabot/A treatment's protocol for patients affected by idiopathic OAB, and what is the more appropriate care of patient before, during and after treatment.
In this retrospective study, we evaluated how the applied treatment protocols changed along time by using Onabot/A intravesical injections in patients affected by idiopathic OAB, in order to propose a standardized treatment's protocol. At the same time, we wanted to evaluate the efficacy and safety of Onabot/A injections in these patients in a long-term follow-up.
Methods
Between January 2001 and December 2014, 87 patients older than 18 years of both sexes, without any congenital or acquired pathology of the muscular system, affected by OAB symptoms, with or without urgency urinary incontinence have been selected and treated. Patients affected by OAB due to bladder outlet obstruction for benign prostatic enlargement or urogenital prolapse and those with recurrent urinary tract infections (UTIs) have been excluded.
Baseline evaluation
After giving their written informed consent, patients underwent history and physical examination, a 3-day voiding diary, with the recording of day-time and night-time urinary frequency and daily episodes of urgency and urgency incontinence, to be completed 7 days before treatment;
urinalyses and cultures at least 5 days before treatment;
interruption of therapy with antiplatelets and anticoagulants at least 10 days before treatment;
wash out of anticholinergics and of other drugs used to control urinary incontinence at least 3 weeks before treatment;
Visual analog scale to score the bother of urinary symptoms on QoL (10);
Uroflowmetry with the evaluation of postvoid residual volume (at least two consecutive tests);
Urodynamics: it was performed in patients with complicated OAB.
Treatment's technical protocol
From the beginning of intradetrusorial injection of Onabot/A at our Clinic, the treatment protocol went through several modifications due to the increasing experience and observations of the results.
In the first stage, selected patients have been treated in as for in-patient setting, in the operating room and under general anesthesia of brief duration. Indwelling catheter was recommended for at least 24 h, and then removed at discharge or later.
More recently and as current clinical practice, patients have been treated as for out-patients setting, according to the following protocol. After confirming negative urinalyses and cultures, an antibiotic prophylaxis is administered (generally ciprofloxacin 500 mg orally). Soon after, patients undergo an intravesical local anesthesia with lidocaine 2% diluted in 50 ml of normal saline, left into the bladder for 20 min. A lidocaine gel is used to insert the catheter. Then, patients are subjected to standard cystoscopy (with a rigid or flexible cystoscope). After removing completely the anesthetic solution from the bladder, we inject the botulinumtoxin according to a previously reported modality (7, 8). Specifically, 100 U Onabot/A diluted in 10 ml of normal saline is injected into the bladder walls, with the exclusion of the trigone. At the end of treatment, the bladder is adequately washed out to completely remove any eventual blood clots.
Posttreatment control
Patients remain under observation for about 30–60 min. Indwelling catheter is not recommended except in cases of severe hematuria. Before discharging, patients have to spontaneously empty their bladder. At discharge, patients are suggested to continue with antibiotic prophylaxis with ciprofloxacin 500 mg twice daily for the next 3 days. In addition, treatment satisfaction is evaluated by Visual Analog Scale (VAS). Patients are asked to score their satisfaction according to the type and duration of treatment (duration of hospital stay, type of anesthesia and degree of bladder and/or urethral pain during and after treatment). A score of 0 indicates ‘absolutely not satisfied’ and a score of 10 indicates ‘most satisfied’. Once at home, patients can contact the specialists in the case of any complications.
Follow-up
After the first injection treatment, an appointment is scheduled within 15 days. Patients undergo uroflowmetry with postvoid residual urine (PVR), VAS to score the bother of urinary condition to QoL (a score of 0 indicates ‘absolutely not satisfied’ and a score of 10 indicates ‘most satisfied’) and their 3-day voiding diary was controlled. Patients are requested to start IC if symptomatic with a postvoid residual volume more than 200 ml. Further controls are scheduled at 3 and 6 months after treatment. For patients who received a repeat set of injections, the first appointment is scheduled within 15 days after treatment. The subsequent evaluations are usually performed on patients’ request, when there is a worsening of urinary symptoms. Injections cannot be repeated before 3 months after the previous one.
Outcomes of the study
Primary outcomes
Evaluation of the length of hospital stay, both as for ‘in-patient’ and ‘out-patient’ setting; frequency and duration of catheterization; frequency of UTIs during the first 60 days after treatment; frequency of intraoperative or postoperative complications; type and duration of antibiotic prophylaxis; and patients’ satisfaction to the proposed treatment protocol.
Seconday outcomes
The secondary outcomes include changes from baseline in daytime and nighttime urinary frequency, episodes of urgency and urgency urinary incontinence, changes from baseline in maximum flow rate and PVR, and changes in the VAS score related to urinary symptoms and QoL.
For the analysis, parameters at baseline (before the first Onabot/A injection) and at the last follow-up have been considered.
Statistical analysis
Statistical analysis was performed using the Friedman, Wilcoxon and Mann-Whitney tests for nonparametric data. The Bonferroni correction was applied to posthoc multiple comparisons. Statistical significance was set at p value less than 0.05.
Results
Between January 2001 and December 2014, 87 patients (73 females/14 males) affected by idiopathic OAB, refractory to treatment with anticholinergics, have been treated with Onabot/A intravesical injections. Here, we report the results of 64 (59 females and five males) evaluable patients, who underwent at least two consecutive Onabot/A injections.
Of the remaining 23 patients, 12 received their first injection and are still under evaluation; 11 had stopped Onabot/A treatment after their first injection due to the initial use of general anesthesia (seven patients) and due to the wish of pregnancy (five women).
Mean age ± SD at the time of the first injection was 54.4 ± 5.6 years and mean ± SD disease duration was 12.3 ± 3.5 years. All the patients had previously been treated with anticolinergics (tolterodine: 34 patients; tolterodine and oxybutynin: 22 patients; tolterodine, fesoterodine and solifenacin: eight patients), to which patients showed a low clinical response in 51% of cases, intolerable side effects in 36% of cases or both these effects in 13% of cases.
Onabot/A treatment
Mean ± SD frequency of injections/patient was 3.01 ± 1.6; mean ± SD interval between two consecutive injections was 7.2 ± 2.1 months. Overall, performed treatments have been: two in 18 patients; three in 31; four in 10; five in two; seven in one; and 10 in two patients. Twenty-one patients have been treated on an inpatient basis during the first three injections; afterwards, they underwent repeat injections in an outpatient setting. Forty-three patients underwent Onabot/A intravesical injections on an outpatient basis.
Primary outcomes
Twenty-one patients received their first three consecutive Onabot/A intravesical treatments on an inpatient basis; for these patients, mean ± SD duration of hospitalization was 39.4 ± 12.6 h; mean ± SD duration of catheterization was 37.8 ± 10.6 h; six out of 21 (28.5%) patients presented with hematuria during or after the intravesical treatment. Mean ± SD VAS value to score treatment was 6.3 ± 1.1. The frequency of UTIs during the 60 days following the last injection was 2.4 ± 1.6. No patient was required to perform intermittent catheterization (Tabs. I and II).
Comparison between primary outcomes in 64 patients with refractory overactive bladder syndrome treated with onabotulinumtoxinA repeat intradetrusorial injections in an ‘inpatient’ or in an ‘outpatient’ setting
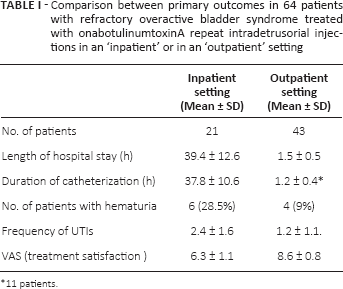
11 patients.
Secondary outcomes in 64 patients affected by refractory OAB treated with onabotulinumtoxinA repeat intravesical injections

Comparison between clinical results before the first and after the last injection in 21 patients treated in an inpatient setting and 43 in an outpatient basis.
VAS to score the bother of urinary symptoms on QoL.
For the 43 patients who had been treated since the beginning on an outpatient basis, the mean ± SD duration of catheterization, the mean ± SD duration as “out-patient” setting and the mean ± SD frequency of hematuria (four of 43 patients: 9%) were significantly reduced as compared with those of patients treated on an inpatient basis (Tab. I). The frequency of UTIs during the 60 days following the last injection was 1.2 ± 1.1. With regard to satisfaction with treatment, the mean ± SD VAS score was 8.6 ± 0.8 (Tab. I).
Secondary outcomes
At baseline, all patients presented with increased daytime and nighttime urinary frequency and daily episodes of urgency, as assessed by the 3-day voiding diary (Tab. II). The mean ± SD frequency of daily urinary incontinence episodes was 5.9 ± 1.4. The mean ± SD of Qmax and PVR were 28.5 ± 2.2 and 20.6 ± 12.4 ml, respectively. The mean ± SD VAS value related to the bother of urinary symptoms was 3.7 ± 1.4. Fifteen days after the last intravesical injections, daytime and nighttime urinary frequency were significantly reduced to 6.9 ± 3.2 and 1.7 ± 1.4, respectively. The daily frequency of urgency and UUI episodes was 5.2 ± 1.8 and 2.4 ± 1.5, respectively. Nineteen patients (29.6%) reached a complete urinary continence. When comparing patients treated in two different settings, we did not observe any significant difference, but only a trend to a better improvement in secondary outcomes in patients treated one an ‘outpatient’ basis than those treated in an ‘inpatient’ setting (Tab. II). The mean ± SD VAS value to score the bother of urinary symptoms on QoL was 8.2 ± 1.3 in patients treated in an inpatient setting and 9.1 ± 0.5 in those treated in an outpatient setting.
Discussion
In this study, we primarily wanted to investigate how the Onabot/A injection's protocol for patients affected by idiopathic OAB changed in our Clinic along time, and what was the level of patients’ satisfaction with the applied treatment modalities. At the same time, we studied the efficacy and safety of Onabot/A repeat injections in these OAB patients. As previously demonstrated, we found that repeat Onabot/A intravesical injections are safe and effective in the long-term follow-up, with a significant amelioration in all the considered OAB symptoms and in QoL as assessed by the VAS scoring (7–9).
What is new in the present study is the comparison between specific characteristics of different treatment modalities. Length of hospital stay, duration of catheterization and antibiotic prophylaxis, assessment of intraoperative and postoperative complications and, finally, level of patients’ satisfaction have been here studied and compared when the injection is performed in an inpatient or in an outpatient setting. Obviously, in this respect, we have to consider that, when this type of treatment was introduced in our Clinic, we started to inject patients in an in patient setting, in the operating room, by using general anesthesia. This approach was used to be extremely cautious with the use of a new pharmacological and potentially dangerous agent. Although some centers continue to perform injections under general anesthesia, to date, in the majority of cases, Onabot/A treatment is performed in an outpatient setting, under local anesthetic, although type and duration of anesthesia, type of cystoscope, antibiotic prophylaxis and use of indwelling catheter can vary from one center to the other. Indeed, in 2006, Kuo treated a total of 75 patients with detrusor overactivity (35 affected by idiopathic DO) with Onabot/A different doses randomly assigned in an inpatient setting, under general anesthesia in the operating room. A 14F urethral Foley catheter was inserted for 1 day and oral antibiotics were prescribed for the next 7 days. Patients were discharged the day after the procedure. Eleven percent of these patients developed mild hematuria that resolved by the next morning and PVR more than 150 ml was noted in a high proportion of patients treated with high Onabot/A doses. In that study, no information was given about the rate of UTIs and satisfaction with treatment. More recently, two multicenter, randomized, placebo-controlled phase III studies reported the results of a single Onabot/A injection in a total of 1105 patients with refractory, idiopathic OAB, treated with both local anesthesia and sedation, presumably in an outpatient setting. No patients’ satisfaction with treatment has been reported in these studies. Data have been given about changes in QoL due to treatment and about PVR and UTIs at week 12, which were less than 50 ml and 15.5-20.4%, respectively. Also, in other published studies, local anesthesia has been used to treat patients with idiopathic OAB, in an outpatient setting (11–15). Nevertheless, all these studies did not mention patients’ satisfaction with treatment, but they often report on satisfaction about changes in urinary symptoms and QoL.
The present study shows that intradetrusorial Onabot/A injection is a simple procedure that can be easily performed in an outpatient setting with the use of local anesthesia, followed by a low rate of intraoperative or postoperative complications. Indeed, only 9% of our patients treated in an outpatient setting showed a light hematuria during or soon after the injection. Also, the frequency of UTIs in these patients during the 60 days following treatment is low as compared with that in other studies (7, 8). More importantly, none of our patients needed to perform IC. These beneficial effects may be due to the use of low dose of Onabot/A since the beginning of treatment in our Clinic, although the increasing experience in performing the injections presumably allowed us to reduce the rate of complications in patients more recently treated. Furthermore, our successful results may also be derived by the fact that all patients eligible for Onabot/A intravesical treatment are at our Centre usually evaluated by a group of specialists in a dedicated clinic. Similar results have been obtained by Malde et al who showed that the use of Onabot/A service with a specialized, dedicated outpatient-based injection clinic can obtain a high satisfaction score on the Client Satisfaction Questionnaire (CSQ-8) questionnaire (16). Similarly, a reduced anxiety score with low pain has been detected by Ballert et al in patients treated with local anesthesia and rigid cystoscope (17). From the results related to the primary outcomes of our study, we can suggest the following rules to treat patients with refractory, idiopathic OAB with Onabot/A injection. Before proposing an injection, we have to control the feasibility and acceptability of catheterization by patient. Urodynamics should be performed before the first injection in cases of complicated OAB. At least one uroflowmetry with PVR is needed to control bladder emptying. Before the injection, any UTI has to be diagnosed and treated. The injection should be performed in the endoscopy room, in an outpatient setting, depending obviously also on local regulations and patients’ characteristics. Local anesthesia is useful to obtain a painless procedure. Antibiotic prophylaxis (usually ciprofloxacin 500 mg orally) should start about 30 min before the injection and can be protracted for the following 3 days. Indwelling catheter after the injection is not necessary when the procedure is without any bleeding. A first appointment should be scheduled within 15 days after treatment, mainly to control the ability of patients to completely empty the bladder. These simple rules are similar to those recently published by Hermieu et al, as French recommendations for practical use of Onabot/A in OAB patients (18).
Conclusion
To date, Onabot/A injection in patients with refractory, idiopathic OAB is a simple and repeatable procedure in the endoscopic room, under local anesthesia. Also, it is followed by low intraoperative and postoperative complications. This is the result of an increasing experience along the time and the availability of a urologic center specifically dedicated to patients needing for an intravesical treatment with Onabot/A.
Footnotes
Financial support: The authors have no financial disclosures to make.
Conflict of interest: Antonella Giannantoni is a Consultant for Allergan.
