Abstract
This is a case report on a patient with non-dialysis chronic kidney disease (CKD) in whom several nutritional issues are briefly discussed from a practical point of view. The article is accompanied by an editorial published in this Journal in relation to the 2nd International Conference of the European Renal Nutrition working group at ERA-EDTA—“Retarding CKD progression: readily available through comprehensive nutritional management?”— and focuses on several practical topics associated with the nutritional approach for the conservative treatment of non-dialysis CKD. The article is divided into 3 sections—basic nutritional assessment, nutritional targets, and nutritional follow-up in non-dialysis CKD—linked to 3 consecutive steps of the clinical follow-up of the patient and the related nutritional concerns and intervention.
What nutritional assessment/monitoring for protein-energy wasting (PEW) should be employed? Is a body mass index (BMI) of 21 kg/m2 adequate? What phosphate target should be pursued? What are the nutritional habits in patients with incident CKD? What protein needs and amount of dietary protein should be pursued? Does the quality of protein matter? What amount of dietary salt should be employed? How should this be obtained? How should normal serum phosphate be achieved? What diet should be recommended? Is a vegetarian diet an option?
Consequences of unintentional weight loss What is the role of the renal dietitian in helping the patient adhere to a renal diet?
What treatment for calcium/parathyroid hormone (PTH) will affect CKD progression? Final visits: Would a dietary recall/intensive dietary education improve adherence with the diet? Would a very-low-protein diet (VLPD)/ketodiet be indicated for this patient?
Keywords
Case report
A 64-year-old male nonsmoker had CKD. In the patient's family, there was a history of diabetes mellitus (DM) but no end-stage renal disease (ESRD) had been reported. Fourteen years previously, the patient had been diagnosed with type 2 DM, and had been on insulin treatment for 10 years; DM was complicated by peripheral neuropathy and retinopathy. The patient had had hypertension for 15 years and had hepatitis C virus-related chronic liver disease. At the first visit, the patient was admitted to the CKD nephrology clinic and underwent comprehensive, multifactorial nephrology care, including nutritional treatment; several nutritional concerns were raised during the follow-up and specific nutritional interventions were applied. These nutritional queries are highlighted in the following.
First visit (admission to CKD clinic: Tab. I)
Case report: clinical and laboratory data during the follow-up
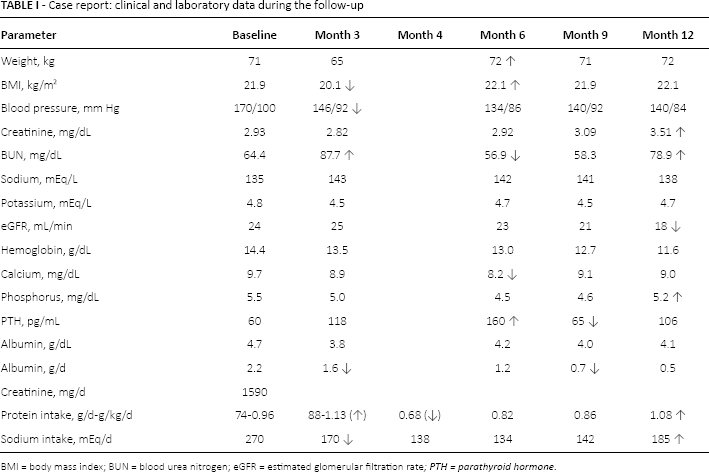
BMI = body mass index; BUN = blood urea nitrogen; eGFR = estimated glomerular filtration rate; PTH = parathyroid hormone.
Weight: 71 kg; height: 180 cm; BMI: 21.9 kg/m2
Blood pressure (BP): 170/100 mm Hg (supine and standing); heart rate: 80 bpm
No edema; no dyspnea; no lung fluids
Ultrasound: kidneys with low-normal size (longitudinal diameter: R: 100 mm; L: 102 mm), mild reduction of cortico-medullary differentiation
Fundus oculi: proliferative diabetic retinopathy
Echocardiography: mild left ventricular hypertrophy, ejection fraction: 55%
Renal function: serum creatinine 2.93 mg/dL, estimated glomerular filtration rate (GFR) 24 mL/min/1.73 m2, urine albumin 300 mg/dL (CKD stage 4)
Multidrug treatment:
Furosemide 50 mg, OID Irbesartan 300 mg, OID Amlodipine 5 mg, OID Nebivolol 5 mg, OID Fast insulin 4+12+10 U/d Retard insulin 26 UI/bedtime
Baseline nutritional assessment and basic nutritional considerations in non-dialysis CKD
At the basal evaluation, major concerns were uncontrolled BP (despite consumption of 3 BP-lowering drugs plus furosemide) and moderate proteinuria (despite the patient was treated with ATII blockade)
After undergoing 24 urine tests, albuminuria was 2.2 g/d, estimated protein intake was approximately 1 g/kg/d, and salt intake was very high (about 15 g/d)
Nutritional considerations:
Low body weight/BMI as compared to guidelines (ideal body weight [BMI 23] = 74.5 kg) Hyperphosphatemia High blood pressure and proteinuria, which may relate to nutrient intake (salt and proteins)
Intervention
Low-protein diet, 0.6 g/kg/d, mixed proteins, 30 kcal/kg/d
Low-sodium diet, 6 g/d salt
Angiotensin-converting-enzyme inhibitor, ramipril 5 mg OID
What nutritional assessment/monitoring for PEW should be employed?
The available recommendations for the assessment of nutritional status in CKD/ESRD are toward an integrated approach to nutritional status evaluation (1–4), which combines evaluation of body mass and anthropometric parameters, biochemistry, and dietary intake assessment. The International Society of Renal Nutrition and Metabolism (ISRNM) recommend the diagnosis of PEW to be established by the presence of at least one criterion in 3 out of 4 categories of nutritional variables (1) (Tab. II).
Criteria for the diagnosis of protein-energy wasting

Laboratory markers have a strong relation to mortality; however, both albumin and prealbumin can be largely influenced by non-nutritional factors, such as inflammation (3–5). Assessment of body mass is widely used and unintentional body weight fluctuations in a short time suggest a high risk for PEW (1, 6).
Assessment of skeletal muscle mass may provide the most reliable information for PEW diagnosis and monitoring (1). Assessment through the currently available gold standard imaging methods (magnetic resonance imaging, computed tomography, dual-energy X-ray absorptiometry) is expensive and/or not suitable for serial routine use. Body mass index does not distinguish between muscle and fat masses and is not a reliable tool. Anthropometric methods, such as assessment of skin fold thickness and circumferences or bioelectric impedance analyses (BIA), can be an option if performed by well-trained professionals (7).
In addition, assessment of dietary history and self-reports of appetite are critical to evaluate the adequacy of nutrient intake. The application of tools for periodic appetite assessment and food diaries also allows early intervention when dietary intake of protein and energy is lower than recommended (<0.8 g/kg/d and < 30 kcal/kg/d, respectively) (1, 3–6).
Is a BMI of 21 kg/m2 adequate?
While BMI is an excellent risk stratification tool in the community, its accuracy in assessing nutritional status and risk of malnutrition in catabolic diseases such as CKD is debatable. The limitations of BMI as a nutritional assessment tool in CKD include the inability to differentiate between body composition compartments (fat, muscle, and bone) or body fat distribution (abdominal vs peripheral) and to account for differences related to age, sex, ethnicity, and physical fitness. Of significance in most settings, BMI is importantly confounded by fluid retention. The ISRNM proposed a BMI lower than 23 kg/m2 as a criterion in the diagnosis of PEW (1). The suggestion of this specific cutoff, besides not being an established WHO BMI risk stratum, was based on the mortality prognostication of BMI in a large cohort of US dialysis patients (8). There are two hindrances in this cutoff:
It assumes that a BMI below this limit is undernutrition, but a study from Brazil showed that patients with CKD with a BMI below 23 kg/m2 did not present any specific signs of PEW and their muscle function was similar to their counterparts (9)
It assumes that a BMI above that limit is good nutritional status; nevertheless, obesity per se represents a nutritional disorder (by overnutrition).
In addition, obesity is not unequivocally exempt from PEW; malnutrition and inflammation-induced hypercatabolism also coexist in these individuals, and obese sarcopenia, that is, diminished muscle mass in obese individuals, should not be ruled out (10).
While this BMI is not abnormally low for the patient's ideal BMI, it is probably not sufficient to make a diagnosis. Information on other nutritional indicators is needed. Recent weight loss is diagnostically more useful than a single BMI value, and whenever possible analysis of body composition by BIA or skin fold thickness can provide a more accurate assessment of the presence and nature of the PEW (11).
What phosphate target should be pursued?
Phosphate metabolism is crucial in the pathophysiology of CKD mineral and bone disorder (MBD) and vascular calcification. High phosphate levels have been consistently associated with unfavorable outcomes in patients with CKD, but several limitations hamper a definition of the optimal targets of phosphate serum levels (12). Nonetheless, hyperphosphatemia is a late marker of phosphate overload.
The patient in this case had high serum phosphorus, out of range of the degree of CKD, in the presence of normal levels of calcium and PTH. Current guidelines suggest initiating dietary and pharmacologic approaches to reduce it (13). Specifically, Kidney Disease/Improving Global Outcomes (KDIGO) guidelines recommend limiting dietary phosphate intake and using phosphate-binding agents in order to reach and maintain phosphorus in the normal range (4.0–4.5 mg/dL). However, the concomitant normality of PTH value reasonably suggests that CKD-MBD is of relatively minor severity. Indeed, high phosphorus levels have a greater negative effect on cardiorenal prognosis when the compensatory increase of PTH is fully displayed (14, 15). On the other hand, it is possible that the elevated phosphorus be at least in part a consequence of albuminuria (>2 g/24 h in this patient); a recent multifaceted study, including experimental and clinical research, has in fact disclosed increased tubular reabsorption of phosphorus linked to abnormal urinary albumin excretion (16).
With the above considerations in mind, in moderate CKD in the presence of normal serum phosphate, it seems wise to postpone therapy with phosphorus-binding drugs while starting a soft approach to hyperphosphatemia (Tab. III) (17).
A soft approach to hyperphosphatemia

What are the nutritional habits in patients with incident CKD?
The nutritional habits in patients with incident CKD largely depend on the level of residual renal function. In early CKD, dietary intake is usually normal. In Western countries, average protein and salt intake, however, overcome the values recommended for the general population, and the phosphate intake is high. In addition, the widespread use of processed foods causes extra phosphate and sodium intake, very commonly among low-income patients. This type of diet impairs effectiveness of renin-angiotensin-aldosterone system inhibitors and antihypertensive drugs, increases proteinuria, and contributes to overt CKD-MBD and metabolic acidosis.
When residual renal function is severely reduced, the above-mentioned uncontrolled usual diet induces retention of nitrogen-derived toxins, which in turn causes loss of appetite, taste abnormality, or anorexia, leading to low food intake. Hence it is likely to result in low energy intake coupled with low protein intake, without any protein selection, so that the essential aminoacid supply is inadequate. This scenario is a high risk of PEW (18).
This patient at baseline had a very high sodium intake (roughly 15 g of salt intake) with a protein intake above the lower suggested level (0.8 g/kg/d). It is remarkable that phosphatemia is very high in respect to residual renal function and to the estimated protein intake: it suggests a large use of processed foods with phosphate-containing preservatives. The high salt intake may be in accordance with this type of dietary habit.
What protein needs and amount of dietary protein should be pursued?
The uremic state is characterized by the retention of waste products and toxins resulting from protein catabolism and excreted with the urine; the low-protein diet, by lowering the dietary intake of protein, counterbalances such metabolic derangements, ameliorating several uremic symptoms and complications. In addition, due to the effects on glomerular hemodynamics, the low-protein diet may help to slow the progressive GFR decline. Overall, protein restriction is able to delay the start of chronic dialysis. As a result, dietary protein restriction is the heart of the nutritional therapy for CKD. Nevertheless, a consensus on the level of GFR and when to start a low-protein diet and the optimal dietary protein amount or quality across the CKD stages has not been reached.
In the general population, the minimum recommended dietary allowance for proteins is 0.8 g/kg body weight/d (19); the protein intake in developed countries, however, is much higher, reaching 3.0 g/kg/d (20). Indeed, in Italy, the mean protein intake is 1.3±0.6 g/kg/d, with 3/4 individuals exceeding the recommendation (21). Guidelines on renal disease advise 0.8 g/kg/d in all adults with GFR lower than 30 mL/min/1.73 m2, and also recommend avoiding a protein intake higher than 1.3 g/kg/d in all patients at risk of CKD progression (22). Recommendations for patients in early CKD stages (1–3) or at low risk of progression are unclear. On this basis, it may be considered a safe, pragmatic approach to suggest that dietary advice established for the general population also suffices most patients with CKD. At the first visit, a low-normal protein diet and counsel to avoid excessive intake of proteins should be suggested as a basic approach to all patients with CKD (23). Of note, in CKD stages 1–3, only 1 out of 2 patients complies with a 0.8 g/kg/d protein intake (24). In CKD more advanced than stage 3b, metabolic derangements and water retention rapidly worsen (25, 26), and a 0.8 g/kg/d diet is no longer effective to obtain striking metabolic and clinical control. At these CKD stages, it is required to adapt the dietary intakes to the impaired renal function and a more restricted nutritional treatment is needed.
In CKD stages 3b or advanced (as in this case), a protein prescription of 0.6–0.7 g/kg/d should improve uremia, metabolic acidosis, and hyperparathyroidism (27). To avoid a negative nitrogen balance with such a reduced protein intake, at least 50% of proteins have to be of high biological value (i.e., meat, fish, eggs), and the energy intake should be normal-high (30–35 kcal/kg/d) (23). Since adherence to nutritional therapy is low in CKD, only patients compliant with these low-protein diets may be switched to more restricted, and potentially more effective, dietary regimens.
Does the quality of protein matter?
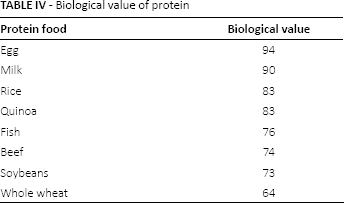
The Kidney Disease Outcomes Quality Initiative guidelines state that when prescribing a low-protein diet, at least 50% to 75% of the protein should be of high biological value. A complete dietary pattern should include all the 8 essential aminoacids plus histidine in the case of renal failure. Lack of essential aminoacids in the human body compromises vital functions. When a protein contains all the essential aminoacids in a proportion similar to that required by the body, it has a high biological value. When one or more of the essential aminoacids are missing or present in low amounts, the protein has a low biological value.
The biological value of a protein is indicated by a number from 100 to 0. More precisely, it is a measure of the percentage of the protein that is actually incorporated into the proteins of the human body (Tab. IV). However, food preparation and cooking methods also have an impact on the biological value of protein and may denature some of the essential aminoacids.
Biological value of protein
What Amount of Dietary Salt should be Employed? How should it be Obtained?
As depicted by 24-h urinary sodium excretion, the patient with CKD in this case is on a high-salt diet, with sodium intakes far above the recommended level for hypertensive patients by current guidelines (around 100 mmol/d; that is, 5–6 g NaCl/d). The patient should reduce his salt intake. A large body of evidence supports the validity of lowering BP to prevent cardiovascular disease in the general population. This issue becomes even more critical in patients with CKD because they carry a greater cardiovascular risk across the entire spectrum of CKD stages (28). In these patients, achievement of lower BP levels also is fundamental to limit the progression of renal damage, especially in the presence of significant proteinuria (as in this case). Although guidelines have repeatedly recommended in the last decade to intensively decrease BP in CKD, management of hypertension in patients with CKD remains inadequate. Armed with the knowledge of the extreme salt-sensitivity of BP in CKD as of the lower anti-albuminuric efficacy of anti-Ras agents if salt intake is high, a low-salt diet should become the basic approach to CKD-related hypertension (22). Indeed, besides the well-known lowering effectiveness of diuretic and anti-albuminuric therapy in the presence of unrestricted salt intake (as in this case), a normal sodium diet also precludes optimal control of BP during pharmacologic treatment with vasodilating agents (as in this case). Furthermore, about one-fourth of hypertensive patients with non-dialysis CKD have resistant hypertension (as in this case) (29). Resistant hypertension is defined by the persistence of office BP ≥130/80 mm Hg despite adherence to ≥3 full-dose antihypertensive drugs including a diuretic or ≥4 drugs regardless of BP level. This condition is more common in patients not adhering to a low-salt regimen and markedly increases the risk of cardiovascular and renal events.
Therefore, lowering salt intake, unless contraindicated (i.e., salt losers, volume depletion), is a cornerstone of conservative therapy in CKD (2). No large randomized study with hard endpoints has been conducted to date on this issue in patients with CKD to determine the optimal amount. However, while waiting for ad hoc trials in CKD, nephrologists should not give up with low-salt diet prescription but rather follow what renal pathophysiology teaches and make efforts to broaden implementation of this nonpharmacologic intervention in hypertensive patients. Rather than pursuing a specific level of salt intake, it is important to introduce a gradual, stepwise sodium reduction aiming at around 5 g/d, as recommended by the US Institute of Medicine (30). Tips to reduce salt are many (28): monitor salt intake (daily salt intake [g] = 24 urinary sodium excretion [mmol]/17); communicate to the patient the daily salt intake estimated by 24-h urinary sodium excretion (target: 100 mmol/d); move the salt shaker away from the table; cook pasta, rice, and cereals without salt, and add it in smaller amounts directly to cooked food; use spices (e.g., herbs, lemon, vinegar, hot pepper) rather than salt or salt-containing condiments (e.g., ketchup, mayonnaise, mustard, barbecue sauce); decide what to eat by looking at the amount of sodium on food labels; choose fresh foods and low-salt bread; cut back on frozen dinners, canned soups, packaged mixes, and cured meat and fish (e.g., ham, bacon, anchovies, salmon); choose fresh (e.g., mozzarella) rather than seasoned cheese; rinse canned foods (e.g., tuna) to remove some sodium; and abolish salty snack foods (e.g., chips, nuts).
How should normal serum phosphate be achieved?
Clinical nephrologists routinely counteract the positive phosphate balance in patients with CKD through nutritional counseling and prescription of phosphate binders (31). However, the superiority against placebo of phosphate control by diet or binders in terms of survival has never been tested in randomized controlled trials (RCTs). There is a rationale supporting the value of a simultaneous intervention against phosphate overload in patients with CKD via the improvement of dietary intake and an individualized choice of phosphate binders (32).
In our patient with CKD, the choice of the phosphate binder is delicate. Calcium-based phosphate binder (calcium carbonate or acetate) should be limited (depending on PTH control and daily dietary calcium intake), since the patient has DM, a condition with an increased risk of developing vascular calcification. When serum phosphate levels are ≥5.5 mg/dL, a calcium-free phosphate binder may also be prescribed to help reduce serum phosphate, either alone or in combination with other phosphate binders.
What diet should be recommended? Is a vegetarian diet an option?
For this patient, a vegetarian diet could be an option. Vegetarian diets present some advantages in CKD-diabetic patients:
Decrease in protein intake from animal origin is recommended. Red meat is associated with risk of type 2 diabetes and greater risk of vascular disease and ESRD (33). In diabetic patients, a decrease in protein intake leads to a decrease in proteinuria. This effect is higher when substituting red meat with chicken and even more with a decrease in protein intake (34). The renal hemodynamic response to a protein meal in a subject with normal renal function is different when using animal or vegetarian protein (35).
The intestinal absorption rate of phosphate is lower with plant protein. A decrease in meat processed food leads to a decrease in phosphate additives as well. A vegetarian diet is associated with lower plasma phosphate and a decrease in FGF23 levels (36).
Vegetarian diets are associated with decreased acid load and acid production. A high intake of plant-based foods in patients with CKD renders an alkaline-rich diet. Plasma bicarbonate increases and renal excretion of acid decreases, which is associated with a lower risk of renal fibrosis and lower progression of renal insufficiency (37, 38).
Beneficial effects of vegetarian diets on insulin resistance have been shown in several studies (39).
Replacing a Western-style diet by a vegetarian diet style is probably too early in this patient. Changing eating habits requires a period of information and adaptation. Nutritional preferences and capacity to adhere to the diet recommendations need to be checked before switching the patient to a vegetarian or low-protein diet with or without supplementation of aminoacids.
Second visit (3rd month during CKD follow-up: Tab. I)
↓ Blood pressure: 146/92 mm Hg
↓, Proteinuria: 1.6 g/d
↓↓ Weight: 65 kg; BMI: 20.1 kg/m2
↑ Blood urea nitrogen (BUN): 88 mg/dL
↑ Protein intake: 1.13 g/kg/d
↓ Sodium intake: 170 mEq/d
Major nutritional targets in non-dialysis CKD
Three months after the introduction of a low-protein, normal-protein, low-salt diet, a reduction of sodium intake was observed with better control of BP and proteinuria
The major nutritional concern was the striking reduction of body weight, though the protein intake seemed to be enhanced (BUN and urinary urea increased)
Nutritional considerations:
In CKD, adherence to the dietary protein targets prescribed is low, but usually such intake is above the prescription In advanced CKD, energy intake is often reduced A low energy intake in the presence of the minimum suggested intake of proteins in CKD causes a negative nitrogen balance The benefits of lowering body weight are debatable
Intervention
Additional tests:
Dietary diary and dietitian interview:
– Protein intake 47 g/d, 0.60 g/kg/d (the patients is adherent to prescription) – Energy intake 1,375 kcal/d, <18 kcal/kg/d (↓↓ prescription) – Assume only half of the prescribed olive oil (≈10% prescribed energy) – Does not assume at all the protein-free food (≈30% prescribed energy)
Increase energy intake
Dietary counseling and diet concordance
Increase amlodipine to 10 mg, OID (moved to bedtime)
Consequences of unintentional weight loss
Any unintentional reduction in body weight should be taken as a potential sign of PEW, requiring additional nutritional assessment and monitoring/follow-up. Unintentional weight loss is a common feature of all nutritional screening tools applied in clinical practice. Also in CKD, unintentional weight losses in a short period of time, even if only a couple of kilograms, is associated with risk of adverse outcomes (40, 41).
Observational studies reported that in a predialysis, metabolically healthy CKD population, overweight or obese individuals had a significantly lower risk of all-cause mortality than normal weight patients but, in presence of metabolic syndrome, no difference in survival was observed. On the other hand, lower BMI is associated with greater mortality risk in patients with predialysis CKD; this relationship persisted when adjusted for malnutrition and inflammation markers (42).
No evidence exists that normal weight or leanness are favorable prognostic factors in the CKD population. Instead, an overweight condition seems to be preferable. Body weight loss is not a priority in absence of severe obesity and/or metabolic syndrome. Weight (fat mass) loss may be useful in patients with CKD mainly as a tool for correction of the associated cardiovascular and renal risk factors (dyslipidemia, hypertension, proteinuria, inflammation, insulin resistance) (43).
Fatty mass loss should be encouraged by means of modifications of lifestyle, increase of physical activity, and limitation of energy intake excess. It is noteworthy that calorie restriction increases protein requirements and then prevents normal adaptation to a low-protein diet. Hence, overweight or obese patients who are on a low-energy diet should not be prescribed protein-restricted (0.3–0.7 g/Kg/d) regimens. Conversely, when a low-protein diet is indicated, energy intake must equal or overcome energy requirements, even in the overweight or obese patient.
Regular physical exercise always should be recommended to prevent loss of lean body mass.
Nonsurgical weight loss interventions are able to reduce proteinuria and arterial blood pressure in patients with CKD and seem to reduce the progression of renal function decline (44). A careful valuation of potential benefits and risks is mandatory before performing bariatric surgery in the patient with CKD. Bariatric surgery normalizes GFR and reduces urine albumin excretion rate and arterial blood pressure levels in comorbid obese subjects with normal or increased GFR (44).
What is the role of the renal dietitian in helping the patient adhere to a renal diet?
The role of the dietitian is to translate the nephrologist's prescription into recipes and meals so that the required restrictions are not a burden but a new eating style for the renal patient. The nutritional intervention for patients with CKD should not be related only to the residual renal function but depends on many aspects such as socioeconomic, psychological, and functional status (45, 46).
The first step for the renal dietitian is knowing the clinical data of the patient but also his or her habits, both food and life, the environment in which he or she lives, and who takes care of him or her, regarding those involved in the preparation of meals in particular so as to ensure an appropriate nutritional intervention without dramatically changing the patient's habits and lifestyle. Another aspect the renal dietitian knows is that the success of the nutritional intervention is conditioned by the presence and cooperation of family members or caregivers; their involvement in the counseling process is essential (Fig. 1).
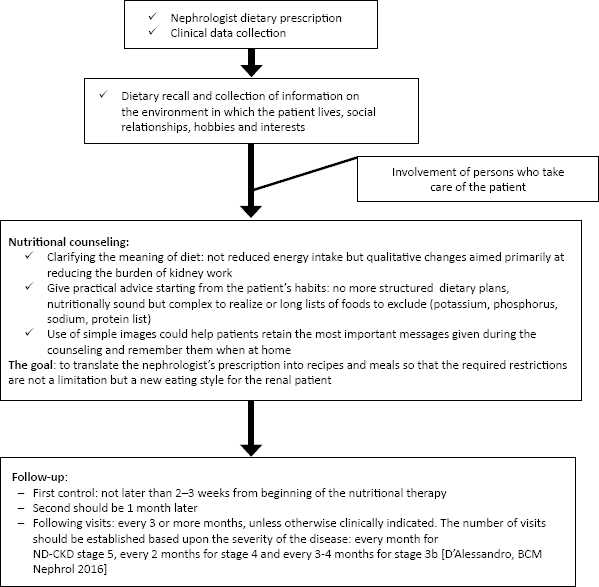
Nutritional counseling for patients with chronic kidney disease: a hypothetical path for the renal dietitian
When a dietitian starts a nutritional intervention, it is important to explain to the patient and relatives/caregivers that when we talk about diet we do not mean reducing energy intake, as this frequently has negative effects on nutritional status and metabolic control. We need to make it clear that we are referring mainly to qualitative changes aimed primarily at reducing the burden of work for the kidney.
Finally, it is important to provide practical advice: no more structured dietary plans, nutritionally sound but complex to realize, or long lists of foods to exclude (potassium, phosphorus, sodium, protein lists), but useful and simplified basic information on how to change habits and help patients combine ingredients to create enjoyable meals that fit recommendations for CKD (47–49).
Intermediate visits (4th and 6th months during CKD follow-up: Tab. I)
After 1 month, the patient again underwent dietary diary and dietitian interview:
Protein intake 0.68 g/kg/d (adherent to prescription)
Energy intake 1,945 kcal/d, 25 kcal/kg/d (↑↑ adherent to prescription)
After 2 more months, nutritional status was restored and some clinical changes occurred:
Weight: 72 kg ↑; BMI: 22.2 Kg/m2
BUN: 57 mg/dL ↑
Blood pressure: 134/86 mm Hg ↓
Serum Calcium: 8.2 mg/dL ↓
PTH: 160 pg/mL ↑
Nutritional follow-up in non-dialysis CKD
After 6 months of nutritional interventions, the patient seems on effective nutrient intake, with stable nutritional status and no major metabolic derangements
Calcium/phosphate/PTH metabolism remains to be improved
Intervention
Paricalcitol, 1 μg OID
Quarterly nutritional/dietitian monitoring planning
What treatment for calcium/PTH will affect CKD progression?
In the present case, the patient developed hyperphosphatemia, hyperparathyroidism, and mild hypocalcemia. Directly treating hyperphosphatemia should be the primary goal in the management of CKD-MBD abnormalities because this can reduce not only serum phosphorus, but also PTH and FGF-23 levels, which are directly involved in poor clinical outcome in patients with CKD (50). To support this approach, large epidemiologic studies consistently show that high serum phosphorus levels are associated with increased renal progression in CKD stage 3 to 5 patients (51–54). This association between phosphorus and worse outcome has been extended to the general population, suggesting that high phosphate levels may impair renal health at all levels of kidney function (55). As a result of these findings, the KDIGO guidelines emphasize the need for treatment to normalize phosphorus levels in CKD stage 3 to 5 (13). Optimal phosphate control involves dietary measures, but these are not always sufficient, and it can also be needed to consider phosphate binder use. All the phosphate binders appear to be equivalent in their ability to control phosphate levels (50). With the limitations inherent to the observational studies, we know that there is a correlation between renal calcium content and renal impairment in human renal biopsies (56). Moreover, at least in experimental studies, calcium-free phosphate binders stabilize renal function and attenuate kidney calcification (57, 58). Limited data from interventional studies support these findings. In an independent study, a nonsignificant trend for dialysis inception was noted in the non-dialysis-dependent patients randomized to sevelamer compared to calcium carbonate (59). In a randomized clinical trial comparing calcium acetate, lanthanum carbonate, sevelamer carbonate, and placebo, only patients treated with sevelamer showed a reduction in FGF-23, whereas treatment with calcium acetate increased FGF-23 levels (50). Whereas the most beneficial approach in the management of CKD-MBD abnormalities for preventing renal progression is maintaining phosphate levels within target range, there is no convincing evidence for treating the mild hypocalcemia in patients with CKD. Data on calcium balance have shown that giving excessive amounts of calcium causes calcium retention, which may lead to vascular calcification, and possibly, to worse renal outcomes (60). In an observational study, the use of phosphate binders (type not specified) was linked to faster CDK progression (61), while in a randomized clinical trial comparing different phosphate binders, only a participant in the calcium acetate group experienced an episode of acute kidney injury (50). Moreover, calcium supplementation could also contribute to progressive renal injury in the general population. Among 1,675 healthy participants in an RCT of colorectal adenoma chemoprevention with calcium and vitamin D, daily supplementation with 1,200 mg of elemental calcium caused a small but significant increase in blood creatinine, which may have potential implications for clinical recommendations for calcium supplementations (62). In line with these findings, the KDIGO guidelines emphasize the need for restricting the dose of calcium-based phosphate binders, especially in the presence of vascular calcification, which is prevalent in patients with CKD (13).
Once the phosphorous level is on target range, our next goal should be treatment of hyperparathyroidism with vitamin D or vitamin D analogues. Although the presence of high PTH levels have been described as an independent predictor of CKD progression, the effect of controlling hyperparathyroidism with vitamin D on renal progression has not been evaluated systematically (63). However, it is well-known that vitamin D may reduce proteinuria, a surrogate marker of renal progression. A systematic review of RCTs has demonstrated the anti-proteinuric effect of active vitamin D (64). However, before starting active vitamin D treatment, it seems reasonable to assess 25-hydroxyvitamin D levels and initiate replacement with native vitamin D if levels are low, as KDIGO guidelines suggest. The fact that the antiproteinuric effect of vitamin D has been also demonstrated after nutritional vitamin D supplementation reinforces this approach (65). Regardless of the type of vitamin D provided, serum calcium and phosphorus levels need to be monitored closely to prevent hypercalcemia and hyperphosphatemia. This effect seems to be less important with paricalcitol (66). Whether vitamin D can slow CKD progression is not yet known.
Therefore, keeping in mind the potential renoprotective effect of controlling CKD-MBD disorders, it seems reasonable for the present patient 1) to consider the use of a phosphate binder for controlling high phosphate levels, with a preferential use of a non-calcium-containing binder if vascular calcification is present; 2) to avoid the overuse of calcium-containing phosphate binders; and 3) to add vitamin D for reducing PTH (and proteinuria), with a preferential use of paricalcitol when active vitamin D preparation is needed. Although there is no randomized clinical trial, an observational study has recently tested this approach, showing better outcome when we use a regimen based on the restricted use of calcium-based binders in combination with calcium-free phosphate binders, low-dose nutritional vitamin D supplementation, and the use of paricalcitol as antiparathyroid agent, compared with unrestricted conventional care, in non-dialysis-dependent patients with CKD (67).
Final visits (9th and 12th months during CKD follow-up: Tab. I)
After 3 months:
Patient was stable, well-being, good control of comorbidities No nutritional changes Improvement of PTH and proteinuria No intervention
After 3 more months:
Stable nutritional status (body weight, serum albumin) Stable clinical conditions and comorbidities Worsening of renal failure Reduced diet adherence:
Serum phosphate: 5.2 mg/dl ↑ Protein intake: 1.08g/kg/d ↑ Sodium intake: 185 mEq/d ↑
Intervention:
Intensive dietitian intervention Consider a more restricted renal diet
Would a dietary recall/intensive dietary education improve adherence with the diet?
A careful evaluation of dietary habits is mandatory for an effective nutritional intervention. Dietary recalls (food diary, 3-day recall) are essential tools for the renal dietitian to estimate habitual energy and nutrient intake, but they allow doing more. While collecting a dietary recall, one can obtain information about the patient's life, practical aspects such who does the shopping and who prepares meals, or the patient's capacity to perform activities of daily living, social relationships, interests, and hobbies. This is important information to provide feasible dietary instructions not too far from the patient's current dietary habits (23).
The timing of the visits is important to verify the efficacy of the counseling and to strengthen some concepts as dietary recommendations in CKD are many. During the first visit, the dietitian gives the main instructions that need to be further clarified during the control visits. Blood and urinary tests are essential to show the patients what is correct and what needs to be adjusted. Do not worry about repeating the same things. The goal of repetition is to find new solutions together with the patient so that he or she perceives that what we ask is not an imposition but what is the best for improving his or her health/nutritional status. A key finding to reach ample adherence to the low-protein diet is to individualize the dietary strategies and at the same time to build up specific intensive counseling/education programs for patients (68). The message for our patients is that we do not need the patient to follow the diet we prescribed perfectly but to work with us to find the best solution for realizing a new style, which should be considered as a therapy to all effects (45, 47).
Would a VLPD/ketodiet be indicated for this patient?
The VLPD (0.3–0.4 g/kg body weight/d protein) is a vegetarian diet that mainly includes vegetables and fruits and is supplemented with essential aminoacids and ketoacids (69). To maintain a neutral nitrogen balance, though the protein intake is below the minimum required threshold, ketoacids are given to allow endogenous nitrogen recycling as a means of building the essential aminoacids from the relative ketoacids, thus further contributing to reduce serum urea levels (70). Because of the very low nitrogen intake, a high energy intake (never lower than 30–35 kcal/kg/d) is mandatory to maintain a neutral body nitrogen balance. Protein-free artificial foods can be added to this diet, helping to reach such a high energy supply, since these products provide high energy amounts (23).
The VLPD has been shown to have further additional positive effects, such as a lower indoxyl sulfate content compared to the usual low-protein diet (71), a reduced need for erythropoietin (72), better control of BP (73), lower urinary and plasma phosphate and FGF23 levels (74), lower proteinuria, and higher antiproteinuric response to renin-angiotensin system inhibitors (75). Also, vegetables and fruits increase plasma bicarbonate levels, improving the metabolic acidosis (38), and fiber intake, both being possibly related to a better CKD outcome.
A major concern of the use of the VLPD is the nutritional risk, since adherence to diet prescription in the CKD population is low. Protein metabolism and nitrogen balance adapt well in patients with CKD on a VLPD (up to 0.3 g protein/kg/d) when supplemented with ketoacids and in presence of high energy supply, allowing a neutral nitrogen balance in the short term (76, 77). Also in the long term (more than 1 year), the adaptive response to the reduced nitrogen load was similarly positive (78). Therefore, VLPDs are, theoretically, nutritionally safe during advanced CKD, given that an adequate amount of calories is provided. Clinical studies have shown that VLPD is safe during either the predialysis CKD period or during the following long-term dialysis period for patients who previously received such a diet and were on dietitian control (79). Indeed, VLPD has recently received a final consensus from experts in this field (80).
Not every patient with CKD may benefit from a supplemented VLPD. A patient with progressive CKD (as in this case), proven high adherence to diet and acceptance of lifestyle changes (somewhat in this case), and a low rate or well-controlled comorbidities (as in this case) is likely to have a positive result from the VLPD (79, 81). A recent prospective RCT assessed the effectiveness and safety of a VLPD supplemented with ketoanalogs in reducing CKD progression in CKD stage 4–5 as compared to a conventional low-protein diet (81); these patients were highly selected, well-nourished, and well-adherent to the low-protein diet, and had low comorbidity levels. In these conditions, the ketodiet was effective and safe in deferring dialysis initiation or reducing the rate of decline in renal function; the key points of such effects were careful patient selection and continuous nutritional monitoring and counseling. Hence, supplemented VLPD management requires high expertise by nephrologists and dietitians, good doctor-patient concordance, family and social support, as well as high doctor-patient empathy.
Footnotes
Acknowledgment
The European Renal Nutrition Working Group is an initiative of and supported by the European Renal Association-European Dialysis Transplant Association.
Financial support: No grants or funding have been received related to this study.
Conflicts of interest: V. Bellizzi: Lectures, consulting and travel grants Fresenius Kabi; consulting Shire; lecture Abbott. P. Chauveau: Lecture and travel grant Fresenius Kabi and Vifor. M. Cozzolino: Lectures Shire, AbbVie, Amgen. A. Cupisti: Lectures, consulting fees and travel grants Shire. L. De Nicola: Consulting and lectures AbbVie, Janssen, AstraZeneca. P. Molina: speaker honoraria from Abbott Nutrition and Vifor-Pharma-Fresenius Medical Care. D. Fouque: Lectures, consulting and travel grants Fresenius Kabi. The other authors did not declare any conflict of interest related to this study to disclose.
