Abstract
Background
Whether anemia and mineral bone abnormalities (chronic kidney disease–mineral bone disorder [CKD-MBD]) are associated still remains to be elucidated. Both anemia and CKD-MBD have been associated with adverse cardiovascular outcome and poor quality of life. However, recent evidence suggests that use of large doses of erythropoietin-stimulating agents (ESAs) to correct hemoglobin (Hb) may be detrimental in CKD. The Optimal Anemia Treatment in End Stage Renal Disease (ESRD) (Optimal ESRD Treatment) study will assess whether lowering of parathyroid hormone (PTH) is associated with a reduction in ESA consumption.
Methods
The Optimal ESRD Treatment study is a pilot single-center open-label study with blinded end point (a prospective randomized open blinded end-point [PROBE] design) enrolling 50 patients on maintenance dialysis. Eligible patients with intact PTH (iPTH) 300–540 pg/mL and Hb 10–11.5 g/dL will be randomized 1:1 to strict PTH control (150–300 pg/mL) versus standard care (PTH range 300–540 pg/mL). Available drugs for CKD-MBD and anemia treatment will be managed by the attending physician to maintain the desired levels of PTH (according to study arm allocation) and Hb (10–11.5 g/dL). Echocardiographic data for cardiac structure and function as well as arterial stiffness will be assessed at study inception and completion.
Conclusions
The Optimal ESRD Treatment study should shed light on the complicated interplay of anemia and CKD-MBD and on the feasibility of clinical trials in this domain. The study results are expected in the spring of 2017.
Introduction
Anemia is one of the most worrisome complications of chronic kidney disease (CKD). Numerous prospective studies have repeatedly documented an increased risk of morbidity and mortality associated with lower levels of hemoglobin (Hb) (1–5). Hence the international guidelines on patient care suggest the use of erythropoietin-stimulating agents (ESAs), iron and folate supplementation for anemia correction in CKD individuals (1–5).
However, recent randomized controlled trials (RCTs) have demonstrated that Hb correction to normal levels increases the risk of major cardiovascular (CV) events (1–5). Although the reasons are still unclear, the cumulative ESA dose and rapid Hb fluctuations may at least partly explain these findings (6–10). Hence, limiting ESA to the minimal dose needed to achieve the suggested Hb targets in end-stage renal disease (ESRD) as well as identifying factors associated with ESA response seems advisable (6–10).
Among other factors, mineral metabolism abnormalities (chronic kidney disease-mineral bone disorder [CKD-MBD]) have repeatedly been associated with poor outcomes and have emerged as a risk factor for severe anemia as well as for use of large ESA doses (11–13). In this regard, a few observational studies have suggested a linear inverse association between parathyroid hormone (PTH) and ESA dose in ESRD patients (11,12), although these findings may be the result of a faster deterioration of the CV system associated with higher PTH levels (14).
Due to the lack of RCTs that clearly demonstrate any survival benefit associated with strict PTH control, the latest Kidney Disease: Improving Global Outcomes (KDIGO) guidelines on CKD-MBD management suggest a higher reference target for intact parathyroid hormone (iPTH) (2–9 times the upper level of the normal range) when compared with the National Kidney Foundation (NKF) guidelines published in 2003 (150–300 pg/mL) (14).
The aim of the Optimal Anemia Treatment in End Stage Renal Disease (ESRD) (Optimal ESRD Treatment) study is to test whether a tighter PTH control is associated with an ESA dose reduction and a slower CV system deterioration in ESRD patients receiving dialysis.
Methods
The Optimal ESRD Treatment study is a pilot, pragmatic, single-center, open-label trial with a blinded end point (prospective randomized open blinded end-point [PROBE] design) aimed at improving patient care. The study protocol was reviewed and approved by the local ethics committee of Azienda Ospedaliera Sant'Anna, Como (Parere Unico 61/13, May 16, 2013) and was registered at https://clinicaltrials.gov/ (NCT02273570), with the official title “Single-center, Open-label, Randomized Study of Anemia Management Improvement in End Stage Renal Disease (ESRD) Patients With Secondary Hyperparathyroidism.” All procedures involved in the study will be conducted in adherence to the Declaration of Helsinki and the Ethical Principles for Medical Research Involving Human Subjects, and according to the principles of Good Clinical Practice.
The Optimal ESRD Treatment study objectives are as follows:
Primary objective: to test whether a tighter PTH control (PTH level <300 pg/mL vs. PTH level between 300 and 540 pg/mL) is associated with a lower ESA dose use to achieve the target Hb of 10.0–11.5 g/dL
Secondary objective: to test whether a tighter PTH control to achieve a PTH level <300 pg/mL vs. PTH level between 300 and 540 pg/mL is associated with a better iron storage mobilization.
Secondary objective: to test whether a tighter PTH control to achieve a PTH level <300 pg/mL vs. PTH level between 300 and 540 pg/mL is associated with cardiac valve deposition and progression attenuation (assessed via echocardiogram).
Secondary objective: to test whether a tighter PTH control to achieve a PTH level <300 pg/mL vs. PTH level between 300 and 540 pg/mL is associated with an increase in arterial stiffness attenuation (assessed via applanation tonometry).
Secondary objective: to test whether a tighter PTH control to achieve a PTH level <300 pg/mL vs. PTH level between 300 and 540 pg/mL is associated with a better CKD-MBD control.
Eligible patients should be adult men and women (i.e., age >18 years), on thrice-weekly maintenance dialysis via arteriovenous fistula. They should be undergoing treatment with an ESA with an Hb level at study entry between 10.0 and 11.5 g/dL, iPTH between 300 and 600 pg/mL and Kt/V >1.2. Finally, all study participants should able to provide a signed informed consent prior to the initiation of the study.
All eligible patients will be randomized (1:1) to one of the study arms in which a different PTH control will be enforced during the 12 months of study follow-up:
Control group (standard care group): the iPTH target in this group is 300–540 pg/mL
Optimal CKD-MBD control group: in this group, the PTH target is 150–300 pg/mL, to be achieved with a therapeutic algorithm.
To achieve the desired PTH (according to study arm allocation) and Hb (10.0–11.5 g/dL) levels, no specific therapeutic algorithm is to be enforced, and the attending physician will be responsible for prescriptions as well as dose titration of the different drugs available on the market for CKD-MBD and anemia management.
To detect meaningful changes in CV structure and function associated with levels of iPTH, the presence of cardiac valve calcification, cardiac function and arterial stiffness will be assessed noninvasively via transthoracic echocardiogram and applanation tonometry at study entry as well as study completion (Tab. I summarizes the Optimal ESRD Treatment study checklist). Briefly, a standard transthoracic echocardiogram will be performed. Parasternal short and long axes as well as 4- and 5-chamber views will be acquired to assess cardiac structure and function. In particular, the presence and extension of cardiac valve calcification will be scored as suggested by Corciu and collaborators (15). Arterial stiffness and thoracic aorta blood pressure will be evaluated noninvasively and expressed as pulse wave velocity, thoracic aorta central blood pressure and augmentation index. To reduce the intra- and inter-reader variability, echocardiogram and applanation tonometry will be performed before the midweek dialysis session, and echocardiogram readings will be centralized.
Optimal ESRD Treatment study checklist
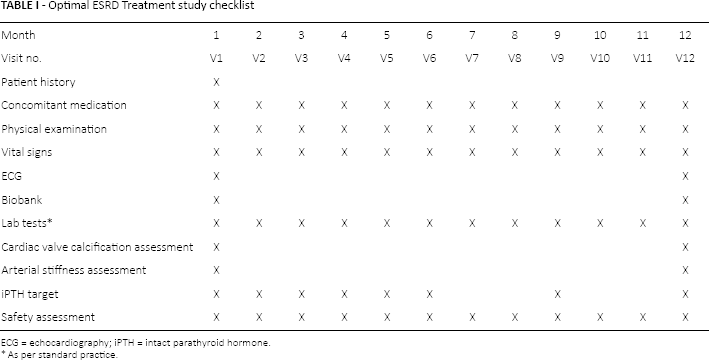
ECG = echocardiography; iPTH = intact parathyroid hormone.
As per standard practice.
It was anticipated that the entire study should take about 18 months (from first patient in to last patient out): 6-month recruitment phase and 12-month active treatment period. Active patient recruitment began in March 2015. However, as of September 2015, only 17 patients had been randomized, due to a slower than anticipated recruitment rate, and it is now anticipated that the study will last until February 2017.
The Optimal ESRD Treatment study is funded by an unrestricted research grant from Amgen and from the Italian Society of Nephrology.
Statistical analysis and sample size estimation
Standard descriptive statistics as well as parametric and nonparametric tests will be used. Linear regression models will be adopted to test the difference in ESA use, cardiac valve calcification and arterial stiffness between groups at study completion. All efficacy analyses will be performed on an intention-to-treat basis.
According to the study protocol, the primary end point is defined as percentage reduction in weekly ESA consumption to maintain Hb levels within the recommended range 10.0–11.5 g/dL.
The following secondary end points will also be investigated:
Change in iron status and storage, defined as change in iron consumption per week, percentage of patients with transferrin saturation >20% between groups at study completion, change in hepcidin levels between groups at study completion. These end points will be compared between and within treatment groups.
Difference (absolute change and percentage increase) in prevalence of valvular calcification deposition and progression as detected by echocardiography between groups. This end point will be compared between and within treatment groups.
Difference (absolute change and percentage increase) in pulse wave velocity as assessed by applanation tonometry between groups. This end point will be compared between and within treatment groups.
Given that that this is a pilot study and there are no data for an accurate sample size estimation, and assuming: (i) 40% ESA dose reduction in the Optimal CKD-MBD control group, (ii) power 80%, (iii) statistical significance 5% and (iv) dropout rate of 10%, it is estimated that 50 patients (n = 25 per study arm) are needed.
Discussion
Observational studies suggest a close association between CKD-MBD and anemia. Nonetheless, no RCT has investigated whether CKD-MBD manipulation may ameliorate anemia control in ESRD (11–13). The aim of the Optimal ESRD Treatment study is to test whether a strict PTH control (i.e., 150–300 pg/mL vs. 300–540 pg/mL) reduces ESA consumption in a cohort of 50 patients on maintenance dialysis. The secondary objectives of the Optimal ESRD Treatment study are to investigate the impact on the CV system, of 2 different iPTH targets within the suggested range of twofold to ninefold the upper level of the laboratory reference for iPTH (14).
The Optimal ESRD Treatment study is a pragmatic trial. Indeed, with the exception of a vitamin D usage limitation (i.e., the cumulative weekly dose of paricalcitol or equivalent should not exceed 6 μ.g), no specific recommendations on drug use to achieve the desired levels of iPTH and Hb are to be enforced. The attending physician is free to prescribe and adjust doses of commonly available drugs for CKD-MBD and anemia management.
Although patient recruitment has been slower than expected and a protocol amendment is under evaluation to not limit patient eligibility to individuals with arteriovenous fistula as vascular access, it is anticipated that the study will be completed and the study results available in spring 2017.
The single-center design and small sample size are indeed limitations of this pilot study. Nonetheless, the Optimal ESRD Treatment study will shed some light on the feasibility of future larger studies in this domain. Far more importantly, it will provide some hints for how to optimize ESRD patient care. An ongoing, large, properly powered, multicenter study is testing the hypothesis that ESA dose is linked to poor outcome in ESRD (16). Knowing what factors are associated with increased use of ESA and knowing that manipulation of bone mineral abnormalities may result in ESA use or dose minimization will ultimately result in improved patient care. Although only 50 patients will participate in the study, the fact that all study participants receive maintenance dialysis in a single center, as well as the centralization of laboratory and imaging procedures, should minimize the between-center and inter-reader variability, thus increasing the chance to detect a meaningful result if it exists.
In summary, the Optimal ESRD Treatment study aims to shed light on any causative association between PTH and anemia. The study results are expected for spring 2017, and it is the authors' opinion that they will be valuable for improved patient care and/or for future larger studies to be undertaken in this domain.
Footnotes
Financial support: The Optimal ESRD Treatment study is funded by an unrestricted research grant from Amgen and from the Italian Society of Nephrology.
Conflict of interest: None of the authors has any financial interest related to this study to disclose.
