Abstract
Purpose
To compare pro re nata (PRN) and bimonthly intravitreal aflibercept injection protocols in the treatment of neovascular age-related macular degeneration (nAMD).
Methods
This was a retrospective, observational, comparative study in 2 centers. All study patients presented with newly diagnosed nAMD and were treated as follows after an induction of 3 consecutive monthly loading doses: the bimonthly group was retreated every 2 months and followed up every 4 months while the PRN group was retreated in case of subretinal exudation or visual acuity decrease and followed up every month. A comprehensive ophthalmic examination was performed at each study visit.
Results
At 1 year, 34 eyes were analyzed. The 19 patients in the bimonthly group had a mean visual acuity improvement of +8.5 Early Treatment Diabetic Retinopathy Study (ETDRS) letters whereas stabilization was observed in the PRN group (-0.47 ETDRS letters; p = 0.027). All patients in the bimonthly group had 4 follow-up visits and 7.5 injections whereas the mean number of follow-up visits in the PRN group (15 patients) was 7.8 with 7.6 injections.
Conclusions
Our study suggests that a fixed bimonthly aflibercept retreatment protocol may provide better visual results than a PRN protocol.
Keywords
Introduction
Age-related macular degeneration (AMD), particularly its neovascular form (nAMD), is a burden to health care systems. It is considered the leading cause of blindness in patients older than 50 years in developed countries despite the development of new effective therapies over the last 15 years (1).
Anti-vascular endothelial growth factor (VEGF) intravitreal injections have greatly modified the prognosis of nAMD and have rapidly become the gold standard for its treatment. Currently, many protocols for injecting and monitoring patients with AMD are available (2). Pivotal studies such as Minimally Classic/Occult Trial of the Anti-VEGF Antibody Ranibizumab in the Treatment of Neovascular Age-Related Macular Degeneration (MARINA) (3), Anti-VEGF Antibody for the Treatment of Predominantly Classic Choroidal Neovascularization in Age-Related Macular Degeneration (ANCHOR) (4), and A Phase IIIb, Multicenter, Randomized, Double-Masked, Sham Injection-Controlled Study of the Efficacy and Safety of Ranibizumab in Subjects with Subfoveal Choroidal Neovascularization with or without Classic CNV Secondary to Age-Related Macular Degeneration (PIER) (5) have demonstrated the efficacy of monthly ranibizumab injections. Ophthalmologists have been able to reduce the number of injections based on the results of the Prospective Optical Coherence Tomography Imaging of Patients with Neovascular AMD Treated with Intraocular Ranibizumab (PRONTO) (6) study using a pro re nata (PRN) regimen. Pro re nata has been demonstrated to be as efficient as monthly injections in at least 2 randomized studies: Comparison of Age-related Macular Degeneration Treatment Trial (CATT) (7) and Alternative Treatments to Inhibit VEGF in Age-related Choroidal Neovascularization (IVAN) (8).
In 2011, the VEGF Trap-Eye: Investigation of Efficacy and Safety in Wet AMD (VIEW) 1 and 2 studies (9) demonstrated that aflibercept could be injected bimonthly for 1 year with results as good as monthly ranibizumab injections. However, few studies have compared the PRN and the fixed bimonthly regimens. Only one recent study reported the effects of these 2 regimens in cases of a switch from ranibizumab to aflibercept in recurrent nAMD. In this study, the bimonthly protocol used after the switch seemed more efficient than PRN usage (10).
To our knowledge, no real-life study of treatment-naive patients has compared these 2 regimens using the same molecule. The objective of the present study was therefore to compare the anatomical and visual outcomes after 1 year of aflibercept treatment using a PRN versus a fixed bimonthly retreatment protocol.
Methods
This was a retrospective, observational, comparative study in 2 retina centers in Strasbourg, France. The study protocol was approved by the Ethics Committee of the French Society of Ophthalmology.
Consecutive patients were included in this study between September 2013 and March 2015 and followed up for 1 year. All patients presented with newly diagnosed subfoveal nAMD and were naive to any treatment before the initiation of aflibercept therapy. At baseline, they had all undergone funduscopy, optical coherence tomography (OCT), fluorescein angiography, and indocyanine green angiography. All clinical types of new vessels were included (Tab. I) except those associated with large atrophic scars and/or subretinal fibrosis, large subretinal hemorrhage, or large pigment epithelial detachments (PEDs). Eyes with retinal diseases other than AMD, such as diabetic retinopathy, retinal vein occlusion, or pathologic myopia, were excluded from the study. Uncontrolled glaucoma, uveitis, or any other ocular disease able to confound the study evaluations were also excluded.
Distribution of neovessels in the bimonthly and PRN groups, n (%)
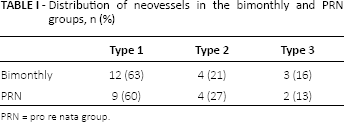
PRN = pro re nata group.
After an induction of 3 consecutive monthly loading doses of aflibercept, patients were randomly allocated to 2 different regimens: group 1 (bimonthly) (Fig. 1) had a fixed follow-up and treatment protocol, treated with fixed bimonthly injections and monitored every 4 months, while group 2 (PRN) (Fig. 2) patients were followed up and treated using a PRN protocol with monthly follow-up. Retreatment criteria were based on the PRONTO guidelines (6): 1) loss of 5 or more Early Treatment Diabetic Retinopathy Study (ETDRS) letters of best-corrected visual acuity (BCVA); 2) OCT signs of onset or persistence of intraretinal/subretinal exudation and/or central retinal thickness increase more than 100 μm; 3) new macular hemorrhage; and 4) new or expansion of new vessels evidenced by fluorescein angiography.
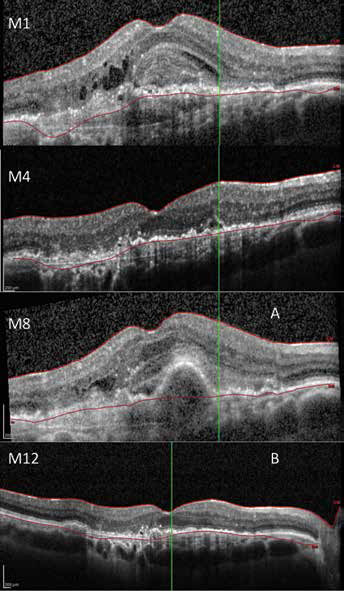
Example of visual and anatomical outcomes with bimonthly protocol from baseline to month 12. (
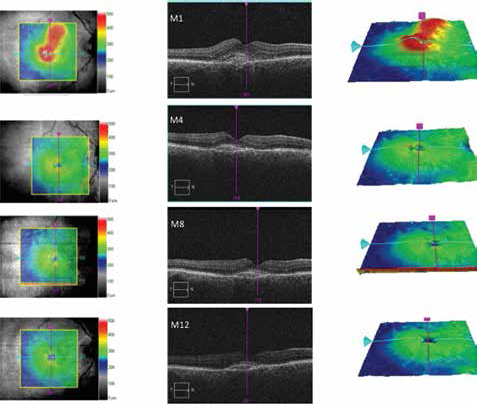
Example of visual and anatomical outcomes with pro re nata protocols over 1 year. Good anatomic and visual outcomes with +10 Early Treatment Diabetic Retinopathy Study letters and a central macular thickness improvement of 176 μm over the year are present. During the year, this patient had 9 injections and 7 follow-up visits.
All the study patients had a comprehensive ophthalmologic examination at every study visit: BCVA using the ETDRS scale, funduscopy, and OCT examination with central macular thickness (CMT) measurement. All patients were followed up over a period of 1 year; the number of follow-up visits and injections given were recorded.
Two different OCT devices were used for CMT evaluation: the Cirrus® OCT (Carl Zeiss Meditec) for the PRN group and the Spectralis HRA® OCT (Heidelberg Engineering) for the bimonthly group (Fig. 3). For comparison purposes, CMT measurement was converted to a Stratus OCT equivalent using the formula described by Smretschnig et al (11). Further, only CMT changes from baseline were compared between the bimonthly and PRN groups to reduce OCT device bias.
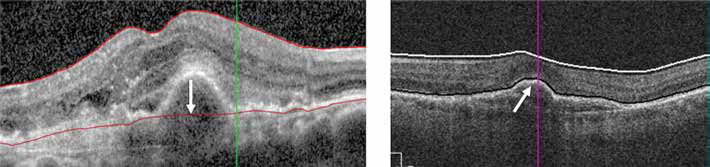
Difference in segmentation of retinal layers with Spectralis® and Cirrus® optical coherence tomography. Example of 2 patients using the automatic segmentation software. Right: On Spectralis® data, pigment epithelial detachments (PED) (arrow) are included in the measurement. Left: On Cirrus® data, PED are excluded from the retinal thickness.
Statistical analysis
In each group, the improvement in mean BCVA and CMT was analyzed using a Wilcoxon matched-pairs test or the paired t test if data passed the normality test. Best-corrected visual acuity and CMT changes were compared between the 2 groups using an unpaired t test. Two statistical software programs were used: GraphPad InStat 3.10 (2009) and Statview 5.0 (SAS Institute). A p value less than 0.05 was considered significant.
Results
Thirty-four eyes (15 in the PRN group and 19 in the bimonthly group) of 31 naive patients (13 male, 18 female) were included in this study and followed up for 1 year. The patients’ baseline characteristics are reported in Table II. Neovascular lesions were classified as type 1 (minimally classic new vessel), type 2 (classic neovascularization), and type 3 (retinal angiomatous proliferation). There was no significant difference between the 2 groups in the distribution of neovascular lesion type (Tab. I). The baseline mean visual acuities in the 2 groups were comparable, with an average BCVA of 57.2 (range 20-72) in the bimonthly group and 58.4 (range 28-80) ETDRS letters in the PRN group (p = 0.8206). A significant difference (p = 0.0092) in baseline CMT (bimonthly group 330.3, PRN group 249) was noted between the 2 groups despite using the CMT conversion formula.
Patient baseline demographic characteristics
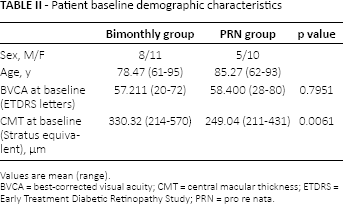
Values are mean (range).
BVCA = best-corrected visual acuity; CMT = central macular thickness; ETDRS = Early Treatment Diabetic Retinopathy Study; PRN = pro re nata.
Changes in visual acuity from baseline to the final visit at month 12 (Fig. 4)
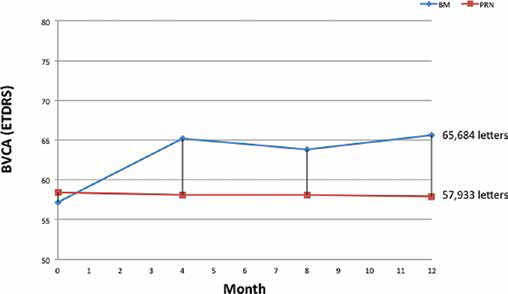
Best-corrected visual acuity progression at 1 year.
In the bimonthly group, the final BCVA (65.7) was significantly better than at baseline (57.2; p = 0.0046). In the PRN group, no statistical changes were observed between the baseline and final values (58.4 vs 57.9; p>0.05). At 1 year, the patients in the bimonthly group had a mean BCVA improvement of +8.5 ETDRS letters (range −12 to +40), whereas patients in the PRN group stabilized their average visual acuity with a small loss of −0.47 ETDRS letters (range −15 to +22). The visual gain was significantly better (p = 0.0278) in the bimonthly group than in the PRN group.
In the PRN group, 8 patients out of 15 lost vision at 1 year. They had a mean vision decrease of 8 letters (range 2-15) despite a mean of 8 follow-up visits (range 7-11). In the bimonthly group, only 2 patients lost vision (12 and 10 letters).
Changes in CMT from baseline to the final visit at month 12 (Fig. 5)
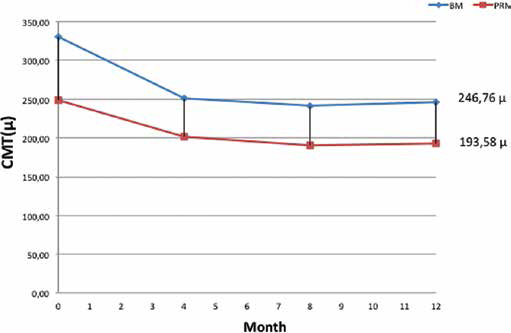
One year central macular thickness evolution.
In the bimonthly group, the final CMT (310.2 µm) was significantly lower than at baseline (404.7 µm; p<0.0001). Similarly, a significant decrease was also observed in the PRN group (289.3 µm vs 238.9 µm; p = 0.0119). The mean CMT change between the baseline and final visit was −97.36 µm (range −266.8 to +20.6) in the bimonthly group and −55.46 µm (range −180.8 to +51.7) in the PRN group. The observed CMT changes were not significantly different (p = 0.1181) between the 2 groups.
The mean number of injections received over 1 year (Tab. III) was similar in the 2 groups: the bimonthly group patients received 7.5 injections while the PRN group received 7.6.
Visual and anatomic outcomes, injections, and number of follow-up visits

BVCA = best-corrected visual acuity; CMT = central macular thickness; ETDRS = Early Treatment Diabetic Retinopathy Study; PRN = pro re nata.
All patients in the bimonthly group had 4 follow-up visits (Tab. III), whereas the mean number of follow-up visits in the PRN group was 7.8 (range 6-11). In the PRN group, patients with 7 or less than 7 follow-up visits over the year had an improvement of +3.3 letters with a mean number of 7.6 injections, whereas patients with 8 or more follow-up visits over the year had a mean decrease of 5.2 letters with 7.7 injections.
Extension of neovessels was not observed in the PRN group or in the bimonthly group.
Retreatment decision was due in all cases to vision loss or presence of intraretinal or subretinal exudation on OCT.
Discussion
The primary objective of this study was to compare the efficacy of the PRN protocol and a fixed bimonthly protocol using aflibercept over 1 year in patients with nAMD since limited data are available for comparing these 2 protocols in real-life situations with treatment-naive patients.
After 1 year of treatment, both protocols had improved anatomic CMT. However, bimonthly injections provided BVCA improvement while patients in the PRN group only had stabilized visual acuity. Our good results with a fixed bimonthly protocol are consistent with a recent study by Ricci and al (10). In this study, the authors compared both treatment protocols, but in eyes treated with aflibercept after a switch from ranibizumab. They observed an improvement of +3 ETDRS letters in the bimonthly group while the PRN group lost 3 ETDRS letters after 1 year of follow-up.
It is worth noting that the BVCA improvement we observed in the bimonthly group was similar to the one observed in the VIEW 1 and 2 studies (+8.1 letters in VIEW 1 and +9.4 letters in VIEW 2 at 52 weeks posttreatment) (9). On the other hand, Ricci et al (10) reported slightly inferior results in their bimonthly group (improvement of only +3 ETDRS letters), probably because their patients were not treatment-naive.
One of the potential advantages of the PRN protocol is the lower number of injections required per year. Surprisingly, we did not observe a difference in terms of the number of injections in the 2 groups (7.5 in the bimonthly group vs 7.6 in the PRN group). The number of injections in the PRN group was similar to the HARBOR (12) and CATT (7) studies, with 7.7 injections and 6.9 injections, respectively, over the year. Other real-life studies have shown that it is difficult to obtain this number of injections. For instance, Cohen et al (13) treated 122 patients with a mean number of 3.79 injections over the year with no visual gain (+0.7 letters) at 1 year. The LUMIERE study (13) reached the same conclusion: 551 patients were treated with a mean number of 5.1 injections per year with only 3.2 letters of visual gain. It therefore seems necessary to monitor patients monthly when using the PRN protocol to give early treatment and obtain optimal outcomes (14). This may be another reason why Ricci et al (10) had inferior outcomes in their PRN group compared with ours (-3 letters vs −0.47 letters) since the mean number of injections was only 3.5 per year in their study.
The different outcomes observed between the 2 protocols in our study are difficult to explain. The reactive PRN protocol of this study could have delayed retreatment. In fact, the theoretical number of monthly follow-up visits, after the induction sequence, should be 10. However, in this real-life series, 14 patients had fewer than 10 visits (6 to 9) and only 1 patient had an additional visit. This lower number of visits could result in delayed retreatment; we observed that patients with inferior visual acuity results had lower attendance than the group with better prognosis. The anxiety of patients with visual symptoms and visual acuity decrease in poor prognosis nAMD may lead patients to optimize their attendance. Another explanation may be the development of subretinal fibrosis following undertreatment; patients with 6 or fewer injections over the year had a mean visual acuity decrease of −3.5 letters.
The difference in outcomes between the 2 protocols may also be due to the small size of our study population and the retrospective design of this study and the fact that the 2 groups were treated and followed in 2 different settings by 2 different physicians. Nevertheless, no significant difference was observed between the 2 groups in the distribution of the type of neovessels. Concerning CMT, the quantum and speed of changes were similar in both the protocols: we observed a substantial CMT decrease after the 3 initial monthly loading doses followed by retinal thickness stabilization during the rest of the year. These results have been widely reported in previous studies (15-16-17-18-19).
We observed a significant difference between the 2 groups in baseline CMT; this difference remained significant during the whole study. The different OCT devices used in the 2 groups probably explains this difference. The Cirrus® OCT was used in the PRN group while the Spectralis HRA® OCT was used in the bimonthly group.
The retinal layer segmentation software of the Spectralis HRA® OCT includes PEDs and large drusen, while these are excluded from the retinal segmentation in the Cirrus® OCT software (Fig. 3) (20).
The mean changes in CMT in the 2 groups were similar over 1 year.
The main advantage of the fixed protocol is the reduction in the need for monthly monitoring. In our bimonthly group, patients were monitored every 4 months while patients in the PRN group had twice as many follow-up visits as those in the bimonthly group (7.8 visits versus 4). Also, patients in the PRN group had variable attendance (from 6 to 11 visits) during the 1-year follow-up. Variable attendance was also reported by Ricci et al (10), with about 15% of the scheduled visits lost during follow-up. The burden of monthly follow-up for both patients and ophthalmologists may induce a certain slackening of monitoring. This may also be a cause of the difference observed between the 2 groups in terms of visual outcomes (14).
Conclusion
Our results suggest that at 1 year of aflibercept treatment for nAMD, the fixed bimonthly retreatment protocol may provide better visual results than a PRN protocol with fewer follow-up visits and no difference in the number of intravitreal injections. This difference may be explained by noncompliance with the required monitoring of patients in the PRN protocol resulting in possible undertreatment in a real-life setting. Our results need to be confirmed by a larger prospective study. Nevertheless, strict bimonthly aflibercept injections, after 3 consecutive monthly loading doses, appears to be a valuable option for treating naive patients with nAMD for at least 1 year. Such a protocol enables patients to be monitored only every 4 months without any detrimental consequence to visual outcomes.
Footnotes
Financial support: No financial support was received for this submission.
Conflict of interest: None of the authors has conflict of interest with this submission.
