Abstract
Purpose
To evaluate the outcomes of deep anterior lamellar keratoplasty (DALK) followed by phacoemulsification and toric intraocular lens (IOL) implantation for the treatment of concomitant stromal disease and cataract.
Methods
In this retrospective non-comparative interventional case series, ten eyes affected by stromal disease and cataract underwent DALK followed by phacoemulsification with toric IOL implantation after a minimum period of 5 months from complete suture removal. In each case, topographic astigmatism, refraction, visual acuity, and endothelial cell density were recorded before DALK and 1, 6, and 12 months after cataract surgery. In addition, IOL rotation was evaluated using anterior segment optical coherence tomography.
Results
Big-bubble DALK was performed in all eyes but one that received manual dissection. Topographic astigmatism averaged 5.6 ± 2.2 diopters (D) after suture removal; refractive astigmatism decreased to 0.55 ± 0.61 D as early as one month after cataract surgery and did not change substantially throughout the follow-up period. In all patients, one month after phacoemulsification uncorrected and best spectacle-corrected visual acuity were, respectively, ≥20/40 and ≥20/25 with a residual spherical equivalent of 0.00 ± 0.84 D. At the latest follow-up visit, in all cases the IOL rotation was ≤5 degrees from the intended position and the endothelial cell loss within 8.5%. No complications were recorded.
Conclusions
DALK followed by phacoemulsification with toric IOL implantation optimizes visual and refractive outcomes in patients with concomitant stromal disease and cataract. In comparison with a combined procedure, the sequential approach offers better predictability of the postoperative refraction in the absence of an increased risk of complications.
Introduction
For many years, a combined surgical procedure including penetrating keratoplasty (PK), cataract extraction and intraocular lens (IOL) implantation, the so-called “triple procedure”, has been considered a valid option for the treatment of eyes with simultaneous stromal disease and cataract (1, 2). A combined approach may offer a visual rehabilitation faster than that obtained with sequential surgery, and avoid the damage to the transplanted endothelium caused by subsequent cataract surgery (3).
More recently, deep anterior lamellar keratoplasty (DALK) has been developed as an alternative to PK for the treatment of stromal diseases with healthy endothelium (4); when cataract is present, DALK in combination with phacoemulsification has been proposed as the surgical treatment of choice to restore vision (5).
Although this approach maintains the advantages of a single procedure, including the main one of a faster visual recovery, unsatisfactory refractive outcomes may result from both the difficulty of accurately calculating the IOL power and the possible occurrence of high-degree postoperative astigmatism (5, 6). In the last few years, phacoemulsification combined with toric IOL implantation has been reported to provide predictable and stable correction in eyes with cataract developing after PK or DALK in eyes with high-degree postoperative astigmatism, but no sequential strategy has been described to date for DALK and cataract surgery in eyes with concomitantly diseased cornea and crystalline lens (7-8-9-10).
To optimize the final refraction in eyes with stromal diseases and cataract, we performed a two-stage procedure including DALK followed by phacoemulsification and toric IOL implantation and report herein the outcomes of this approach.
Patients and methods
We reviewed the charts of 10 eyes of 10 patients with stromal disease and cataract, who underwent a two-step surgical treatment consisting of DALK (big-bubble technique) followed by phacoemulsification with a toric IOL implantation in the capsular bag. Inclusion criteria for implantation of a toric IOL were senile cataract and post-keratoplasty stable and regular topographic astigmatism higher than 2.00 diopters (D), as measured no earlier than five months after complete DALK suture removal.
Institutional review board approval was obtained from the University of Magna Graecia; the study followed the tenets of the 1964 Declaration of Helsinki and all patients had signed a detailed informed consent.
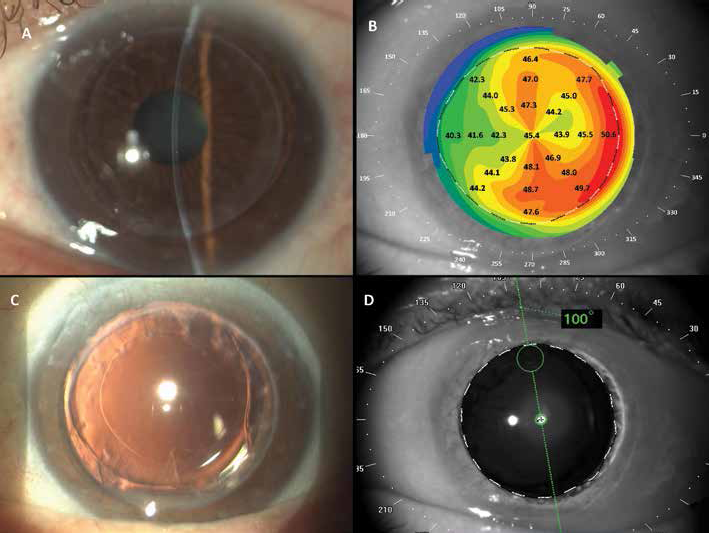
Before and after DALK, as well as 1, 6, and 12 months after phacoemulsification a complete ophthalmologic examination was performed including assessment of uncorrected (UCVA) and best spectacle-corrected visual acuity (BSCVA), refraction, slit lamp biomicroscopy (Fig. 1A and C), tonometry, and fundus examination in mydriasis; when preoperative corneal opacity did not allow fundus examination, a B-scan echography was performed.
(
The amount and meridian of post-DALK corneal astigmatism were determined by means of anterior segment optical coherence tomography (AS-OCT, SS-1000 CASIA, Tomey, Enlargen, Germany) (Fig. 1B). Optical partial coherence interferometry (IOLMaster, Carl Zeiss Meditec AG, Jena, Germany) was used for biometric purposes; all data were forwarded to the IOL manufacturer, who calculated the dioptric power of the lens aiming at postoperative emmetropia. Postoperatively, an internal AS-OCT software tool, included in the “Corneal map” section, was used to evaluate the toric IOL position and assess postoperative rotation through a dilated pupil (Fig. 1D), by simply aligning a linear cursor passing across the IOL center on the peripheral marks of the lens.
The targeted toric correction, residual postoperative astigmatism and spherical equivalent were recorded in each case and the accuracy of cylinder correction was calculated as percentage value of the achieved toric correction from the intended value. Endothelial cell density (ECD) was calculated with the cornea module of the HRT-II device (Heidelberg Technology, Heidelberg, Germany).
An analysis of variance (ANOVA) test was used to determine the significance of changes in UCVA, BSCVA, ECD, and IOL rotation. Statistical analysis was performed using GraphPad (GraphPad Software, La Jolla, California, USA); differences were considered statistically significant when the p value was <0.05.
Surgical technique
DALK was performed according to the classic big-bubble technique described by Anwar and Teichmann (11); all patients were treated on by a single surgeon (VS), under local anesthesia obtained with a peribulbar injection of a ropivacaine 0.75% solution.
In detail, a Hessburg-Barron suction disposable trephine (Katena Products Inc., Denville, NJ, USA) was used to create a circular incision in the recipient cornea, 8.00-8.25 mm in diameter and approximately 80% of corneal thickness in depth. A blunt thin spatula (model AE-2900, Asico, IL, USA) was inserted at the bottom of the circular incision and advanced centripetally into the stroma for 2-3 mm. Next, a blunt 27-gauge Fogla cannula (Bausch & Lomb Storz Ophthalmics, USA), connected with a 5-mL syringe filled with air, was inserted in the previous fashioned track and air was gently injected until a pneumatic dissection was obtained; the bubble was then opened and the floor of the bubble exposed by removing the detached stroma. When the big bubble was not obtained, surgery was completed by manual dissection of the residual bed.
The donor cornea was punched from the endothelial side with a Barron punch (Katena Products Inc., Denville, NJ, USA) to the same diameter of the trephination; the endothelium-Descemet membrane complex was gently stripped off and the graft secured in place with two 10-0 nylon running sutures. Starting from the next morning, eyedrops containing dexamethasone phosphate 0.1% and tobramycin sulfate 0.3% were administered every 2 hours, then tapered off over a 3-month period; in each patient, all sutures were removed within 12 to 15 months after DALK.
Cataract surgery was performed in all eyes under topical anesthesia with lidocaine 4% eyedrops; before surgery, the patient was positioned at the slit-lamp and the horizontal meridian marked with a sterile pen. Standard bimanual phacoemulsification was performed through a 2.2 mm limbal tunnel positioned as posterior as possible in order to minimize the astigmatic effect induced; a customized hydrophilic acrylic toric IOL (AT Torbi 709M, Carl Zeiss Meditec AG, Jena, Germany) was implanted in the capsular bag and then aligned with the corneal meridian previously marked by means of a Mendez ring (model AE-2765N, Asico, IL, USA).
Results
Ten eyes of 10 patients were enrolled (6 males and 4 females) with a mean age of 65.7 ± 4.7 (mean ± standard deviation) years. Indications for initial DALK surgery were: keratoconus (n = 4), scars of various origin (pterygium n = 2; post-PRK [Photo-Refractive Keratectomy] n = 1; post-infection n = 2), and corneal granular dystrophy (n = 1). A big bubble was achieved in 9 of 10 eyes; in the only case where air injection failed to yield a big bubble, surgery was completed with layer-by-layer manual dissection aiming to reach a predescemetic plane. No intra- or postoperative complications were recorded and the grafts remained clear during the entire follow-up period.
The average time elapsed between DALK and phacoemulsification was 18.0 ± 1.2 months (range = 17 to 20 months). After removal of all sutures, the mean topographic post-DALK corneal curvature was 44.7 ± 1.3 D and mean astigmatism measured 5.6 ± 2.2 D (range = 9.8 to 2.8 D); both corneal curvature and entity of astigmatism did not change substantially one year after phacoemulsification measuring 44.7 ± 1.1 D and 5.6 ± 2.3 D, respectively. The mean value of targeted residual cylinder (as provided by the manufacturer) was 0.28 ± 0.21; the refractive astigmatism and the residual spherical equivalent recorded one month after phacoemulsification with toric IOL implantation were 0.55 ± 0.61 D (range = 0 to 1.50 D) and 0.00 ± 0.84 D, respectively; both values did not change substantially throughout the follow-up period. The accuracy of cylinder correction was 89.9 ± 9.0% (ranging from 67.6 to 99.0). A significant improvement in UCVA was recorded between the average pre-DALK value (1.60 ± 0.37 logMAR) and the values recorded one year (0.16 ± 0.11 logMAR) after phacoemulsification; no eyes lost any lines of UCVA or BSCVA. The mean ECD recorded preoperatively was 2472.2 ± 352.4 cells/mm2 (range: 1703 to 2782 cells/mm2) and decreased to an average value of 2330.3 ± 345.6 cells/mm2 (endothelial cell loss = 5.8 ± 1.8%) at the last scheduled visit.
One month after implantation, the toric IOL showed a mean rotation from the intended position of 1.8 ± 1.5, (ranging from 0 to 5 degrees); no significant variation was recorded at latest follow-up. No eye had intraoperative or postoperative complication and all grafts remained clear up to last scheduled visit.
Table I summarizes the main pre- and postoperative data.
Pre-operative indications for surgery and main post-operative outcomes recorded 12 months after phacoemulsification
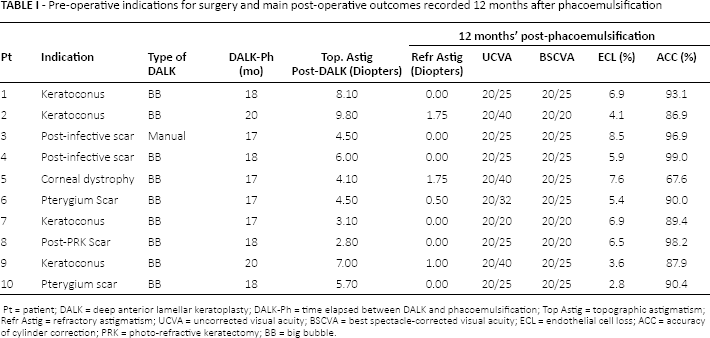
Pt = patient; DALK = deep anterior lamellar keratoplasty; DALK-Ph = time elapsed between DALK and phacoemulsification; Top Astig = topographic astigmatism; Refr Astig = refractory astigmatism; UCVA = uncorrected visual acuity; BSCVA = best spectacle-corrected visual acuity; ECL = endothelial cell loss; ACC = accuracy of cylinder correction; PRK = photo-refractive keratectomy; BB = big bubble.
Discussion
In the past, combined cataract surgery and keratoplasty have been performed routinely in eyes with concomitant corneal disease and cataract (1, 2). This type of approach offered several advantages over a sequential one (faster visual rehabilitation, elimination in general of all risks related to a procedure subsequent to the keratoplasty and specifically of possible damage to the donor endothelium), but did not allow a good predictability of the final postoperative refractive error, thus being complicated in a relatively high percentage of cases by high-degree astigmatism and anisometropia (12-13-14).
Moreover, so long as keratoplasty of the penetrating type was the gold standard, the surgeon could choose between performing phacoemulsification under “close system” conditions and removing the cataract manually “open sky”, mainly depending on corneal clarity. With DALK, the open sky approach is not possible and phacoemulsification can be performed only if the stromal disease is compatible with good visualization of the anterior segment structures (5). Also, for those eyes that can undergo combined phacoemulsification with DALK, the poor predictability of the postoperative refractive error and the incidence of high-degree astigmatism would prevent a satisfactory visual outcome in a relevant number of the patients, who would then require either contact lens fitting or additional refractive surgery (laser or incisional surgery) (15, 16).
In addition, as the patient's endothelium is left in place, even a mild visual hindrance to phacoemulsification may result in a loss of recipient endothelial cells higher than that possibly caused by the same procedure being performed at a later time after reestablishing normal corneal conditions with DALK.
In an attempt at both minimizing the damage to the recipient endothelium and optimizing the postoperative refractive error in eyes with concomitant stromal disease and cataract, we have performed DALK and phacoemulsification with implantation of a toric IOL in a sequential fashion.
In terms of safety for the recipient endothelium, the sequential approach has proved an optimal choice yielding a final endothelial cell loss (ECL) of 5.8% from the pre-DALK measurement, thus comparing favorably with any other type of combined or sequential corneal and cataract surgery (17). This value actually shows that ECL is minimally affected by DALK and phacoemulsification can be performed in post-DALK eyes with an ECL substantially equal to that recorded in eyes not operated previously. In addition, the ECL after DALK stabilizes relatively early after surgery, while it continues to decrease for many years after PK (18). A loss of 5.8% in the eyes of our series is not expected to vary substantially over time and is therefore compatible with long-term corneal transparency.
Postponing phacoemulsification after the removal of all DALK sutures would allow the measurement of a stable post-PK corneal curvature and therefore a more appropriate IOL selection. However, residual astigmatism would not be corrected by the implantation of a conventional monofocal IOL, thus reducing the potential advantage of a sequential approach.
In the last decade, toric IOLs have been implanted to correct moderate and high-degree post-PK astigmatism with excellent results in terms of refraction, visual acuity, and rotation stability (7-8-9). In our series, sequential surgery with implantation of a toric IOL after DALK was associated with optimal and stable refractive outcomes: the predictability was good with 8 of 10 patients within one D from the predicted postoperative refractive astigmatism (mean value of 0.50 ± 0.74 D) with a mean accuracy of cylinder correction of 89.9 ± 9.0%; in six eyes UCVA was ≥20/25, and patients elected not to wear any kind of correction. During the year following phacoemulsification, topography did not change substantially and the toric IOL showed a minimal rotation from the intended meridian (average value = 1.8 ± 1.5 degrees); as a consequence, refraction was stable throughout the whole post-phacoemulsification period considered in our study.
All eyes in our series showed a regular topographic pattern of post-DALK astigmatism and toric IOLs could be used without reservation. Instead, if the post-DALK astigmatism were of the irregular type, in particular when the hemi-meridians are not aligned forming an angle, incisional surgery in the DALK wound after stabilization of the post-phacoemulsification refraction would be preferable.
In keratoconus eyes, corneal curvature has been shown to change even to a great extent up to 17 years after PK and therefore toric IOLs should be considered with caution after PK performed for this indication (19, 20). However, these changes, often considered as a “recurrence of keratoconus” were seen in the presence of small grafts (usually not larger than 7 mm in diameter) with improper donor/host wound apposition, usually in young patients (21, 22). Refractory stability of DALK performed in keratoconus eyes has been reported with a 10-year follow-up (23). In addition, the larger graft diameters used for DALK in our series, the improved alignment between donor and host created by the maintenance of a recipient posterior layer and the older age of our patients, may all contribute to minimize this risk.
In our series, the average time elapsed between complete DALK suture removal and phacoemulsification was 5.6 ± 0.7 months (range = 5 to 7 months); no case of wound dehiscence or topographic astigmatism modification was recorded throughout the post-DALK follow-up period before phacoemulsification. After PK, the corneal graft-host junction was shown to heal usually within one year from surgery, yielding a stable corneal shape and anterior curvature as early as 3 to 4 months after complete suture removal (24); after DALK the shorter period of postoperative steroidal treatment and the non-penetrating type of wound could further reduce the time necessary for stabilization, possibly allowing cataract surgery even after a shorter time interval (25).
Although several surgical procedures could be employed to correct post-PK astigmatism (photo-refractive keratectomy, laser in situ keratomileusis, corneal arcuate keratotomy), the amount of correction is limited, especially if a relevant spherical ametropia is associated; in addition, their results are poor, predictable and repeated surgery is often required (24-25-26).
Implantation of a monofocal IOL still remains a good refractory option, especially in those patients who need spectacle correction for the fellow eye; however, in these cases a secondary astigmatic correction could be achieved by implantation of a “piggyback” toric IOL in the sulcus (27, 28).
A limitation of this study is the small cohort of patients and the relative short follow-up period; further studies with a larger number of patients and a longer follow-up period are required to confirm our positive experience. In conclusion, for patients with concomitant stromal disease and cataract, the prolonged visual rehabilitation induced by a two-step DALK and phacoemulsification should be weighed against the benefit of optimizing the postoperative refractory error by allowing the use of a toric IOL, while minimizing ECL. Especially for elderly patients like those included in our series, eliminating the need for contact lens fitting or additional refractive surgery is of crucial importance.
Footnotes
Financial support: No grants or funding have been received for this study.
Conflict of interest: Massimo Busin receives travel expenses reimbursement and royalties from Moria (Antony, France). Vincenzo Scorcia, Andrea Lucisano, Vincenzo Savoca Corona, Valentina De Luca, and Adriano Carnevali have no financial interest to disclose.
