Abstract
Purpose
To evaluate the short-term clinical outcomes of a specific toric diffractive trifocal intraocular lens (IOL) implanted following an optimized clinical protocol in a large population.
Methods
Retrospective analysis of 337 eyes of 231 patients (mean age, 62.2 years) undergoing cataract surgery with implantation of the trifocal diffractive IOL AT.LISA tri toric 939M/MP (Carl Zeiss Meditec). A strict and careful clinical protocol was followed, including an accurate measurement of corneal astigmatism, use of a latest generation IOL power calculator, photography-based method intraoperative control of IOL alignment and IOL reposition at 1 week postoperatively if needed. Clinical outcomes in terms of visual acuity, refraction, efficacy of astigmatic correction analysed by vector analysis and patient satisfaction were evaluated during a 3-month follow-up.
Results
A total of 82% and 98% of eyes achieved a postoperative uncorrected distance visual acuity of 0.00 and 0.10 logMAR or better, respectively. Furthermore, 99.7%, and 100.0% of eyes showed a postoperative spherical equivalent within ± 0.50 D and ± 1.00 D, with 97.9% of eyes having a postoperative cylinder ≤ 0.50 D. Uncorrected near and intermediate visual acuities were 0.2 logMAR or better in 89.0% and 99.1% of eyes, respectively. Mean difference vector, magnitude of error and angle of error were 0.02 ± 0.14 D, 0.02 ± 0.13 D and 0.11 ± 1.18°. Patient satisfaction was referred as high or very high by 97.6% of patients.
Conclusions
The implantation of the trifocal toric IOL evaluated following a careful clinical protocol provides an efficacious visual rehabilitation and astigmatic correction, leading to high levels of patient satisfaction.
Introduction
The correction of corneal astigmatism when implanting a multifocal diffractive intraocular lens (IOL) for providing a complete visual rehabilitation after cataract surgery has been shown to be crucial as it can induce a significant degradation of the visual acuity and quality. 1 Specifically, it has been demonstrated that uncorrected corneal astigmatisms of more than 0.75 D and high order aberrations (HOAs) can significantly limit the capability of diffractive multifocal IOLs ability to increase the depth of focus. 1 For this reason, toric multifocal IOL designs have experienced significant development in the last few years, with a great variety of studies reporting the efficacy and safety of this type of implant.2–12
Despite the excellent optical performance of current multifocal toric IOLs,13,14 some factors can lead to a limitation of the visual outcomes with this type of implant. 15 Specifically, the main sources of error in toric IOL power calculation have been found to be the preoperative corneal measurement (27%) and IOL tilt (11.3%) and misalignment (14.4%). 15 Likewise, pupil size, angle kappa, axial eye length, anterior chamber depth, surgically induced astigmatism (SIA), and decentration have been shown to be also potential contributors to the presence of residual refractive astigmatic error. 15 For this reason, more sophisticated clinical protocols are being developed to promote the optimization of clinical outcomes with multifocal toric IOLs, considering aspects such as the centroid SIA calculated by vector analysis, the use of advanced IOL power calculators, the intraoperative use of imaging systems allowing the surgeon to perform a highly accurate IOL alignment or the creation of corneal incisions with femtosecond laser technology.16,17 The aim of the current large population study was to evaluate the short-term clinical outcomes in terms of visual acuity, refraction, efficacy of astigmatic correction evaluated by vector analysis, and patient satisfaction of a specific toric diffractive trifocal IOL implanted following an optimized preoperative and intraoperative protocol based on a careful candidate selection, a comprehensive evaluation of corneal astigmatism, use of a latest generation IOL power calculator and an accurate intraoperative IOL alignment.
Methods
Patients
This study consisted of a retrospective analysis of 337 eyes undergoing cataract surgery with implantation of the trifocal diffractive intraocular lens AT.LISA tri toric 939 M/MP (Carl Zeiss Meditec, Jena, Germany) in our clinic. Inclusion criteria were subjects with unilateral or bilateral cataracts or subjects with unilateral or bilateral incipient cataracts with concomitant night vision disturbances that underwent routine cataract surgery. Exclusion criteria included history of ocular trauma, any inflammation or oedema (swelling) of the cornea, including but not limited to the presence of keratitis, keratoconjuntivitis or keratouveitis, subjects with macular degeneration or other retinal disorders, presence of epithelial, stromal, or endothelial dystrophy, previous retinal or ocular surgery, optic nerve atrophy, uveitis, iris neovascularization, iris atrophy, use of systemic or topical medication with the potential of interfering with visual performance, and active systemic diseases increasing surgical risks. The study was conducted according to the tenets of the Declaration of Helsinki, being approved by the medical ethics committee of AVER clinic.
Preoperative protocol
The preoperative examination consisted of medical history, measurement of monocular and binocular uncorrected visual acuity (UDVA), measurement of corrected distance visual acuity (CDVA), autorefraction, manifest refraction, measurement of monocular and binocular uncorrected (UNVA) and distance-corrected near visual acuity (DCNVA) at 40 cm, measurement of monocular uncorrected intermediate visual acuity (UIVA) at 66 cm, Goldman tonometry, pupillometry, slit lamp biomicroscopy, optical coherence tomography (Cirrus 500, Carl Zeiss Meditec, Jena, Germany), corneal topography (Orbscan, Bausch & Lomb Inc, Rochester, NY, USA), optical biometry (IOL-Master 500, Carl Zeiss Meditec AG, Jena, Germany), and fundus evaluation.
In all patients, measurements of astigmatism obtained by the optical biometer, autorefraction and corneal topography were recorded and compared. At least, four consecutive biometric measurements were taken in at least 2 different days for IOL power calculations that had to be consistent in terms of axial length (AXL) (±0.03 mm), anterior chamber depth (ACD) (±0.06 mm) and corneal astigmatism (magnitude and axis) (±0.25 D and ± 3°). 18 If they were not consistent, more consecutive measurements were taken until 4 of them were consistent according to our criteria. Finally, the biometric measurement in which the astigmatism has the greatest concordance with the rest of the astigmatisms measured (in value and axis) was selected for IOL power calculations. The preoperative calculation of the toric IOL was performed with the online ZCALC 1.5 calculator (Carl Zeiss Meditec, Jena, Germany) (https://zcalc.meditec.zeiss.com/), considering a historical level of surgically induced astigmatism (SIA) by the incision of less than 0.25 D for the surgeon that performed all procedures. The IOL power was selected to target as closest as possible to emmetropia.
Intraoperative protocol
The same experienced surgeon (BP) performed all surgeries using a standard sutureless phacoemulsification procedure. Before entering the operating theatre, a facial photograph of the patient was taken to rule out the presence of asymmetries. Subsequently, in the slit lamp, we checked alignment of the head and marked the 0–180° axis of the eye to be operated and took a photo that was opened in the Goniotrans application (https://www.goniotrans.com/), which is a free access tool to reference the angular position of the ocular structures on an image of the eye. Specifically, the photographic image of the eye is copied and pasted into the program, which superimposes a template that simulates a protractor with two superimposed hemimeridians with marks from 0° to 180° in steps of 5° or 10°. Once the marks had been visualised with Goniotrans, we remark the 0°-180° meridian, and mark the lens implantation axis and the incision axis at 140° under the microscope with a chopper to make the marks as fine as possible and then facilitate checking the alignment of the IOL. In all cases, a 2.2 mm corneal incision was made at 140°, as well as a 60–90 degrees paracentesis clockwise from the main incision. After performing a large capsulorhexis (between 5.5 and 6.0 mm), insertion of the IOL and its alignment, the surgical procedure was completed. On leaving the operating theatre, a photograph of the eye was taken at the slit lamp to confirm the position of the IOL with the help of the Goniotrans application, which should be within ± 2° of the intended axis. If the level of IOL alignment was poor, the IOL was repositioned at 1 week after surgery.
Postoperative protocol
All patients were evaluated the day after surgery, and at 1 week, 1 month and 3 months postoperatively. At 1 week after surgery, if the visual or refractive outcomes were not acceptable, the pupil was dilated and the position of the IOL evaluated. If needed, the IOL was repositioned. In the last postoperative visit of this postoperative follow-up, the following tests had been performed in all cases: measurement of monocular and binocular UDVA, CDVA, UNVA and DCNVA (40 cm), measurement of monocular UIVA (66 cm), manifest refraction, slit lamp biomicroscopy, corneal topography, optical coherence tomography, and evaluation of patient's satisfaction using a self-developed scale. Specifically, patient satisfaction level was scored from one to five, being 1 the lowest level and 5 the highest: 1, very low; 2, low; 3, moderate; 4, high; 5, very high.
Statistical analysis
The software SPSS version 22.0 for Windows (SPSS, Chicago, Illinois, USA) was used for data analysis. First, the normality of all data distributions was assessed using the Kolmogorov-Smirnov test. The analysis of differences between preoperative and postoperative visits was done with the paired Student t or Wilcoxon tests if the data samples were or were not normally distributed, respectively. Pearson or Spearman correlation coefficients (with or without the assumption of normality of data samples, respectively) were used to analyse how strong was the relationship between different variables. The Chi-square test was used for the comparison of percentages. The criterion for the presence of statistical significance was defined as a p-value of less than 0.05.
For the analysis of the efficacy of the astigmatic correction, the Alpins vector analysis method was used.19,20 The following vectors were calculated with this methodology: the targeted induced astigmatism (TIA), that represents the intended change in cylinder in each case, the surgically induced astigmatism (SIA), that represents the real astigmatic change achieved and the difference vector (DV) that is equivalent to the astigmatic change required allowing the initial surgery to reach the initial intended target. Additionally, the angle of error (AE) (angular difference between SIA and TIA) and the magnitude of error (ME) (difference between the magnitude of SIA and TIA) were calculated.
All refraction notations were converted into a vectorial notation according to the power vector method described by Thibos and Horner.
21
Therefore, the spherocylindrical refractive error was expressed by a vector in a 3-dimensional dioptric space:
M/SE: spherical equivalent J0, J45: the two Jackson crossed cylinders equivalent to the conventional cylinder B: length of the vector representing the overall blurring strength
Finally, the standard graphs for reporting astigmatism outcomes of refractive surgery were adopted to present the results of the current series, according to the procedure described by Reinstein et al.
22
Results
A total of 337 eyes of 231 patients with mean age of 62.2 years (SD: 10.0, median: 62.0, range: 26 to 92 years) were enrolled. Unilateral surgery was performed in a total of 125 patients (54.1%), while bilateral surgery was performed in the rest. In all patients with unilateral surgery, the fellow eye had not undergone cataract surgery previously. The sample was comprised of 92 males (40.0%) and 139 females (60.0%), and of 177 (53.0%) right eyes and 160 (47.0%) left eyes. Mean AXL and ACD were 23.53 mm (SD: 1.51, median: 23.27, range: 20.74 to 29.76 mm) and 3.23 mm (SD: 0.40, median: 3.23, range: 2.11 to 4.38 mm), respectively. Mean preoperative corneal astigmatism was 1.77 D (SD: 0.92, median: 1.59, range: 0.61 to 7.01 D). Mean spherical and cylindrical powers of the IOLs implanted were 19.61 D (SD: 5.03, median: 20.50, range: 0.00 to 31.00 D) and 2.00 D (SD: 1.23, median: 1.50, range: 1.00 to 8.50 D), respectively. Reposition of the IOL at 1 week after surgery was needed in a total of 5 eyes (1.5%).
Table 1 shows a summary of the preoperative and postoperative visual and refractive data obtained in the current study. A statistically significant improvement was found after surgery in monocular and binocular UDVA and CDVA, monocular UIVA, and monocular and binocular UNVA and CNVA (p ≤ 0.001). A total of 82% and 98% of eyes achieved a postoperative monocular UDVA of 0.00 and 0.10 logMAR or better, respectively (Figure 1). This visual change was associated with a significant reduction in manifest sphere, cylinder, SE, J0 and B (p ≤ 0.030). However, the change in J45 did not reach statistical significance (p = 0.103). A total of 99.7% and 100.0% of eyes showed a postoperative spherical equivalent within ± 0.50 D and ± 1.00 D, respectively (Figure 1). Likewise, a total of 330 eyes (97.9%) had a postoperative cylinder of 0.50 D or below.
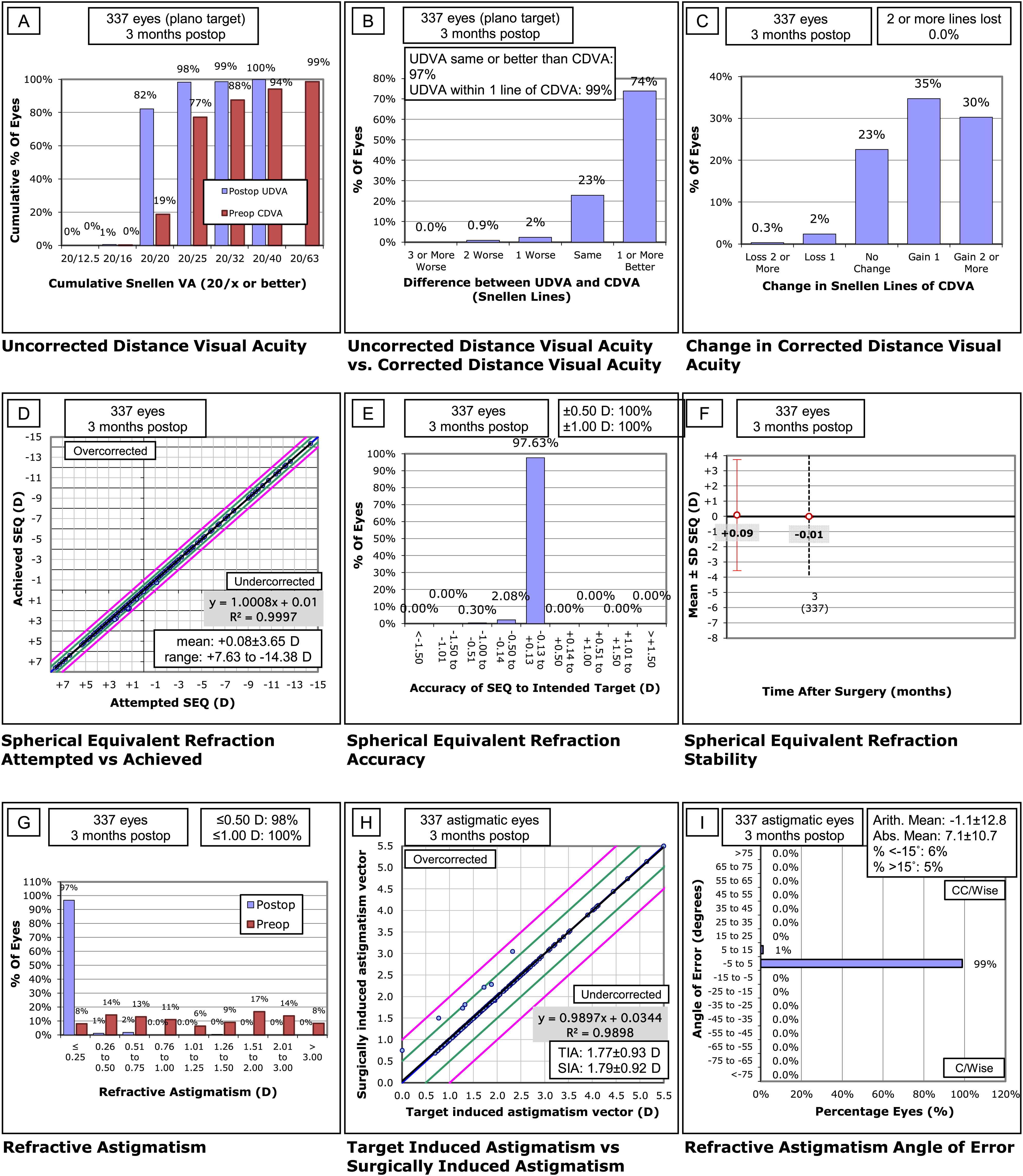
Standard graphs for reporting the visual and refractive outcomes following the guidelines described by reinstein et al. 22 .
Summary of preoperative and postoperative visual and refractive data in the whole sample. The corresponding p-values for the comparison between visits are shown for each parameter evaluated.

*Abbreviations: SD, standard deviation; D, diopters; UDVA, uncorrected distance visual acuity; UIVA, uncorrected intermediate visual acuity; UNVA, uncorrected near visual acuity; CDVA, corrected distance visual acuity; CNVA, corrected near visual acuity; J0 and J45, power vectors; B, overall blur strength; IOL, intraocular lens.
Concerning intermediate and near visual outcomes, a total of 89.0% and 99.1% of eyes achieved a postoperative monocular UIVA and UNVA of 0.2 logMAR or better, respectively. These percentages changed to 43.3% and 93.8% for monocular UIVA and UNVA values of 0.10 logMAR, respectively. Postoperative monocular CNVA was 0.10 logMAR or better in 90.0% of eyes. Likewise, postoperative binocular UNVA was 0.1 logMAR or better in 97.9% of cases.
According to the vector analysis performed, mean values for TIA and SIA were 1.77 D (SD: 0.93, median: 1.59, range: 0.50 to 7.00 D) and 1.79 D (SD: 0.92, median: 1.60, range: 0.61 to 7.00 D), respectively (Figure 1). Mean value of DV and ME was 0.02 D (SD: 0.14; median: 0.00; range: 0.00 to 1.50 D) and 0.02 D (SD: 0.13; median: 0.00; range: −0.24 to 1.44 D), respectively. Concerning the AE, it ranged from 13.99° to −4.68°, with a mean value of 0.11° (SD: 1.18; median: 0.00°). An AE within ± 3° and ± 5° was found in a total of 96.7% and 98.8% of eyes, respectively. The preoperative magnitude of corneal astigmatism did not correlate significantly with DV (r = 0.08, p = 0.14), ME (r = 0.08, p = 0.16) or AE (r = 0.02, p = 0.75).
Concerning patient satisfaction score (1, lowest level; 5, highest level) with the vision achieved after surgery, a mean value at 3 months of 4.7 (SD: 0.5; median: 5.0; range: 3.0 to 5.0) was reported (Figure 2). The level of satisfaction with the vision achieved at the end of the follow-up was high or very high in 97.6% of patients.
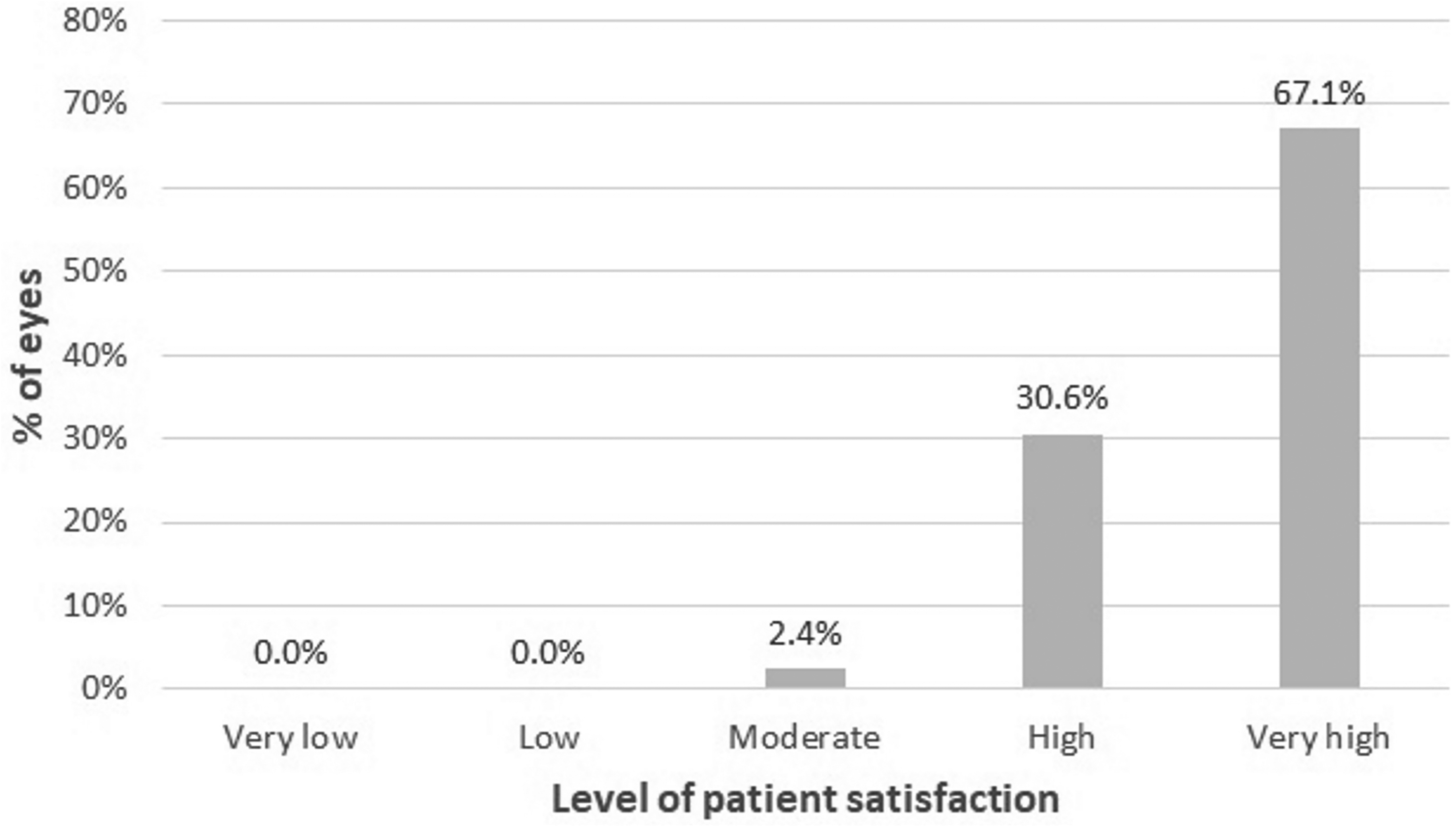
Distribution of patient satisfaction scores at the end of the follow-up.
Discussion
In the current sample, the clinical outcomes of the AT LISA trifocal toric 939MP diffractive IOL have been evaluated in a large population. Several studies have also investigated the outcomes of this specific type of multifocal IOL previously in other samples, reporting good but variables outcomes.2,8,11,12,23–28 Different factors might have contributed to this variability among authors, such as the type of toric IOL power calculator used (considering or not the contribution of the posterior corneal surface), the type of measurement of anterior corneal astigmatism considered, the intraoperative IOL alignment procedure or the mode of calculating and considering in IOL power calculations the SIA. 15 Indeed, there is evidence of the benefit of following a strict preoperative and intraoperative control of all these variables with the potential of affecting the final clinical outcomes when implanting toric IOLs.16,17 In the current study, we report our clinical experience during the last years with the AT LISA trifocal toric 939MP IOL using a very careful preoperative, intraoperative and postoperative protocol, including a comprehensive and very strict procedure for obtaining the anatomical measures required for IOL power calculations, the use of ZCALC calculator, an intraoperative photography-based control of IOL alignment, and a strict criterion for IOL rotation in the very early postoperative period.
In the sample evaluated, the change in refraction was statistically significant, with a mean 3-month postoperative sphere and cylinder of 0.00 ± 0.06 D and −0.03 ± 0.14 D, respectively. Likewise, almost all eyes (99.7%) showed a postoperative spherical equivalent within ± 0.50 D, and 97.9% of eyes had a postoperative cylinder of 0.50 D or below. This confirms that the implantation of the multifocal toric IOL evaluated using the clinical protocol described allows a highly efficacious correction of pre-existing corneal astigmatism. These results are better than those reported by previous authors implanting the same model of multifocal toric IOL.8,12,23,27 Piovella et al 8 reported with the same model of toric multifocal IOL a percentage of eyes with a postoperative manifest cylinder ≤ 0.50 D of 79.7%. Similarly, Mojzis et al, 12 in another series, found that 80% of eyes implanted with the toric multifocal IOL AT LISA tri 939 had a 3-month postoperative refractive cylinder of 0.50 D or below. Furthermore, with the same type of toric multifocal IOL, Almunhim et al 23 found that 87.5% of eyes had a postoperative spherical equivalent within ± 0.50 D and Kretz et al 27 reported a percentage of eyes with a 3-month postoperative cylinder of 0.50 D or below in 78.6% of eyes. However, other authors have reported more predictable outcomes close to those found in the current series.11,26,28 This confirms that some level of variability in the outcomes can be expected with the implantation of the toric multifocal IOL evaluated depending on the protocol followed for IOL power calculation, the selection of the corneal astigmatism to correct, intraoperative IOL alignment or IOL reposition during the follow-up. With the protocol followed in the current study, the predictability of the refractive correction achieved was high, which can be attributed to the combination of various factors considered preoperatively, intraoperatively and postoperatively.
The excellent level of refractive correction achieved was consistent with the optimum distance, intermediate and near visual outcomes obtained. This confirms the ability of the toric multifocal IOL of providing not only an effective distance visual restoration, but also a functional visual rehabilitation at intermediate and near distances, as in previous studies.2,8,11,12,23–28 Mean monocular UIVA (66 cm) and UNVA (40 cm) values of 0.14 ± 0.11 and 0.03 ± 0.06 logMAR were obtained in the sample evaluated in the current study. Bilbao-Calabuig et al 11 found in a sample of 4282 eyes implanted with the AT LISA trifocal toric IOL a mean binocular UNVA measured at 40 cm of 0.05 ± 0.08 logMAR as well as a mean UIVA measured at 80 cm of −0.05 ± 0.14 logMAR. Mojzis et al 12 found in a sample of 30 eyes implanted with the same trifocal toric IOL mean monocular UNVA (40 cm) and UIVA (66 cm) values of 0.16 ± 0.09 and 0.09 ± 0.11 logMAR, respectively. Kretz et al 27 reported in another case series mean 3-month postoperative UIVA and UNVA values of 0.08 ± 0.15 and 0.13 ± 0.18 logMAR, with all eyes achieving distance-corrected intermediate and near visual acuities of 0.10 logMAR or better. In our sample,
89.0% and 99.1% of eyes achieved a postoperative monocular UIVA and UNVA of 0.2 logMAR or better, respectively. Binocularly, a total of 97.9% of patients achieved a postoperative UNVA of 0.1 logMAR or better
The efficacy of the correction of astigmatism was analysed by vector analysis, obtaining mean values of DV, ME and AE of 0.02 D, 0.02 D, and 0.11°, which confirms the low level of error of the astigmatic correction and the ability of the implant of generating an astigmatic correction on axis for any type of astigmatism as no correlation was found between vector analysis parameters and the magnitude of corneal astigmatism. This outcome was slightly better than those reported in previous studies evaluating by vector analysis the efficacy of the astigmatic correction achieved with the same trifocal toric IOL used in the current series. Piovella et al 8 reported a mean ME of −0.16 ± 0.48 D, corresponding to a slight trend to undercorrection, whereas Mojzis et al 12 obtained a mean ME of 0.06 ± 0.30 D (mean AE 5.80 ± 8.47° and mean DV 0.35 ± 0.25 D), which represents a slight trend to overcorrection. This confirms the possibility of variability in the efficacy of astigmatic correction achieved with the toric IOL evaluated that can be attributed to several factors that have been controlled in the current series, such as the selection of the method for IOL power calculations or the intraoperative control of IOL alignment. Only 5 degrees of misalignment of a toric IOL reduces the efficacy of cylindrical correction by about 15%. 29 In our study, the Goniotrans application was used to allow reference the angular position of the IOL axis on an image of the eye. Likewise, the ZCALC calculator was used that have been shown to provide reliable and accurate refractive outcomes in toric IOL calculation,30,31 introducing some adjustments to consider the potential contribution of posterior corneal astigmatism to total corneal astigmatism. Furthermore, IOL reposition was always performed in those cases showing a clinically significant level of IOL rotation in the early postoperative period. Oshika et al 32 concluded in a multicenter retrospective study that there was a relationship between the timing of repositioning surgery and surgical outcome, recommending that repositioning surgery should be performed 1 week after IOL implantation.
Finally, the level of patient satisfaction with the surgical outcomes was evaluated. A mean score of 4.7 on a scale from 0 to 5 was obtained at 3 months postoperatively, being the level of satisfaction with the vision achieved high or very high in 97.6% of patients. These results are consistent with those reported in previous studies evaluating the outcomes of the same toric multifocal IOL used in the current series despite the presence in some cases of lower levels of efficacy for the astigmatic correction achieved.8,11,23,25 Piovella et al 8 reported that 93.3%, 89.4% and 84.6% of patients implanted in their series with the AT LISA trifocal toric 939MP IOL referred to good or very good satisfaction with distance, intermediate and near vision. Bilbao-Calabuig, 11 in their series of 4282 eyes, found that 98% of patients were satisfied or very satisfied with the implantation of the same trifocal toric IOL. In the sample of Almunhim et al, 23 nearly all the patients were satisfied with the surgical outcome achieved with this IOL, and in the study from Brozková et al, 25 all patients were satisfied with their vision.
This study has several limitations that should be mentioned, such as its retrospective nature, the measurement of UIVA at 66 cm rather than 80 cm (manufacturer recommended), and the use of self-developed questionnaire as opposed to a validated questionnaire. Future prospective studies should be performed to confirm the data obtained using a validated questionnaire. Likewise, comparative studies of the outcomes with the same trifocal toric IOL using different preoperative, intraoperative, and postoperative protocols should be additionally performed to define the most optimal approach.
In conclusion, cataract surgery with implantation of the AT LISA tri toric 939MP IOL following a careful preoperative, intraoperative, and postoperative protocol allows a very efficacious correction of pre-existing corneal astigmatism and a complete visual rehabilitation, leading to high levels of patient satisfaction. The excellent results obtained with this toric IOL in the current series compared to some previous studies can be explained by the combinations of two factors: the use of a strict pre- and intraoperative protocol, and the early intervention for IOL rotation.
Footnotes
Disclosure
The authors have no proprietary or commercial interest in the medical devices that are involved in this manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Support for this publication was provided by an unrestricted grant from Carl Zeiss Meditec, Inc. Carl Zeiss Meditec, Inc had no role in the design, data collection, analysis, decision to publish, or manuscript preparation. The authors alone are responsible for the content and writing of the paper
The author David P Piñero has been supported by the Ministry of Economy, Industry and Competitiveness of Spain within the program Ramón y Cajal, RYC-2016-20471
