Abstract
Diabetic nephropathy is one of the most frequent microvascular complications in diabetic patients. This report describes a case of diabetic nephropathy in an male adult (diabetic) patient treated with standard therapy and the contribution of the new antidiabetic drugs on the progression of the disease. We will deal the following questions:
What do we know about diabetic nephropathy and its natural history? How should we manage diabetic nephropathy and what do the guidelines suggest on hyperglycemia? How do we manage hyperglycemia in diabetic patients with chronic kidney disease (CKD)? Standard treatment. What's news about antidiabetic medication? New treatment. What's next?
Case Report
A 49-year-old man was admitted to the nephrology department. He was referred to hospital by his general practioner (GP) for a history of diabetes, diagnosed at the age of 43, associated with renal impairment. The patient underwent nephrectomy due to a renal cell carcinoma and experienced two heart attacks. His mother was diabetic. On admission to hospital the patient was in good general condition and the physical examination showed blood pressure 125/80 mmHg under control with angiotensin-converting enzyme inhibitors (ACEi) (ramipril 5 mg), absence of edemas, body weight 104 kg, height 181 cm, body mass index (BMI) 31.25 kg/m2. The blood parameters showed serum creatinine (sCr) of 1.81 mg/dL, blood urea nitrogen (BUN) 25 mg/dL, fasting plasma glucose (FPG) 100 mg/dL, glycated hemoglobin 1c (Hb1Ac) 44 mmol/mol, trygliceridemia 405 mg/dL, cholesterolemia 281 mg/dL, low-density lipoprotein (LDL)-cholesterol 145 mg/dL, and microalbuminuria 136 mg/L. The glomerular filtration rate (GFR) calculated by the modification of diet in renal disease (MDRD) equation was 40 mL/min/1.73 m2. The echotomography of the kidney showed a compensatory growth with normal general characteristics. With regard to organ impairment, the fundus oculi showed an initial damage with rare microaneurysm and intraretinal hemorrhages, without edema or exudates. The echocardiogram showed normal values for the whole cardiac measurement except for the initial left ventricular hypertrophy (LVH), with a normal result of 60% for the left ventricular ejection fraction (LVEF) and without valvular abnormalities. Finally, the results of the arterial venous Doppler ultrasound examination of the neck and lower limbs and of the upper and lower limbs electromyography (EMG) were normal. The glycemic parameters were kept under control with metformin 1000 mg twice a day. The patient was a nonsmoker, not addicted to alcohol, but his overweight suggested that a prompt lifestyle change was mandatory. He started taking omega-3 (1000 mg twice a day) and a combination of ezetimibe-simvastatin 10/20 mg once a day.
What do we know about Diabetic Nephropathy and its Natural History?
The incidence and prevalence of type 2 diabetes mellitus (T2DM) will continue to increase and it's widely recognized that the number of adults with diabetes worldwide will rise from 382 million in 2013 to 592 million in 2035, especially in developing countries (1). Diabetic nephropathy (DN) is one of the most frequent microvascular complications of diabetes. Whereas in the USA has been estimated that 50% of prevalent patients under dialysis are affected by diabetes (2), in Italy the prevalence of diabetic patients on dialysis has been estimated at 25.3% (3). Fortunately, only one-third of patients with T2DM experience DN (4).
According to medical literature, diabetes leads to significant renal abnormalities in a period of approximately 20 years and the reduction in estimated glomerular filtration rate (eGFR) can change from −3.57 to −1.31 mL/min/1.73 m2 every year due to several concurrent factors (5). The first mechanism starts with both hypoperfusion and hyperfiltration stimulating albumin loss across the glomerular capillaries. Also, we see structural changes that lead to kidney fibrosis as the thickening of the glomerular basement membrane, glomerulosclerosis, and expansion of the mesangial cells (6). Although the clinical manifestations of DN include the reduction in GFR and the increase of urinary albumin excretion, many diabetic patients do not show albuminuria (7). On the other hand, poor glucose control, arterial hypertension, hypercholesterolemia, and the activation of mediators of inflammation and oxidative stress promote the nephropathy progression through the advanced stages (8).
DN can definitely be divided into five stages: (i)
Moreover, it is well known that in dialysis patients and after renal transplantation the pathophysiology of diabetes changes during the chronic kidney disease (CKD) stages. During this stage, many variations occur in glucose sensitivity, insulin production and insulin resistance, which lead to different therapeutic approaches. Moderate-to-severe CKD seems to be associated with reductions in insulin sensitivity and clearance but not in a CKD-specific deficit in insulin secretion. Moreover, the combination of insulin resistance and inadequate augmentation of insulin secretion led to a high prevalence of impaired glucose tolerance (11). Furthermore, insulin resistance has been reported to be a strong risk factor for cardiovascular disease and ESRD in diabetic patients (12). On the other hand, in transplant population the hyperglycemia appears rapidly and the transition to diabetes is clearly much faster than in T2DM due to a variety of
How should we Manage Diabetic Nephropathy and what do the Guidelines Suggest on Hyperglycemia?
Hyperglycemia, blood pressure control, proteinuria, dislipydemia, and change of lifestyle represent the target of diabetes care. The recent guidelines of the American Diabetes Association (ADA) highlighted the concept of “Patient-Centeredness”. It is noteworthy that all the diagnostic, therapeutic and clinical approaches should be tailored to the patient preferences, literacy and numeracy, and address the cultural barriers to care (14). Since diabetes represents part of the so called “metabolic syndrome”, it is worldwide recognized that a multidisciplinary approach to manage the disease and its microvascular and cardiovascular complications is necessary (15). In this context, the definition of risk factors related to DN and the interventions necessary to slowing its progression is important. The first step to reach the goal is the change in lifestyle: smoking cessation, increase of physical activity, and establish adequate dietary guidelines (14, 16, 17). However, the next step, that should be considered “interventional”, consists of all the medical treatment required to prevent the development of complications based on the target defined firstly by the guidelines, and secondly by the clinician's experience (14, 18).
Glycemic control: The intensive treatment of hyperglycemia, cause of vascular target organ complications, seems to prevent elevated albuminuria or delays the kidney failure progression, even in patients treated with normoglycemic approaches who may be at risk of severe hypoglycemia (19). The National Kidney Foundation Diabetes Guidelines recommend a target glycated hemoglobin (HbA1c) of <7.0% to prevent or delay progression of the microvascular complications of diabetes, including DN and not treating to an HbA1c target of <7.0% in patients at risk of hypoglycemia. Guidelines also suggest that target HbA1c should be extended above 7.0% in individuals with co-morbidities or limited life expectancy and high risk of hypoglycemia (14, 18). Also, the ADA recommends a premeal glycemic target to 80-130 mg/dL (4.4-7.2 mmol/L) and a postprandial peak capillary plasma glucose <180 mg/dL (10.0 mmol/L) (14, 19). Moreover, since cardiovascular disease (CVD) is a more common cause of death than microvascular complications in diabetic patients, the cardiovascular benefit of intensive glycemic control after long-term follow-up of study in cohort patients early treated in the course of type 1 and type 2 diabetes (20) was demonstated.
In a healthy population the maintenance of the stability of plasma glucose is predominantly due to the counterregulatory system. The counterregulatory system is altered in patients with diabetes. The hypoglycemia prevention is a critical component of diabetes management and the presence of CKD adds additional risk factors (21). In fact, in moderate and severe CKD stages the reduction in renal mass and renal gluconeogenesis leads to a reduced capacity for renal glucose excretion (22). Also, a decrease in renal clearance of insulin is evident when GFR falls below 15-20 mL/min/1.73 m2 (23). Therefore, in people with diabetes affected with CKD, the antidiabetic dose adjustment is mandatory.
How do we Manage Hyperglycemia in Diabetic Patient with CKD? Standard Treatment
Management of hyperglycemia requires a multifactorial approach that includes medicines, nutrition planning and physical activity. Each of these approaches may need to be modified in the setting of CKD. With regard to medicines in type 1 diabetes, insulin is the mainstay of therapy and can be associated with metformin to reduce insulin requirements and improve metabolic control especially in overweight/obese patients (24, 25). On the other hand, in T2DM all the other conventional therapies, except pioglitazone (and insulin), must be reduced or withdrawn as eGFR declines. At present we know that many details of the conventional therapy of T2DM and treatment regimen have been evolving daily. Conventional and newer antidiabetic therapy is described below and its dose reductions in CKD is summarized in Table I.
Antidiabetic medication and dose adjustment in CKD

CKD = chronic kidney disease; GFR = glomerular filtration rate; SGLT-2 = sodium-glucose co-transporter-2; GLP-1 = glucagon-like peptide-1; DDP-4 = dipeptidyl peptidase-4.
Metformin is widely sustained as initial therapy by clinicians because of its low cost, safety profile, and potential cardiovascular benefits (26, 27). If not contraindicated and if tolerated, metformin is preferred as initial pharmacological agent for T2DM. In a recent review based on 179 trials and 25 observational studies, the use of metformin as monotherapy or in combination as first-line treatment of hyperglycemia has been strongly recommended, given its relative safety and beneficial effects on hemoglobin A1c, weight, and cardiovascular mortality (compared with sulfonylureas) (28). Furthermore, metformin has been extensively studied to suppress tumorigenesis by direct and indirect mechanisms (29).
Since the only route of elimination is via the kidneys, it may accumulate in people with impaired renal function. Guidelines suggested the use of metformin in diabetic patients when GFR >45 mL/min/1.73 m2 and its discontinuation in patients with GFR <30 mL/min/1.73 m2 due to the recurrence of metformin-related lactic acidosis (14, 18).
The sulphanylureas glipizide, glibenclamide, gliclazide, and glimepiride can be used as monotheraphy as well as in combination, but should be used with caution in CKD patients. Hypoglycemia risk is increased as a consequence of accumulation of the sulfonylurea and/or its active metabolites’ duration of action (30). Glibenclamide is cleared by the kidneys and its use is not recommended in people with eGFRs <60 mL/min/1.73 m2. Glimepiride and gliclazide can be used with caution in people with mild-moderate renal insufficiency, and dose reduction is required when eGFR is <30 mL/min/1.73 m2. Finally, glipizide is the preferred sulfonylurea, due to its liver metabolism, but usually at a reduced-dose, in people with CKD (21).
Meglitinides, such as repaglinide, are metabolized by the liver and, with dose adjustment, may be used in patients with CKD (31).
Also, thiazolidinediones, such as pioglitazone, do not require dose adjustment with impaired renal function (stages 2-5 CKD), because they are metabolized mainly by the liver. However, fluid retention is a major limiting side effect and they should not be used in advanced heart failure and CKD (18, 32).
There are no restrictions on the use of insulin in patients with renal disease. Nevertheless, people with severe renal disease (eGFRs <30 mL/min/1.73 m2) may have reduced capacity of the liver and therefore the kidney is not able to release glucose and reverse insulin-mediated hypoglycemia, so insulin requirements may decrease by 20% or more when GFRs decrease below 45 mL/min/1.73 m2. Moreover, insulin requirements are often lower the day after hemodialysis (18, 21).
What's new about Antidiabetic Medication? New Treatment
Diidro-peptidil-peptidase IV (DPP-IV) inhibitors and glucagon-like peptide-1 receptor agonists (GLP-1RAs), which develop their action by stimulating the incretin system, and sodium-glucose cotransporter-2 (SGLT-2) agonists are the most recent classes of antidiabetic agents licensed for use in T2DM in Europe. Also, it is important to consider if these newer therapies can be used safely and effectively in CKD patients, especially because the reduction in albuminuria was observed in many of these drugs. With regard to the DPP-IV inhibitors and GLP-1RAs, it is important to understand the physiology of the incretin system (Fig. 1) (33). DPP-IV inhibitors, such as sitagliptin, vildagliptin, saxagliptin, alogliptin, and linagliptin, reduce the physiological breakdown of native GLP-1, which increases the secretion of insulin and promotes satiety, effectively decreasing blood glucose levels. All of them can be used in CKD patients but where sitagliptin, saxagliptin, and vildagliptin need downward dose adjustments as detailed in Table I linagliptin can be administered without dose reduction at all CKD stages (34). However we need to take into account that no clinical studies related to the safety profile in dialysis patients are available. Linagliptin, mainly used in our country as monotheraphy or combined therapy, shows the best clinical profile especially because is predominantly eliminated by a hepatobiliary route. In addition to this, a recent clinical study showed a positive effect on albuminuria reduction in patients treated with linagliptin and ACEi (35). Also, positive effects on cardiovascular outcomes have been demonstrated and other results will be available in the next few years (33). In controlled clinical studies of DDP-IV inhibitors, the overall incidence of adverse reactions, such as hypoglycemia, pancreatitis, nasopharyngitis, upper respiratory tract infection and headache, were similar to those reported with placebo (36). GLP-1RAs are a relatively new class of glucose-lowering agents that are increasingly used as second- or third-line therapy in patients with T2DM. GLP-1RAs, such as exenatide and liraglutide, are injectable incretin mimetics that stimulate insulin release from the pancreas and suppress glucagon secretion. Also, GLP-1RAs are associated with high glycemic efficacy, weight loss, and low risk of hypoglycemia, but with some risk of gastrointestinal side effects such as pancreatitis (37). Exenatide is eliminated by renal mechanisms and should not be given to patients with severe CKD. Liraglutide is not eliminated by the kidney, but it should be used with caution because of the limited experience in patients with CKD (38). The National Kidney Foundation Diabetes Guidelines definitely underline the few data on GLP-1RAs long-term use and the manufacturer recommends avoiding this medicine when GFR is <60 mL/min/1.73 m2 (18, 31). SGLT-2 inhibitors inhibit glucose reabsorption and induce excretion of glucose in the urine. The kidneys play a major role in the regulation of glucose in humans, reabsorbing 99% of the plasma glucose that filters through the renal tubules. The glucose transporter SGLT2 accounts for 90% of the glucose reabsorption in the kidney (39). Treatment with SGLT-2 inhibitors is associated with reductions in HbA1c levels and weight with a low risk of hypoglycemia. Adverse events include urinary and genital tract infections, usually not severe, especially in female patients. Also, SGLT-2 inhibitors appear to be associated with a small increased risk of euglycemic diabetic ketoacidosis (40, 41). As their efficacy is important for the renal function, SGLT-2 inhibitors are generally contraindicated in patients with eGFR <60 mL/min/1.73 m2, mainly because of reduced efficacy. Furthermore, in June 2016 the Food and Drugs Administration (FDA) announced the current warning about the risk of acute kidney injury for drug products containing canagliflozin and dapagliflozin (42). In the EMPA-REG OUTCOME trial, empagliflozin, in comparison with placebo, showed a reduction in risk of major adverse cardiovascular events in patients with T2DM and a slower progression of kidney disease in patients with pre-existing CKD. This study takes into account all patients with at least 30 mL/min/1.73 m2 of body-surface area who receive once a day either empagliflozin (at a dose of 10 mg or 25 mg) or placebo (43). Moreover, the Italian Medicines Agency (AIFA), in accordance with the FDA decision, recently approved the use of empaglifozin in patients with T2DM for the prevention of cardiovascular death, in adult with cardiovascular disease (44).
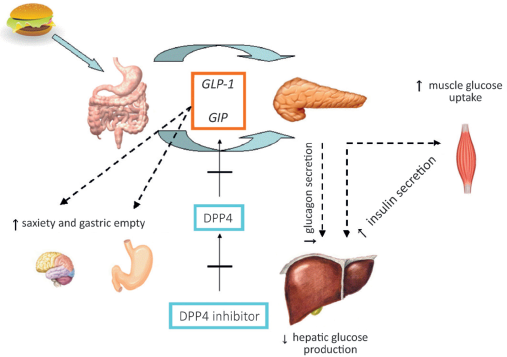
After meal ingestion, as blood glucose levels rise, neuroendocrine cells of the intestine secrete the incretin hormones GLP-1 and GIP. Both stimulate insulin secretion by pancreatic beta cells, in a glucose-dependent fashion. GLP-1 additionally decreases glucagon secretion from pancreatic alpha-cells, slows gastric emptying and increases satiety, allowing for reduced food intake. Physiologically, the enzyme dipeptidyl peptidase (DPP)-4 rapidly degrades both GLP-1 and GIP to their inactive metabolites. Pharmacological competitive inhibition of DPP-4 increases the half-life and bioavailability of active incretin hormones, enhancing their physiological effect. GIP = gastric inhibitory polypeptide; GLP-1 = glucagon-like peptide-1.
What's Next?
Further research, aimed at determining the safety and efficacy profile of DPP-4 inhibitors, GLP-1RAs, and SGLT-2 inhibitors in patients with T2DM and CKD, such as CARMELINA (Id: NCT 01243424), MARLINA (Id: NCT 01792518), ELIXA (Id: NCT 01147250), AWARD-7(Id: NCT; 01621178), CREDENCE (Id: NCT 02065791), and CANVAS-R (Id: NCT01989754), is ongoing (except AWARD-7). These studies will help to confirm the position of these new therapy classes in diabetic patients with CKD.
In recent years, the discovery of a family of small (~22 nucleotide) single-stranded, non- coding, endogenous RNAs, called microRNAs (miRNAs), has been a focus of attention for researchers. The function of miRNAs is the inhibition of the expression of specific mRNAs and one miRNA may control multiple genes, and one gene may be controlled by multiple miRNAs. Since the discovery of the first miRNA in 1993, several miRNAs have been identified as having a physiological role in tissues in which diabetes complications occur (45, 46). miR-375 was the first mi-RNA cloned from a mouse insulinoma pancreatic β-cell line and was identified as the most abundant and better conserved, islet-specific miRNA in the humans. miR-375 regulates the glucose-stimulated insulin secretion in a negative manner. Mice with a homozygous deletion of miR-375 appear to have hyperglycemia due to decreased total pancreatic β-cell mass and insulin levels, whereas genetic deletion of miR-375 in obese mice reduces the proliferative capacity of the endocrine pancreas and results in a more severe diabetic phenotype (46).
Although many diabetes-related mi-RNAs have been discovered in the last two decades, research has just begun and it is out of question that in the near future it will allow the diagnosis and prevention of many diseases including diabetes and its complications.
Conclusion
Although the patient GFR was close to the limit suggested by the guidelines in relation to the dose reduction, and the home glycemic measurement was controlled, we decided to maintain the metformin therapy without dose adjustment. After two months, the patient repeated the blood analysis that showed a worsening of renal function: sCr was 2.4 mg/dL, BUN was 41 mg/dL, microalbuminuria 375 mg/L, total cholesterol 212 ml/dL, LDL-cholesterol 110 mg/dL. The GFR calculated with MDRD equation was 29 mL/min/1.73 m2. The patient discontinued metformin and, in agreement with the diabetologist, he started linagliptin 5 mg daily. After 20 months since the patient started linagliptin, the renal function was stable and the therapy modification revealed a positive impact on the patient's general health.
In conclusion, the DPP-IV inhibitors, the GLP-1RAs and the SGLT-2 inhibitors would be a valid and strong alternative to the other antidiabetic drugs and in the near future they will likely completely substitute the “old guard” of diabetic therapy.
Footnotes
Financial support: No grants or funding have been received for this study.
Conflicts of interest: None of the authors have any financial interest in connection with to this study.
