Abstract
The role of vitamin D receptor (VDR) activators for the control of secondary hyperparathyroidism has been clarified during the last few decades; however, their possible activity in conditioning cardiovascular comorbidity and mortality has become of interest more recently. On the basis of experimental studies showing that VDR activating therapy is associated with a reduction of cardiac hypertrophy, the PRIMO Study (an international randomized controlled trial [RCT]) was carried out a few years ago, but the results were disappointing, as the group of uremic patients on dialysis treated for 48 weeks with paricalcitol showed no differences in comparison with controls in terms of regression of heart hypertrophy. The aim of this editorial is to analyze the possible reasons for such results, and to help understand the actual role of VDR activators in dialysis patients in controlling cardiovascular morbidity and mortality.
Activating VDR for controlling cardiovascular morbidity and mortality: evolution of a myth
During the first quarter of the past century, vitamin D deficiency was identified as responsible for several metabolic bone diseases, and later it was observed more specifically in patients with chronic kidney disease (CKD). In this metabolic setting, intact parathyroid hormone (iPTH) serum levels are elevated, and an important influence has been attributed to reduced 1,25-dihydroxyvitamin D3 (1,25(OH)2D3) synthesis leading to hypocalcemia; however, there is a general consensus that the primary event of such disorders is represented by the expanded pool of phosphate responsible for the increased FGF23 generation by bone structures (1, 2).
On this basis, in addition to a strict control of serum phosphate, the role of vitamin D receptor (VDR) activators is widely considered to be important in renal patients when chronic renal failure occurs; the clinical benefits of vitamin D supplementation in controlling renal osteodystrophy (ROD) were observed at all levels of CKD, mainly in patients on regular dialysis treatment (RDT) where ROD is an overt complication.
More recently, in the last 2 decades, it was also understood that VDR activation in CKD patients (in addition to a suppressive control of PTH secretion) was accompanied by several other advantages in terms of morbidity and mortality (3–6) and that these effects were independent of serum Ca, P and iPTH profile. One of the explanations was the direct effects of vitamin D on cardiovascular structures where VDRs were found at different levels (vascular smooth tissue, endothelial cells and cardiac tissue) (7, 8).
Based on the experimental data showing that VDR activating therapy is associated with a reduction of left ventricular hypertrophy, improved left ventricular diastolic dysfunction and reduced rates of heart failure (9), it was an obvious decision to promote a prospective trial (The PRIMO Randomized Controlled Trial; clinicaltrials.gov:NCT00497146) to clinically confirm this fascinating hypothesis.
A good idea with a naïve design
This was the project of an international team who enrolled 227 patients with CKD stage 3 and 4 (with mild to moderate left ventricular hypertrophy) with the aim of determining the effects of paricalcitol (an active and selective vitamin D analog) on left ventricular mass over a period of 48 weeks. The patients were randomly assigned to receive orally 2 μg/day of paricalcitol or placebo.
The unexpected results showed that, after 48 weeks of observation, the left ventricular mass index did not change between the 2 groups (paricalcitol group 0.34 gr/m2.7 vs. placebo group −0.07 g/m2.7). Also inconclusive were the Doppler measures of diastolic lateral mitral annular tissue velocity (paricalcitol group −0.01 cm/s vs. placebo group 0.30 cm/s). In conclusion, the 48-week therapy with paricalcitol did not alter left ventricular mass index or improve diastolic dysfunction in patients with chronic renal failure.
In practice, the protocol had some weak points that could have played a role in conditioning the results, such as the short period of observation and the magnitude of cardiac hypertrophy which was too mild in a large part of the cohort studied.
VDR and cardiac hypertrophy in CKD patients: the end of a myth?
Taken together, these data produced a fall in enthusiasm and interest among the clinical experts in the field. However, the data set of the PRIMO Study generated additional results.
Considering that left atrial enlargement is a well-recognized index associated with cardiovascular morbidity and mortality, the group of investigators of the PRIMO study produced a post hoc analysis of the same data by evaluating the modification of left atrial volume index (LAVi) (9). Interestingly, 2-dimensional echocardiography performed at baseline and at 24 and 48 weeks after initiation of therapy was able to show a significant decrease in LAVi in the paricalcitol group (-2.79 ml/m2) compared with the placebo group (-0.70 ml/m2; p = 0.002). In addition, paricalcitol therapy was also significantly associated with a reduced rise in serum brain natriuretic peptide (BNP) level (10.8% in paricalcitol group vs. 21.3% in placebo; p< 0.02). Considering the entire population, the changes in serum BNP correlated with change in LAVi (r = 0.17, p = 0.03) (Figs. 1 and 2).
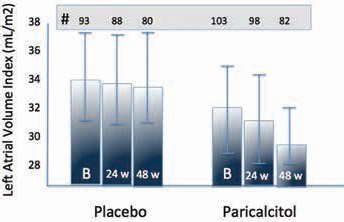
Adjusted mean left atrial volume index (LAVi) at baseline, after 24 and 48 weeks (w) of observation (modified from (10)).
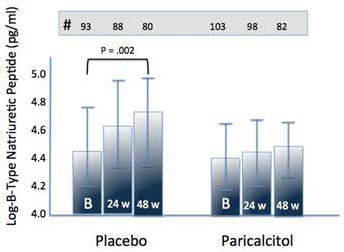
Adjusted mean brain natriuretic peptide (BNP) at baseline, after 24 and 48 weeks (w) of observation (modified from (10)).
The mechanisms explaining the effects of paricalcitol in reducing LAVi and serum BNP levels are not clear: it has been argued that any VDR activators can directly inhibit the renin-angiotensin-aldosterone system (RAAS), thus adding its effects to angiotensin-converting enzyme (ACE) inhibitors or sartans (the largest decline in LAVi was in fact observed in patients also receiving RAAS inhibitors) (11, 12). Additionally, vitamin D could affect cardiac remodeling by attenuating collagen deposition and structure oxidation (13) and, as observed in an animal model, can regulate myocyte growth (14).
One obvious concern and criticism was addressed to the possible and confounding body fluid changes related to the use of diuretics; however, the authors clarified that the groups were similar in terms of body weight and that LAVi changes were independent of extracellular body volume variations.
Paricalcitol and Endothelial function
After the PRIMO Study, more evidence dealing with the clinical effects of paricalcitol appeared in the literature, and recently, interesting data about the positive role of paricalcitol in conditioning endothelium-dependent vasodilatation (in patients with CKD stages 3 to 4) were published (15).
This study (the Paricalcitol and Endothelial Function in Chronic Kidney Disease [PENNY] trial; clinicaltrials.gov: NCT01680198) was a double-blind RCT designed to investigate the possible role of paricalcitol (oral 2 μg daily × 12 weeks) on endothelium-dependent and endothelium-independent vasodilation. The protocol included 88 patients (with CKD 3 to 4) with mildly elevated serum iPTH (>65 pg/mL). The effect of paricalcitol on endothelium-dependent vasodilation was significant (p = 0.016), and the mean change in flow-mediated dilation was 61% higher in the paricalcitol group in comparison with the placebo group. So, considering that endothelium plays a major role in accelerated vascular aging, on the basis of these results, can we assume that VDR activators should be included in the routine therapeutic armamentarium of clinical practice?
VDR activation at a point of care: open questions and paradoxes
The possibility of having reliable tools able to modify cardiac structure and reduce cardiovascular morbidity and mortality in patients on RDT represents a continuous field of interest for the nephrological community. However, prospective RCTs are lacking, and the PRIMO study (the most appropriate approach for solving this question) gave disappointing, negative results: on the other hand, can we imagine that the data of the PRIMO post hoc analysis (dealing with atrial volume) are robust enough to encourage the use VDR activators in any dialysis point of care?
And again: if any, should therapy with VDR activators begin at a early stage of chronic renal failure when cardiac geometry is still normal, or should we wait until the appearance of overt cardiac hypertrophy? In other words, VDR activators should be considered for prophylaxis or for therapy of dialysis cardiomyopathy?
Also several paradoxes are to be considered: among them, the fact that VDR activator therapy is associated with FGF23 serum increase, and excess FGF23 levels have been independently linked with cardiovascular disease and mortality, suggesting that chronically elevated FGF23 levels may directly contribute to adverse CKD outcomes.
On the other hand, a solid amount of scientific literature (although mostly related to retrospective studies) is supporting the wide use of vitamin D in any renal patients for its beneficial effects on mineral metabolism, as well as for being associated with reduced morbidity and mortality in dialysis patients.
In conclusion, should we also consider the use of vitamin D, or of its synthetic analogs, for controlling or even preventing on a large scale changes of cardiac remodeling?
Again, more RCTs are needed before giving a solid answer to these questions.
Footnotes
Financial support: None.
Conflict of interest: None
