Abstract
A case of critical ischemia presenting with pain and finger gangrene in the right hand of a patient on hemodialysis is presented. The multifactorial origin of ischemia and its pathophysiology are discussed, together with the most appropriate diagnostic approach. Innovative endovascular treatment of such conditions is also described.
Case report
Mr. M is a 58-year-old man. He developed type 1 diabetes mellitus when he was 20 years old, and started hemodialysis at the age of 52. The last 3 years have been a real ordeal: angina pectoris resolved with a triple coronary bypass graft, an isolate episode of transitory brain ischemic attack, multiple laser treatments of retinopathy, gangrene and repeated infections of the feet that, after several unsuccessful attempts at revascularization and removal of necrotic tissue, led to a bilateral below-the-knee amputation of the legs.
Now Mr. M is in a wheelchair and hopes to get a limb prosthesis soon, to be able to drive and to go back to work in his company. He does not give up and wants to live.
In September 2010, during the dialysis sessions, he began to feel pain in his right hand. Mr. M had dialysis through a proximal radiocephalic fistula. The fistula flow was estimated to be approximately 275 ml/min, therefore a steal effect was excluded, and the symptoms were underestimated. In October, due to an accidental trauma, an ulcerated lesion appeared on the fourth finger of his right hand (Fig. 1), which did not heal despite daily medications and antibiotic therapy. Multiple treatments with hyperbaric oxygen therapy were given without any improvement. In November, the situation had become unbearable: Mr. M had constant pain, was unable to sleep at night and the lesion progressed into gangrene and osteomyelitis of the proximal and middle phalanx of the fourth finger (Fig. 2). Hand surgeons recommended amputation of the finger after angiography and closure of the fistula. On January 17th, 2011, an angiography was performed (Fig. 3) showing a normally functioning fistula, obstruction of the distal radial and ulnar arteries and diffuse disease of the blood vessels of the hand. A central venous catheter was positioned through the right internal jugular vein, the fistula was surgically closed and the fourth finger was amputated.
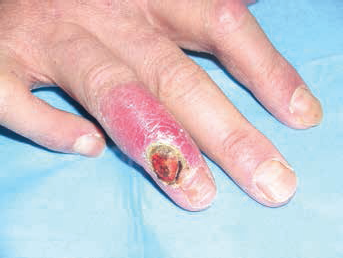
The first lesion: an ischemic ulcer of the fourth finger with surrounding infection.
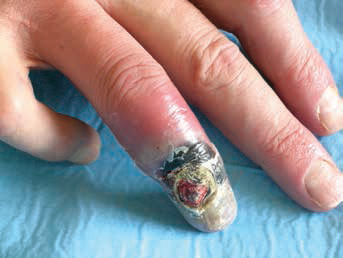
Evolution of gangrene of the distal phalanx.
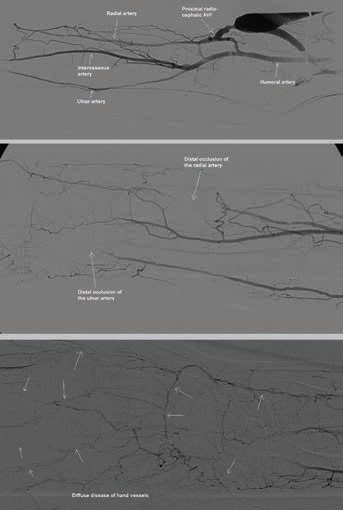
Angiographic study of the forearm showing a normally functioning fistula, obstruction of the distal radial and ulnar arteries, diffuse disease of the blood vessels of the hand. AVF = arteriovenous fistula.
Despite these treatments, in the following weeks, the situation did not improve: The pain persisted, surgical wounds did not heal but were reinfected and showed dehiscence (Figs. 4 and 5). Mr. M was desperate: He was in a wheelchair, in constant pain; unable to sleep, to use his right hand or to work; and depended in every detail of his life, from personal hygiene to eating, on other people. In late March, the patient was transferred to our center to seek a solution to an apparently intractable condition.
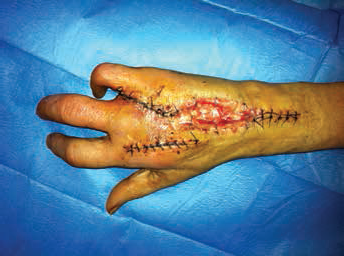
Dorsal view of the hand after amputation of the fourth finger, showing dehiscence of the wound.
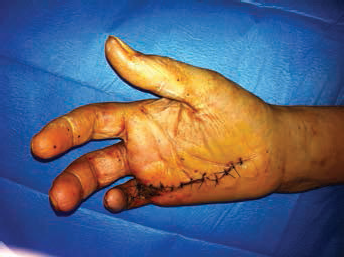
Palmar view of the hand after amputation of the fourth finger.
Diagnosis
The causes of critical hand ischemia in patients in hemodialysis are essentially 3 in number (1–3):
The presence of an arteriovenous fistula that steals blood from the natural antegrade flow
The presence of disease of the great vessels of the upper limb, which prevents the blood to reach the hand directly, diverting the flow onto small collateral branches with high hydraulic resistance.
The presence of obstructive disease of the small vessels of the hand (arches, metacarpal and digital vessels) which is a further obstacle to the final distribution of blood flow to the tissues.
In our experience (over 130 ischemic hands), only rarely have we found a single cause of critical hand ischemia; in most cases there is a combination of several factors that determine the poverty of the blood supply to the distal tissues.
The pathophysiology of ischemia in Mr. M's hand was complex, because he presented simultaneously with all of the possible causes. The proximal radiocephalic fistula was a standard hemodialysis fistula, its flow was not pathological and, by itself, it would not have been sufficient to cause hand ischemia. However, we must consider that at the level of the fistula, the blood flow is roughly distributed on the basis of the ratio of the resistances offered by the 2 circuits, the venous retrograde and arterial antegrade, as described in detail by various mathematical models (4). In the case of Mr. M, retrograde venous flow was meeting a standard resistance, because the anastomotic mouth and the vein circuit were of the proper size. In contrast, the antegrade arterial circuit offered a high resistance, because it presented 2 sequential barriers: at the level of the great vessels of the forearm and of the small vessels of the hand. Beyond refined functional or angiographic investigations, a simple radiography of the hands of Mr. M pointed out that every vessel was encased in a shell of calcium, spreading, fiercely and ubiquitously, up to the extreme arteries of the fingertips (Fig. 6). To close the fistula was therefore a proper action but by itself insufficient to provide an adequate flow to guarantee the healing of the hand.
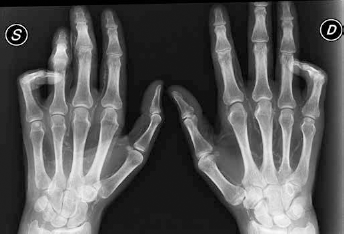
X-ray of the hands showing vessel calcification extending to the fingertip arteries.
Treatment
On March 23rd, the patient underwent an angiography of the right forearm by a humeral antegrade approach carried out with a small caliber (4 French) sheath. In the same session, radial and ulnar arteries were reopened by percutaneous angioplasty (Fig. 7); we used long and thin balloons inflated to 14 atm (5, 6). The inflation of the balloons in the forearm and hand usually causes unbearable pain, so we made a prediction with undersized (1.5–2.0 mm) balloons to reopen the vessel and restore blood flow, followed by an intra-arterial injection of 50 mg of lidocaine that anaesthetized the area and allowed the subsequent dilation using proper sized balloons (2.5–3.0 mm). The treatment was completed with the use of drug-eluting balloons that released paclitaxel on the vessel wall, giving a true local chemotherapy whose purpose was to block the reparative processes that would have led to restenosis (7). After the procedure the patient had both the radial and ulnar pulses well palpable. Dual antiplatelet therapy (Clopidogrel and aspirin) was maintained for 1 month.
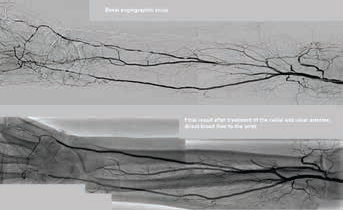
Angiographic study after closure of the arteriovenous fistula and after angioplasty of the radial and ulnar arteries.
In the following days, we proceeded to the debridement of the tissue lesions. The surgical treatment was carried out with particular delicacy, due to the persistence of the intractable disease of the small vessels of the hand (Fig. 8), which conditions a reduced perfusion. In these cases, it is essential to avoid sites of elevated tissue pressure (tight suture, folded flaps) that could be foci of further ischemic necrosis of the tissue. The use of dermal substitutes based on hyaluronic acid allows for a tailored reconstructive surgery aimed to preserve the hand functions (gripper, prehension).
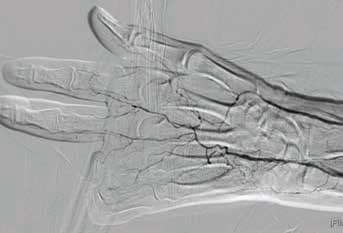
Final angiogram of the hand, showing diffuse disease of the metacarpal and finger vessels.
In the following week, we revascularized also the left forearm, because it mirrored the right, presenting small digital lesions.
After 2 months, Mr. M had healed (Figs. 9 and 10) and resumed the normal use of his hand.
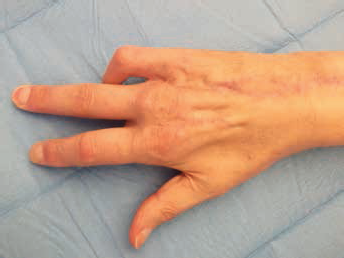
Dorsal view of the hand after healing.
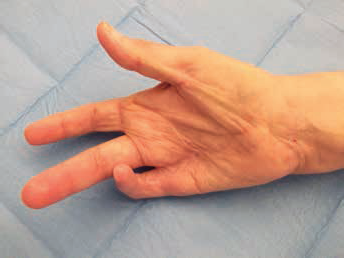
Palmar view of the hand after healing.
Follow-up
Patients with critical hand ischemia and diffuse calcifications present a mortality of almost 40% per year; despite this, Mr. M is still alive after 3 years. The hand lesions never returned, and he is fully autonomous, driving a car and working every day in his company. Walking with the leg prostheses is very tiring; Mr. M prefers to move in a wheelchair. The central venous catheter, fortunately, still works well.
Footnotes
Financial support: None.
Conflict of interest: None.
Below-the-knee and below-the-elbow vessel disease: are drug-eluting balloons the solution? My point of view
Diabetic patients in hemodialysis presenting critical limb ischemia have a favorite localization of the disease in the below-the-knee and below-the-elbow vessels. Whereas calcific small vessel disease of the foot and of the hand is generally considered intractable by surgical or endovascular approaches, obstructive disease of the tibial and forearm arteries is nowadays considered an attainable target of revascularization procedures, mainly endovascular. Due to the typically long extension of the diseased segments to dilate and to the small size of the vessel, restenosis is huge and precocious, about 70% at three months [Schmidt et al Catheter Cardiovasc Interv. 2010;76:1047-54.]. For these reason, the debate regarding the real value of extreme endovascular procedures is still open: on one hand, the restoration of blood flow to distal tissues is a key factor for wound healing, on the other hand endovascular procedures are technically demanding and the rate of target lesion revascularization is high (>50% at one year). Every one of us knows very well how difficult it is to heal a skin lesion in a diabetic patient in hemodialysis, and how frequent reulceration is.
Drug eluting balloons (DEBs) appear to be a promising solution: paclitaxel is embedded on the balloon surface and is released during inflation into the vessel wall to prevent or to moderate the cellular response leading to restenosis. DEBs maintain the antiproliferative properties of drug eluting stents without the metallic burden of the stent, which is unsuitable in the long diseased vessels of the leg or the forearm. Despite several studies having clearly demonstrated that DEBs represent one of the best treatment options in superficial femoral and popliteal arteries, data concerning below-the-knee arteries are scarce and conflicting. Two studies by Schmidt et al [J Am Coll Cardiol. 2011;58: 1105–1109] and Liistro et al [Circulation. 2013;128:615-21] demonstrated a reduction in restenosis rate after DEBs treatment from 70% to <30%; however the In Pact Deep trial [Zeller et al. JACC. 2014;64:1568-76], a prospective, multicenter, randomized trial, failed to confirm these data, raising a safety signal towards major amputations and leading to market withdrawal of the Medtronic DEB. Today we have many DEBs on the market dedicated to tibial vessels, but their use is more justified by the hope of the operators than scientific data.
