Abstract
Nonadherence is a widespread problem among patients receiving hemodialysis, while contributing factors are underexplored due to lack of a validated instrument. The study aimed to develop and validate a new disease-specific instrument determining the factors affecting treatment adherence behaviors among patients receiving hemodialysis in a Sri Lankan hospital. The content validation process using a modified Delphi technique suggested 42-item instrument with an average Item-level Content Validity Index (I-CVI) of 0.94. Subsequent principal component analysis (PCA) revealed 5 components with 17 items (patient-centered psychological and physical factors, factors associated with health care, hemodialysis therapy-related factors, socioeconomic factors, and factors associated with the continuity of the treatment), explaining 67.24% total variance. The reliability estimate based on Mislevy and Bock for each component greater than 0.8 and overall standardized Cronbach's alpha of 0.605, indicated acceptable internal consistency reliability. Root mean square residual (RMSR) and weighted root mean square residual (WRMR) indices of 0.084 and 0.059, respectively, indicated acceptable model fit. The final 17-item instrument provides a valid measure for the factors affecting treatment adherence behaviors of the patients receiving hemodialysis in a Sri Lankan hospital.
Introduction
Chronic kidney disease (CKD) is a growing public health problem which requires renal replacement therapy when it progresses into the end stage (1). Of about 500 million patients diagnosed with CKD globally, approximately 80% live in developing countries such as in Sri Lanka, with a significant prevalence of related morbidity and mortality (2). Hemodialysis remains as the most prevailing treatment regimen among patients with CKD (3) because of its accessibility and availability (1).
Nonadherence is a widespread problem among patients diagnosed with many chronic diseases (4) and is reported in approximately 50% of the patients treated with long-term therapies in developed countries. In fact, the magnitude and impact of poor adherence in developing countries are assumed to be comparatively higher due to the paucity of health resources and limited access to health care (5).
According to Ozen et al., as described by the World Health Organization, treatment adherence can be described as “the correspondence level of a person receiving medication, following the recommended diet or implementing lifestyle modifications according to the health care providers’ recommendations” (6). Accurate assessment of adherence behavior is necessary for effective and efficient treatment planning and to ensure changes in health outcomes (5). It was reported that nonadherence among patients with CKD for hemodialysis treatment ranged from 8.5% to 22.1%, (7) whereas missing hemodialysis sessions were reported at a rate of 0% to 18% (8, 9). Nonadherence to medication, fluid restrictions, and dietary recommendations were ranging from 3% to 80%, 9.8% to 75.3%, (8, 9) and 2% to 81.4%, (7, 8) respectively.
In chronic disease conditions, poor adherence leads to adverse events and is the primary reason for disease progression, frequent hospitalizations, and reduced quality of life among patients (10). In fact, poor adherence often causes medical and psychosocial complications of the disease and wasting of health care resources (5).
Nonadherence is a multifactorial problem and was roughly divided into social and economic, health care system, health condition, therapy, and patient-related dimensions (4, 5). Besides, sociodemographic variables, complexity of treatment, health beliefs, social support, health literacy, patient satisfaction (11), and the relationship between patient and the health care provider were also observed as key determinants in improved treatment adherence among patients (12).
Informal clinical observations and the concurrent research in the Sri Lankan renal units found poor adherence among patients, particularly for fluid restrictions and prescribed diet (Table 2). Though nonadherence is rampant among patients receiving hemodialysis, periodic assessment of their adherence behavior and contributing factors is lacking in Sri Lankan hospitals. Besides, treatment adherence plays a critical role in decreasing the economic burden of the disease to both families and society (6). Though the health care team fosters adherence, the ability to follow medical advice and recommendations entirely depends on the patients’ level of understanding and motivation (13). Thus, before planning any interventions to improve the clinical outcomes of the patients on dialysis, all the potential determinants that influence their adherence behavior should be taken into consideration (11). Periodic assessment and identification of the factors associated with nonadherence permit planning future interventions (14) while providing a basis to make alterations of the present treatment regimen to deliver quality and safe patient care. Very few studies explored adherence to the hemodialysis treatment regimen in Asian populations (11). In this sense, the availability of a psychometrically sound instrument in assessing the determinants of treatment adherence behaviors is a timely need and is also lacking in the literature. Thus, the study aimed to develop and validate a new disease-specific instrument to determine the factors affecting treatment adherence behaviors among patients receiving hemodialysis in a Sri Lankan hospital.
Methods
Study Design and Setting
This methodological study involved 3 phases: (1) item development (2) scale development, and (3) scale evaluation (15). A descriptive cross-sectional study was conducted in a selected tertiary care, teaching hospital which is located in the North Western province, approximately 100 km faraway from the capital city of Sri Lanka to evaluate the instrument's construct validity using principal component analysis (PCA). The selected hospital has access to the patients coming from different ethnic backgrounds, different areas of the country, and diagnosed with different etiologies (both CKD and CKD of unknown etiology).
Phase 1: Item Generation and Questionnaire Development
Since there was no widely used gold standard or existing validated instrument in the literature, domain identification and item generation of the instrument was done using the deductive method based on an in-depth literature review. The initial 49-item instrument was developed mainly upon the theoretical underpinnings of adherence to long-term therapies: Evidence for action by the World Health Organization (5).
Face and Content Validity
In order to evaluate content validity of the instrument, modified Delphi technique which is a widely used method to obtain unbiased expert consensus was used (16). The Delphi method is “a process of gaining consensus through controlled feedback from a group of experts via multiple iterations” (17).
The opinions of 5 to 10 experts are adequate in evaluating content validity of a questionnaire (18) and 10 to 15 participants from 2 relevant expert groups (professionals and academics) are adequate if their background is homogenous for a Delphi technique (17). Hence, the opinions of the 10 experts in the health field, who had more than 5-year experience in research background, expert clinicians and nurses in the field of hemodialysis (2 nephrologists, 1 clinician, 1 university lecturer, and 6 dialysis nurses) were involved in evaluating face and content validity of the newly developed instrument using modified Delphi technique (Table 1). In the present study, initially, a list of factors affecting treatment adherence was identified from the existing literature and consensus was built upon the experts’ opinions. The modified Delphi method was comprised of 3 rounds as described below.
Characteristics of Participants.

Abbreviations: CKD, chronic kidney disease; PCA, principal component analysis.
Level of Treatment Adherence Based on End-Stage Renal Disease—Adherence Questionnaire Score (3) of Patients Receiving Hemodialysis in the Same Dialysis Unit.

Delphi Round 1: Emailing or Survey Administration of the Instrument to Build the Consensus
In Delphi round 1, panelists were administered a structured questionnaire consisted of 49 items related to contributing factors for treatment adherence. They were requested to review the questionnaire overall or suggest any changes to the questions. In this round, experts suggested to include 3 new items to the 49-item instrument, including “Do the members of the health care team allow you to ask questions to resolve any queries you have regarding your treatments?”, “Does the hospital with hemodialysis unit locate faraway from your home?”, and “Do you receive adequate family support?”. The two items “Have you ever smoked?’ and “Are you currently smoking?” were combined as 1 question, “Have you ever smoked cigarettes? or Are you a current smoker?”. A 51-item instrument was produced after Delphi round 1.
Delphi Round 2: Ranking the Items of the Instrument Based on the Importance by the Experts
In Delphi round 2, panelists voted for each item of the 51-item questionnaire whether to keep, remove, modify, or to state that they have no exact opinion on the item. The panelists were given an option to provide free-text comments on their decision if necessary. Repeating items were removed from the instrument based on expert opinion. Panelists were provided 5-point Likert type scale (1 = Strongly disagree, 2 = Disagree, 3 = Neutral, 4 = Agree, and 5 = Strongly agree) to rate each item of the instrument based on their relevance, appropriateness, and acceptance of each item to determine treatment adherence of the patients receiving hemodialysis. Item relevance was decided if the experts rated each item as agree or strongly agree (score: 4-5). According to Lynn, 80% agreement among the experts was considered in retaining an item in the questionnaire. Item-level Content Validity Index (I-CVI), (18) Item-level Content Validity Ratio (I-CVR), (19) and modified Kappa statistic coefficient (K) (20) based on the ratings of the experts were calculated for each item using the relevant formula. The 42 items which yielded excellent content validity were retained in the instrument after Delphi round 2.
Final Face-to-Face Meeting With the Experts
Experts discussed and agreed with all the 42 items to retain in the final instrument during the face-to-face interviews.
To ensure less bias, partial or “quasi anonymity” was assured during the entire process of the modified Delphi technique where experts did not have interaction among them (17). Thus, it was not affected the opinions of the individual panelists. In addition, use of multiple rounds and controlled feedback ensured less bias during the process.
Phase II: Scale Development Phase
Construct Validity
The resulting 42-item instrument was administered to a group of patients (n = 5) for pretesting to check for poorly worded items and whether the items were well understood by the patients. Even though it is recommended to recruit around 30 participants for pretesting a questionnaire (21), the present study could only approach 5 patients due to safety considerations during the COVID-19 pandemic. The instrument included only closed-ended questions in a simple language without medical jargons and with yes/no answer format. During pretesting, there were no major modifications indicated. The construct validity of the instrument was evaluated using PCA aiming to reduce the dimensionality of a large data set, to a smaller number of components increasing interpretability and minimizing the information loss. The minimum sample size recommended for PCA is 150 cases or 5 to 10 cases per variable (22). Thus, the content validated and pretested 42-item instrument was administered to a consecutive sample of 150 patients receiving hemodialysis at the Teaching Hospital, Kurunegala which has one of the major hemodialysis units in Sri Lanka (Table 1). All the patients more than 18 years old and able to understand and speak the Sinhala language were included in the study after obtaining written and verbal informed consent. Patients who were critically ill were excluded.
Phase III: The Scale Evaluation Phase
The reliability of the final instrument was evaluated using internal consistency reliability.
Data Analysis
Descriptive statistics and content validity indices were analyzed using Statistical Package for Social Sciences (SPSS) version 25.0 software (IBM Corp.) and Microsoft Office Excel for Windows, respectively. The items with I-CVI ≥ 0.79, (18, 20) I-CVR ≥ 0.49, (19, 20) and Kappa index> 0.74 (20) were considered valid to be retained in the final instrument. PCA was performed using FACTOR 10.10.3 software (23).
Prior to performing PCA, data were screened for missing data, normality, outliers, and for the suitability to conduct factor analysis. PCA was carried out considering the nominal nature of the variables (dichotomous items with yes/no responses) on a polychoric (tetrachoric) correlation matrix. Mardia's multivariate test for skewness was not statistically significant (test statistic = 26525.632, P = 1.00). Though, there was evidence of excessive kurtosis (test statistic = 9.068, P < .001) indicating non-normality of the survey data providing further indication to conduct the factor analysis using polychoric (tetrachoric) correlation matrix. The adequacy of the correlation matrix (sampling adequacy) was assessed by Kaiser–Meyer–Olkin (KMO) test and the value greater than 0.6 was considered for a good factor analysis (24). Bartlett's test of sphericity is used to assess an adequate amount of correlation between items. The KMO value for the 17-item instrument was 0.743 and Bartlett's test of sphericity was significant (Bartlett's statistic = 1640.6; df = 136; P = .000010 < .001) indicative of meeting criteria for good factor analysis.
High values of bivariate correlation scores indicate multicollinearity between the items (25). Thus, after initial analysis, 17 items were removed including one of a pair of items with bivariate correlation scores greater than 0.8 and with very low variance (<0.1) as suggested by the FACTOR software. In the subsequent analysis, 5 items with communalities less than 0.3 were removed and analysis was rerun. Then, 3 items with cross-loadings of close factor scores were observed and removed from the instrument and resulted the final 17-item instrument. An average communality value between 0.5 and 0.6 was considered acceptable for sample sizes between 100 and 200 while the items with a factor loading greater than 0.4 (lowest acceptable threshold) was considered to be retained in an instrument (25).
The parallel analysis method (Horn, 1975) in which the widely accepted Monte Carlo simulation technique and Kaiser criterion (retaining components with an eigenvalue greater than 1.0) (26) were applied to estimate the exact number of components to be extracted. Oblique rotation (Robust Promin) which is recommended in exploratory factor analysis with social science data (27) and best applicable for a multidimensional instrument (28) was applied to improve the interpretability and to achieve simplicity of the extracted components. The adequacy of component structure was assessed by the percentage of variance explained. Bentler's simplicity index (S) and the loading simplicity (LS) index were used to evaluate the simplicity of the extracted component solution. The value of 1.0 represents the maximum simplicity while greater values indicate simpler and more interpretable solutions (29). The measure of fit between the measurement model and the observed covariance matrix was evaluated with root mean square of residuals (RMSR) and weighted root mean square residual (WRMR) indices.
Reliability Analysis
The internal consistency reliability of the extracted components was evaluated using the reliability estimates based on Mislevy and Bock (1990), generated through a formula based on the standard error of factor scores solution (27). Standardized Cronbach's alpha calculated for ordinal data and McDonald's ordinal Omega coefficient (McDonald, 1999), which is a practical alternative to Cronbach's alpha in estimating measurement reliability of the total score for the whole instrument, were evaluated. The value of 0.6 was considered the minimum acceptable value of Cronbach's alpha (30) and value greater than 0.7 was considered acceptable for McDonald's Omega (31).
Results
Content Validity
I-CVIs of both 42-item and 17-item instruments ranged between 0.8 and 1.0. The calculated average CVI for the 42-item and final 17-item instrument (S-CVI) was 0.94 and 0.95, respectively. I-CVR for all the 17 items ranged between 0.6 and 1.0. Modified Kappa statistic coefficient values ranged between 0.79 and 1.0, indicating good content validity of the instrument (Table 3).
Content Validity of 51-Item new Disease-Specific Instrument Based on Modified Delphi Method.
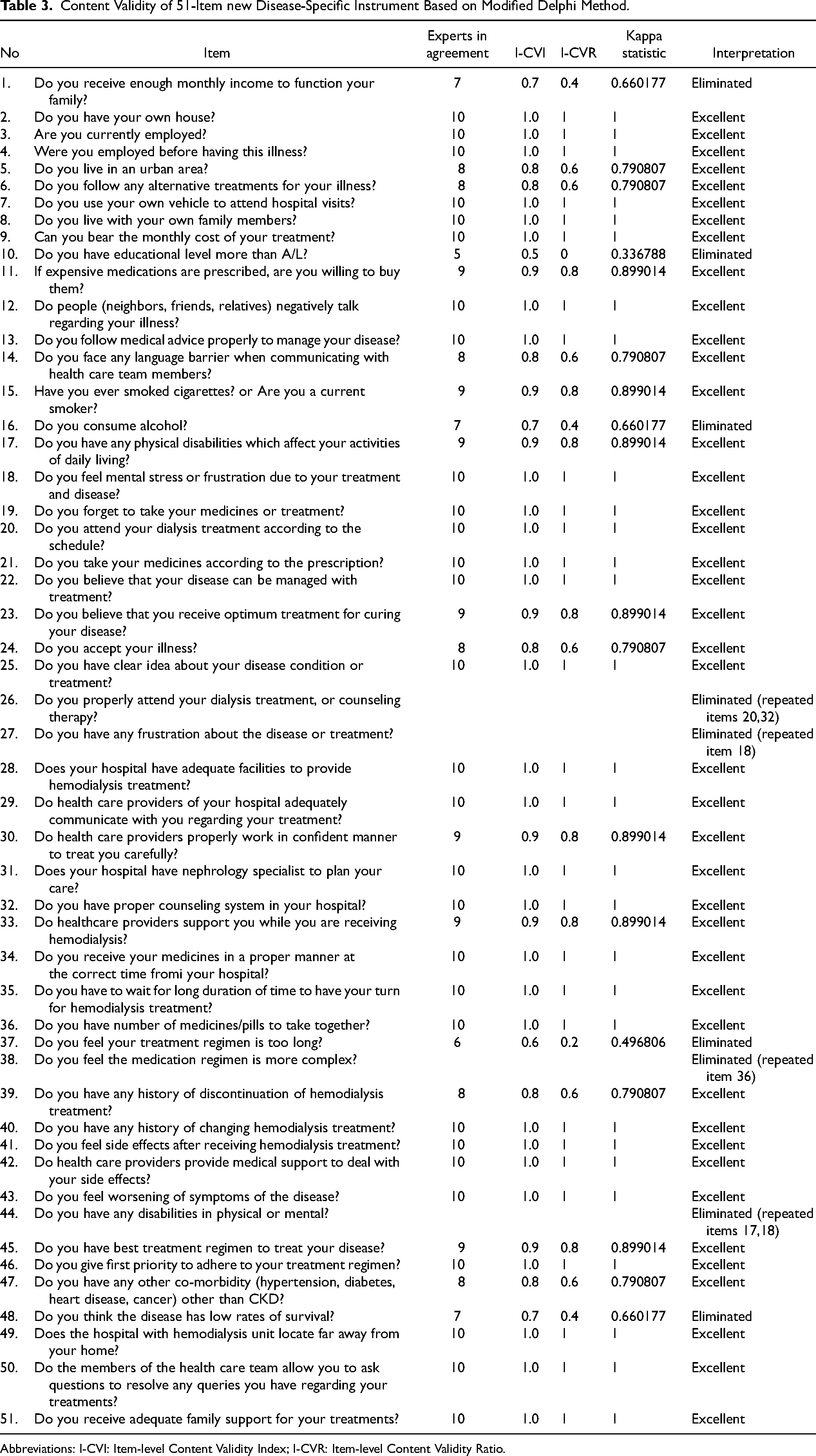
Abbreviations: I-CVI: Item-level Content Validity Index; I-CVR: Item-level Content Validity Ratio.
PCA
PCA of the final 17-item instrument extracted 5 components (eigenvalue greater than 1) explaining the total variance of 67.24%. With respect to the individual factors, the fourth extracted component (C4) accounted for the highest explained common variance of 16.6% while the fifth component (C5) accounted for the least (9.3%). Eigenvalues ranged from 2.91 to 1.50 (Table 4). After subsequent oblique (Robust Promin) factor rotation, items V7, V8, and V17 were loaded into the first component (C1), items V9, V10, V11, and V13 were loaded into the second component (C2), items V15 and V16 were loaded into the third component (C3), items V1, V2, V3, V4, and V5 were loaded into the fourth component (C4), and items V6, V12, and V14 were loaded into the fifth component (C5). Factor loadings and communality of all the 17 items were greater than 0.5 except item V12 in which the communality was 0.371(Table 5).
Explained Variance of the Components Based on Eigenvalues.

Advised number of dimensions when mean is considered: 5.
Advised number of dimensions when 95 percentile is considered: 4.
Bold values indicate the eigenvalues of the components extracted when mean is considered.
Final Rotated (Robust Promin) Component Solution of the Instrument.

Reliability Analysis of Component Structure
The Mislevy and Bock (1990) reliability estimates for each component ranged between 0.809 and 0.938 (Table 6). The standardized Cronbach's alpha coefficient calculated for the ordinal data for the total 17-item instrument was 0.605, indicating an acceptable level of reliability. McDonald's ordinal Omega coefficient of 0.52 was below the acceptable threshold of 0.7.
Explained Variance and Reliability of Rotated Components [(Mislevy and Bock (1990)].

Factorial/Component Simplicity and Model Fit
The Bentler's simplicity index (S) (0.915, p100) and the LS index (0.464, p100) indicated a good structural solution with its maximum simplicity (29). The RMSR value was 0.0846, indicating the marginal acceptability of the model [as this value lies on the upper margin of the expected mean value of 0.0819 for an acceptable model by Kelley's criterion (Kelley, 1935)]. The WRMR value was 0.0595, which is below 1.0 representing a good fit of the model (32).
Discussion
The psychometric evaluation of the newly developed disease-specific instrument comprised 17 items, in which the most parsimonious solution determining factors affecting treatment adherence behaviors among patients receiving hemodialysis in a Sri Lankan hospital.
After content validation, both 42-item and 17-item instruments had I-CVI for all the items greater than 0.8, I-CVR ranging from 0.6 to 1.0 and modified Kappa statistic coefficient ranging between 0.79 and 1.0, demonstrating excellent content validity. The five-dimensional solution yielded from PCA explained 67.24% total variance. Of the 5 components, the first component was “patient related-psychological and physical factors (3 items)“ which explained 12.9% proportion of the common variance and was mainly focused on the perceived psychological and physical barriers to perform normal day-to-day activities by the patients. Communication between health care team and the patient and complexity of the medication regimen was described in the second component, namely “factors associated with health care” (4 items) and were explained 16.0% of the common variance. Feeling side effects after receiving hemodialysis treatment and support extended by the health caregivers in dealing with the side effects were included in the third component namely, “hemodialysis therapy related factors” (2 items) and was accounted for 12.3% of the common variance. The fourth component namely, “Socio-economic factors (5 items)” accounted for the highest proportion of common variance (16.6%) and mainly focused on the status of current employment, location of the residence, and economic status of the patient. The fifth component was a new dimension related to “factors associated with continuity of the treatment” (3 items) and was identified with the least proportion of the common variance (9.3%). It described the influence of others’ negative attitudes on disease and the treatment, following alternative treatments and availability of history of discontinuation of hemodialysis treatment.
The reliability of the final 17-item instrument was confirmed with internal consistency reliability with standardized Cronbach's alpha greater than 0.6, indicative of the items in the instrument reflecting the same underlying construct (30, 33). The reliability estimates proposed by Mislevy and Bock for each component also achieved the acceptable threshold, which reflects “the proportion of variability in the factors scores explained by the latent variable” (34). Nonetheless, McDonald's ordinal Omega coefficient for the final component structure was 0.52 which was below than the acceptable threshold. The validity of Omega coefficient does not require the items to be true score equivalent, but the items required to be homogenous or unidimensional. In fact, Omega coefficient is the better choice than alpha when the presence of skew items in the instrument even in small samples (35). In the present study, the instrument is not unidimensional since it measures different aspects of factors affecting treatment adherence while the sample size of the study was also relatively small (n = 150). According to Mardia's multivariate test for skewness, there were no significantly skewed items in the instrument. Thus, the low Omega coefficient demonstrated relatively minor importance over other reliability estimates in the present study.
A few studies in determining the factors affecting treatment adherence behaviors among patients receiving hemodialysis were found among the published literature. Of them, a systematic review of contributing factors for nonadherence among patients with CKD revealed 6 categories of factors, including patient-related, socioeconomic, psychological, therapy-related, pathophysiological (disease) related, and health care system-related (9). Similarly, the present study identified a component structure with similar scope, though pathophysiological-related factors were not evident in the final 17-item instrument.
Limitations
This study was conducted in a single hospital in Sri Lanka. Therefore, a multicenter study is recommended in future studies with a large sample size to confirm the final component solution of the instrument and necessary adaptations are also encouraged. We assessed only the internal consistency reliability of the instrument. Thus, different reliability assessments are also recommended in future studies.
Conclusion
The new disease-specific instrument provides a valid and reliable measure in determining factors affecting treatment adherence among patients receiving hemodialysis in a Sri Lankan hospital. The instrument will help health care providers and policymakers to identify reasons for nonadherence in different aspects and to develop relevant health policies and interventions to overcome the factors affecting nonadherence among patients.
Footnotes
Acknowledgments
The authors would like to express their gratitude to all the patients who participated in the descriptive cross-sectional study and all the experts who participated in the modified Delphi technique in the instrument validation process.
Author’s Contributions
KW, UH, TDA, CSEG, and CL contributed to the concept and design of the study. CL carried out data collection, data analysis, data interpretation, and drafting of the manuscript. KW, UH, TDA, and CSEG assisted in writing and revising the manuscript. All authors have read and approved the final manuscript to be published.
Declaration of Conflicting Interests
The author(s) declare that there is no conflict of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the University Research Grants, University of Sri Jayewardenepura, Sri Lanka (Grant number ASP/ 01/RE/MED/2017/61).
Ethical Approval and Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the approved protocols and guidelines of the Ethics Review Committee, Faculty of Medical Sciences, University of Sri Jayewardenepura, Sri Lanka (ERC No. 06/19) and Committee of Ethics Review on Scientific Research, Teaching Hospital, Kurunegala, Sri Lanka (THK/CERSR/2019/12).
Statement of Informed Consent
Written and/or verbal informed consent was obtained from the patients for their anonymized information to be published in this article.
