Abstract
Interleukin 6 (IL-6) is a 184-amino acid protein cytokine that is produced by many types of cells and is expressed during states of cellular stress, such as inflammation, infection, wound sites, and cancer. IL-6 levels may increase several thousand-fold in these states and may help to coordinate the response to dysregulation of tissue homeostasis. IL-6 acts through a membrane-bound IL-6 receptor (mIL-6R), which, together with a second receptor, glycoprotein 130 (gp130), leads to the initiation of intracellular signaling (classic signaling). Given that IL-6R is expressed on only a few types of cells, though all cells express gp130, direct stimulation by IL-6 is limited to cells that express mIL-6R. However, IL-6R is also produced as a soluble, secreted protein that, together with IL-6, can stimulate all gp130-expressing cells by a process termed IL-6 trans-signaling. IL-6 trans-signaling can be blocked without affecting IL-6 classic signaling through mIL-6R. IL-6 has major effects on the adaptive and innate immune system and on mesenchymal and stromal responses during inflammation. It promotes the development of pathogenic T-helper 17 T cells and the maturation of B lymphocytes. Many innate immune cells, neutrophils, and monocytes/macrophages produce and respond to IL-6, resulting in autocrine feedback loops that amplify inflammation. IL-6 has been implicated in the pathogenesis of fibrotic diseases in which IL-6 trans-signaling has been shown to stimulate the proliferation of fibroblasts and the release of procollagen and fibronectin.
Introduction
Interleukin-6 (IL-6) is a proinflammatory cytokine that was first identified as a B-cell stimulatory factor (1). The mature protein consists of 184 amino acids that form a four-helix bundle with the up-up-down-down topology common to most cytokines (2). On elucidation of its cDNA sequence (1), it became apparent that IL-6 was identical to hepatocyte-stimulating factor (3), hybridoma/plasmacytoma growth factor (4), interferon-β2 (5), and a 26-kDa protein with unknown function (6). Although this early observation provided initial clues to the pleiotropic nature of IL-6, it is now known that IL-6 has many functions in the regulation and coordination of the immune system, metabolism, and nervous system (7, 8). Not only is it involved in many autoimmune diseases, it also plays a role in the body's defense against infection, in many regenerative processes, and in the regulation of body weight (8). IL-6 is produced by activated immune cells and stromal cells, including T cells, monocytes/macrophages, endothelial cells, fibroblasts, and hepatocytes. This article provides an overview of the pleiotropic effects of IL-6, focusing on its involvement in mediating both normal and pathological processes related to the immune system.
Interleukin-6 classic signaling and trans-signaling
Stimulation of cells by IL-6 is initiated by binding of IL-6 to the IL-6 receptor (IL-6R), which belongs to the family of type 1 cytokine receptors (2). The complex of IL-6 and IL-6R then associates with a second receptor protein, glycoprotein 130 (gp130), which dimerizes and initiates cellular signaling (9, 10). Because IL-6 exhibits measurable affinity to IL-6R but not to gp130, only cells that express IL-6R and gp130 are capable of responding directly to IL-6 (11, 12). Although gp130 is expressed on all cells of the body, IL-6R is found on only a few cell types, including hepatocytes, some epithelial cells, and some leukocytes (9). However, soluble IL-6R (sIL-6R) is generated by proteolytic cleavage and, to a minor extent, by alternative splicing (13–14–15). This sIL-6R binds IL-6 with an affinity comparable to that of membrane-bound IL-6R (mIL-6R), and the complex of IL-6 and sIL-6R can also bind to membrane-bound gp130, inducing dimerization and signaling (16). Furthermore, cells that do not express IL-6R and that are therefore not directly responsive to IL-6 can be stimulated by the complex of IL-6 and sIL-6R (9). This process, which has been termed IL-6 trans-signaling, dramatically increases the spectrum of IL-6 target cells (13). It has been shown that IL-6 trans-signaling through sIL-6R is mainly proinflammatory, whereas IL-6 classic signaling through mIL-6R is associated with protective effects against infections and regenerative activities (8). IL-6 trans-signaling can be efficiently blocked by the soluble gp130Fc protein, which consists of the entire extracellular portion of gp130 dimerized by the Fc portion of a human immunoglobulin G1 (IgG1) antibody (9, 11). Blockade of IL-6 trans-signaling by the soluble gp130Fc protein leaves all classic IL-6 signaling through mIL-6R intact (11) (Fig. 1). As a result, it has been proposed that selective inhibition of IL-6 trans-signaling might be a more prudent mode of IL-6 inhibition because its protective and regenerative activities are not compromised by this strategy (17, 18).
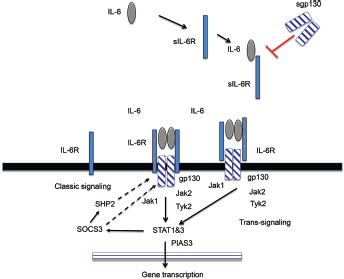
IL-6 classic signaling. gp130 = glycoprotein 130; IL-6 = interleukin-6; IL-6R = interleukin-6 receptor; JAK1/2 = Janus kinase 1/2; PIAS3 = protein inhibitor of activated STAT3; sgp130 = soluble glycoprotein 130; SHP2 = Src homology region 2-containing protein tyrosine phosphatase 2; sIL-6R = soluble interleukin-6 receptor; SOCS3 = suppressor of cytokine signaling 3; STAT1 & 3 = signal transducer and activator of transcription 1 and 3; Tyk2 = tyrosine kinase 2.
Intracellular signaling
The dimerization of gp130 leads to activation of Janus kinase (JAK) tyrosine kinases (19), which are constitutively bound to the intracellular portion of the gp130 protein (20) (Fig. 1). There are five tyrosine residues within the cytoplasmic tail of gp130 that are phosphorylated primarily by JAK1 (20). The membrane proximal tyrosine residue acts as a docking site for SHP2, which is an adapter protein and a tyrosine phosphatase, leading to the activation of mitogen-activated protein kinase and phosphoinositide 3-kinase signaling (19, 21). The membrane proximal tyrosine residue is also a target of the negative regulator of gp130 signaling known as suppressor of cytokine signaling 3, or SOCS3, which is rapidly upregulated by IL-6 and leads to the inhibition of gp130 signaling (19, 22). The four-membrane distal tyrosine residues of the cytoplasmic portion of gp130 are docking sites for signal transducer and activator of transcription 1 (STAT1) and 3 (STAT3), which become phosphorylated by JAK1, dimerize, and translocate to the nucleus, whereupon they act as transcription factors and lead to the transcription of IL-6 target genes, such as c-myc, bcl2, cyclin D1, MCP-1, Gremlin-1, several matrix metalloproteinases, and Notch (20, 23). Transcription of STAT3 is negatively regulated by protein inhibitor of activated STAT (PIAS).
Intracellular IL-6 classic signaling and trans-signaling are qualitatively identical, though it has been shown that IL-6 trans-signaling leads to higher signal amplitudes because most IL-6R–expressing cells exhibit higher levels of gp130 than IL-6R (9). Therefore, stimulation of such cells leads to fewer activated gp130 molecules than stimulation with the complex of IL-6 and sIL-6R, which can stimulate virtually all membrane-bound gp130 molecules (9). Moreover, it was demonstrated that stimulation of IL-6R–expressing cells with IL-6 results in rapid internalization and degradation of IL-6, whereas the complex of IL-6 and sIL-6R is internalized ineffectively (24). This results in longer signaling induced by the complex of IL-6 and sIL-6R than signaling induced by IL-6 alone (24). The higher signaling amplitude and the longer duration of signaling may result in qualitatively different IL-6 responses mediated by classic signaling and trans-signaling. For example, chemically induced liver cancer was recently shown to be dependent on IL-6 trans-signaling even though hepatocytes are one of the few cell types that express mIL-6R (25).
Interleukin-6 and the acute phase response
Stimulation of hepatocytes by IL-6 leads to initiation of the acute phase response and release of acute phase proteins, including C-reactive protein (CRP), serum amyloid A, haptoglobin, ferritin, and fibrinogen (26). Elevated levels of acute phase proteins are the hallmark of inflammation and are routinely measured when monitoring inflammation in clinical practice (27). IL-6 is the principal driver of the acute phase response in the pathogenesis of rheumatoid arthritis (RA), as evidenced by neutralization of IL-6 by the anti–IL-6R antibody tocilizumab, which led to the normalization of CRP (28). Hepcidin is an acute phase protein that regulates iron metabolism; it reduces intestinal iron absorption and sequesters iron into macrophages (29), which can lead to the development of anemia of chronic diseases (29). In RA, the role of IL-6 signaling in the development of inflammatory anemia is supported by evidence showing that IL-6R blockade with tocilizumab reduces hepcidin and haptoglobin, as well as CRP, subsequently increasing hemoglobin levels (30).
Innate immunity
Many innate immune cells and stromal cells produce and respond to IL-6 (17, 31), highlighting the prominent role that this cytokine plays in the innate immune response and in its interaction with stromal cells (32). Autocrine feedback can amplify inflammation (33), and IL-6 is thought to be involved in the transition from acute to chronic inflammation. During acute inflammation, monocytes, macrophages, and endothelial cells produce IL-6, leading to the recruitment of neutrophils through activation of a subset of chemokines and adhesion molecules by endothelial cells, smooth muscle cells, and fibroblasts (31, 34). IL-6 has also been shown to prolong neutrophil survival through regulatory effects on neutrophil apoptosis (35). The shedding of mIL-6R from neutrophils during chronic inflammation allows stromal cells that do not express IL-6R to respond to IL-6, leading to the production of monocyte-specific chemoattractants (36, 37) and increasing monocyte influx (36, 38).
Adaptive immunity
T and B cells are the main orchestrators of the adaptive immune response, which is central to the pathogenesis of many autoimmune diseases. Naive T and B cells are activated in the germinal centers of secondary lymphoid organs (39). These activated effector T and B cells drive cellular- and humoral-mediated immunity to amplify inflammation and produce autoantibodies (39). IL-6 has important roles in both T- and B-cell activation, as evidenced by the observation that IL-6–deficient animals are resistant to the development of antigen-induced arthritis (40).
B cells
On engagement with an antigen through the B-cell receptor, B cells are activated, differentiate into plasma cells, and produce antibodies. IL-6 was originally identified as a T-cell-derived soluble factor that induced the maturation of B cells into plasma cells (41). In addition, IL-6 causes CD4+ and CD8+ T cells to differentiate into subsets that produce IL-21, which in turn stimulates B-cell maturation (42, 43). IL-6–deficient animals have diminished antibody responses and are susceptible to infection (44). A recent study has demonstrated a role for IL-6 in the induction of regulatory B cells (45). IL-6 also allows the adaptive immune response to reciprocally impact the innate immune response because activated mature B cells and regulatory B cells produce IL-6, which then triggers innate immune cells and stromal cells to produce cytokines, chemokines, and other proinflammatory factors (32).
T cells
IL-6 plays a pivotal role in T-cell–mediated immunity, in part by promoting the differentiation of T-helper 17 (Th17) cells (46), which are characterized by the production of IL-17. Countering the pro-autoimmune Th17 cells are CD4+CD25+Foxp3+ regulatory T (Treg) cells. Transforming growth factor-beta (TGF-β) is a critical differentiation factor for the generation of Treg cells. Bettelli et al (46) showed that the presence of IL-6 inhibits TGF-β-induced differentiation of Treg cells. Furthermore, the presence of both IL-6 and TGF-β drives naive T cells to differentiate into Th17 cells (46–47–48). IL-6 is also produced by Th17 cells, further promoting differentiation into Th17 cells (49) and the development of autoimmune diseases (50). IL-6 and IL-17 released by Th17 cells induce fibroblasts to produce IL-6, further amplifying the immune reaction (51).
Endothelial cell function
IL-6 and IL-6R have been implicated in the pathogenesis of cardiovascular diseases. Human endothelial cells express gp130 but not IL-6R (9, 37, 52), whereas sIL-6R–mediated trans-signaling is known to increase the expression of adhesion molecules by endothelial cells and the binding of neutrophils, thereby causing vascular inflammation (37). In collagen-induced arthritis—an animal model of RA—inflammatory arthritis is associated with impaired vascular contractile responses when stimulated by 5-hydroxytryptophan (53). Blocking IL-6 trans-signaling with the soluble gp130Fc protein reverses vascular dysfunction to a normal state (54).
Fibroblasts and fibrosis
IL-6 has been implicated in the pathogenesis of fibrotic diseases, including pulmonary (55) and hepatic (56) fibrosis and systemic sclerosis (SSc) (57). Fibrosis is a common feature in chronic inflammatory diseases, whereas in fibrotic diseases, such as SSc, inflammatory infiltrates and cytokines are often found in affected organs (58). Fibroblasts and macrophages, especially activated M2 macrophages, produce IL-6 and TGF-β, which are profibrotic cytokines (59, 60). IL-6 trans-signaling leads to increases in collagen 1 synthesis by STAT3 and SMAD3 activation and production of Gremlin-1, a bone morphogenetic protein antagonist (59). Inhibiting Gremlin-1 reduced collagen synthesis. Serum amyloid A, an acute phase reactant produced by the liver, can induce dermal fibroblasts to produce IL-6 (61). IL-6–deficient mice were resistant to fibrosis in a peritoneal model of inflammation-mediated fibrosis; transfer of polarized Th1 cells reversed this effect, suggesting IL-6 caused fibrosis by shifting from chronic to acute inflammation through the induction of Th1 responses (62). In hypertrophic scars, dermal fibroblasts express a high level of cell surface gp130, and IL-6 trans-signaling leads to the proliferation of fibroblasts and the excessive production of extracellular matrix, including procollagen and fibronectin (63).
In patients with SSc, IL-6 levels are elevated in serum and in bronchoalveolar lavage fluid (64, 65). Serum IL-6 levels correlate with skin scores (66). Peripheral blood monocytes from patients with SSc produce more IL-6, either spontaneously or when stimulated (65, 67). Elevated sIL-6R and sgp130 levels have been reported in 20 patients with localized scleroderma (68). Fibroblasts from diseased skin sites of patients with SSc produce from 6- to 30-fold higher levels of IL-6 than fibroblasts from unaffected skin and healthy controls (69).
Conclusion
IL-6 is produced by a number of cell types and exerts pleiotropic effects. In particular, IL-6 plays an important role in mediating innate and adaptive immune responses. Many innate immune cells—neutrophils and monocytes/macrophages—produce and respond to IL-6, which can result in the amplification of inflammation and a switch from an acute to a chronic inflammatory state. IL-6 is also involved in mediating the activation of T and B cells, which are the main drivers of adaptive immune responses and key cells in the pathogenesis of many autoimmune diseases. IL-6 has been implicated in the pathogenesis of fibrotic diseases, including SSc. Therefore, targeting IL-6 signaling may be a rational therapeutic target for the treatment of patients with these diseases.
Footnotes
Acknowledgments
Medical writing and editorial assistance was provided by Sara Duggan, PhD, of ApotheCom (Yardley, PA, USA).
Conflict of interest: EC has received research grants from and has served on advisory boards and speaker bureaus for Amgen, Biogen, Bristol Myers Scripps, Celgene, Chugai Pharma, Eli Lilly, Janssen, Novimmune, Novartis, Pfizer, Regeneron, Roche, R-Pharm, Sanofi-Aventis, Tonix, and UCB. SR-J has served as a consultant to and speaker for Chugai, Genentech Roche, AbbVie, Sanofi, and Pfizer. He is the inventor of the sgp130Fc protein, and he is listed as an inventor on patents owned by CONARIS Research Institute, which develops the sgp130Fc protein (Olamkicept) together with Ferring Pharmaceuticals. He also has stock ownership in CONARIS.
