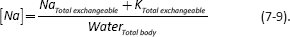Abstract
Purpose
Both hyponatremia and hypernatremia occur frequently in patients with cancer. To understand these disorders, investigators have mathematically modeled the serum sodium concentration in humans.
Methods
We expanded the model to incorporate time, in a “kinetic sodium” equation, and then a term explicitly appears for the rate of sodium correction, a key consideration to avoid treatment complications. The equation is solved for the fluid administration rate to assist the clinician with the hospital order in the management of dysnatremia.
Results
With baseline data on the input and output rates of Na/K/water along with the patient's body weight, a clinician can use the kinetic equation to aim for a desired [sodium] over a safe timeframe. The formula returns the infusion rate for any treatment fluid, such as normal saline, hypertonic saline, or 5% dextrose in water. The formula can also calculate treatment in terms of the dosage of salt tablets. If an abrupt water diuresis is anticipated, the equation should be entered with the predicted urine [Na+K] and flow rate to avoid overcorrection of hyponatremia. To demonstrate efficacy, we applied the kinetic equation to real-life cases of hypo- and hypernatremia.
Conclusions
The kinetic equation improves upon existing sodium equations by potentially accounting for all inputs and outputs, if known, and incorporating a rate of correction. The quantitative approach provides a basis for prescribing intravenous fluids and salt tablets.
Introduction
Dysnatremias are commonly associated with cancer (1, 2). Hyponatremia, already the most common electrolyte disorder, may be seen in as many as 46% of hospitalized patients in a cancer center (3). The etiologies are myriad. Certain cancers cause a paraneoplastic syndrome of inappropriate antidiuretic hormone (SIADH). Tumor involvement, in the brain and lungs commonly, can result in SIADH as well. Certain chemotherapies elicit antidiuretic hormone (ADH) release (4). ADH is also stimulated for nonosmotic reasons such as nausea, pain, narcotics, and hypovolemia, all of which are common in cancer patients (5). At times, patients overdo the well-meaning advice of avoiding chemotherapy toxicity by excessively hydrating. As for hypernatremia, which is much less common at 2.6%, some etiologies include cancer-related hypercalcemia that induces nephrogenic diabetes insipidus (3). Cancer can also interrupt the hypothalamic-pituitary axis and cause central diabetes insipidus. For non-diabetes insipidus reasons, perhaps having to do with diuretics and chemotherapy, the majority of hypernatremia is hospital-acquired (3). Tubulotoxins, such as ifosfamide, may impair the renal concentrating ability and lead to unregulated water loss (6). Neutropenic fever can also contribute to hypernatremia.
The basic serum sodium equation is
But this “static” equation only reflects a patient's sodium concentration at one cross-section in time. Nevertheless, we can convert the fundamental sodium equation into a “kinetic” version that describes how the serum sodium would evolve over time (10-12). The strategy is to express each input or output as a function of time. Since
Deriving a kinetic sodium equation is important because the treatment of the dysnatremias hinges upon the rate of correction. We err on the side of increasing the serum sodium at no faster than +8-10 mEq/L in 24 h for hyponatremia that is assumed to be chronic (>48 hours) unless proven otherwise (13-18). We aim for a similar rate of correction for hypernatremia, although some leeway is given for a faster rate of correction in the first 24 hours (19, 20). At the very least, doctors want to prescribe a therapy that keeps the dysnatremia from worsening. This situation may arise when normal saline (NS) is given in the setting of SIADH (13, 21-24). Correcting a dysnatremia at close to the desired rate can be achieved in a more technical way with the kinetic sodium equation (11, 12).
One variable that doctors have the most control over is the fluid rate (25-27). FlRate specifies how fast a fluid, such as NS or dextrose 5% water (D5W), should be infused. These prescriptions must compensate for the variables out of our control, such as urine [electrolyte] and urine flow rate, in such a way as to correct the serum sodium at a safe rate (10, 28-30). Thus, we solved the kinetic sodium equation explicitly for FlRate.
Methods
Incorporating time
Physicians are familiar with the role of time, because they prescribe intravenous (IV) fluids at a rate, usually in units of mL/h. Multiplying the rate and time by the fluid concentration yields a quantity of solutes to add to the numerator of the sodium equation. Likewise, multiplying rate and time yields the volume of water to add to the denominator. For example, NS at 42 mL/h means that in one day
The urine flow rate is also coded in terms of time. The kidney produces urine at more or less a continuous rate. That is belied by the episodic nature of urination, since urine accumulates in the bladder to be expelled in a large volume when it is convenient (unless there is a Foley catheter). But the sum of all the urine volumes in a day can give an accurate measure of the urine production rate. That information is found in the input/output section of the hospital chart. Knowing the urine flow rate, OutRate, we can deduce that the urinary loss of cationic solutes equals OutNa+K OutRatet, where OutNa+K = urinary [Na+K], and t = time. The volume of urinary water is OutRatet.
Similarly, oral intake of fluids can be converted into a continuous rate. Although the drinking of fluids is truly episodic, we can pretend that the total volume drunk in 1 day yields the average rate of fluid consumption for the purposes of the kinetic equation. Physicians are also familiar with the concept of average fluid rate, because they prescribe fluid restriction in terms of mL/day. For example, 1 liter per day is as if the patient were drinking
Behind the math
Having codified the net balance of Na/K and water from the various inputs (IV fluids, oral intake, salt tablets, etc.) and outputs (urine, gastrointestinal, insensible, etc.) in terms of time, we can use conservation of mass to assert that the rate of change in the Na/K amount is equal to the rate of Na/K coming in minus the rate of Na/K going out. The solution to that differential equation is
Spreadsheet
Once the principle of sodium kinetics is understood, the calculations can be done on a spreadsheet or other programmable app. This portability is especially helpful on the wards. To show how the equation may be used in practice, we entered the relevant clinical data into the spreadsheet and used the result to take care of real patients with dysnatremia.
Results
Normal saline
L.P. is a 63-year-old woman with metastatic epithelioid hemangioendothelioma, who is being treated with cyclophosphamide. Her hyponatremia was of relatively recent onset, so it was attributed to cyclophosphamide-induced SIADH. Nephrology was consulted because the serum sodium was slowly decreasing despite her receiving IV fluids.
Gathering data
Her body weight was 54.4 kg. If roughly half of the weight is total body water (TBW), then we can input 27 L. A recent urine Na was 83 mEq/L and urine K was 71.0 mEq/L for a sum of 154 mEq/L. Urine output was charted as 600 mL in 24 hours for a rate of 25 mL/h. She was receiving IV fluid in the form of D5NS + 20 mEq/L KCl for an [Na+K] of 174 mEq/L. The infusion rate was 75 mL/h. She was nil by mouth and was not taking salt tablets.
Goal and analysis
We wished to increase her serum sodium from 120 to 124 mEq/L over 24 hours. The current IV fluid was acceptably more hypertonic compared to her serum sodium of 120 mEq/L, so it was decided to simply adjust the infusion rate. The kinetic sodium spreadsheet was generated as follows (Fig. 1).
Normal saline-like intravenous (IV) fluid. In this case of cyclophosphamide-induced syndrome of inappropriate antidiuretic hormone leading to hyponatremia, the kinetic sodium spreadsheet was used to determine how the infusion rate of the current IV fluid, D5NS + 20 mEq/L KCl, should be adjusted. The solicited input data are above (see text for details), and the calculated output result is below.
To replicate the spreadsheet answer, the equation would be populated as follows:
The calculation said that an additional 30 mL/h of the treatment fluid was needed, going from 75 to 105 mL/h. We settled on changing the prescribed rate to 100 mL/h.
Reality Check and Commentary
In reality, the serum sodium went from 120 mEq/L at 00:01 to 124 mEq/L at 00:41 the next day. NS is often given to treat hyponatremia, as NS is hypertonic compared to any level of hyponatremia (31). However, NS can occasionally exacerbate hyponatremia, especially when SIADH is severe (13, 22, 32), as was the case here. The risk of a paradoxical lowering of sodium with NS is greater the more concentrated the urine electrolytes are (154 mEq/L) versus the serum sodium (120 mEq/L), so our patient was at risk (21, 23, 33). It took an increase in the rate of D5NS + 20 mEq/L KCl to overcome the SIADH and start to raise the serum sodium (31).
Hypertonic saline
J.A. is a 58-year-old woman with small cell lung cancer and paraneoplastic SIADH. She was hospitalized for hyponatremia to a serum sodium of 117 mEq/L. When fluid restriction to 1 L/day did not improve her [sodium], the nephrology consult team decided to try 3% saline.
Gathering data
The patient's body weight was 65.4 kg, so TBW was inputted as 33 L. The urine Na was 147 mEq/L. Urine K was not checked, but based on the urine osmolality of 393 mOsm/kg H2O the urine K could be 49.5 mEq/L at most, if no urine urea nitrogen or glucose was present. We decided to input the urine [Na+K] as 155 mEq/L. Urine output was recorded as 500 mL over the past 24 h for a rate of ~20.8 mL/h. She was not receiving IV fluids. No oral intake was assumed, and she was not taking salt tablets.
Goal and analysis
The goal was to raise the serum sodium from 117 to 125 mEq/L in 24 hours. Since 3% saline was being used, the treatment fluid [Na+K] was entered as 513 mEq/L. The spreadsheet was filled in (Fig. 2), and based on the calculation 3% saline was ordered at 30 mL/h.

Hypertonic saline. A woman with small cell lung cancer, hyponatremia, and the syndrome of inappropriate antidiuretic hormone was to be treated with 3% NaCl intravenous fluid. With the solicited data entered, the kinetic sodium spreadsheet calculated that the 3% saline should be infused at ~30 mL/h to raise the serum sodium from 117 to 125 mEq/L in 24 hours.
Reality Check and Commentary
In reality, the sodium went from 117 mEq/L at 05:15 to 122 mEq/L at 07:53 the next day. To be fair, 3% saline was not begun until about 15:00 of day 1, and a little less than 24 hours later at 12:21 of day 2 (~4.5 h after the 07:53 draw) the serum sodium was 125 mEq/L. Hypertonic saline was stopped.
D5W
T.C. is a 73-year-old man with a clival chordoma that was causing central diabetes insipidus leading to hypernatremia, with serum sodium of 152 mEq/L. He was receiving D5W at 100 mL/h, but the sodium lowering had stalled, so the renal fellow was planning to empirically titrate the D5W to 110 mL/h.
Gathering data
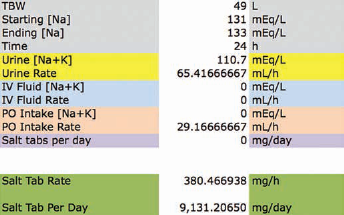
The patient's body weight was 78.7 kg. This was multiplied by 0.6 to get ~47 L for the TBW. The urine Na was 51 mEq/L and urine K was 11.0 mEq/L. The urine output was recorded as 3,120 mL in 24 hours for a rate of 130 mL/h. As mentioned above, D5W with an [Na+K] of 0 mEq/L was being infused at 100 mL/h. The oral intake of water was estimated as 150 mL in 24 hours for a rate of 6.25 mL/h.
Goal and analysis
The fellow wanted to bring the [Na] down from 152 to 146 mEq/L in 24 hours. Setting the treatment fluid [Na+K] to zero for D5W, we obtained the following spreadsheet result (Fig. 3). Based on the calculation, the fellow convinced the intensive care unit team to increase the D5W rate from 100 to 150 mL/h.

Dextrose 5% in water (D5W) was being given to a man with central diabetes insipidus and hypernatremia, but the serum sodium was not improving. The kinetic sodium spreadsheet informed the physicians that the D5W infusion rate should be increased by ~50 mL/h to achieve the desired lowering of sodium.
Reality Check and Commentary
In reality, in the next ~24 hours from 06:13 to 07:01 the next day, the serum sodium went from 152 to 144 mEq/L. As an aside, the rate calculation for D5W could have been extrapolated to free water flushes, as both are effectively salt free. However, with flushes, the gain of water is not as certain as with the IV infusion.
Salt tablets
The kinetic sodium equation can be adapted to calculate the amount of salt tablets to give. Tablets are not in fluid form, but multiplying FlRate
Then multiply by the molecular weight of NaCl, 58.5, to get the salt tablet rate in mg/h, assuming that all the tablets are fully absorbed.
L.G. is an 81-year-old female with melanoma, newly metastatic to the brain with some of the lesions being hemorrhagic. She was diagnosed with hyponatremia due to SIADH. Her serum sodium was 131 mEq/L when nephrology was consulted.
Gathering data
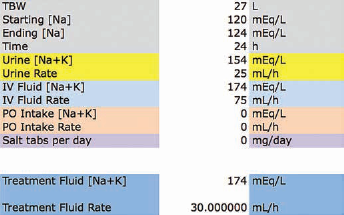
Her body weight was 98.5 kg, so we could input 49 L for TBW. The urine Na was 76 mEq/L and the urine K was 34.7 mEq/L for a sum of 110.7 mEq/L. Urine output was recorded as 1,570 mL over the past 24 hours for a rate of ~65.4 mL/h. No IV fluids or salt tablets were being given. She was drinking approximately 700 mL per day, so the oral intake of fluid was at ~29.2 mL/h.
Goal and analysis
We decided to treat the hyponatremia with salt tablets. The goal was to raise the serum sodium from 131 to 133 mEq/L in a day. The kinetic sodium spreadsheet (Fig. 4) gave the salt tablet rate at ~380.5 mg/h, which, over 24 hours, would be ~9,131 mg. Thus, the patient was prescribed NaCl tablets at 3 g by mouth 3 times a day.
Salt tablets. A woman with melanoma metastatic to the brain had hyponatremia and the syndrome of inappropriate antidiuretic hormone. The kinetic sodium spreadsheet was used to calculate the dosage of salt tablets to give in 1 day, in order to increase the serum sodium from 131 to 133 mEq/L.
Reality Check and Commentary
In reality, her serum sodium went from 131 mEq/L at 04:19 to 134 mEq/L at 04:37 the following day. This exercise shows that the required sodium load can far exceed a typical starting dose of NaCl, like 1 gram by mouth 3 times a day. There is even a precedent for salt tablets being given hourly (34). Inadvertent underdosing may explain why salt tablets may not seem to work at times in hyponatremia (35).
Water diuresis
So far, the urine [Na+K] was assumed to be constant over the time period of the analysis, usually 24 hours. The assumption worked well enough, as the kinetic sodium equation yielded sensible prescriptions that, when acted upon, nearly achieved the desired serum sodium. In reality, the urine [Na+K] can fluctuate, but in the cases above it probably oscillated around an average value. That average [Na+K] ends up being measured by the laboratory due to the way that urine is pooled prior to specimen collection, whether in the patient's bladder or in a Foley bag. An average UNa+K can be fairly consistent in the short-term, such as in SIADH (36), but sometimes the average UNa+K can change drastically. In the following case, SIADH was treated with tolvaptan, resulting in an abrupt water diuresis. This scenario requires a modification in how the kinetic equation is used.
S.S. is a 73-year-old female who has small cell lung cancer of the right upper lobe with a secondary malignant neoplasm of the liver and intrahepatic bile duct. Nephrology was consulted for hyponatremia that was due to SIADH, despite being on 7.5 mg tolvaptan daily as an outpatient. She was admitted for a [sodium] that had decreased to the 120s mEq/L range.
Gathering data
Her body weight was 48 kg for a TBW of ~24 L. Serum sodium was 122 mEq/L at 03:16. The urine Na was 146 mEq/L and the urine K was 24.0 mEq/L, measured at 09:02. Urine flow was 25 mL/h. Given the concentrated urine, nephrology recommended that an additional 15 mg tolvaptan be given that day (total 22.5 mg). The primary team also ordered salt tablets at 1,000 mg by mouth 3 times a day. An aquaresis ensued, and the urine Na dropped to 54 mEq/L while the urine K dropped to 17.0 mEq/L. Urine flow increased to 65 mL/h.
Goal and analysis
Instead of a well-defined IV fluid, tolvaptan was the therapy, and we lacked control over its eventual effect on the urine [Na+K] and the flow rate. We could not aim for a desired sodium, but we could still use the kinetic equation retrospectively to see how close it came to the actual [sodium]. Tolvaptan has a rapid onset and sustained action (37). The patient probably had diluted urine and increased flow for the bulk of the time period. Thus, for an abrupt water diuresis, the modification of the kinetic equation, in this case, was to input the post-tolvaptan urinary parameters. The aquaresis here was purposely induced by a V2-receptor antagonist (38, 39), but other cases can be triggered by hypertonic saline (30).
Reality Check and Commentary
In reality, the patient's sodium went from 122 mEq/L at 03:16 to 126 mEq/L at 19:57. If we enter the actual 126 mEq/L for the ending [sodium] and the actual 16.7 hours for the time, the kinetic equation should report that no additional treatment is needed. The infusion rate should be close to zero for any IV fluid we choose to prescribe (Fig. 5). NS is shown, but similar results are seen with 3% saline or salt tablets (not shown).
Water diuresis. Tolvaptan was being given to treat hyponatremia and the syndrome of inappropriate antidiuretic hormone, so an abrupt aquaresis was anticipated. As expected, the urine became diluted and the urine volume increased. Over the 16.7 hours, her serum sodium had increased from 122 to 126 mEq/L. In retrospect, this increase is nearly what the kinetic sodium equation would predict if the water diuresis were continued throughout the time period. The observed rise in serum sodium could be entirely explained by the effect of tolvaptan, which is supported by the kinetic sodium spreadsheet. It shows that 0.9% saline would only need to be given at ~1 mL/h. Although not shown, other intravenous fluids or salt tablets would also be calculated at close to 0.
Using the equation prospectively, clinicians do not have the benefit of knowing the future urine [Na+K] or the urine flow rate. However, if these urinary parameters are anticipated to change quickly and durably to their future values, we can enter our best educated guesses on the effect of tolvaptan, for example. As this case demonstrates, entering the predicted (dilute) urine values is preferable for an acute change in renal response. If the pre-tolvaptan (concentrated) urine values were used, the equation would yield a substantially higher treatment fluid rate, which, if acted upon, could have overcorrected the serum sodium.
Conclusions
Dysnatremia frequently warrants nephrology consultation at cancer centers and hospitals in general. However, the day-to-day treatments by the primary team and the recommendations by nephrology vary widely. When serum sodium goals are not achieved and IV fluids need to be titrated, we can make sound judgment on how to adjust the infusion rate qualitatively, up or down. However, quantitatively, we tend to change the infusion rate in multiples of 5 mL/h. A gradual path to improvement may be indicated, but it could prolong a hospital stay, at best, or actually set the patient back, at worst (e.g., NS paradox in SIADH). We can do better from a quantitative standpoint by understanding the basic sodium equation and utilizing the formulas derived from these first principles to guide our therapies (10, 11, 25).
The kinetic sodium equation is also based on the same fundamental physiology and would yield the same answers as the existing formulas. However, our equation explicitly incorporates time, which will elevate the role of the rate of sodium correction. Previous equations took the volume and composition of IV fluids (25), paired with urinary parameters (10), and then calculated how much the serum sodium would change. If it was too much or too little, the infusion rate was adjusted proportionally, which was not necessarily an easy computation. For treatment purposes, clinicians would likely prefer the equations to work in the opposite direction. Knowing the serum sodium to aim for, they would like to know the IV fluid volume to give (11). Our equation refines that further and reports the IV fluid rate, which is what the hospital orders require, while also taking into consideration the rate of sodium correction.
Admittedly, the kinetic sodium equation appears more complex than past formulas. However, the complexity is necessary, in part, to accommodate all of the potential sources of inputs and outputs that affect the cationic solute-to-water balance. When these multiple sources are not included, our equation reduces to the Adrogué-Madias formula, for example (15, 26). Besides, the complexity does not have to be intimidating, as the kinetic equation can be shrouded behind a user-friendly spreadsheet or an app that could make the adoption curve less steep. Also, by varying the input values one-at-a-time (e.g., TBW, correction rate, urine [Na+K], urine flow rate, etc.), our fellows have been able to develop an intuition for how the IV fluid prescription ought to change.
The renal response to dysnatremia is not static. The urine [Na+K] and flow rate measured at one point in time do not always remain around those values. However, these fluctuations are represented in aggregate by the average values of the urinary parameters. In practice, the averages are the data being entered into the kinetic equation, because (i) urine is mixed to give the average UNa+K; and (ii) the charted daily urine volume divided by 24 hours is the average URate. The use of averages results in less skewing and helps the accuracy of the kinetic equation. In a sense, the average UNa+K and URate are also being used in the case of water diuresis. However, the UNa+K and URate during an aquaresis will not be measured until a few hours later. The best we can do is to predict what the average values will be, and the kinetic spreadsheet is equipped to run the worst-case and other “what-if?” scenarios. Our predictive approach is consistent with other strategies to avoid too rapid a correction of hyponatremia (30) and to mitigate the risk of osmotic demyelination (40-42).
The kinetic and other sodium equations do not eliminate the need for vigilant monitoring of serum sodium and urinary parameters. Correction may not proceed as expected, because some variables may have changed or unknown variables may have a significant effect. Nevertheless, the most current or predicted clinical data can be entered into the kinetic equation to see how treatment should be adjusted. The calculations and aggregation of data are simplified by the use of a spreadsheet, which gives immediate and actionable results that facilitate the decisions on IV fluid orders during rounds. We hope that the kinetic sodium equation will assist in the management of dysnatremia in cancer centers and beyond.
Footnotes
Appendix
The kinetic sodium model as a function of time flows from the mass balance principle that the rate of change in the bodily Na/K content equals the rate of Na/K inputs minus the rate of Na/K outputs. For example, a dynamic equilibrium is achieved when InNa+K InRate (units of mEq/h) matches the OutNa+K OutRate, and then the net rate of change in the total body Na+K content equals zero.
The differential equation becomes [Na]t = serum concentration of sodium as a function of time Volt = volume that the Na and K are dissolved in. (It is also a function of time) t = time InNa+K = generic input's Na+K concentration InRate = volume rate at which the generic input is being given (e.g., mL/h) OutNa+K = generic output's Na+K concentration OutRate = volume rate of the generic output (e.g., urinary flow rate)
Every term on the right-hand side is considered to be a constant for the duration of the analysis. If and when the variables do change, the equation can be recalculated with the new lab values.
Use initial conditions to solve for C, the arbitrary constant of integration. At time t = 0, let [Na]t = [Na]0 and Volt = TBW, where [Na]0 = initial serum sodium, and TBW = total body water that the patient starts with. Then
Finally, concentrations are in mEq/L, all the volume ratesVolt is a linear function of time that is described by the baseline TBW plus Input volume minus Output volume, or
If we express all the variables in the standard units encountered in clinical practice, then the unit conversions turn the equation into
Therapeutically, physicians want to be able to control where a patient's serum sodium will be after a certain time. We can add a “treatment fluid” and solve for its administration rate. This is the mathematical equivalent of a physician choosing an IV fluid like NS, and then calculating the NS infusion rate, in theory, to achieve the desired change in the serum sodium at a safe correction rate.
The
As seen above, inputs are subtracted and outputs are added, counterintuitively. That rule (and an umbrella term for all other inputs/outputs) can be denoted by ∓… For clarity, we can salvage one generic input term and one major output term of urine, denoted by U.
Summarizing, the fundamental kinetic sodium equation solved for the IV fluid rate is
Financial support: None.
Conflicts of interest: None.