Abstract
Introduction
The aim of this study was to investigate the diagnostic accuracy of anaplastic lymphoma kinase (ALK) immunohistochemistry (IHC) for ALK gene rearrangement in non-small cell lung cancer (NSCLC) through systematic review, meta-analysis and diagnostic test accuracy review.
Methods
The current study included 11,806 NSCLC cases in 42 eligible studies. We performed concordance analyses between ALK IHC and fluorescence in situ hybridization (FISH). The diagnostic accuracy of ALK IHC was analyzed based on ALK IHC criteria and antibodies.
Results
The overall ALK IHC results were positive in 13.2%. The overall concordance rate between ALK IHC and FISH was 0.950 (95% confidence interval [CI], 0.927-0.966). In the ALK IHC-positive and negative groups, the concordance rates were 0.805 (95% CI 0.733-0.861) and 0.985 (95% CI 0.978-0.990), respectively. The ALK FISH-positive rates were 0.009 (95% CI 0.004-0.023), 0.378 (95% CI 0.217-0.572), 0.628 (95% CI 0.420-0.796) and 0.900 (95% CI 0.840-0.939) in the ALK IHC 0, 1+, 2+ and 3+ groups, respectively. In diagnostic test accuracy review for ALK IHC, the pooled sensitivity and specificity were 0.92 (95% CI 0.89-0.94) and 0.91 (95% CI 0.90-0.91), respectively. The diagnostic odds ratio and the area under the curve on the summary receiver operating characteristic curve were 266.56 (95% CI 110.83-641.14) and 0.983, respectively.
Conclusions
Our results suggested that ALK IHC equivocal (score 1+ and 2+) cases should not be considered as IHC-negative in screening for ALK gene rearrangement. Additional detailed criteria for ALK IHC equivocal cases are necessary to determine how to best apply this approach in daily practice.
Keywords
Introduction
Lung cancer is the leading cause of cancer-related death worldwide (1, 2). Many targeted therapies have been developed and applied for lung cancer patients in daily practice. The discovery and establishment of epidermal growth factor receptor and anaplastic lymphoma kinase (ALK) treatments have advanced targeted therapies in non-small cell lung cancer (NSCLC). Before the application of targeted therapies, molecular tests are required for patient selection. Thus, the accuracy and effectiveness of molecular tests is important in targeted therapy for NSCLC patients.
ALK gene fusion in lung cancer is strongly associated with adenocarcinoma and never smoking (2, 3). EML4-ALK fusion is the most common ALK gene rearrangement in lung adenocarcinoma and accounts for more than 90% of ALK gene rearrangements (1, 2). The diagnostic molecular test for ALK gene rearrangement is fluorescence in situ hybridization (FISH), which is a unique FDA-approved assay (1). In addition, reverse transcription-polymerase chain reaction (RT-PCR) is not recommended as a first-line diagnostic test for detecting ALK gene rearrangement due to the possibility of false negative results and loss of other ALK fusion partners. ALK immunohistochemistry (IHC) as a screening tool for ALK gene rearrangement may be useful for selecting patients, similar to HER2 IHC in breast and gastric cancers. In most pathology laboratories, IHC is more readily available than in situ hybridization (ISH) or RT-PCR (4). In addition, ISH or RT-PCR tests are expensive, require multiple steps, and are time-consuming compared with IHC. Before application of ALK IHC in daily practice, pathological validation is needed (1). The definition of ALK IHC positive/negative differs depending on previous studies, so it is necessary to determine how to evaluate ALK IHC in terms of its diagnostic role in screening (5-6-7-8-9-10-11-12-13-14-15-16-17-18-19-20-21-22-23-24-25-26-27-28-29-30-31-32-33-34-35-36-37-38-39-40-41-42-43-44-45-46).
In this study, we performed a systematic review and meta-analysis to determine the concordance between ALK IHC and FISH in NSCLC. In addition, the diagnostic role of ALK IHC as a screening tool for ALK gene rearrangement was investigated through diagnostic test accuracy review.
Materials and methods
Published Study Search and Selection Criteria
Relevant articles were obtained by searching the PubMed and MEDLINE databases through October 31, 2015. These databases were searched using the following keywords: “lung or NSCLC”, “ALK” or “anaplastic lymphoma receptor tyrosine kinase” and “immunohistochemistry or FISH”. The titles and abstracts of all searched articles were screened for exclusion. Review articles were also screened to find additional eligible studies. Search results were then reviewed and included if 1) the study was performed in human NSCLC and 2) there was information about the correlation between ALK IHC and FISH, and excluded if the articles were 3) case reports or non-original articles or 4) non-English-language publications.
Data Extraction
Data from all eligible studies were extracted by 2 independent authors. The included data were extracted from each of the eligible studies (5-6-7-8-9-10-11-12-13-14-15-16-17-18-19-20-21-22-23-24-25-26-27-28-29-30-31-32-33-34-35-36-37-38-39-40-41-42-43-44-45-46): the first author's name, year of publication, antibody clone and manufacturer, study location, antibody dilution ratio, evaluation criteria, and number of patients analyzed. For meta-analysis, we extracted all data associated with IHC results (scores 0, 1+, 2+ and 3+) and FISH results (positive and negative).
Statistical Analysis
For the meta-analysis, all data were analyzed using the Comprehensive Meta-Analysis software package (Biostat). We investigated the rates of ALK gene rearrangement according to the ALK IHC results. In this study, ALK IHC 1+ cases were considered to be positive. The concordance rates were measured by agreement rates between ALK IHC and FISH. Because eligible studies used various antibodies and evaluation criteria, a random-effects model was more suitable than a fixed-effects model. In addition, subgroup analyses based on tumor type and ALK antibody were performed. Heterogeneity between the studies was checked using the Q and I2 statistics and presented using p values. Additionally, sensitivity analysis was conducted to assess the heterogeneity of eligible studies and the impact of each study on the combined effect. To assess publication bias, a Begg's funnel plot and Egger's test were used. If significant publication bias was found, the fail-safe N and trim-fill tests were performed to confirm the degree of publication bias. The results were considered statistically significant at p<0.05.
Diagnostic test accuracy review was carried out using the Meta-Disc program (version 1.4) (47). Forest plots and summary receiver operating characteristic (SROC) curves were obtained. The area under the curve (AUC) on the SROC curve and the diagnostic odds ratio (OR) were calculated using the Meta-Disc program.
Results
Selection and Characteristics
A total of 309 reports were identified in the database search. Among them, 183 were excluded because of insufficient or lack of information on the correlation between ALK IHC and FISH. In addition, 46 reports were excluded because they reported the results of other diseases, or used animals or cell lines, and 38 were excluded because they were non-English or non-original articles. Finally, 42 studies were included in this systematic review and meta-analysis (Fig. 1 and Tab. I). There were 185 subsets from 42 eligible studies that included separate counting of IHC scores and ALK antibodies. The total number of patients was 11,799. Eligible studies used different manufacturers’ antibodies, as shown in Table I. In addition, eligible studies used many cutoff values for ALK IHC positivity, varying from a few percent to 15%. The overall ALK IHC-positive rate was 13.2% in the current meta-analysis.
Main characteristics of the eligible studies
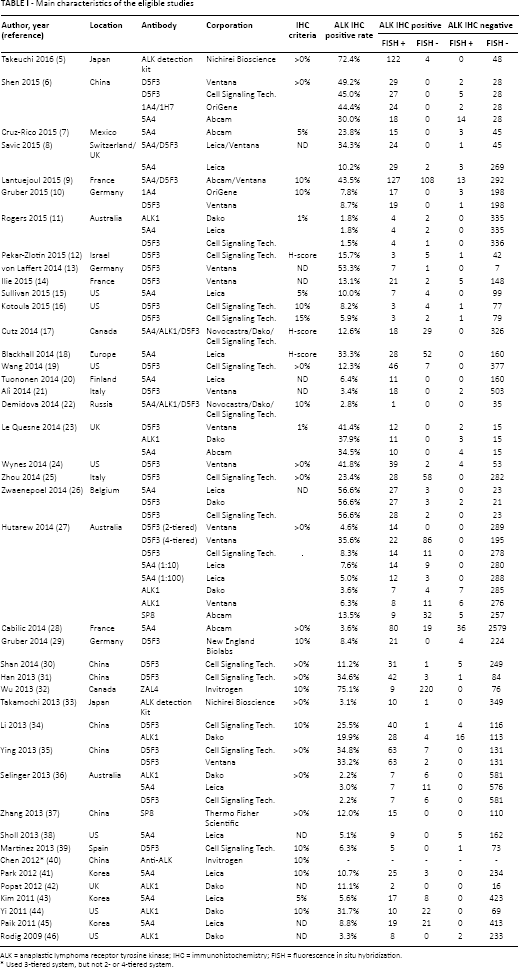
ALK = anaplastic lymphoma receptor tyrosine kinase; IHC = immunohistochemistry; FISH = fluorescence in situ hybridization.
Used 3-tiered system, but not 2- or 4-tiered system.

Flowchart of study search and selection methods.
Systematic Review and Meta-Analysis
Correlation between ALK IHC and FISH
The overall concordance rate was 0.950 (95% confidence interval [CI] 0.927-0.966) in 127 ALK IHC-positive and negative subsets of eligible studies. In the ALK IHC-positive and negative cases, the concordance rates were 0.805 (95% CI 0.733-0.861) and 0.985 (95% CI 0.978-0.990), respectively (Tab. II). Some of the eligible studies included ALK IHC 1+ cases in the ALK IHC-negative groups. However, in our meta-analysis, ALK IHC 1+ cases were considered positive for a 4-tiered evaluation system, regardless of the 2-tiered evaluation system. There was no significant difference in the concordance rate when adenocarcinoma and NSCLC were compared (0.949, 95% CI 0.904-0.974 vs. 0.951, 95% CI 0.921-0.970). However, in the IHC-positive group, the concordance of adenocarcinoma was significantly lower than that of NSCLC (0.713, 95% CI 0.583-0.815 vs. 0.849, 95% CI 0.759-0.909), but not in the IHC-negative group (0.987, 95% CI 0.980-0.992 vs. 0.982, 95% CI 0.970-0.990).
Concordance analysis between ALK immunohistochemistry and in situ hybridization in non-small cell lung carcinoma
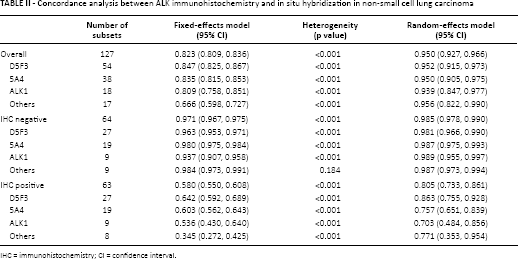
IHC = immunohistochemistry; CI = confidence interval.
Next, we investigated the ALK FISH-positive rates according to the ALK IHC scores, using a 4-tiered system. The ALK FISH-positive rates were 0.009 (95% CI 0.004-0.023), 0.378 (95% CI 0.217-0.572), 0.628 (95% CI 0.420-0.796) and 0.900 (95% CI 0.840-0.939) in the IHC score 0, 1+, 2+ and 3+ groups, respectively (Tab. III). The 95% CI of the ALK FISH-positive rates of IHC score 1+ and 2+ cases overlapped. The overall concordance rate and ALK FISH-positive rate were slightly lower in studies that used the 4-tiered system than in the overall eligible studies (0.852, 95% CI 0.770-0.908 vs. 0.951, 95% CI 0.928-0.966). However, the concordance rate of the ALK IHC score 0 group in the 4-tiered system was higher than in the overall eligible studies.
ALK positive rates based on ALK immunohistochemistry in non-small cell lung carcinoma
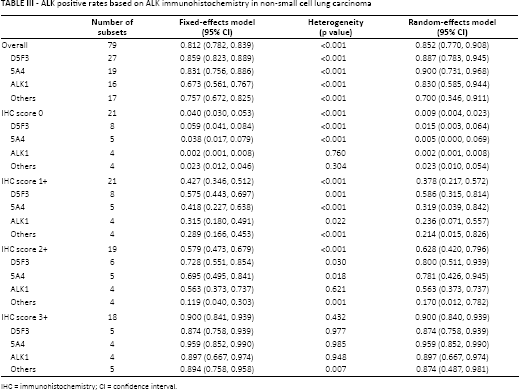
IHC = immunohistochemistry; CI = confidence interval.
The impact of ALK antibodies on the concordance between ALK IHC and FISH was investigated through subgroup analysis. In the 2-tiered system (Tab. II), there was little difference in the concordance rates between ALK antibodies. However, in studies that used the 4-tiered system (Tab. III), the overall concordance rate of the ALK 5A4 antibody was the highest among the ALK antibodies. In the ALK IHC score 0 group, there were no differences in concordance rates between ALK antibodies (range 0.002 to 0.023). In the ALK IHC 1+ and 2+ groups, D5F3 had the greatest sensitivity for detecting ALK gene rearrangement among various ALK antibodies. The ALK1 antibody had significantly lower ALK FISH-positive rates than 5A4 and D5F3 in the ALK IHC 1+ and 2+ groups. Additionally, the ALK 5A4 antibody showed the highest concordance rate in the ALK IHC 3+ group. Each eligible study may have the possibility of selection bias. However, meta-analysis cannot check each eligible study's selection bias, but considers publication bias as selection bias. In our meta-analysis, there was no significant publication bias in Begg's funnel plot, Egger's test, fail-safe N and trim-fill tests.
Diagnostic Test Accuracy Review
To confirm the ALK IHC score classification of 1+ and 2+ cases required application of ALK IHC for predicting ALK gene rearrangement. We performed a diagnostic test accuracy review based on the coverage of ALK IHC-positive cases, such as IHC 1+, 2+ and 3+ vs. IHC 2+ and 3+ vs. IHC 3+. The pooled sensitivity and specificity were highest in the IHC 1+, 2+ and 3+ and IHC 3+ subgroups, respectively. The highest diagnostic OR was identified in the IHC 1+, 2+ and 3+ subgroup (Tab. IV). However, there was no significant difference in the AUC on the SROC curve between the coverage of ALK IHC-positive cases (from 0.9830 to 0.9964). In the ALK IHC 1+, 2+ and 3+ subgroup, the pooled sensitivity and specificity and diagnostic OR were 0.92 (95% CI 0.89-0.94), 0.91 (95% CI 0.90-0.91) and 266.56 (95% CI 110.83-641.14), respectively. Our data suggested that ALK IHC-intermediate cases (score 1+ and 2+) may be considered as IHC equivocal in NSCLC and further confirmative molecular tests will be needed in such cases.
Sensitivity, specificity, diagnostic odds ratio and area under curve of summary receiver operation characteristics curve for prediction of ALK positivity

CI = confidence interval; AUC = area under the curve; OR = odds ratio.
A diagnostic test accuracy review based on ALK antibodies was conducted. The highest diagnostic OR was observed with the 5A4 antibody compared to various other antibodies (604.51, 95% CI 160.67-2274.40). The ALK1 antibody was the most sensitive (1.00, 95% CI 0.92-1.00) for ALK gene rearrangement (Tab. V). However, there was no significant difference in specificity between 5A4, D5F3 and ALK1 (from 0.93 to 0.96).
Sensitivity, specificity, diagnostic odds ratio and area under curve of summary receiver operation characteristics curve based on ALK antibodies for prediction of ALK positivity

CI = confidence interval; AUC = area under the curve; OR = odds ratio.
Discussion
The importance of targeted therapy using tyrosine kinase inhibitors has increased, especially in lung cancer. Precise evaluation of genetic alterations, such as ALK gene rearrangement, is important in determining targeted therapies for patients. Because a FISH, confirmative molecular test cannot be applied for all lung cancers at every pathology laboratory, effective screening tests are needed. The present study is the first meta-analysis to assess the association between ALK IHC and FISH and the diagnostic accuracy of ALK IHC in NSCLC. There were 4 major findings of this study. First, there was a significantly high concordance rate between ALK IHC and FISH in NSCLC. Second, our meta-analysis showed information for various ALK antibodies, such as D5F3, 5A4 and ALK1. Third, equivocal cases including ALK IHC 1+ and 2+ showed higher positive rates of ALK FISH compared to HER2 IHC in breast and gastric cancers (4). Fourth, consideration of ALK IHC 1+, 2+ and 3+ cases as positive could be helpful for detecting ALK gene rearrangement in diagnostic test accuracy review, unlike HER2 IHC in breast and gastric cancers.
ALK gene rearrangement was identified in 3%-13% of NSCLC (48-49-50-51-52-53-54). In the current systematic review, the overall rate of ALK gene rearrangement detected by FISH was 9.5%. However, the FISH-positive rate ranged from 1.2% to 70.1%. Additionally, the eligible studies that were included in this analysis showed 13.2% ALK IHC positivity (range 1.5%-72.4%). The positive rate of ALK IHC was slightly higher than that of ALK FISH. Drastically higher and lower ALK IHC and FISH positive rates may be caused by patient selection bias. However, the overall concordance rate between ALK IHC and FISH was statistically significantly high at 0.950 (95% CI 0.927-0.966). The concordance rate of ALK IHC-negative cases was higher than that of ALK IHC-positive cases (0.985, 95% CI 0.978-0.990 vs. 0.805, 95% CI 0.733-0.861). The false negative rate was significantly low at 1.5%, similar to HER2 IHC-negative cases in gastric cancer (3.1%) (4). Therefore, our data suggested that ALK IHC could be useful in screening for ALK gene rearrangement.
In the current systematic review, detailed information for evaluating the criteria for ALK IHC could not be obtained from some eligible studies. Specifically, in studies that used a 2-tiered evaluation system, information about the classification of equivocal cases (such as ALK IHC 1+ or 2+) could not be found. However, subgroup analysis is required to confirm the criteria for selecting patients who need additional FISH among ALK IHC-positive cases. In screening tests for ALK rearrangement, false positive or false negative cases may be important in decisions on patient management. So, the concordance between ALK IHC and FISH should be investigated to diminish the false positive and false negative cases. In the 4-tiered system, the concordance rates between ALK IHC and FISH were 0.991 (95% CI 0.977-0.996), 0.378 (95% CI 0.217-0.572), 0.628 (95% CI 0.420-0.796) and 0.900 (95% CI 0.840-0.939) in ALK IHC score 0, 1+, 2+ and 3+ subgroups, respectively The equivocal cases (score 1+ and 2+) can affect the overall concordance rate, as shown in Table IV. In the previous meta-analysis for HER2 in breast and gastric cancers (4, 55), the concordance rates of HER2 IHC 0/1+ and 3+ with HER2 ISH were about 96% and 91%, respectively. Indeed, cases with HER2 IHC scores of 0/1+ or 3+ can be confirmed as being negative or positive without additional ISH tests. According to our data, ALK IHC 1+ and 2+ cases could be considered as equivocal, like HER2 IHC 2+ cases. The definitive difference between ALK IHC and HER2 IHC results was the concordance rate in IHC 1+ cases.
Some of the eligible studies included in this analysis used various criteria to combine intensity and distribution, such as the Allred score, Histoscore (H-score) or Immunoscore (I-score). In addition, many cutoff values for ALK IHC 1+ have been used in previous studies, varying from a few percent to more than 50%. There may be limitations for applying our data due to various criteria and antibodies for ALK IHC. As shown in Table IV, criteria with the highest sensitivity and specificity were the IHC 1+, 2+ and 3+ group (0.92, 95% CI 0.89-0.94) and the IHC 3+ group (1.00, 95% CI 1.00-1.00), respectively. Tests with higher sensitivity might be more suitable for screening and tests with higher specificity might be more suitable for confirmation. In evaluating the impact of the equivocal cases, a diagnostic OR was also the highest in the ALK IHC 1+, 2+ and 3+ group. However, there was no difference in the AUC on the SROC curve between the ALK IHC criteria groups. The IHC score 1+ and 2+ subgroups could be heterogeneous compared with other IHC score groups, such as the HER2 IHC score 2+ group of breast and gastric cancers.
In the current systematic review and meta-analysis, the pooled concordance rate of eligible studies using the 4-tiered evaluation system was lower than that of eligible studies that used the 2-tiered evaluation system. The discrepancy in the concordance rates between studies that used 2- and 4-tiered evaluation systems may be caused by a difference in the inclusion criteria for equivocal cases (ALK IHC 1+ and 2+). In the current meta-analysis, the false negative rates were 1.5% and 0.9% in the 2- and 4-tiered evaluation systems, respectively. Therefore, ALK IHC-negative cases could be excluded for selection of TKI treatment in NSCLC. ALK IHC 1+ and 2+ cases might be considered as IHC equivocal and need further confirmative molecular tests such as FISH. However, similar to HER2 tests for breast and gastric cancers, whether ALK IHC 3+ cases required additional FISH was unclear. Because false negative cases do not provide opportunities for targeted therapy, additional FISH tests for all ALK IHC-equivocal and positive cases could be more helpful for patient treatment. Cumulative studies to determine ALK IHC evaluation criteria are necessary before this can be applied for use in clinical practice.
Although many studies have investigated the concordance of various ALK antibodies, we have not yet determined the optimal antibody and IHC methods. In previous studies, various antibodies were used, including D5F3, 5A4, ALK1 and others. In Takeuchi's study, IHC with the ALK1 antibody failed to detect EML4-ALK rearrangement (56). However, in IHC using the iAEP (intercalated antibody-enhanced polymer) method, the ALK1 antibody also showed positivity for EML4-ALK rearrangement (56). We performed an additional subgroup analysis of IHC with the ALK1 antibody based on the use of an amplification method to investigate the impact of ALK antibodies on the concordance between ALK IHC and FISH (data not shown). In the subgroup where the ALK1 antibody was used, the ALK FISH-positive rate of ALK IHC score 1+ cases was 0.236 (95% CI 0.071-0.557). ALK IHC score 1+ cases showed a higher false positivity rate in IHC using the ALK1 antibody compared to other antibodies (D5F3 and 5A4). However, all ALK IHC score 1+ cases were not false positive. So, an ALK inhibitor in patients with an ALK IHC 1+ score using the ALK1 antibody may have a therapeutic effect. There was no significant difference in the discordance rates between different antibodies, such as 5A4, D5F3, ALK1 and others, in ALK IHC-negative cases. However, significant differences between antibodies were detected in the ALK IHC score 1+, 2+, and 3+ subgroups. Differences in the pooled concordance rates between antibodies were 0.372, 0.611, and 0.085 in the ALK IHC score 1+, 2+, and 3+ subgroups, respectively. In equivocal cases with ALK IHC 1+ and 2+ scores, the most sensitive antibody was D5F3. In cases with ALK IHC 3+, 5A4 had the highest sensitivity among the various antibodies. In the previous guidelines, conclusive sensitivity and specificity data for various antibodies were not available (1). Because the concordance rate of the ALK1 antibody using the amplification kit was lower than without the amplification kit (data not shown), the usefulness of the ALK1 antibody might be limited compared to that of the other antibodies, 5A4 and D5F3. Therefore, further cumulative studies are also needed to identify the ideal ALK antibody and immunohistochemical methods for NSCLC.
There were some limitations to the current meta-analysis. First, some of the included studies showed extremely low or high positive rates of ALK IHC and FISH, which may have influenced the pooled values. Some studies showed higher ALK IHC and FISH positive rates, up to 70% in some cases. In addition, patient selection bias in the eligible studies could affect the variability of ALK IHC and FISH positive rates. Second, our data showed that consideration of an ALK IHC-equivocal diagnosis for ALK IHC 1+ cases could be useful for screening. However, because there is currently no consensus on detailed ALK IHC criteria, the interpretation of ALK IHC 1+ may have been limited in the present study. Finally, the current meta-analysis could not be performed to assess the correlation between FISH and other ISH methods due to insufficient information. Kim et al (43) reported that chromogenic ISH was concordant with FISH (ĸ = 0.92).
In conclusion, this study showed that ALK IHC was strongly concordant with ALK FISH and had high diagnostic accuracy for ALK gene rearrangement. In addition, our data suggested that ALK IHC equivocal cases (score 1+ and 2+) should not be considered as negative in NSCLC. These ALK IHC equivocal cases will need further confirmative molecular tests. However, more cumulative studies will be required to obtain detailed criteria for ALK IHC diagnoses.
Footnotes
Financial support: None.
Conflict of interest: The authors declare they have no conflict of interest related to this study.
