Abstract
Introduction
Papillary thyroid cancer is a disease that has been associated with chronic inflammation. The purpose of this study is to measure the production of the proinflammatory cytokines IL-1β, IL-6 and IL-8 and neopterin, which is a novel biomarker for cellular immune response in papillary thyroid cancer.
Materials and methods
The serum IL-1β, IL-6, IL-8 and neopterin values of 31 papillary thyroid cancer patients undergoing bilateral total thyroidectomy were measured before and 20 days after surgery. The values were compared with those of 39 healthy controls.
Results
Serum IL-1β levels were similar across groups. IL-6 (p<0.001), IL-8 (p = 0.015) and neopterin levels (p = 0.002) were higher in presurgical samples and returned to normal following surgery.
Conclusions
The proinflammatory cytokines IL-6 and IL-8, but not IL-1β, were produced in greater amounts in papillary thyroid cancer. Serum neopterin seems to be a valid biological marker supporting the presence of papillary thyroid cancer.
Introduction
Thyroid carcinoma is a rare malignancy, accounting for only 1%-2% of cancer cases (1). Ninety to ninety-five percent of thyroid cancers are categorized as well-differentiated tumors arising from follicular cells. Papillary cancers, among the follicular and Hürthle cell subtypes, are included in this category (2). Inflammation is a physiological, protective process that the organism activates in response to tissue damage. The persistence of stimuli that induce an inflammatory response or the failure of the mechanism that ends it may result in chronic inflammation even among patients with cancer, as first proposed by Virchow in 1863 (3). The concept that inflammation can cause cancer is further supported by the observation that polymorphisms in genes encoding proinflammatory cytokines are associated with an increased risk of some cancer types (3, 4).
The inflammatory response has been associated with carcinogenesis in the thyroid. Most thyroiditis-associated carcinomas are papillary, but other types have also been reported (3, 5). It has been suggested that about 20%-50% of papillary thyroid carcinomas occur within the setting of chronic inflammation (6).
Cytokines are molecules that influence the activation, growth and differentiation of various target cells. They are produced by different cell types including cancer cells. Several cytokines, such as interleukin-1 beta (IL-1β), interleukin-6 (IL-6) and interleulin-8 (IL-8), are proinflammatory cytokines, inducing a systemic inflammatory response reflected by increased levels of neopterin or tumor necrosis factor-alpha (TNF-α), while other cytokines such as the IL-1β receptor antagonist are secreted as a feedback control mechanism of the systemic inflammatory response (7). Proinflammatory cytokines produced at tumor sites play key roles in cancer progression. The production of these cytokines by epithelial cancer cells depends mainly on the NFκB transcription factor. Among the cytokines induced by NFκB, IL-6 has been identified as one of the most relevant myeloid-derived factors that promote tumor formation (8). It has been reported that several growth factors stimulate the growth of normal thyroid epithelial cells and that cultured healthy thyroid follicular cells are able to produce high levels of IL-6 and IL-8. Both IL-1β and interferon gamma (IFN-γ) are anticancer factors that are able to suppress the proliferation of papillary thyroid carcinoma cells (9).
Neopterin serves as a marker for cellular immune system activation. It is produced by monocytes/macrophages upon stimulation with IFN-γ, interferon-alpha (IFN-α) and endotoxins (10). Therefore, measurement of neopterin concentrations in body fluids like serum, cerebrospinal fluid or urine provides information about T-helper-cell type 1-derived cellular immune activation (11).
According to our hypothesis, serum proinflammatory cytokines and neopterin levels may play a role in the pathogenesis of papillary thyroid cancer. The aim of the present study was to investigate the levels of IL-1β, IL-6, IL-8 and neopterin in patients with papillary thyroid carcinoma prior to and following surgical treatment.
Materials and methods
This is a sex-matched case-control study, conducted at the Department of General Surgery of Ankara Oncology Hospital and the Department of Biochemistry of Celal Bayar University. The study was approved by the hospital ethics committee. All patients and volunteers involved in the study gave their informed consent.
Patients
Recruitment of study participants was performed at the Department of General Surgery, Ankara Oncology Hospital, by trained physicians between 2013 and 2015. The patients whose fine-needle aspiration revaled papillary thyroid carcinoma were enrolled into the study. Nodule size, capsule invasion, multicentricity and T stage according to the AJCC staging system (12) were evaluated. Since follicular thyroid carcinomas cannot be diagnosed by fine-needle aspiration, they were not included in the study (13). Patients with lymph node metastasis and previous neck radiotheraphy were also excluded. Only female patients were included in the study, as prognosis in men is known to be poor compared to that of women (14, 15), and serum neopterin levels are also known to differ depending on age and gender (16). Other exclusion criteria included diabetes mellitus, hypertension, hormonal therapy such as oral contraceptives or estrogen replacement therapy, treatment with thyroxin derivates, and coexistence of active infectious and chronic diseases. All patients had normal complete blood counts and C-reactive protein (CRP) values before surgery. Basically we tried to exclude all situations associated with a known or possible increase in immune activation.
Eventually 39 patients were chosen for the study group. Blood was collected from each patient the day before surgery. All patients were clinically euthyroid before surgery. Bilateral total thyroidectomy was performed in all patients. All patients were asked to come to the control visit on the 20th postoperative day. Eight patients were excluded from the study group because they did not attend the postoperative control visit for the blood test.
Thirty-nine healthy volunteers who had normal thyroid ultrasonography findings were chosen from the hospital personnel as the control group. Individuals with chronic diseases, abnormal complete blood counts and high CRP values were excluded. Thus 39 control group subjects were compared with 31 study group subjects. Leukocyte and thrombocyte counts, hematocrit, hemoglobin, serum protein, serum urea, alkaline phosphatase, gamma-glutamyl transferase and aspartate aminotransferase at diagnosis were measured by routine techniques (data not shown). From these data and from the following routine checks, signs of infection in the patients could be excluded.
Assay
IL-1β, IL-6, IL-8 and neopterin were evaluated in serum samples isolated from peripheral blood and banked at -80°C. All samples from each patient were run in the same assay. Serum thyroid hormones were measured using radioimmunoassay kits (TOSOH Bioscience). Patient plasma samples were analyzed by an enzyme-linked immunosorbent assay (ELISA) with commercial kits for IL-6 and IL-8 (Invitrogen), IL-1β (IBL-Hamburg) IBL International and neopterin (Shanghai Yehua Biological Technology Co., Ltd.). The values were measured in triplicate and the mean concentrations were determined from the standards provided.
Statistical Analysis
The software SPSS for Windows, version 16.0 was used in the statistical analysis of the data. Associations between continuous variables were assessed by the Kruskal-Wallis test. In all analyses, a p value of 0.05 was used as the cutoff for significance.
Results
Median age was 44 years (range: 18-74) in the study group and 41 years (range: 20-68) in the control group (p = 0.254). The mean size of the nodule that led to the diagnosis was 3.01 ± 0.85 cm (range: 1.5-4.5). Twenty-five patients had stage T2 tumors (25/39; 64%), 9 patients had stage T3 tumors, and 5 patients had stage T1 tumors. Twenty-one patients (53.8%) had multicentric disease. Only 2 patients had capsule invasion.
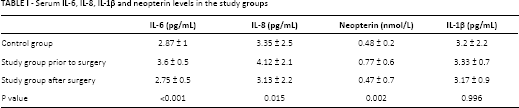
Serum IL-1β, IL-6, IL-8 and neopterin levels and their comparisons are presented in Table I. There was no statistical difference between serum IL-1β levels (control group: 3.2 pg/mL; study group: 3.17 pg/mL; after treatment: 3.33 pg/mL; p = 0.99). The IL-6 level was higher in the study group than the control group (3.6 pg/mL vs. 2.87 pg/mL, p<0.01) and was similar between the control and treatment groups (2.87 vs. 2.75 pg/mL). The same pattern was observed for IL-8 and neopterin. The IL-8 level was higher in the study group than in the control group (4.12 pg/mL vs. 3.35 pg/mL, p = 0.015) and there was no difference between the control and treatment groups (3.35 pg/mL vs. 3.13 pg/mL). The neopterin level was also higher in the study group than the other 2 groups (0.77 nmol/L, p = 0.002). The serum neopterin level was approximately the same in the control and treatment groups (0.48 nmol/L vs. 0.47 nmol/L).
Serum IL-6, IL-8, IL-1β and neopterin levels in the study groups
We evaluated the correlations of histopathological parameters with the levels of IL-1β, IL-6, IL-8 and neopterin. None of these were statistically significant. P values for nodule size were 0.282 for IL-1, 0.377 for IL-6, 0.087 for IL-8, and 0.749 for neopterin. P values for T stage were 0.399 for IL-1, 0.744 for IL-6, 0.482 for IL-8, and 0.44 for neopterin. P values for multicentricity were 0.106 for IL-1, 0.17 for IL-6, 0.767 for IL-8, and 0.489 for neopterin. Since only 2 patients had capsule invasion, we did not perform statistical analysis for this parameter.
Discussion
Neopterin is a biological marker for cellular immune activation and has been shown to increase in numerous diseases such as viral infections (e.g., HIV) (17), allograft rejection episodes (18), cardiovascular disease (19), and neurological diseases (20). Increased neopterin production has been detected in cancer patients and has been proposed as a prognostic indicator (21-22-23).
Neopterin levels have also been evaluated in thyroid diseases. Wagner et al (24) found Graves’ disease and nontoxic multinodular goiter to be associated with increased neopterin levels. There was no neopterin increase in autoimmune thyroiditis and hyperthyroidism. Kondera-Anasz et al (25) evaluated 33 patients with thyroid neoplasms, 30 with nontoxic goiter and 8 with thyroiditis and found increased serum neopterin production in goiter and thyroid cancer patients. Sahin et al (26) measured urinary neopterin in 24 patients with thyroid papillary cancer and 44 patients with benign thyroid disorders. Both groups had higher urinary neopterin levels than the control group and there was a difference between malignant and benign thyroid disorders. The authors suggested that neopterin can be a differential biomarker for malignant and benign thyroid disorders. Inancli et al (27) also found malignant thyroid disorders to have higher neopterin levels than benign disorders and both disease groups to have higher levels than healthy controls. They also compared patients with and without chronic thyroiditis within the malignant group and found no difference. Our study group consisted of only papillary thyroid cancer patients and the neopterin levels were similarly higher than those in healthy controls. All patients had normal thyroid hormone tests, so we are not able to analyze the effect of hyperthyroidism. Since our study group involved only papillary thyroid cancer we could not compare malignant versus benign thyroid diseases.
While acute inflammation is part of the body's defense mechanism, chronic inflammation may also lead to cancer. Several inflammatory interleukins, such as IL-1, IL-6, IL-8 and IL-18, and certain chemokines have been linked to tumorigenesis (28). IL-1 is a major proinflammatory cytokine that mainly acts by inducing cascades of cytokine- and inflammation-promoting mediators. It is produced by both malignant and microenvironmental cells. IL-1β is a major agonist of IL-1 and is only active in its inflammasome. It is considered to be a major mediator of inflammation (29). IL-1β has been previously evaluated in papillary thyroid cancer. Kammoun-Krichen et al (30) measured serum levels of 17 cytokines in 115 patients with a variety of thyroid disorders (including 15 papillary thyroid cancers) and compared them with those of 30 healthy controls. They found IL-1β to be underexpressed when compared to other thyroid disorders and healthy controls. They even concluded that IL-1β can be used to discriminate papillary thyroid cancer from atrophic thyroiditis. It has been reported earlier that IL-1 affects thyroid cells by downregulating the expression of thyroglobulin and thyroperoxidase by inhibiting iodide organification (31-32-33). This downregulating effect is probably the reason IL-1β was not found to be increased among cancer patients where other interleukins were. Since IL-1β is a proinflammatory cytokine, we expected it to increase along with other interleukins. In this regard, our findings are similar to those of previous reports.
IL-6 is a multifunctional cytokine which plays important roles in a wide range of biological activities in different types of cells including tumor cells. IL-6 is the primary mediator of the acute-phase inflammatory response with effects characterized by fever, chills, fatigue and increased hepatic synthesis of acute-phase reactants (34). IL-6 levels in thyroid cancer have been evaluated in numerous studies. Lumachi et al (35) showed several cytokines including IL-6 to be expressed in thyroid cancer cell lines, thus confirming the role of IL-6 in papillary thyroid cancer. Zivancevic-Simonovic et al (36) evaluated several cytokine levels in papillary thyroid cancer patiens with and without Hashimoto's thyroiditis. They reported that papillary thyroid cancer patients with Hashimoto's thyroiditis produced increased amounts of IL-4, IL-6, IL-9, IL-13 and IFN-γ compared to patients without thyroiditis. Provatopoulou et al (37) measured serum levels of 10 interleukins in malignant and benign thyroid diseases. They found that IL-6 levels (along with IL-7, IL-10 and IL-13) were significantly higher in malignant and benign thyroid diseases compared to healthy controls, which is similar to our results. Unlike our results they found IL-8 levels to be lower among patients with thyroid disorders.
IL-8 is a proinflammatory chemokine which is associated with neutrophil chemotaxis and degranulation. The biological effects of IL-8 are mediated through the binding of IL-8 to 2 cell surface receptors, CXCR1 and CXCR2. The increased synthesis and secretion of IL-8 from tumor cells has wider significance to the tumor microenvironment given the characterized expression of CXCR1 and CXCR2 on cancer cells, endothelial cells, and neutrophils/tumor-associated macrophages (38). IL-8 is the first chemokine shown to be secreted by thyrocytes. Even though some evidence points to a role of IL-8 as a tumor-promoting agent in several cancers, there are few reports to support this in thyroid cancer (39). Kobawala et al (40) measured serum IL-8 and IFN-α levels in 69 patients with thyroid disorders (32 with thyroid cancer) and compared the values with those of 19 healthy controls. They reported that the levels of both IL-8 and IFN-α were higher in individuals with thyroid disorders than healthy controls. Furthermore, serum IL-8 levels showed a significant positive correlation with disease stage in thyroid cancer patients. Our results are similar in this regard, as both IL-6 and IL-8 levels were higher in the cancer group and returned to normal values after surgery.
In conclusion, our data support that the proinflammatory cytokines IL-6 and IL-8 are produced in greater amounts in papillary thyroid cancer. However, a lower amount of IL-1β is produced in this cancer group. Serum neopterin seems to be a valid biological marker supporting the presence of papillary thyroid cancer. To our knowledge, this is the first study to analyze these cytokines following surgery. Return of interleukins to normal levels following surgery suggests normalization of immune reactivation to cancer. Further studies are needed to monitor serum levels prior to recurrence.
Footnotes
Acknowledgment
We would like to thank Ayse Seval Ozgu-Erdinc for her contribution to statistical analysis.
Financial support: None.
Conflict of interest: The authors have no conflict of interest to disclose.
