Abstract
Background
We evaluated the role of single nucleotide polymorphisms in the CYP17A1 gene for predicting clinical outcome in castration-resistant prostate cancer (CRPC) patients treated with abiraterone.
Methods
Sixty-four patients were genotyped for the selected polymorphisms (rs743572, rs10883783, rs17115100 and rs284849) in CYP17A1. We hypothesized that different genotypes could be associated with progression-free survival (PFS) and overall survival (OS).
Results
Statistical analyses highlighted no significant associations between these polymorphisms and clinical outcome. However, individuals with the most common TT genotype for rs10883783 had a 3 months’ longer PFS than individuals with the TA + AA genotype.
Conclusions
With the limitation of the retrospective study design and the small sample size, the analyzed polymorphisms do not seem to be correlated with clinical outcome of CRPC patients treated with abiraterone.
Introduction
Hormone therapies lead to a decrease in testosterone and dihydrotestosterone synthesis and patients with metastatic prostate cancer only benefit temporarily from these therapies, subsequently progressing to a castration-resistant status. Such resistance could be due to persistent androgen signaling and to its de novo synthesis (1, 2).
Abiraterone is a potent oral selective inhibitor of the cytochrome P450 17α-hydroxylase (CYP17A1) which leads to decreased androgen production in the adrenal glands, testicles and tumor microenvironment (3-4-5-6). The CYP17A1 gene is located on chromosome 10q24.3 and encodes an enzyme that catalyzes key reactions in sex-steroid biosynthesis, mediating 17α-hydroxylase and 17,20-lyase activities (7). The identification of somatic alterations in the specific target of abiraterone could help to select patients who will really benefit from this type of therapy. Intratumoral CYP17A1 overexpression has been detected in prostate cancer biopsies from patients treated with abiraterone, suggesting that upregulation of the enzyme could play a key role in resistance to treatment (8). Despite its potential key role, there are few literature data regarding CYP17A1 genetic alterations and their potential application for prostate cancer prognosis. Carey and collaborators (9) identified a common single base pair substitution, -34T>C (rs743572) in 5’-UTR CYP17A1, defining patients with homozygosis for the common allele as “A1A1”, those with heterozygosis as “A1A2” (genotype TC), and individuals with homozygosis for the variant allele as “A2A2”. The authors hypothesized that this promoter variant has an effect on the level of the transcript. However, it is still not understood how this alteration affects the protein expression and, consequently, testosterone levels in serum (10, 11). The single nucleotide polymorphism (SNP) rs743572 has also been correlated with the clinical outcome of patients who are resistant to hormone therapy, and men with the A2A2 genotype have a longer survival than those with the common allele (12). Another important SNP would appear to be rs10883783, although few data are available on it. Wright and coworkers found that men with the minor variant allele A in rs10883783 showed a 56% lower risk of prostate cancer-specific mortality (13).
Since there is very little evidence of a correlation between CYP17A1 gene polymorphisms and clinical outcome with abiraterone therapy, we decided to evaluate different patient genotypes and to verify their impact on treatment efficacy.
Methods
Case Series and Study Design
Sixty-four castration-resistant prostate cancer (CRPC) patients treated with abiraterone at the Istituto Scientifico Romagnolo per lo Studio e la Cura dei Tumori (IRST) IRCCS, Meldola, were included in this retrospective study. The eligibility criteria comprised histological confirmation of adenocarcinoma of the prostate without neuroendocrine differentiation or small cell histology progressing on androgen deprivation. Patients were required to have received at least 1 but not more than 2 cytotoxic chemotherapy regimens for metastatic CRPC. At least 1 regimen should have contained docetaxel. Prior ketoconazole therapy was not permitted. Additional eligibility criteria included Eastern Cooperative Oncology Group (ECOG) performance status ≤2, adequate cardiac, renal, hepatic and bone marrow function, a serum potassium level ≥3.5 mmol/L, and ongoing androgen deprivation with serum testosterone <50 ng/dL. The protocol was approved by our institutional review board. Written informed consent was obtained from all patients.
Clinicopathological characteristics of CRPC patients

CRPC = castration-resistant prostate cancer; PSA = prostate-specific antigen.
Treatment consisted of 28-day cycles of abiraterone acetate 1,000 mg taken daily on an empty stomach with prednisone 5 mg twice daily. Treatment continued until there was evidence of disease progression or unacceptable toxicity. Before starting treatment, patients underwent a baseline PSA blood test and a CT scan of the chest and abdomen. Patients were evaluated monthly for PSA response and toxicity. A CT scan was performed every 3 months during treatment with abiraterone. Disease progression was defined according to the Prostate Cancer Working Group 2 (PCWG2) criteria (14). Adverse events were graded using the National Cancer Institute Common Terminology Criteria for Adverse Events (CTCAE), version 3.
Genotyping
DNA was extracted from peripheral blood samples collected in PAXgene blood DNA tubes using the PreAnalytiX kit (Qiagen) according to the manufacturer's instructions. Two SNPs (rs743572 and rs10883783) were genotyped by the ABI 3130 Genetic Analyzer (Applied Biosystems) with a fluorescence-based capillary electrophoresis system. Purified DNA was amplified for the CYP17A1 gene using the following primer sequences: rs743572 forward 5’-TTGGGCCAAAACAAATAAGC-3’, reverse 5’-GGGCTCCAGGAGAATCTTTC-3’; rs10883783 forward 5’-CTATGGCAGGATGAGGGTGT-3’, reverse 5’-TGAGTTTGCTGTGGACAAGG-3’. PCR results were verified by agarose gel electrophoresis and sequenced using a BigDye Terminator 3.1 sequencing kit (Applied Biosystems) with the same PCR primers. The sequences were then analyzed with sequencing analysis software (Applied Biosystems).
The third polymorphism, rs17115100, was genotyped using a commercial TaqMan SNP genotyping assay (assay ID: 25597854_10). All DNA samples were analyzed in duplicate with TaqMan PCR Master Mix on a 7500 real-time PCR cycler according to the manufacturer's instructions. The allele calls were identified by specific software.
Immunohistochemistry
FFPE tissue samples were deparaffinized and rehydrated. Five-micron sections were incubated for 1 hour at room temperature with the anti-CYP17A1 antibody (ab134910, Abcam) at a dilution of 1:250.
Statistical Analysis
Progression-free survival (PFS) was defined as the time from the starting date of abiraterone treatment to the first observed progression, relapse or death (whichever came first). Overall survival (OS) was defined as the time from the starting date of abiraterone treatment to the date of death from any cause. Patients who did not experience the outcome of interest were censored at the time of last follow-up. Kaplan-Meier methods were used to estimate PFS and OS. The log-rank test was calculated to compare the curves of the different patient genotypes. For each polymorphism, differences in the allelic frequencies between our case series and the worldwide population were evaluated by the chi-square test and Hardy-Weinberg equilibrium was assessed. Allelic frequencies were determined by dbSNP short genetic variations.
A value of p<0.05 was considered statistically significant. All p values were 2-sided. Data were analyzed using the SAS 9.3 software (SAS Institute).
Results
Sixty-four Caucasian patients with metastatic CRPC treated with abiraterone were genotyped for 4 polymorphisms in the CYP17A1 gene. The clinical and pathological characteristics of the patients recruited for the study are summarized in Table I.
There were no statistically significant differences in the allele frequencies of our case series with respect to those of the worldwide population for all the analyzed polymorphisms (Tab. II). All studied polymorphisms were in Hardy-Weinberg equilibrium (Tab. II).
Allele and genotype frequencies for each polymorphism
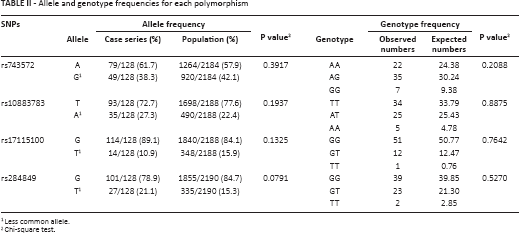
Less common allele.
Chi-square test.
The CRPC patients treated with abiraterone had a median PFS of 7.2 months (95% CI: 5.3-10.5) and a median OS of 17.6 months (95% CI: 10.8-21.9) (Fig. 1). The association between each CYP17A1 gene polymorphism and PFS and OS was evaluated (Figs. 2 and 3). No statistically significant differences were found in either PFS (log-rank test p = 0.6474) or OS (log-rank test p = 0.4825) for the rs743572 polymorphism. For rs10883783 the PFS of individuals with the most common TT genotype was 3 months longer than that of patients with the TA+AA genotype: 8.5 vs. 5.4 months, respectively, even if the difference was not statistically significant (log-rank test p = 0.5586). This difference was not observed in the OS curve: median OS 17.7 months for the TT genotypes vs. 17.6 months for the TA + AA genotype (log-rank test p = 0.7668). The rs17115100 polymorphism showed no statistical differences in either PFS (log-rank test p = 0.4054) or OS (log-rank test p = 0.9825). Similarly, we found no differences for the rs284849 polymorphism with regard to PFS (log-rank test p = 0.8282) or OS (log-rank test p = 0.5188).
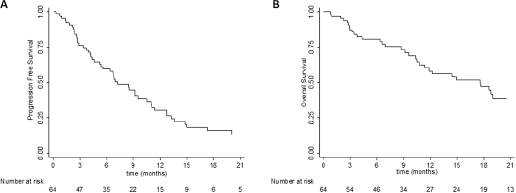
Progression-free survival. Kaplan Meier curves for all samples (
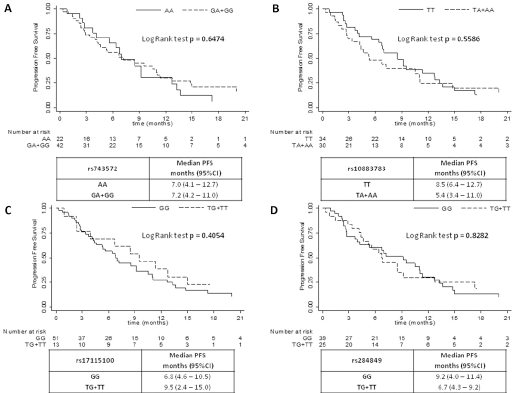
Progression-free survival curves for rs743572 (
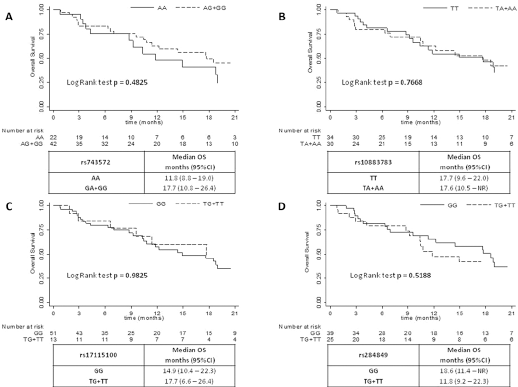
Overall survival curves for rs743572 (
In order to verify how the polymorphisms could affect the CYP17A1 protein expression, we also performed immunohistochemical analyses on a small series of 17 paraffin-embedded prostate cancer tissues. All patients (6/6, 100%) with the TA genotype for rs10883783 showed CYP17A1 expression on the tissue level, whereas only 6/11 (54%) patients with the TT genotype for the same polymorphism showed CYP17A1 expression (Supplementary Figure S1 - Immuhistochemical staining of CYP17A1 in prostate cancer tissues. A) protein expression in a patient with AT genotype for rs10883783; B) loss of protein expression in a patient with normal genotype for the same polymorphism. Available online at www.biological-markers.com). Given the small number of patients analyzed for CYP17A1 expression, it was not possible to obtain statistical evidence and the results must be confirmed in a larger case series.
Discussion
The genotyping of genes involved in CRPC carcinogenesis, cancer progression and drug metabolism could help us to better understand the behavior of CRPC and, consequently, patient outcome (15). Abiraterone showed impressive results, substantially increasing the PFS and OS of CRPC patients pretreated with docetaxel (14, 15). Several biomarkers related to tumor characteristics have been studied and have shown a predictive role in these patients, but to date no studies have investigated the effect of genotype characteristics on outcome prediction (16, 17).
The aim of the present paper was to evaluate the impact of 4 selected SNPs (rs10883783, rs17115100, rs284849 and rs743572) of the CYP17A1 gene on the clinical outcome of CRPC patients treated with abiraterone. With the limitation of the retrospective nature of the study and the relatively small sample size, our analysis revealed no associations between the polymorphisms and patients’ outcome, even if patients with the rs10883783 TA + AA genotype had a shorter PFS.
A single nucleotide variation in a gene may exert an effect on its expression level in different ways, e.g., by altering the splicing process (18, 19). One of the polymorphisms we analyzed (rs743572) is located at the 5’-UTR of CYP17A1 and can lead to promoter activity alterations. The others are intronic and may be involved in splicing mechanisms. Using a splicing motif predictor tool (Human Splicing Finder, http://www.umd.be/HSF3), rs10883783 would seem to be located in a branch point motif and may thus be involved in a variation in consensus sequences required for correct splicing with possible effects on gene expression. For this reason, we have evaluated CYP17A1 expression by immunohistochemistry in a small series of FFPE samples. We showed that the less common A allele for rs10883783 could be implicated in CYP17A1 protein expression changes, as all patients with the TA genotype for whom immunohistochemical results were available showed enzyme expression at the primary tissue level.
Specific tumor-related characteristics such as epigenetic modifications and genetic alterations are known to predict clinical outcome in patients treated with abiraterone (19). Thus, it would be interesting to focus on CYP17A1 gene polymorphisms in groups of CRPC patients subdivided on the basis of tumor-related characteristics in blood and/or tumor tissue. Abiraterone is currently being investigated in combination with other drugs including hormone therapies and inhibitors of the PI3K-AKT-mTOR signaling pathways, making the identification of genetic biomarkers increasingly important (20-21-22). The genetic characterization of CYP17A1 could facilitate our understanding of patient response/resistance to abiraterone therapy. In our case series of 64 treated patients no significant associations were found between the selected polymorphisms (rs743572, rs10883783, rs17115100, rs284849) and clinical outcome of CRPC treated with abiraterone.
Genetic polymorphisms of proteins involved in drug targeting and drug metabolism (e.g., CYP17A1) are likely to be the most important sources of individual variability in drug efficacy, but some drugs have a wide therapeutic window, defined as a wide interval between a drug dose having a therapeutic effect and causing an apparent adverse reaction (23). Leibowitz-Amit et al have recently shown that low doses of abiraterone (250 or 500 mg daily in spite of the standard 1,000 mg daily) have not been associated with poorer outcome in patients with metastatic CRPC treated with abiraterone (24).
Thus, the therapeutic window of abiraterone has been shown to be extremely wide. These observations may have implications even for the interpretation of the results of our study: it is plausible that the outcome of CRPC patients treated with abiraterone, due to its very wide therapeutic window, cannot be affected by CYP17A1 gene polymorphisms.
Moreover, we analyzed 4 selected SNPs (rs10883783, rs17115100, rs284849 and rs743572) with a high likelihood of being correlated with abiraterone activity (19), but other CYP17A1 polymorphisms could be studied as well, including rs6162, which was previously shown to be associated with plasma dehydroepiandrosterone-sulfate (DHEA-S) levels in prostate cancer patients on androgen deprivation therapy (25).
In addition, our study only has sufficient statistical power to detect very large clinical differences between genotype groups, especially after the different comparisons are taken into account.
In conclusion, this is the first study to explore the role of CYP17A1 polymorphisms in outcome prediction of abiraterone, a new hormonal therapy in CRPC patients. CYP17A1 is the direct target of abiraterone and may be the principal cause of therapy response. No associations were found, probably due to the drug's wide therapeutic window. However, we believe that the same SNP analysis could lead to different results in larger cohorts of patients including other ethnicities.
We analyzed only 4 CYP17A1 SNPs, but full-gene genotyping could undoubtedly help to identify other important alterations that may be involved in protein expression. Further studies of larger case series regarding the pharmacokinetics and pharmacodynamics of abiraterone are needed to better investigate the therapeutic window of the drug and the potential impact of SNPs.
Footnotes
Acknowledgments
The authors thank Grainne Tierney and Ursula Elbling for editorial assistance.
Financial support: No grants or funding have been received for this study.
Conflict of interest: The authors declare no conflict of interest.
