Abstract
Background
Abiraterone acetate is a second-generation androgen biosynthesis inhibitor widely used in the treatment of metastatic castration-resistant prostate cancer (mCRPC). However, resistance to abiraterone remains a major clinical challenge. Polymorphisms in genes involved in steroidogenesis, such as CYP17A1, may influence abiraterone therapeutic outcomes.
Methods
A systematic review of PubMed, Scopus, and ScienceDirect was conducted to examine the association between CYP17A1 polymorphisms and abiraterone treatment outcomes.
Results
Out of 518 articles initially identified through keyword search, five studies were eligible for qualitative analysis. Among them, three studies reported an association between polymorphisms rs2486758, rs743572 and abiraterone outcome—the associations are biochemical response, Progression-free survival (PFS), Overall survival (OS) and toxicity. Other polymorphisms, including rs4919685, rs17115100, rs743572, rs10883783, rs284849 did not show consistent associations.
Conclusion
Due to the heterogeneity in study designs, it would be difficult to state with conviction whether or not there is a statistically significant association between these polymorphisms and response to abiraterone. Larger prospective studies are needed. However, rs2486758 is the most consistently associated CYP17A1 variant with abiraterone response.
Keywords
Introduction
Prostate cancer is one of the most frequently diagnosed malignant tumors in men, with an estimated 1,467,854 new cases worldwide in 2022.1, 2 In the same year, there were approximately 397,430 deaths, 2 driven primarily by metastatic or recurrent disease. At this stage, the standard treatment is androgen deprivation therapy which provides effective control.3, 4
The mode of action of androgen deprivation therapy or chemical castration is to block the production of testosterone and other male hormones, preventing them from feeding the cancer cells, which can only be achieved by chemical or surgical castration.5, 6
The first-generation anti-androgens used for many years have proved effective against most tumors, but others progress to resistance within 18-24 months. 4 New or rescue processes are engaged by cancer cells, such as, amplifications and mutations on the androgen receptor (AR), amplifications on the steroid synthesis regulatory enzyme CYP17A1, which can activate de novo androgen synthesis.6–8 Given their dependence on the androgen receptor AR axis, new second-generation agents have been developed to target this axis and inhibit androgen production. These include inhibitors of the CYP17A1 enzyme, such as abiraterone.
The CYP17A1 enzyme, located in the endoplasmic reticulum, plays an important role in androgen biosynthesis. 9 Abiraterone, a second-generation anti-androgen, reduces the growth of castration-resistant metastatic prostate cancer (mCRPC) by inhibiting CYP17A1. 10 The aim of abiraterone suppression of CYP17A1 is to suppress intra-tumoral androgens plus inhibition of AR. 4
Nonetheless, abiraterone too does not seem to escape cases of resistance. Studies suggest that resistance to abiraterone may occur through mechanisms including upregulation of CYP17A1 and/or induction of variant AR splicing, which confers ligand-independent transactivation to the receptor 11 or a mutation of the androgen receptor. 12
However, the search for biomarkers useful for optimizing treatment with abiraterone could be an asset in establishing targeted therapy.
In this review, we aim to investigate the association between variations in CYP17A1 activity and response to abiraterone.
Methods
The search was conducted by accessing the PubMed, Science Direct and Scopus databases using keywords such as: Prostate cancer AND abiraterone efficacy, polymorphism CYP17A1 AND abiraterone, CYP17A1 AND Abiraterone. The search focused on articles published from inception to October 2024. The study excluded articles on neoplasms other than prostate cancer, as well as studies investigating the association of CYP17A1 polymorphisms in response to other treatment, and those reporting insufficient information, or unrealistic.
Results and Discussion
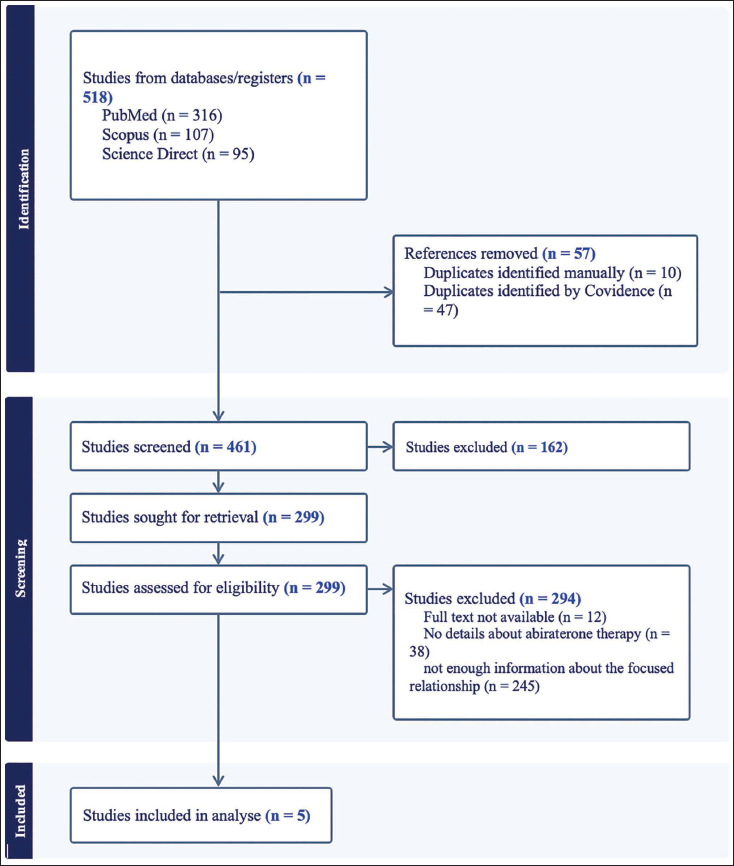
The systematic review followed the guidelines of the Preferred Reporting Items for Systematic Reviews and Meta-Analysis or Flow Diagrams. The systematic search conducted in PubMed, Scopus and Science Direct identified 518 studies overall. Among these, 57 duplicates were excluded. The titles and abstracts of the remaining 461 studies were examined, leading to the exclusion of 162 articles deemed irrelevant. This left 299 studies for further evaluation. In the assessment for eligibility (n = 299), 294 studies were excluded. Specifically, those studies were removed due to the absence of the complete text and for lacking pertinent information on the relationship between genetic polymorphisms and clinical outcome or about the focused therapy.
Consequently, 5 studies were included in the review, each meeting the defined eligibility criteria and providing comprehensive information on the focused relationship. The contents of these studies were subsequently analyzed and discussed in this academic article. The flow diagram is below in Figure 1.
Flow Diagram.
In castration-resistant prostate cancer (CRPC), cancer remains fueled by androgen production and signaling pathways.11, 13
This highlights the need for further development of new strategies to target resistance mechanisms.
CYP17A1
The CYP17A1 enzyme, encoded by a single gene on chromosome 10q24.3, is mainly expressed in the adrenal gland and in prostate tumor tissue. 14 Physiologically, CYP17A1 plays an important role in the process of maturation and sexual differentiation mediating the synthesis of circulating adrenal and intra-tumoral de novo androgens, that can activate the androgen receptor (AR) to promote prostate cancer growth despite clinical castration.15, 16
For androgen biosynthesis, CYP17A1 catalyzes two sequential reactions necessary for cancer proliferation: a 17α-hydroxylation reaction and a 17,20-lyase reaction in which the C17-C20 bond is cleaved, leading to the eventual production of potent androgens. 17 These two reactions contribute to the formation of DHEA (dehydroepiandrosterone) and androstenedione from 17α-hydroxypregnenolone and 17α-hydroxyprogesterone. DHEA and androstenedione are then converted into testosterone, 18 making it a key gene in the production of androgens and glucocorticoids. 19
The CYP17A1 gene has numerous single-nucleotide polymorphisms (SNPs). It has been suggested that some of these mutations have a functional impact, to varying degrees, and also influence patient survival. 20
In view of this, considerable interest and effort have been devoted to the discovery of compounds that regulate CYP17A1 activity, with the specific aim of finding drugs useful in the treatment of castration-resistant prostate cancer.
Abiraterone and CYP17A1 Polymorphism
Abiraterone acetate is the first drug to be developed from the class of second-generation androgen biosynthesis inhibitors, 21 administered in association with prednisone to prevent hypocorticoidism. 22 Abiraterone, developed from pregnenolone, a CYP17A1 substrate, blocks the 17,20 lyase region of the CYP17A1 enzyme, reducing circulating androgen levels and thus improving survival.13, 23 As a result, abiraterone enhances anti-tumor efficacy and provides a survival advantage for patients with castration-resistant prostate cancer.24, 25
Recommended as standard treatment for castration-resistant prostate cancer,25, 26 abiraterone confers an advantage on patients due to its effectiveness. This effectiveness has been observed in several studies, including a European study conducted in 4 countries, which was maintained by an increase in overall survival in medical practice after chemotherapy treatment. 27 Similarly, several other studies have been used to demonstrate this increase in survival.28–31 There is considerable evidence that CYP17A1 polymorphism is associated with risk and progression to prostate cancer.21, 32
However, the prognosis after abiraterone treatment varies from one individual to another. 33
Abiraterone resistance mechanisms have been associated with steroidogenesis, a systemic and intra-tumoral biosynthesis of androgens that is up-regulated. 34 A study by Mostaghel et al. reports that upregulation of CYP17A1 and induction of androgen receptor splice variants are mechanisms of resistance to abiraterone. 7
Five separate studies reporting a correlation between CYP17A1 polymorphisms and abiraterone outcomes are presented in Table 1. All patients with metastatic castration-resistant prostate cancer included in the studies received abiraterone acetate. Three studies reported a statistically significant association between CYP17A1 gene polymorphisms and abiraterone clinical outcome,3, 11, 20 while two studies found no significant association.35, 36 The polymorphisms (SNPs) that were analyzed in the 5 studies were essentially: rs2486758, rs4919685, rs17115100, rs743572, rs10883783, rs284849. Out of these studies, only the rs2486758 and rs743572 polymorphisms were associated with statistically significant results.3, 11, 20 In the Binder et al. cohort of 87 patients with castration-resistant prostate cancer, rs2486758 was associated with a lower risk of biochemical response and shorter time to biochemical progression on abiraterone/prednisone (AA/P), even after adjustment for multiple tests and potential confounding clinical variables. 3 This suggests a more than four-fold decrease in the odds of biochemical response to AA/P in patients harboring the rs2486758 minor allele. It should be highlighted that in this cohort, the rs2486758 variant was harbored in only 29 of 87 patients, or 33%. 3 While this aspect may have impacted the study’s quality, it is noteworthy that a key strength of this research lies in its prospective design, distinguishing it from others studies.
Summary Results of the Association Between CYP17A1 Polymorphisms and Outcomes of Prostate Cancer Patients Treated with Abiraterone.

Cruccita et al. in a retrospective study of 60 patients, showed a significant correlation between rs2486758 variant mutant genotype and shorter radiological progression-free survival (PFS). In contrast to the findings of Binder et al. in the multivariate analysis, prior docetaxel use was identified as an independent predictor of progression-free survival (PFS), indicating a potential limitation related to both the study design and sample size. 11 The prospective multicenter Abigene study confirms the interest in this variant, reporting a significant association with biological progression-free survival in multivariate analysis based on a larger population than the two previous studies. 20 However, with regard to the association between the rs743572 variant and abiraterone toxicity, this study contradicts the two other studies that did not exhibit any associations and that of Binder et al. which reported no association between this variant and response to abiraterone.3, 36, 35 In the case of Salvi et al., patients with the TT genotype of rs10883783 showed a longer progression-free survival than the AA and AT genotypes. 36 Wright et al., without distinguishing between treatments, reported a 56% reduction in prostate cancer specific mortality in carriers of the A allele of the same CYP17A1 variant. 37
The impact of CYP17A1 polymorphism on abiraterone treatment appears to be controversial. The absence of correlation observed in certain studies may be attributable to several factors. These include abiraterone’s broad therapeutic window, evidenced by the finding that low-dose abiraterone acetate does not necessarily correlate with poor outcomes in patients with castration-resistant prostate cancer as well as limitations such as small sample sizes within these studies. However, CYP17A1 remains a significant potential biomarker, knowing that genome-based predictors for abiraterone—like serum CYP17A1 copy number variations (CNV)—may indicate early abiraterone resistance and poorer PFS and OS. 38 In addition, one of the polymorphisms analyzed by Salvi et al. (rs743572) is located in the 5’-UTR region of CYP17A1 and may lead to alterations in promoter activity. 36 In an earlier study, carriers of the A2 allele of this variant had significantly longer survival than those expressing the A1 allele in prostate cancer patients who had not necessarily received abiratone. 39 The others, intronic, may be involved in splicing mechanisms.
It would also be interesting to study other CYP17A1 polymorphisms, such as rs6162 or rs6163 in the Abigene study. 20
Conclusion
Among the CYP17A1 polymorphisms investigated, rs2486758 shows the strongest evidence as a predictive biomarker for abiraterone efficacy. Larger prospective studies are needed to validate these findings. Incorporating pharmacogenomic testing into routine clinical practice could ultimately guide treatment decisions and improve patient outcomes.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Patient Consent
This study is based on the analysis of many others studies or previous research without the direct involvement of human participants. Therefore, it does not require approval or consent.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
