Abstract
Purpose
HPV-related locally advanced head and neck cancers (LA-HNCs) show a good prognosis. This study aimed to investigate the HPV prevalence in LA-HNCs and compare the prognostic value of E1, E6 and L1 genomic viral fragments and p16, individually and in combination, in order to find the best prognosticator in terms of overall survival (OS) and progression-free survival (PFS).
Patients and Methods
HPV16 was searched in 255 LA-HNC formalin-fixed paraffin-embedded tumor tissues, 89 oropharyngeal cancers (OPCs), and 166 non-OPCs by DNA-PCR with 3 primer pairs. p16 was analyzed by immunohistochemistry in 235 patients.
Results
The prevalence of positive samples decreased constantly from E6 to L1 and E1 in both OPCs and non-OPCs. Each LA-HNC patient highlighted variable positivity for each fragment. OPCs showed a higher prevalence of positive samples compared to non-OPCs.
Positive coexistence of all the fragments was more common in OPCs (31.5%) than non-OPCs (4.2%), and E1 detection was always associated with E6 and L1. E1-positive OPCs showed improved OS (p = 0.012) and PFS (p = 0.036), while L1- or E6-positive ones did not. p16-positive patients were more prevalent in the OPC (29.8%) than the non-OPC group (7.3%) (p<0.0001) and its prognostic value was not superior to that of E1. However, the multivariate Cox analysis which included E1, L1, E6 status and p16 expression did not show a significant p value.
Conclusions
Though HPV16 positivity measured by DNA-PCR was higher for L1 and E6, they performed weakly as prognosticators; E1 might become a strong prognostic marker for OS and PFS in OPCs.
Introduction
Head and neck (HN) cancer is the sixth most common cancer worldwide (1). Tobacco and alcohol abuse remain the strongest established risk factors, but accumulating data indicate that oncogenic HPVs, particularly HPV16 (2, 3), are associated with squamous cell lesions of HN (4). The oropharynx is the favorite HPV infection site (5) and it is widely accepted that HPV-positive patients show a better survival than HPV-negative ones (6), with the depiction of a distinct tumor entity (7).
The strong relationship between HPV and oropharynx cancers (OPCs) is not so evident in other HN squamous cell carcinoma (SCC) primary sites (8, 9). We included as OPCs tumors originating from the tonsil, base of the tongue and soft palate. Non-OPCs included carcinoma of the hypopharynx (piriform sinus), larynx (glottis, vocal cords), oral cavity (tongue, floor of mouth, palate and cheek mucosa) and nasopharynx (nasal cavity).
A prerequisite for the development of lesions and invasive cancers is persistent infection, which is generally associated, albeit not obligatorily, to integration of the HPV16 genome in the host chromosome (10-11-12-13). Integration leads usually to disruption of the circular genome in the E1 or E2 open reading frames (14), inducing transcriptional activation of the oncogenic viral proteins E6 and E7 (15) and interference of viral DNA replication control (16-17-18-19). Nevertheless, other studies have proposed that the HPV16 genome is integrated within the host chromosome through disruption of the L1 and L2 genes as well (16, 20, 21). However, limited information about HPV integration in HNSCC is available (22).
The HPV detection rate varies based on the technique used for identification and on the viral life cycle. Polymerase chain reaction (PCR) is a very sensitive test for HPV identification (23), and though it is the most feasible, available and cost-effective method, its main disadvantage is its limited ability to distinguish clinically relevant HPV infections (24). In this sense, E6/E7 mRNA quantification in frozen specimens is considered the gold-standard test for relevant infections, but it does not seem generally feasible in retrospective clinical studies, where testing is usually performed on formalin-fixed paraffin-embedded (FFPE) archival tissue samples (24).
In clinical trials, the combination of immunohistochemical staining (IHC) for p16, a surrogate marker for the transforming activity of high-risk HPVs, together with HPV in situ hybridization (ISH), is a very specific and sensitive approach (24, 25). However, ISH is not a standard procedure since the commercially available tests are poorly reliable and the reliable in-house modified tests are unlikely to be extendable to routine settings (26).
Therefore a single, adequate marker to identify HPV-induced HNSCC is still lacking and the expression of p16, when used in combination with HPV DNA detection or HPV-specific antibodies, could be of importance in standardized testing (27, 28).
In this study we investigated the prevalence of HPV16 by DNA-PCR in a series of locally advanced (LA) HNCs and compared the prognostic value of E1, E6 and L1 genomic viral fragments with each other and with p16 expression in order to find the best prognosticator for overall survival (OS) and progression-free survival (PFS). Moreover, we evaluated these viral markers in non-OPCs to establish also a possible role of HPV outside OPCs.
Patients and Methods
Patients and Therapy
We retrospectively collected a series of 297 patients with histologically confirmed LA-HNC stage III-IV who had been treated with curative intent by chemoradiation (CRT) between 1997 and 2013 at the S. Croce & Carle Teaching Hospital, Cuneo, Italy. To identify LA-HNC we extracted categories C00-C14 and C30-C32 included in the 10th edition of the International Classification of Diseases and Related Health Problems (ICD-10) of the World Health Organization (WHO). Informed consent for tissue collection and use for scientific purposes was obtained from each patient enrolled in the study. The study was conducted according to the guidelines of the Helsinki Declaration.
All patients underwent the same combined treatment consisting of radiotherapy alternated with chemotherapy regimens according to our center's experience (29). SCC of the maxillary sinus and undifferentiated carcinoma of the nasopharyngeal type (WHO types II and III) were excluded.
HPV Detection by PCR, IHC and ISH
DNA samples were extracted from FFPE tissues at diagnosis by an in-house standard procedure (30) that included pretreatment with xylene and ethanol to physically remove the paraffin wax and proteinase K employment in the lysis buffer (EuroClone).
HPV fragments were detected by DNA-PCR, using 3 specific primer pairs for type 16. The forward primers for L1 and E6 were designed using the consensus degenerated ones (My11 and WD76, respectively) as references (31). The reverse primers were constructed for each fragment in order to get smaller fragments from FFPE tissues (80 bp for E6 and 97 bp for L1). E1 primers were designed inside the coding region, most frequently disrupted during viral integration in the host chromosome (14, 32), in order to get a fragment of 168 bp.
IHC for p16 was performed on 4-micron sections from the 235 still available FFPE tissue blocks and positivity was defined as ≥70% positive cells. ISH for HPV DNA was performed in FFPE tissue sections from 29 OPCs.
(Detailed information on methods and statistical analyses is reported in the Supplementary Methods. Available online at www.biological-markers.com.)
Results
Demographic and Clinicopathological Characteristics
Tissues from 297 LA-HNC patients of Caucasian origin were available for this study. Forty-two patients were excluded because of poor quality tissue specimens, lack of PCR amplification in both HPV16 and housekeeping genes, or missing information in the patient's clinical history. The remaining 255 LA-HNC patients were evaluable for HPV detection by PCR and had a median follow-up of 25 months (range 1-206). Patients’ age at treatment start ranged from 21 to 86 years, with a median of 61 years. Eighty-nine (34.9%) LA-HNCs arose in the oropharynx and 166 (65.1%) in non-oropharyngeal sites. The baseline patient and tumor characteristics are shown in Table I.
Characteristics of LA-HNC patients (n = 255) and tumors
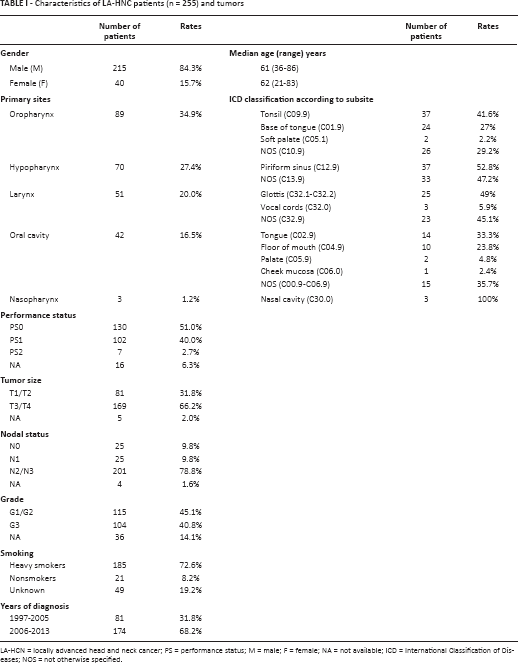
LA-HCN = locally advanced head and neck cancer; PS = performance status; M = male; F = female; NA = not available; ICD = International Classification of Diseases; NOS = not otherwise specified.
Overall, no significant differences were observed in baseline clinical and pathological characteristics including performance status (PS), tumor size (T), histological grade (G) and smoking habit among all the primary tumor sites, while for nodal status (N) a higher percentage of negative lymph nodes was observed in larynx tumors (N0 = 29.4%) compared with the other sites (p<0.0001). Regarding gender, though there was a higher overall proportion of men than women, the prevalence of women among patients with cancer of the oropharynx and oral cavity was higher than among patients with larynx and hypopharynx as the primary sites (p = 0.004) (Supplementary Table S1 – Characteristics of patients and tumors according to primary tumor sites. Available online at www.biological-markers.com).
Data on smoking showed that 72.6% of patients were smokers (185/255) and men were both more frequent (76.7% vs. 50.0%, p<0.0001) and heavier smokers than women (median of 35 pack-years vs. 28 pack-years, p = 0.012) (Supplementary Table S2 – Characteristics of patients and tumors according to gender: 215 males and 40 females, in the whole cohort of LA-HNCs. Available online at www.biological-markers.com).
HPV Prevalence in LA-HNC Patients and Correlation with Tumor Characteristics
Overall, 136 of 255 patients (53.3%) were HPV16 positive with at least one of the primer pairs used. An increasing number of positive samples was detected from E1 (35/255; 13.7%) to L1 (76/255; 29.8%) and E6 (130/255; 51.0%) (Tab. II). Each positive patient therefore displayed variable positivity for E1, L1 and E6 viral fragments. Interestingly, E1 detection was always linked to E6 and L1 positivity.
Comparison of the power of detection between E1, E6 and L1 viral fragments and p16 expression in OPC and non-OPC patients
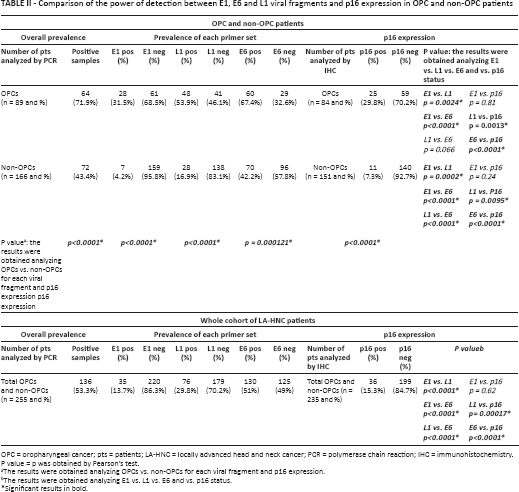
OPC = oropharyngeal cancer; pts = patients; LA-HNC = locally advanced head and neck cancer; PCR = polymerase chain reaction; IHC = immunohistochemistry.
P value = p was obtained by Pearson's test.
The results were obtained analyzing OPCs vs. non-OPCs for each viral fragment and p16 expression.
The results were obtained analyzing E1 vs. L1 vs. E6 and vs. p16 status.
Significant results in bold.
Within the group of 136 HPV16-positive patients we identified 2 subgroups with different viral positivity: 35 patients were simultaneously positive for all 3 viral fragments (E1+L1+E6+), while 101 patients were positive only for 2 viral fragments (L1+ and E6+) or at least 1 viral fragment (L1+ and E6- or L1- and E6+) and never for E1 (identified as E1- and/or L1+E6+). One hundred nineteen patients (46.7%) were HPV16 negative by all primer pairs (E1-L1-E6-).
E1+L1+E6+ tumors were more common in patients with a better PS, in patients who had never smoked, and in those who had a good prognosis when compared to E1- and/or L1+E6+ or E1-L1-E6- tumors [Supplementary Table S3 – Clinical and pathological characteristics of patients defined as (A) simultaneously positive for the 3 viral fragments (E1+L1+E6+), (B) positive only for 2 (L1+E6+) and/or at least 1 viral fragment (L1+E6- or L1-E6+) and never for E1 (E1- and/or L1+E6+); (C) negative for the 3 viral fragments (E1-L1-E6-). Available online at www.biological-markers.com].
HPV Prevalence in OPC and non-OPC Patients and Correlation with Tumor Characteristics
Tumors were grouped into OPCs (n = 89) and non-OPCs (n = 166). Overall, OPCs showed a higher HPV16 prevalence (64/89; 71.9%) than non-OPCs (72/166; 43.4%) (p<0.0001). Moreover, the positive sample proportions for each fragment analyzed were maintained in both groups, with the highest percentage for E6 and the lowest for E1 (Tab. II).
OPC patients showed also a higher prevalence of simultaneous positivity for the 3 viral fragments studied (E1+L1+E6+ = 28/89; 31.5%) and a lower prevalence of E1-L1-E6- tumors (25/89; 28.1%) than non-OPC patients (7/166; 4.2% for E1+L1+E6 and 94/166; 56.6% for E1-L1-E6-) (p<0.0001), while there was no difference in E1- and/or L1+E6+ tumors in the 2 groups (36/89; 40.4% for OPCs and 65/166; 39.2% for non-OPCs; p = 0.84) (Fig. 1). Positivity distributions in OPCs and non-OPCs site by site are depicted in Supplementary Figure S1 - Distribution of positive fragments in OPC and non-OPC patients. Available online at www.biological-markers.com. Among OPC patients, E1+L1+E6+ tumors were more common in patients with a better PS and nonsmokers, while E1- and/or L1+E6+ tumors were more frequent in patients with the worst prognosis.
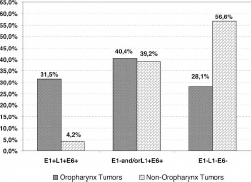
Distribution of combined positive viral fragments by DNA-PCR in OPC and non-OPC patients. Histograms show a higher rate of combined positive fragments (E1+L1+E6+) in OPC patients (28/89; 31.5%) than non-OPC patients (7/166; 4.2%) and a lower rate of combined negative fragments (E1-L1-E6- or HPV16-) in OPC patients (25/89; 28.1%) than non-OPC patients (94/166; 56.6%). No difference was observed for combined variable fragments (E1- and/or L1+E6+) between the 2 groups (36/89; 40.4% for OPCs and 65/166; 39.2% for non-OPCs).
In non-OPC patients we did not find any difference in the clinical and pathological characteristics between E1+L1+E6+ and E1-L1-E6- tumors, but heavy smokers (81.5%) were the most prevalent in E1- and/or L1+E6+ tumors (p = 0.014) (Supplementary Table S4 – Characteristics of patients and tumors according to site and viral status. Available online at www.biological-markers.com).
Survival Analysis in OPC and non-OPC Patients
To determine whether HPV16 detection by DNA-PCR, found with variable degrees of positivity, had prognostic significance, we analyzed the cohort of patients in terms of OS and PFS. OPC patients having E1+L1+E6+ tumors had the best OS (p = 0.037) and PFS (p = 0.056), with the median not reached in both cases, while those having E1- and/or L1+E6+ tumors had the worst survival figures (median 20 and 27 months for OS and PFS, respectively). Moreover, E1-L1-E6- patients had shorter OS and PFS (median 24 and 102 months for OS and PFS, respectively) than E1+L1+E6+ patients but longer than E1- and/or L1+E6+ patients (Fig. 2A and 2B).
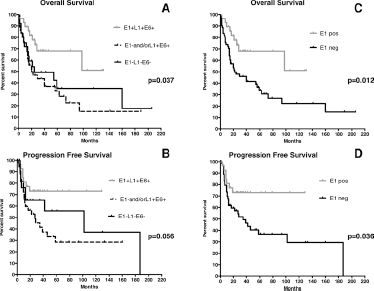
Kaplan-Meier curves in OPC patients (n = 89) according to tumor HPV status defined by E1, taken in combination with L1 and E6 for OS (
Therefore, since E1 detection was always associated with L1 and E6, its positivity alone might be sufficient to correlate with increased OS (p = 0.012) and PFS (p = 0.036) (Fig. 2C and 2D). By contrast, when L1 and E6 fragments were assessed individually using Kaplan-Meier analysis, we found that positive patients for both fragments had longer OS than negative patients, but neither L1 nor E6 gave any significant gain in survival.
Among non-OPC patients we detected only few fully HPV16-positive cases (E1+L1+E6+), most likely not sufficiently powered to detect a survival advantage. However, the 7 non-OPC E1+L1+E6+ patients performed better in both OS (median 77 months) and PFS (median not reached) than E1- and/or L1+E6+ and E1-L1-E6- patients (32 vs. 36 months for OS; 24 vs. 36 months for PFS, respectively; p = NS). When each fragment was assessed individually, neither E1 nor L1 or E6 reached any significant difference in OS and PFS.
Comparison of DNA-PCR and p16 IHC Methods
Two hundred and thirty-five out of 255 LA-HNC patients were tested for p16 by IHC. Overall, 36 LA-HNC patients were found positive for p16 (15.3%), which indicated a stronger association in OPCs (25/84; 29.8%) than non-OPCs (11/151; 7.3%) (p<0.0001).
The concordance between p16 detected by IHC and E1 detected by DNA-PCR was high since the HPV power of detection was similar, although positive samples did not always overlap. Significant differences were instead evident when L1 or E6 was matched with p16 both in OPC and non-OPC patients (Tab. II and Supplementary Table S5 – Combinations of results obtained from DNA-PCR for E1, L1 and E6 viral fragments and p16 expression by IHC. Available online at www.biological-markers.com).
When the p16 by IHC and DNA-PCR results were matched to assess their association with OS and PFS in OPCs, we grouped patients according to their HPV DNA-PCR and outcome. Therefore E1+L1+E6+ patients were labeled as E1pos, while E1-negative patients, irrespective of their L1 and E6 positivity, or E1-L1-E6- were designated as E1neg. Twenty-one tumors were E1pos/p16pos (25.0%), 55 were E1neg/p16neg (65.4%), 4 were E1pos/p16neg (4.8%), and 4 were E1neg/p16pos (4.8%), with a concordance rate between the 2 methods of 90.5% (76/84).
In OPCs, positivity for p16 by IHC, both individually (Fig. 3A and 3B) and in combination with positive E1 status (Fig. 3C and 3D), was associated with the most favorable OS and PFS. Moreover, patients with discordant results (8/84; 9.5%) and those negative for both methods had the worst survival, with no substantial differences between the latter 2 strata (Fig. 3C and 3D). When the 2 markers were evaluated individually also for L1/p16 and E6/p16 combinations, only the L1/p16-positive group was associated with better OS but not PFS, while the E6/p16 combination did not provide any improved OS or PFS [Supplementary Figure 2A – Kaplan-Meier curves in OPC patients (n = 84) according to p16 by IHC, taken in combination with L1 and E6 status for OS, and supplementary Figure 2B – Kaplan-Meier curves in OPC patients (n = 84) according to p16 by IHC, taken in combination with L1 and E6 status for PFS. Available online at www.biological-markers.com].
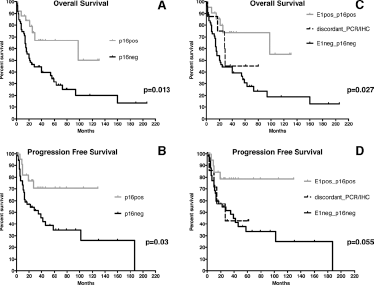
Kaplan-Meier curves in OPC patients (n = 84) according to p16 status determined by IHC taken individually for OS (
Afterwards, in order to define the best prognostic marker in OPC patients, in whom we found the best concordance between PCR and IHC for HPV16 detection, we performed a multivariate Cox analysis in which we included E1, L1, E6 status and p16 expression, but the cumulative p value was not significant (p = 0.061) (Fig. 4).
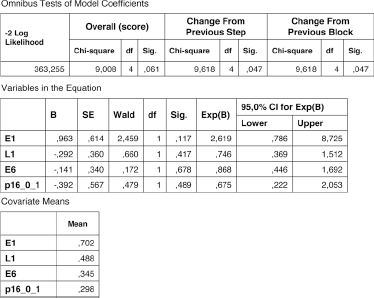
Multivariate Cox analysis showed no cumulative value. p16_0_1: positive samples (1) with ≥70% of positive cells.
Concordance between HPV PCR and ISH/IHC
All ISH-positive samples (14/29=48.3%) were also PCR positive, showing episomal and integrated HPV forms. In particular, the sole integrated form (n = 5) was found in 3 E1+L1+E6+ and in 2 E1- and/or L1+E6+ samples, while the accumulation of episomal sequences mixed with the integrated genome (n = 9) were always associated with E1+L1+E6+ samples [Supplementary Figure S3 – (A) Representative PCR results performed with the 3 couples of HPV primers (E1, L1 and E6) and (B) with primers for a housekeeping gene (GAPDH) in #1-5 OPC patients. Available online at www.biological-markers.com]. In contrast, 13 cases were negative with ISH but HPV positive with PCR for L1 and/or E6 fragments and never for E1 (E1- and/or L1+E6+) and only 2 cases were negative with ISH and PCR (E1-L1-E6-).
In order to verify any differences between DNA-PCR for E1, L1 and E6 fragments taken individually, and the combination of IHC with ISH, considering patients as HPV positive when at least 1 test was positive (concordance 82.8%), we matched the data. Crude concordance between ISH/IHC and DNA-PCR showed declining agreement from E1 (85.7%; kappa = 0.72) to L1 (64.3%; kappa = 0.25) and E6 (53.6%; kappa = 0.01). No difference in HPV detection was found between E1 by PCR and the ISH/IHC combination (p = 0.62), while McNemar's test indicated significant differences when we matched L1 (p = 0.02) or E6 (p = 0.002) with ISH/IHC.
Discussion
This study showed a different HPV16 detection rate in LA-HNCs by DNA-PCR using 3 specific primer pairs targeting relevant viral sequences involved in oncogenesis (E6), capsidation (L1), viral replication and episomal maintenance (E1) (16, 31, 32). In our hands E6 showed the highest HPV16 prevalence (51.0%) compared to L1 (29.8%) and E1 (13.7%). This might be due to the generally retained intact E6 sequence expressed in tumors (31, 33, 34). The E6 gene may be present in both episomal and integrated forms, playing a key role in malignant transformation (15), or may remain just as a persistent and harmless DNA fragment after a resolved infection (16, 35). Integration usually hits other, less attractive targets at different sites in E1, E2, L1 and L2 regions (10, 14, 20, 21, 36) and potentially reduces the sensitivity in HPV detection (37).
However, while disruption in the L1 gene could not be critical, as capsid-encoding sequences are not necessary for the presumed tumorigenic functions (38, 39), disruption events within the E1 and E2 genes have been strongly associated with integration of HPV16 DNA into the host chromosome, immortalization of primary human keratinocytes (40), and progression to cancer (10-11-12-13-14).
Today, although studies focusing on the role of the E1 gene as a prognosticator in HNC are limited (8, 27), there is some evidence that alterations in the E1/E2 regions, mainly E1 disruption (14), are associated with poor prognosis as well as a low disease-free survival rate in cervical cancer (41-42-43-44-45). Increased disease-free survival was reported, instead, in patients with relevant viral episomal patterns, an intact E1/E2 open reading frame, and high HPV16 load (41-42-43), also in tonsillar cancers (46).
Although integration breakpoints of both the viral and cellular genome are different in all samples (21), the most frequent site of disruption within the E1 gene has been identified between nucleotides 1059 and 1323 (14), in which we performed our PCR analysis. This could partly explain the low E1 prevalence, otherwise due to an insufficient viral load. However, since no integration assay and no viral load quantification were performed, we cannot state that E1-positive patients do not have another break at a different point of the viral genome or that E1 negativity indicates only a low viral load condition. These aspects lie beyond the aim of this work, which found that E1 detection was always linked to E6 and L1 positivity, and strengthens its value in predicting the prognosis in OPC patients, as p16 detection by IHC did.
We are aware that the use of more than 1 set of primers might increase the overall prevalence, since we considered as HPV16 positive those patients with at least 1 viral fragment detected in their tumor tissue. In our study we actually found an overall prevalence of 53.3%, and a stronger association with OPC patients (71.9%) than non-OPC patients (43.4%), as had already been reported (7, 47-48-49); we also observed a higher detection rate of E6 compared to the other fragments (L1 and E1) in both OPCs and non-OPCs. However, the use of multiple primers seems not to be informative in predicting prognosis.
Indeed, survival analysis showed that only the E1-positive OPC patients had longer OS (p = 0.012) and PFS (p = 0.036) than E1-negative patients, while no other tumor conditions, associated with viral fragment and/or status (E1 in non-OPC, L1 and E6 in OPC and non-OPC), taken individually or in combination, correlated with a significant difference in survival.
Moreover, although the multivariate Cox analysis failed, most likely due to the small number of OPC samples, we could establish through Kaplan-Meier analysis that within the HPV16-positive patients (as defined in Fig. 2) E1-positive patients showed significantly better OS than E1-negative ones.
Analyzing p16, we found that positivity was more frequent in OPCs, with 29.8% of p16-positive samples versus 7.3% in non-OPCs (p<0.0001).
We observed good concordance between p16 and E1 in the whole population as well as in OPC and non-OPC patients separately. This was not the case when we compared p16 IHC either with E6 or with L1. This supports the use of p16 as a reliable surrogate marker of HPV positivity in OPCs.
Of course, when the use of a new marker is suggested to perform better, or at least to be as reliable as an existing standard, it seems mandatory to compare the techniques. Thus, we compared DNA-PCR with IHC/ISH, since the combination of sensitive (IHC) and specific (ISH) tests is reported to have the best potential in establishing HPV status (24, 25).
Our study on a pilot cohort of 29 OPC patients underlined that HPV16 detection by E1 primers gives equivalent results to the combination of IHC/ISH in FFPE tissues. This was not the case when L1 and E6 primer sets were used, which have much higher sensitivity than ISH.
Although the small number of OPC patients evaluated by ISH did not allow us to perform any statistical analysis, the strong association of E1-positive samples with accumulation of episomal sequences related to a better prognosis in HNC (46) might suggest that E1 selects patients with the best prognosis among the HPV-positive ones. However, further studies on a larger number of LA-HNC patients would be worth considering for in-depth comparison between ISH, whose results are often influenced by subjective interpretation (24, 50), and E1 by DNA-PCR.
Outside the oropharynx, we cannot establish a clear relationship between HPV positivity and clinical outcome, as we were not able to analyze the other primary sites singularly. We are aware that the lack of analysis by site may be a weakness of this study, but the small numbers of p16-positive tumors ruled out any convincing subgroup analysis.
In conclusion, E1 detection by DNA-PCR in FFPE archival tissues is of clear importance in predicting survival and might represent, if validated on a larger number of patients with a proper follow-up and survival analysis, a clinically useful marker for the identification of OPC patients who have a better prognosis and may be candidates for deintensified treatments. Future studies on non-OPC patients should be powered to address the clinical prognostic value of HPV status, analyzing if possible each non-OPC primary site.
Footnotes
Acknowledgment
We thank Professor Jan B. Vermorken from the Department of Medical Oncology, Antwerp University Hospital, Edegem, Belgium for helpful discussions. We are grateful to Fondazione Veronesi that granted D. Vivenza and M. Monteverde in 2014 with Post-Doctoral Fellowship 12 months, supporting the progression of this work.
Abbreviations
Financial support: This work has been partially supported by AIRC (Associazione Italiana per la Ricerca sul Cancro) (to C. Lo Nigro and M.C. Merlano) and by an institutional grant from the Fondazione IRCSS Istituto Nazionale Tumori Milano “Validation of a new algorithm for HPV status assessment in head and neck carcinoma” (to A. Gloghini and C.C. Volpi).
Conflict of interest: None of the authors has any financial interest related to this study to disclose.
