Abstract
Objective
This study aimed to validate new staging of Japanese patients with human papillomavirus-mediated oropharyngeal cancer (HPV+-OPC) and the effectiveness of transoral surgery.
Methods
Fifty-three patients with HPV+-OPC were classified according to the seventh and eighth editions of the American Joint Committee on Cancer (AJCC) staging, along with assessment of prognosis, smoking and alcohol status, and treatment outcome. Transoral videolaryngoscopic surgery (TOVS) was performed as transoral surgery.
Results
The 5-year overall survival (OS) rate was 77.3% in all patients. Five-year OS rates of seventh edition staging were as follows: II, 100%; III, 100%; IVa, 75.9%; and IVb and IVc, 0%. Five-year OS rates of the eighth edition clinical staging were as follows: I, 89.3%; II, 88.9%; III, 46.3%; and IV, 0%. In non-smoking and smoking patients with HPV+-OPC, the 5-year OS rates were 90.9% and 70.7%, respectively. TOVS was used for treating 20 smokers with HPV+-OPC (5-year OS: 85.7%), while 12 were treated with chemoradiotherapy (5-year OS: 75.0%).
Conclusions
The eighth edition of the AJCC determines the prognosis of HPV+-OPC more accurately than the seventh edition in clinical staging. Smokers with HPV+-OPC have a poor prognosis, but TOVS might improve the prognosis.
Keywords
Introduction
The rate of human papillomavirus-mediated oropharyngeal cancer (HPV+-OPC) has increased in the past 10 years. 1 HPV+-OPC has different oncogenic mechanisms from HPV-negative, tobacco-related, and alcohol-related oropharyngeal cancer (OPC), and conventional staging is inadequate to predict prognosis of HPV+-OPC.1–3 The eighth edition of the American Joint Committee on Cancer (AJCC) cancer staging manual was published in 2016 in which the staging of HPV+-OPC was newly established.4,5 Several studies have shown that the eighth edition of the AJCC is adequate for predicting prognosis of HPV+-OPC in Japanese patients,6–8 but only a few studies have evaluated the pathological staging and outcome of treatment. Several studies have shown that there is a higher rate of smokers in Japanese patients with HPV+-OPC than in other countries, and the positivity rate of HPV+ among OPC is lower.6–8 Because of the different backgrounds, Japanese patients with HPV+-OPC may have a different prognosis or outcomes of treatment.
Patients with HPV+-OPC respond better than human papillomavirus (HPV)-negative patients to the current treatment modalities. Therefore, the former group of patients have a more favorable prognosis than the latter group.6,9 However, several studies have shown that smoking continues to pose a great risk for cancer progression and mortality, even in HPV+-OPC.10–13 Another important issue in the treatment of patients with HPV+-OPC is the effect of the treatment methods. Several researchers have investigated the contribution of various treatment methods, and some reports have shown the value of transoral surgery, regardless of poor prognostic factors. 12
The present study aimed to assess the validity of the new staging for HPV+-OPC in our hospital. We also aimed to assess the value of transoral surgery in the treatment of patients with HPV+-OPC, especially for smokers with a poor prognosis.
Material and methods
Patients
This study was conducted after approval of the National Defense Medical College Ethical Committee (No. 2013). A retrospective study was performed on 123 patients with OPC at the National Defense Medical College Hospital between January 2005 and December 2016. The HPV status was defined by p16 immunostaining. We prospectively followed up patients for 3–115 months from the time of diagnosis (median follow-up time: 34.5 months). HPV+-OPC was classified according to both of the seventh and eighth editions of the AJCC staging, and the prognosis was assessed. The tobacco consumption status was defined categorically as never (non-smokers with HPV+-OPC) or as current/prior users of cigarettes (smokers with HPV+-OPC). The amount of smoking was assessed by the Brinkman Index (BI). The alcohol status was defined as never and rarely (non-drinkers with HPV+-OPC) or as current/prior (drinkers with HPV+-OPC). Finally, the outcome of the investigation was evaluated by the overall survival (OS) rate and disease-specific survival (DSS).
Treatment options
After explaining the treatment options for surgery and chemoradiotherapy (CRT), informed consent for treatment was obtained from the patients. The radiation schedules were 60–70 Gy in 30–35 fractions over 6 weeks. Concomitant CRT was scheduled every 3 weeks when cisplatin (80 mg/m2) was administered or every week when docetaxel (10 mg/m2) was administered. The general indications for surgery were as follows: T1, T2, selected T3, restricted rT1 and rT2 stage cancers for transoral surgery, and T3 and T4 stages for open surgery. Node-positive patients were also included if the tumors were resectable by neck dissection. Our procedure for transoral surgery was transoral videolaryngoscopic surgery (TOVS). TOVS is an original transoral surgical setting that was established by the authors and it uses almost the same procedure as transoral robotic surgery using an endoscope and surgical forceps for laparoscopy.14–16 Patients with stages higher than N2b in the seventh edition, and/or with a positive/close margin had postoperative radiation therapy performed.
Immunohistochemistry
HPV status was evaluated by p16 immunostaining of paraffin-embedded tissue samples. Anti-p16 (sc-56330; Santa Cruz Biotechnology Inc., Santa Cruz, CA, USA) was used as the primary antibody and the VECTASTAIN Elite ABC kit (PK-6100; Vector Laboratories Inc., Burlingame, CA, USA) was used as the secondary antibody. ImmPACT DAB (SK-4105; Vector Laboratories Inc.) was used as the color developing agent. The definition of p16-positive was staining of >70% of the cancer nucleus or cytoplasm, and this was judged by two double-blinded doctors.
Statistical analysis
OS and DSS were calculated using the Kaplan–Meier method using the log-rank test for comparison of OS and DSS. The chi-square or Fisher’s exact test was used to determine significant differences in discrete variables between the groups. Statistical significance was set at a value of p < 0.05 for all tests. JMP statistical package (SAS Institute Inc. Cary, NC, USA) version 13 was used.
Results
HPV and other variables of patients
There were 53 patients with HPV+-OPC among 123 patients (45 men and 8 women) with OPC, and the prevalence of p16 was 43.1%. The number of patients with HPV+-OPC in each clinical stage of the seventh edition was as follows: I, 0; II, 7, III, 5; IVa, 33; IVb, 7; and IVc, 1. The number of these patients in each clinical stage of the eighth edition was as follows: I, 25; II, 13; III, 14; and IV, 1. We could assess the pathological staging for 32 HPV+-OPC cases. In the seventh edition, there were many N2 patients (38/53; 71.7%), and most of the patients were classified as stage IVa, whereas the eighth edition showed an even distribution in each stage (Table 1). The frequencies of the affected sub-sites are shown in Table 2.
Number of patients in each stage in HPV+-OPC.

HPV+-OPC: human papillomavirus-mediated oropharyngeal cancer; AJCC: American Joint Committee on Cancer.
Patients’ characteristics.

TOVS: transoral videolaryngoscopic surgery.
The performance status of all patients in this study was 0 or 1. No patients showed findings of carotid artery invasion and/or cranial nerve paralysis, including the vagal nerve, accessory nerves, or hypoglossal nerve. Twelve patients were evaluated as having positive extra-nodal extension by physical, imaging, or pathological findings. The percentage of extra-nodal extension was 20.0% (4/20) in the CRT group, 24.2% (8/33) in the surgery group, and 20.6% (6/29) in the TOVS group.
There were 51 patients whose smoking status was known, including 34 (64.2%) smokers with HPV+-OPC and 17 (32.1%) non-smokers with HPV+-OPC (Table 2). The number of smokers with HPV+-OPC in each clinical and pathological stage of the seventh and eighth editions is shown in Table 3. There were no significant differences in the clinical and pathological stages between smokers and non-smokers with HPV+-OPC. There were 52 patients whose alcohol status was known, including 43 (81.1%) drinkers with HPV+-OPC and only nine (17.0%) non-drinkers with HPV+-OPC (Table 2). As a result of the few non-drinker with HPV+-OPC, we did not assess the relationship between alcohol consumption and prognosis.
Number of patients in each stage of smokers versus non-smokers with HPV+-OPC.

HPV+-OPC: human papillomavirus-mediated oropharyngeal cancer; AJCC: American Joint Committee on Cancer; TOVS: transoral videolaryngoscopic surgery; CRT: chemoradiotherapy.
Postoperative radiation therapy was performed in 19 of 33 surgical cases. Although radiation therapy alone was performed in most of these cases, only three patients with a positive margin (2 cases) and unresectable metastasis in the retropharyngeal lymph node (1 case) were treated with CRT (Table 2).
Comparison of prognosis of HPV+-OPC between the seventh and eighth editions
The 5-year OS was 77.3% in all patients with HPV+-OPC. In the seventh edition clinical staging, the OS rates were as follows: II, 100%; III, 100%; IVa, 75.9%; and IVb and IVc, 0%. The 5-year DSS rates were as follows: II, 100%; III, 100%; IVa, 83.3%; and IVb and IVc, 0%. The 5-year OS rates in the eighth edition clinical staging were as follows: I, 89.3%; II, 88.9%; III, 46.3%; and IV, 0%. The 5-year DSS rates were as follows: I, 95.0%; II, 100%; III, 57.3%; and IV, 0%. In the eighth edition, the 5-year OS and DSS rates were significantly different between clinical stages I + II and II I+ IV (p < 0.001), and between stages I and III (p < 0.001). However, in the seventh edition, the 5-year OS rate was not significantly different among the clinical stages. In the eighth edition pathological staging, the 5-year OS rate in each stage was 83.3% in stage I, 85.7% in stage II, and 100% in stage III, and the 5-year DSS rate was 83.3% in stage I, 100% in stage II, and 100% in stage III. There were no significant differences between the pathological stages for either OS or DSS (Figures 1, 2). The eighth edition clinical staging was more appropriate than the seventh edition to predict the prognosis of Japanese patients with HPV+-OPC, but the eighth edition pathological staging did not represent the prognosis in our results.
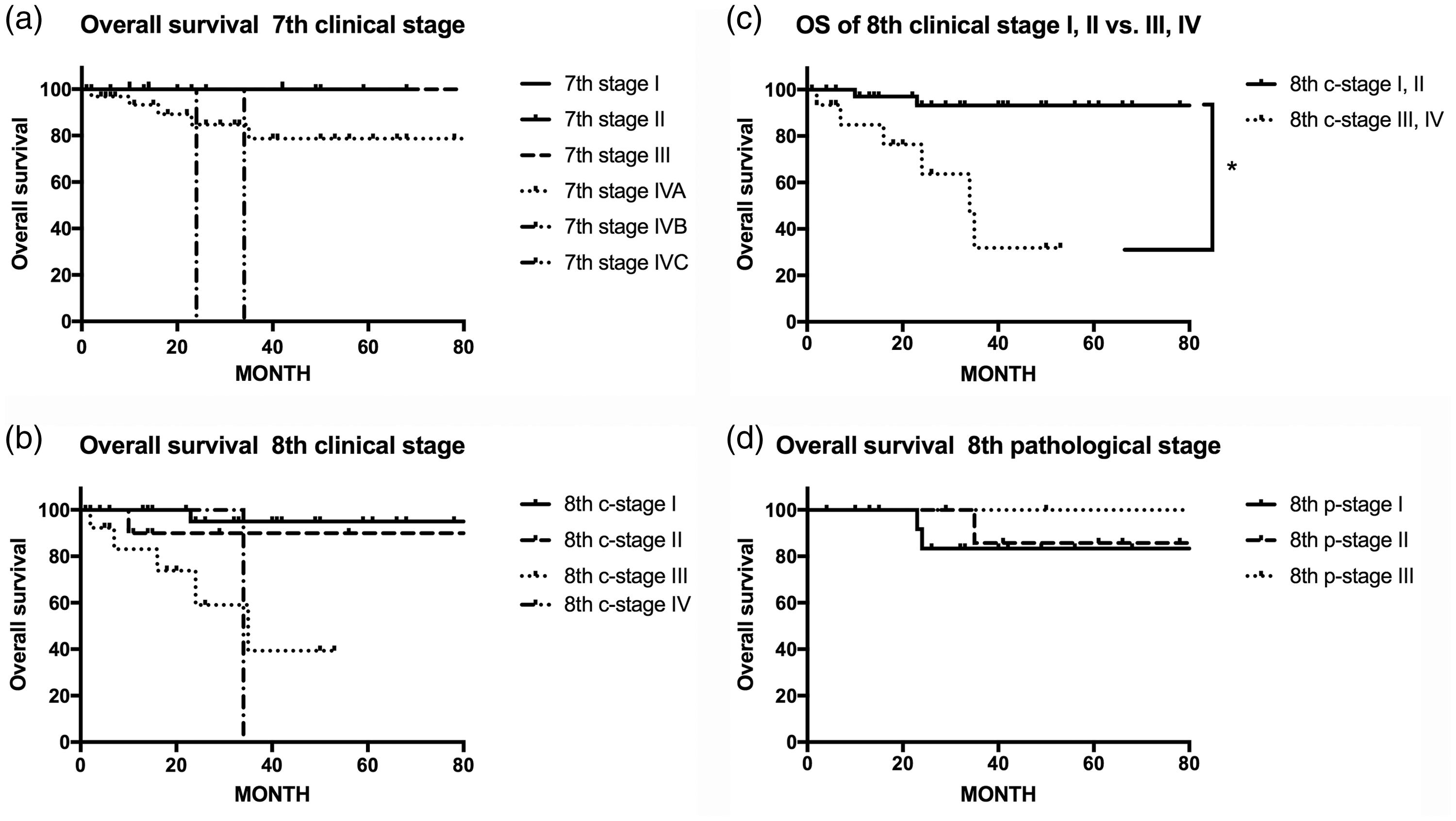
OS in the seventh edition versus the eighth edition of the American Joint Committee on Cancer in HPV+-OPC. (a) The 5-year OS rate was 77.3% in all patients with HPV+-OPC. (a) Five-year OS in the seventh edition clinical stage. (b) Five-year OS in the eighth edition clinical stage. (c) Five-year OS in the eighth edition clinical stage with comparison between the stages. Five-year OS rates were significantly different between clinical stages I+II and stages III+IV (p < 0.001), and between stages I and III (p < 0.001) in the eighth edition. (d) Five-year OS in the eighth edition pathological stage. *p < 0.001. OS: overall survival; HPV+-OPC: human papillomavirus-mediated oropharyngeal cancer.

DSS in the seventh edition versus the eighth edition of the American Joint Committee on Cancer in HPV+-OPC. (a) The 5-year DSS rate was 84.5% in all patients with HPV+-OPC. (a) Five-year DSS in the seventh edition clinical stage. (b) Five-year DSS in the eighth edition clinical stage. (c) Five-year DSS rates were significantly different between clinical stages I+II and stages III+IV (p < 0.001), and between stages I and III (p < 0.001) in the eighth edition. (d) Five-year DSS in the eighth edition pathological stage. *p < 0.001. DSS: disease-specific survival; HPV+-OPC: human papillomavirus-mediated oropharyngeal cancer.
Effect of smoking and treatment options on prognosis
The median BI was 780 (200–2400) in smokers. The 5-year OS rate was 75.0% in six patients with a BI <400 and 81.9% in 24 patients with a BI >400. There was no significant difference in prognosis between the two groups.
The 5-year OS rates were 90.9% and 70.7% in non-smokers and smokers with HPV+-OPC, respectively. The 5-year DSS rates of smokers and non-smokers with HPV+-OPC were 90.9% and 79.7%, respectively (Figure 3). Smokers with HPV+-OPC appeared to have worse OS than did non-smokers with HPV+-OPC, even though there was no significant difference in clinical and pathological stages between these two groups. TOVS was performed for 20 smokers with HPV+-OPC and CRT was performed for 12 smokers with HPV+-OPC.
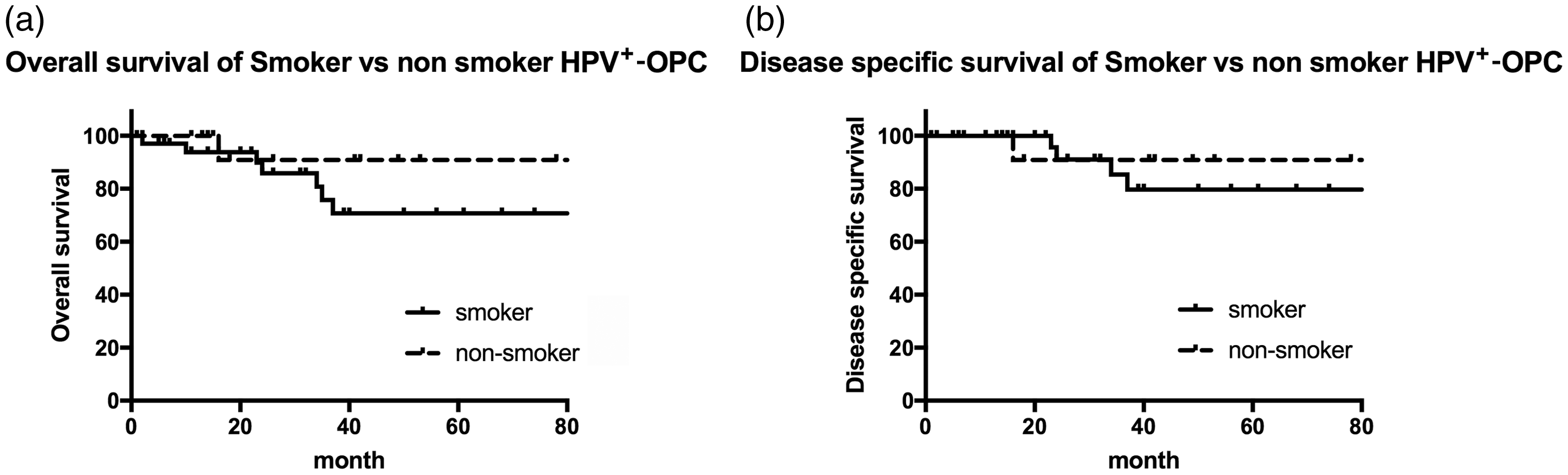
Survival rates of smokers versus non-smokers with HPV+-OPC. The 5-year OS was 90.9% and 70.7% in non-smoker and smoker HPV+-OPC, (Figure 3a), and the 5-year DSS of smoker and non-smoker HPV+-OPC was 90.9% and 79.7% (Figure 3b). Smoker HPV+-OPC had worse OS than non-smoker HPV+-OPC (p = 0.250).
To investigate whether TOVS improved the prognosis in smokers with HPV+-OPC, smokers with HPV+-OPC were classified according to the eighth edition of the AJCC, and the prognosis was assessed by dividing them into the TOVS group and the CRT group. There was no significant difference in the distribution of stages in smokers between the TOVS and CRT groups (Mann–Whitney test) (Table 3). The 5-year OS rate tended to be better in smokers in the TOVS group than in the CRT group (85.7% versus 75.0%, p=0.070) (Figure 4a). When limited to stages I and II, the 5-year OS rate was 100% in the TOVS group and 64.3% in the CRT group (p = 0.071). Although a significant difference was not clear, TOVS appeared to show a better prognosis than did the CRT group. Additionally, six (6/16) patients had a favorable prognosis without postoperative radiation therapy.

Survival rates of TOVS versus CRT in smokers with HPV+-OPC. In smokers with HPV+-OPC, 20 patients were treated with TOVS and 12 were treated with CRT. (a) In smokers with HPV+-OPC, the TOVS group tended to show a higher 5-year OS rate than did the CRT group (p=0.070). In non-smokers with HPV+-OPC, nine patients were treated with TOVS and six were treated with CRT. (b) In non-smokers with HPV+-OPC, 5-year OS was not significantly different between the TOVS and CRT groups. TOVS: transoral videolaryngoscopic surgery; CRT: chemoradiotherapy; HPV+-OPC: human papillomavirus-mediated oropharyngeal cancer; OS: overall survival.
In non-smokers with HPV+-OPC, the numbers of patients in each of the eighth clinical stages were as follows: I, 8; II, 1; III, 0; and IV, 0 in the TOVS group; and I, 2; II, 1; III, 3; and IV, 0 in the CRT group (Table 3). The 5-year OS was 100% in the TOVS group and 75.0% in the CRT group, with no significant difference between the groups (Figure 4b). This lack of a difference could be because of the small number of non-smoker HPV+-OPC cases.
Discussion
Validation of the eighth edition of AJCC staging
The eighth edition of AJCC staging was based on patients in North America and Europe. We investigated validation of this staging for Japanese patients with HPV+-OPC who belonged to different races and backgrounds. The proportion of Japanese patients with HPV+-OPC among OPC has been reported to be 40%–60%,7,8 which is less than that in North America and Europe. This rate was 43.9% in our study, which is within the range found in other countries. 17 In the seventh edition of AJCC staging, most HPV+-OPC is classified under stage IVa because of advanced N staging. In our study, there were no stage I patients, and the prognosis was not significantly different between stages II, III, and IVa. However, N3 and M1 staging had a poor prognosis, as well as stages IVb and IVc. In the eighth edition, HPV+-OPC was distributed evenly in each stage, and stages I and II had a significantly better prognosis than did stages III and IV. The International Collaboration on Oropharyngeal cancer Network for Staging (ICON-S) study showed that T3 and N2 of the eighth edition conferred a higher risk, and stage II patients had a poorer prognosis than did stage I patients. Our study did not show a significant difference in prognosis between stages I and II, probably because of the smaller amount of stage II patients than in the ICON-S study. There have also been few stage II cases in other studies in Japan,7,8 and a significant difference in prognosis was not found between stages I and II. The number of Japanese stage II cases is less than that in Europe and America. Therefore, more cases of stage II HPV+-OPC, including T3 and N2, are required for further evaluation.
In the eighth edition, pathological N staging was significantly revised. T staging was the same as conventional staging, but the metastatic node number defines N staging as follows: N1, four or fewer; and N2, more than four nodes.4,18 There have been few reports that examined the validity of pathological staging, 19 and the prognosis was not different between each stage in our study. We assumed that some clinical N3 cases, which have a poor prognosis, were classified as pathological N1 and this exacerbated the prognosis of stage I. In clinical staging, a lymph node with a major axis of ≥6 cm is regarded as a poor prognostic factor (N3). A previous report on matted nodes, where lymph node metastasis is consistent with fusion of multiple metastases, showed that matted nodes were an independent prognostic factor. 19 Not only the number of lymph node metastases, but also the size and shape of the lymph nodes, need to be added to prognostic factors in the pathological stage. Resectable T3 and T4 were not poor prognostic factors in our study. When surgery is performed, we can conduct appropriate additional treatment based on the pathology results, and treatment intensity can be increased for high-risk cases. We added radiation therapy for pathological T4 or N2 patients after surgery, which could improve the prognosis of pathological T4 and N2. Therefore, further validation is necessary for the validity of pathological staging.
Prognosis in smokers with HPV+-OPC
Several reports have shown that the 5-year OS rate of HPV+-OPC is approximately 80%,2,3,19 and HPV+-OPC is widely known to have a good prognosis. However, several studies have shown that smoking continues to pose a great risk of cancer progression and mortality, even in HPV+-OPC.11,20 Cerezo et al. 10 reported that the 3-year OS rate of smokers with HPV+-OPC, who were treated by radical radiotherapy or CRT, was 67.4%. They also showed that smokers with HPV+-OPC had a poorer prognosis than did non-smokers with HPV+-OPC. In their study, all patients with HPV+-OPC were treated with radiation therapy. HPV+-OPC expresses a wild-type p53, which is thought to be associated with radiosensitivity. Some reports have also suggested that smoking is associated with mutation of TP53 in head and neck cancer, which reduces radiosensitivity. 21 Therefore, smokers with HPV+-OPC are thought to have a poor prognosis and are treated only by CRT.
In our study, 54.7% of patients were treated with TOVS, and we evaluated the potential of TOVS in smokers with HPV+-OPC. We performed TOVS for T1, T2, and selective T3 stages of HPV+-OPC, and performed neck dissection simultaneously in case of node metastasis. Therefore, TOVS had a good indication for treatment of HPV+-OPC because of the small primary tumors and high frequency of metastasis in most cases. In smokers with HPV+-OPC, the prognosis of patients in the CRT group in our study is comparable with that in Cerezo et al.’s 10 report. However, the 5-year OS rate in the TOVS group was 85.7%, which is as good as that in previous reports of HPV+-OPC.6,9 Surgery can improve the prognosis of smokers with HPV+-OPC, regardless of radiosensitivity, and transoral surgery can resect with minimal invasion and less functional loss compared with conventional surgery. 22 Moreover, appropriate adjuvant treatment can be provided after surgery on the basis of pathological findings with higher intensity treatment with postoperative CRT for high-risk patients and with lower intensity of treatment by skipping radiation therapy for low-risk patients. We believe that providing appropriate post-surgical treatment is an important and large advantage of TOVS.
Our findings suggest that TOVS improves the prognosis of smokers with HPV+-OPC by increasing intensity of treatment, without functional loss and regardless of the smoking status. When limited to stages I and II, 16 smokers with HPV+-OPC were treated with TOVS, and the 5-year OS rate was 100%. Additionally, six of these 16 cases avoided postoperative radiotherapy. Smoking is a risk factor for secondary cancer in patients with head and neck cancer, 12 and preservation of radiotherapy is important to salvage second primary tumors. This suggests that transoral surgery, including TOVS and TORS, has great potential in treating smokers with HPV+-OPC who have a high probability of secondary cancer, and preserving radiotherapy for better prognosis.
This study has some limitations. Our study divided patients with HPV+-OPC into two groups of smokers with HPV+-OPC and non-smokers with HPV+-OPC. We also divided smokers and non-smokers with HPV+-OPC into two groups of the TOVS and CRT groups. Consequently, there were few patients in the TOVS and CRT groups. Therefore, increasing the number of patients with HPV+-OPC in this study is required in the future. Moreover, this study was a single-center, observational study, with a small number of cases. Therefore, further study in this area is necessary.
Conclusions
The eighth edition of the AJCC is more appropriate than the seventh edition for predicting the prognosis of Japanese HPV+-OPC in clinical staging. However, pathological staging was not associated with prognosis in our study. Smokers with HPV+-OPC have a poor prognosis, but TOVS might improve the prognosis in these patients. Some smokers with HPV+-OPC can be treated while preserving radiotherapy with transoral surgery.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This work was supported by a Special Research Grant on Defense Medicine from the Ministry of Defense of Japan, Grants-in-Aid for Scientific Research (C) (JSPS KAKENHI Nos. 18K09390, 17K11415, 16K01488, and 26462627), and a Grant-in-Aid for Challenging Exploratory Research (JSPS KAKENHI No. 16K15726) from the Japan Society for the Promotion of Science.
