Abstract
Background
Studies evaluating the role of telomerase activity in pancreatic adenocarcinoma are inconsistent and a systemic review of the available literature may shed new light on this issue.
Objective
To systematically review the usefulness of telomerase activity in distinguishing pancreatic cancer from other pancreatic diseases.
Methods
A comprehensive search of the PubMed and Embase databases was conducted to identify eligible studies. Only studies evaluating telomerase activity in patients with suspected or previously diagnosed pancreatic adenocarcinomas versus nonpancreatic adenocarcinomas and published in English with a sufficient number of cases were included. The hierarchical summary receiver operating characteristic (HSROC) model was used to establish the potential value of telomerase activity in the diagnosis of pancreatic adenocarcinoma.
Results
A total of 19 studies qualified for this meta-analysis. In distinguishing pancreatic adenocarcinoma from benign diseases, the pooled sensitivity and specificity of telomerase activity were 0.81 (95% CI, 0.68-0.90) and 0.97 (95% CI, 0.93-0.98), respectively; the diagnostic odds ratio (DOR) was 126.62 (95% CI, 49.94-320.99); beta was −1.16 (95% CI, −3.62-1.29), Z was −0.93, p was 0.35>0.1, and lambda was 6.86 (95% CI, 1.01-12.70). In distinguishing pancreatic adenocarcinoma from chronic pancreatitis, the pooled sensitivity and specificity of telomerase activity were 0.77 (95% CI, 0.61-0.88) and 0.97 (95% CI, 0.91-0.99), respectively; DOR was 117.28 (95% CI, 32.25-426.53); beta was −0.38 (95% CI, −1.89-1.13), Z was −0.49, p was 0.62>0.1, and lambda was 5.30 (95% CI, 3.37-7.24).
Conclusions
The present meta-analysis demonstrates that telomerase activity could be a useful biomarker for the differential diagnosis of pancreatic adenocarcinoma and benign pancreatic diseases.
Introduction
Pancreatic cancer, with approximately 90% of pancreatic tumors being pancreatic adenocarcinoma, is a highly malignant disease and has an extremely poor prognosis with a 6.7% 5-year survival rate (1, 2). Its poor prognosis is mainly due to the failure of diagnosis at an early stage. Early diagnosis and radical resection can improve the survival rate significantly. The incidence of pancreatic adenocarcinoma ranks second after colorectal cancer among gastrointestinal malignancies in industrialized countries (3). It is estimated that there will be 46,420 new cases of pancreatic cancer and 39,590 deaths from this disease in the United States in 2014 (4). Unfortunately, only 10%-15% of patients with pancreatic cancer have resectable disease at diagnosis (5). Thus, early diagnosis is the key to improving survival, but preoperative diagnosis of pancreatic lesions remains challenging.
Telomerase, which is composed of telomerase RNA (hTR), telomerase protein 1 (TP1) and telomerase reverse transcriptase (hTERT), is an important reverse transcriptase that is responsible for the synthesis of telomeric DNA (6). Several studies suggest that hTERT is the most important unit and reflects the activity of nuclear telomerase (7). Telomerase activity is absent in most somatic cells, but not in germ cells and stem cells (8). Interestingly, it is highly expressed in the majority of human cancers including lung cancer, hepatocellular carcinoma and breast cancer (9), suggesting that it plays an important role in human carcinogenesis. Telomerase has also been reported to be aberrantly expressed in patients with pancreatic adenocarcinoma (10, 11). However, the findings from studies evaluating the role of telomerase activity in pancreatic adenocarcinoma are inconsistent. Some reports indicate that telomerase activity can predict the prognosis of pancreatic adenocarcinoma (12) and can be a therapeutic target (13). Although some reports have demonstrated that telomerase activity is an effective diagnostic biomarker for pancreatic adenocarcinoma (14, 15), bladder cancer (16), prostate cancer (17), and cervical cancer (18), its accuracy remains uncertain. To the best of our knowledge, there has been no comprehensive meta-analysis to evaluate the diagnostic accuracy of telomerase activity for pancreatic adenocarcinoma. Hence, it is necessary to conduct a systematic review and meta-analysis to compare telomerase activity with other diagnostic markers and to establish its value in the diagnosis of pancreatic adenocarcinoma.
Methods
Database Search
We searched the PubMed and Embase literature databases for publications prior to June 10, 2015, using the following keywords: “pancreatic cancer”, “pancreatic neoplasms”, “pancreatic carcinoma”, “pancreas cancer”, “telomerase”, “human telomerase reverse transcriptase”, “human telomerase RNA”, and their combinations. The search was limited to human studies that were published in English. If the same population was the subject of multiple publications, only the most complete or most recent publication was selected. We also hand-searched reference lists from publications to identify additional potentially relevant studies. This process was performed iteratively until no additional articles could be identified.
Inclusion and Exclusion Criteria
We included studies that met the following criteria: (1) the purpose of study was to evaluate telomerase activity in patients with suspected or previously diagnosed pancreatic adenocarcinomas, compared with chronic pancreatitis or benign lesions; (2) the total number of cases was more than 20; (3) the final diagnoses were confirmed by histology, pathology, and imaging data or follow-up longer than 6 months; and (4) the publication was in English. Studies were excluded on the basis of the following criteria: (1) the publication belonged to the categories of case reports, case-only studies, letters, reviews or meta-analyses; (2) the sample size was not sufficient (n<20); (3) the study concerned other kinds of pancreatic cancer or other controls; (4) the study was published in duplication or with data deficiency or duplication; and (5) the full publication was not in English.
Data Extraction and Management
Data from each of the included studies were extracted independently by 2 reviewers (HG and XX) and any conflicting data were settled by discussion between the reviewers until consensus was reached. Data on first author, publication year, study region, ethnicity, period of study, age, detection method, specimen source, specimen collection, diagnostic criteria, source of controls, number of cases and controls, true positive, false positive, true negative, and false negative results were extracted and tabulated in a standard format. For publications including multiple diseases, cases of other kinds of pancreatic cancer were excluded.
Assessment of Quality of Evidence in Included Studies
The aforementioned 2 reviewers (HG and XX) independently assessed the quality of evidence in the included studies using the tool QUADAS-2 (Quality Assessment of Diagnostic Accuracy Studies-2) (19), which comprised 4 key domains including (i) patient selection, (ii) index test, (iii) reference standard flow and timing of the index tests, and (iv) reference standard for judging bias and applicability. The assessment process was conducted with the RevMan 5.2 software. We resolved disagreement about the selection of studies by discussion and consensus among the investigators.
Statistical Analysis
For each of the included studies, the data were abstracted as a 2 × 2 contingency table recording the presence of telomerase activity (positive vs. negative) and the histological diagnosis (benign or chronic pancreatitis). We calculated the true positive, true negative, false positive and false negative rates. When the table had one 0 cell, a correction of the calculation was made by adding a value of 0.5 to all cells of the table to avoid potential problems in odds calculation for sensitivities or specificities of 100%; if a study contained two 0 cells, it was excluded from the meta-analysis (20). The hierarchical summary receiver operating characteristic (HSROC) model was used to establish the potential value of telomerase activity in the diagnosis of pancreatic adenocarcinoma. The beta (β) and Z values assess the symmetry of SROC; the lambda (λ) value reflects the veracity of the diagnostic test discriminant ability. The pooled sensitivity, pooled specificity, diagnostic odds ratio (DOR), positive likelihood ratio (LR+), and negative likelihood ratio (LR-) were used to demonstrate the potential value of telomerase activity in discriminating pancreatic cancer from benign diseases (21). Publication bias was evaluated using the Deeks test. A p value of less than 0.1 (p<0.1) indicated an existing publication bias.
The statistical analyses were performed using STATA 12.0 (StataCorp, College Station, TX, USA). The whole statistical process was conducted independently by the reviewers (HG and YP); discrepancy in results was resolved by discussion with XX. A p value of less than 0.05 (p<0.05) was considered statistically significant with the exception of the Deeks test, as mentioned above.
Results
Included Studies and Quality Assessment
The search of the aforementioned databases identified 207 articles. After the screening of titles and abstracts, 172 articles were excluded and 35 potentially eligible articles were retrieved for analysis of the full text. Among them, 19 eligible articles (8, 10, 11, 14, 15, 22-23-24-25-26-27-28-29-30-31-32-33-34-35) qualified for the meta-analysis. Of the 16 excluded, 9 were not appropriate in their design (12, 36-37-38-39-40-41-42-43), 3 were duplicated publications (9, 44, 45), 2 included fewer than 20 cases (46, 47), and 2 were review articles (48, 49) (Fig. 1).
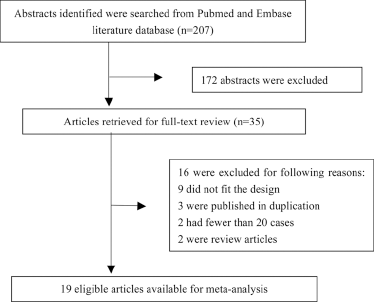
Flowchart of article screening and selection processes.
The characteristics of the 19 articles for pancreatic adenocarcinoma and index tests, detection method, specimen source, reference standard, and number of subjects are shown in Tables I and II. For the differential diagnosis of pancreatic cancer versus benign lesions, a total of 798 subjects were incorporated, while 567 subjects were included in the differential diagnosis of pancreatic cancer versus chronic pancreatitis.
Characteristics of included studies on telomerase activity and pancreatic adenocarcinoma
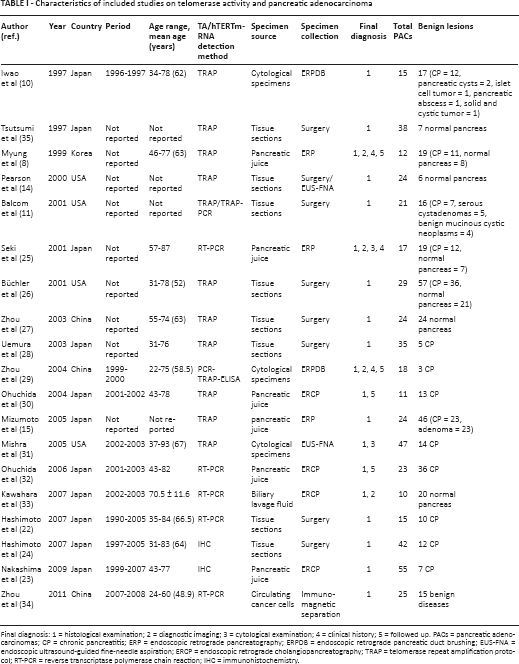
Final diagnosis: 1 = histological examination; 2 = diagnostic imaging; 3 = cytological examination; 4 = clinical history; 5 = followed up. PACs = pancreatic adenocarcinomas; CP = chronic pancreatitis; ERP = endoscopic retrograde pancreatography; ERPDB = endoscopic retrograde pancreatic duct brushing; EUS-FNA = endoscopic ultrasound-guided fine-needle aspiration; ERCP = endoscopic retrograde cholangiopancreatography; TRAP = telomerase repeat amplification protocol; RT-PCR = reverse transcriptase polymerase chain reaction; IHC = immunohistochemistry.
Contingency tables for pancreatic adenocarcinoma and chronic pancreatitis
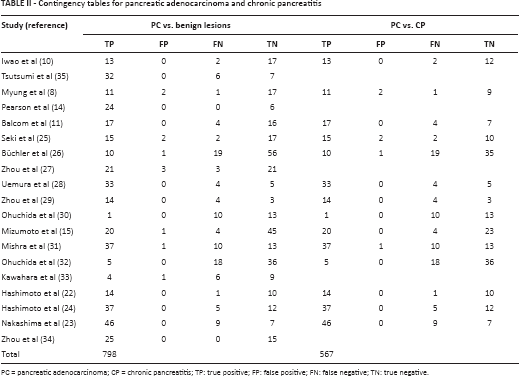
PC = pancreatic adenocarcinoma; CP = chronic pancreatitis; TP: true positive; FP: false positive; FN: false negative; TN: true negative.
Figure 2 shows the quality of the 19 eligible articles assessed by QUADAS-2 criteria. Thirteen studies (8, 14, 22-23-24-25-26-27-28-29, 33-34-35) were regarded as being at high risk of patient selection bias due to the case-control design; 9 studies (8, 11, 14-15, 25-26-27-28, 35) enrolled nonconsecutive patients; and 1 study (29) was classified as high risk in the index test domain due to the lack of the pre-specified threshold. All patients of 7 studies (8, 25, 29-30-31-32-33) received more than one reference standard and therefore these studies were classified as high risk in the flow and timing domain.
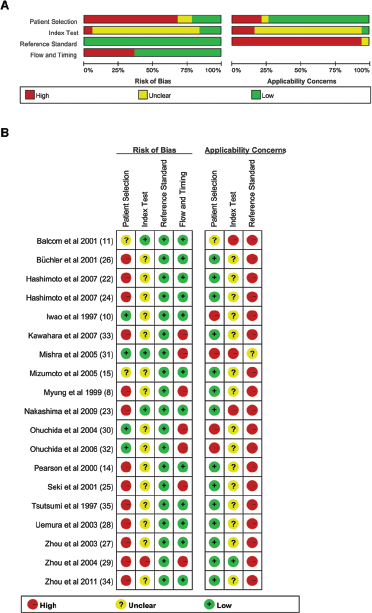
Risk of bias and applicability concerns graph: the review authors’ judgments about each domain are presented as percentages across the included studies (
Accuracy of Telomerase Activity in Differential Diagnosis of Pancreatic Adenocarcinoma versus Benign Lesions
The telomerase activity analysis was feasible in all 798 patients. The sensitivity for pancreatic adenocarcinoma detection ranged from 0.22 to 1.00 and the specificity ranged from 0.88 to 1.00; β = −1.16 (95% confidence interval [CI], −3.62-1.29), Z = −0.93, p = 0.35>0.1, λ = 6.86 (95% CI, 1.01-12.70). The pooled sensitivity, specificity, and DOR were 0.81 (95% CI, 0.68-0.90), 0.97 (95% CI, 0.93-0.98), and 126.62 (95% CI, 49.94-320.99), respectively. LR+ and LR- were 24.51 (95% CI, 11.88-50.56) and 0.19 (95% CI, 0.11-0.34), respectively (Fig. 3A and 3C). The Deeks test revealed no significant publication bias (t = −0.07, p = 0.943; Fig. 4A).
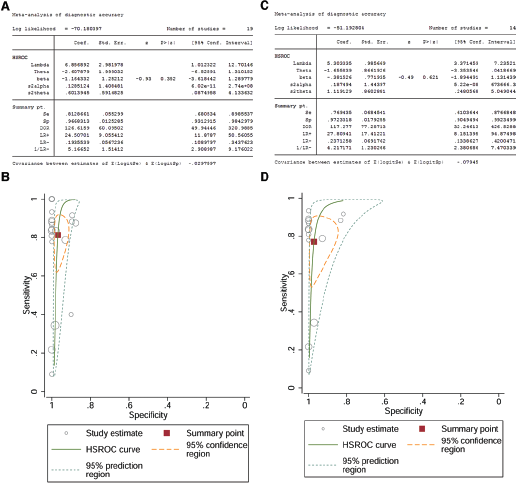
HSROC model for evaluating the potential value of telomerase activity in the diagnosis of pancreatic adenocarcinoma vs. benign lesions (
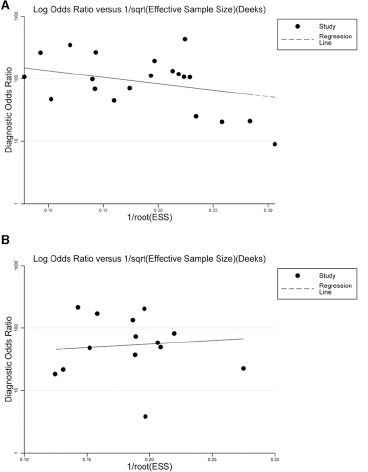
Deeks’ funnel plot of publication bias for pancreatic adenocarcinoma vs. benign lesions (
Furthermore, the subgroup analysis was stratified by specimen source (tissue sections: β = −0.25 [95% CI, −2.60-2.10]; Z = −0.21, p = 0.84>0.1; λ = 5.98 [95% CI, 3.77-8.20]; pooled sensitivity = 0.86 [95% CI, 0.72-0.94]; pooled specificity = 0.98 [95% CI, 0.80-0.998]; DOR = 295.68 [95% CI, 22.43-3897.01]; LR+ = 41.11 [95% CI, 3.79-446.58]; LR- = 0.14 [95% CI, 0.06-0.30]; pancreatic juice: β = −0.09 [95% CI, −1.49-1.31]; Z = −0.13, p = 0.90>0.1; λ = 4.92 [95% CI, 3.38-6.46]; pooled sensitivity = 0.68 [95% CI, 0.33-0.91]; pooled specificity = 0.98 [95% CI, 0.86-0.998]; DOR = 117.70 [95% CI, 18.34-755.53]; LR+ = 37.86 [95% CI, 5.43-263.97]; LR- = 0.32 [95% CI, 0.12-0.88]) (Fig. 5). Other studied specimen sources involving cytological specimens, biliary lavage fluid, and circulating cancer cells could not be combined because the HSROC model requires a minimum of 4 studies.
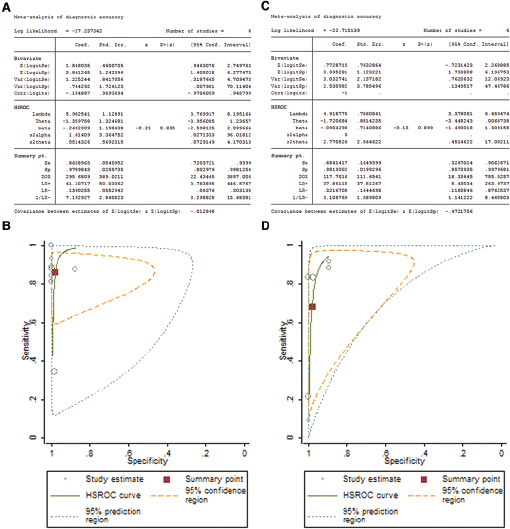
HSROC model for evaluating the potential value of telomerase activity in the diagnosis of pancreatic adenocarcinoma from tissue specimens (
Accuracy of Telomerase Activity in Differential Diagnosis of Pancreatic Adenocarcinoma versus Chronic Pancreatitis
Of the 19 studies, 14 (8, 10, 11, 15, 21-22-23-24-25, 27-28-29-30-31) involved 678 subjects in whom pancreatic cancer was differentiated from chronic pancreatitis; β = −0.38 (95% CI, −1.89-1.13), Z = −0.49, p = 0.62>0.1. Compared with the above results in studies differentiating pancreatic cancer from benign lesions, the λ value (5.30, 95% CI, 3.37-7.24) was slightly lower. The pooled sensitivity, specificity, and DOR were 0.77 (95% CI, 0.61-0.88), 0.97 (95% CI, 0.91-0.99), and 117.28 (95% CI, 32.25-426.53), respectively. LR+ and LR- were 27.81 (95% CI, 8.15-94.87) and 0.24 (95% CI, 0.13-0.42), respectively (Fig. 3B and 3D). Deeks test again revealed no significant publication bias (t = 0.30, p = 0.767; Fig. 4B).
Discussion
The results of this meta-analysis indicated that telomerase activity had good diagnostic accuracy to distinguish benign lesions from malignant tumors. For the differential diagnosis between pancreatic adenocarcinoma and chronic pancreatitis, telomerase activity also demonstrated good diagnostic value, although the values were not as good as those observed for the differential diagnosis between pancreatic cancer and benign lesions. In the specimen subgroup analysis, telomerase activity had better diagnostic value with tissue specimens than pancreatic juice. However, all sensitivity values were not higher as specificity for the effective clinical application.
The relationship between telomerase activity and pancreatic adenocarcinoma has been intensively studied, but the conclusions are not consistent. Some studies showed no obvious link between telomere length and pancreatic cancer (21, 34). Others (11, 26, 34) found that increased telomerase activity was related to clinical manifestation of pancreatic cancer, a finding that was contradicted by yet other studies (25, 27, 45). With regard to the diagnostic value, a larger number of studies have indicated that positive telomerase activity is found in pancreatic adenocarcinoma and that detecting telomerase activity can help diagnose pancreatic adenocarcinoma (14, 30). The results of the present meta-analysis suggested that telomerase activity is a powerful diagnostic tool for pancreatic cancer. Several types of cells such as hematopoietic stem cells, lymphocytes, basal skin cells and intestinal stem cells can also be characterized by low positive telomerase activity. Therefore, when detection of telomerase activity is used in the diagnosis of pancreatic cancer, close attention should be paid to its false positivity, especially in chronic pancreatitis and in pancreatic parenchyma where extensive lymphocyte infiltration may occur. The specimen source may also be a contributing factor, and detection of telomerase activity is more reliable in tissue specimens than in pancreatic fluid, because the composition of the latter is complex and unstable.
For a better comprehensive understanding of the effects of various diagnostic methods on the diagnostic accuracy, we systematically summarized the evidence from other meta-analyses and studies (Tab. III). Benign pancreatic diseases such as chronic pancreatitis and pancreatic cysts can be a confounding factor. Several imaging technologies including ultrasound (50-51-52), computed tomography (53, 54), magnetic resonance imaging (55), and endoscopic retrograde cholangiopancreatography (50) play an important role in identifying pancreatic cancer, but none of them is sufficiently sensitive and specific or has a high positive likelihood ratio. Meanwhile, a wide range of biochemical tumor markers have been found to be elevated in patients with pancreatic cancer (56). For example, CA199 is widely used for pancreatic cancer diagnosis, but it lacks specificity on account of its increased values in benign or malignant biliary diseases and the overall diagnostic accuracy is low (57, 58). CA242 (macrophage inhibitory cytokine-1) also has some disadvantages (58, 59). Additionally, of the genetic alterations, mutated
Accuracy of detection methods for pancreatic cancer (evidence from meta-analyses)
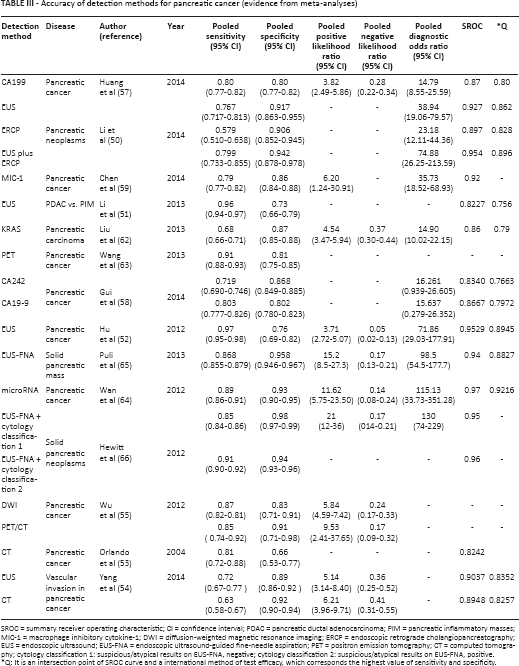
SROC = summary receiver operating characteristic; CI = confidence interval; PDAC = pancreatic ductal adenocarcinoma; PIM = pancreatic inflammatory masses; MIC-1 = macrophage inhibitory cytokine-1; DWI = diffusion-weighted magnetic resonance imaging; ERCP = endoscopic retrograde cholangiopancreatography; EUS = endoscopic ultrasound; EUS-FNA = endoscopic ultrasound-guided fine-needle aspiration; PET = positron emission tomography; CT = computed tomography; cytology classification 1: suspicious/atypical results on EUS-FNA, negative; cytology classification 2: suspicious/atypical results on EUS-FNA, positive.
Q: It is an intersection point of SROC curve and a international method of test efficacy, which corresponds the highest value of sensitivity and specificity.
Hence, it is necessary to investigate a new diagnostic marker with high sensitivity, specificity and stability for early diagnosis of pancreatic cancer. Many studies have demonstrated that PET (63), PET/CT (55), and microRNA (64) may be useful in the early diagnosis of pancreatic cancer. The diagnostic efficacy of 2 or more methods combined may be better than that of 1 method alone. For example, endoscopic ultrasound-guided fine-needle aspiration cytology yields better results (65, 66) than some of the biomarkers including telomerase activity. Nevertheless, telomerase activity can be a candidate marker, and its diagnostic effect can be improved when combined with other methods.
In addition to the small sample size, there were several other limitations in the present meta-analysis. First, English language-only publications were included, which potentially caused publication bias. Second, some low-quality studies included may have influenced the reliability of the results. Third, the cutoff values of telomerase among the included studies differed or were not reported. In addition, different methods were used in the analysis of different specimens, which may lead to significant variations in the results obtained. Publication bias is an inherent limitation of all meta-analyses, irrespective of the outcomes of the Deeks test.
In conclusion, telomerase activity is likely to play an important role in the diagnosis of pancreatic adenocarcinoma, and the diagnostic effect would be improved when it is combined with other diagnostic tools. Future prospective, large-scale multicenter studies are needed to generate higher quality evidence necessary to verify these conclusions. In addition, PET/CT and microRNA may be useful for the early diagnosis of pancreatic cancer.
Footnotes
Financial support: The work was supported by International S&T Cooperation Program of China (Grant No. 2014D FA31420).
Conflict of interest: The authors declare no competing interest.
